Abstract
Introduction
The impact of in vitro resistance on initially appropriate antibiotic therapy (IAAT) remains unclear. We elucidated the relationship between non-IAAT and mortality, and between IAAT and multi-drug resistance (MDR) in sepsis due to Gram-negative bacteremia (GNS).
Methods
We conducted a single-center retrospective cohort study of adult intensive care unit patients with bacteremia and severe sepsis/septic shock caused by a gram-negative (GN) organism. We identified the following MDR pathogens: MDR P. aeruginosa, extended spectrum beta-lactamase and carbapenemase-producing organisms. IAAT was defined as exposure within 24 hours of infection onset to antibiotics active against identified pathogens based on in vitro susceptibility testing. We derived logistic regression models to examine a) predictors of hospital mortality and b) impact of MDR on non-IAAT. Proportions are presented for categorical variables, and median values with interquartile ranges (IQR) for continuous.
Results
Out of 1,064 patients with GNS, 351 (29.2%) did not survive hospitalization. Non-survivors were older (66.5 (55, 73.5) versus 63 (53, 72) years, P = 0.036), sicker (Acute Physiology and Chronic Health Evaluation II (19 (15, 25) versus 16 (12, 19), P <0.001), and more likely to be on pressors (odds ratio (OR) 2.79, 95% confidence interval (CI) 2.12 to 3.68), mechanically ventilated (OR 3.06, 95% CI 2.29 to 4.10) have MDR (10.0% versus 4.0%, P <0.001) and receive non-IAAT (43.4% versus 14.6%, P <0.001). In a logistic regression model, non-IAAT was an independent predictor of hospital mortality (adjusted OR 3.87, 95% CI 2.77 to 5.41). In a separate model, MDR was strongly associated with the receipt of non-IAAT (adjusted OR 13.05, 95% CI 7.00 to 24.31).
Conclusions
MDR, an important determinant of non-IAAT, is associated with a three-fold increase in the risk of hospital mortality. Given the paucity of therapies to cover GN MDRs, prevention and development of new agents are critical.
Similar content being viewed by others
Introduction
Antimicrobial resistance is a growing challenge in the care of critically ill patients, among whom the burden of infection remains high. Escalating rates of antibiotic resistance add substantially to the morbidity, mortality, and cost related to infection in the ICU [1]. Traditionally, most efforts to understand issues of resistance and ICU outcomes have addressed Gram-positive organisms, such as methicillin-resistant Staphylococcus aureus [2],[3]. However, in the United States, alarming trends in resistance are now also reported for a number of Gram-negative pathogens. For example, extended-spectrum beta-lactamase (ESBL) organisms are now endemic in many ICUs, and 15 to 20% of all Pseudomonas aeruginosa isolates from serious infections are categorized as multidrug resistant (MDR) because of reduced in vitro susceptibility to three or more classes of antibiotics [4]-[6]. Of even more concern are pathogens for which clinicians have few antibiotic options, namely Acinetobacter baumanii and carbepenemase-producing Enterobacteriaceae (CPE) [4]-[6]. In the case of these Gram-negative organisms, studies also point to an association between resistance and both clinical and economic outcomes [1].
The mechanism for poor outcomes with resistant Gram-negative organisms is not completely clear. In general, these bacteria are not believed to be inherently more virulent than similar susceptible species. Resistance and its rapid evolution, however, make efforts to insure initially appropriate antibiotic therapy (IAAT) more difficult, and IAAT is a key determinant of outcome in severe infection [7]-[10]. IAAT has consistently been shown to reduce mortality rates in severe sepsis and septic shock, and the Surviving Sepsis Campaign Guidelines strongly support initiatives to guarantee that patients receive timely antibiotic treatment [11]-[16]. However, it remains unclear what proportion of IAAT is driven by in vitro resistance. Appreciating this relationship may facilitate efforts to improve outcomes by helping clinicians determine how to apply newer diagnostic modalities and therapeutic options.
We sought to confirm the importance of IAAT in severe sepsis and septic shock due to Gram-negative bacteria and to estimate the impact of initially inappropriate antibiotic therapy (non-IAAT) on mortality in these syndromes. More importantly, we aimed to identify variables associated with IAAT and to elucidate the relationship between IAAT and in vitro antimicrobial resistance. To accomplish this we conducted a large retrospective analysis of subjects with severe sepsis or septic shock and Gram-negative bacteremia.
Materials and methods
Study design and ethical standards
We conducted a single-center retrospective cohort study from January 2008 to December 2012. Barnes-Jewish Hospital is a 1,200-bed urban academic medical center located in St. Louis, MO, USA. The study was approved by the Washington University School of Medicine Human Studies Committee and informed consent was waived since the data collection was retrospective without any patient-identifying information. The study was performed in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments.
Study cohort
All consecutive adult ICU patients between January 2008 and December 2012 were included if: they had a positive blood culture for a Gram-negative organism; and there was an International Classification of Diseases, Version 9, Clinical Modification (ICD-9-CM) code corresponding to an acute organ dysfunction [17]. Only the first episode of sepsis was included.
Definitions
To be included in the analysis, patients had to meet criteria for severe sepsis based on discharge ICD-9-CM codes for acute organ dysfunction [17]. Patients were classified as having septic shock if vasopressors (norepinephrine, dopamine, epinephrine, phenylephrine, or vasopressin) were initiated within 24 hours of the blood culture collection date and time. Antimicrobial treatment was deemed IAAT if the initially prescribed antibiotic regimen was active against the identified pathogen based on in vitro susceptibility testing and was administered within 24 hours following blood culture collection. Combination therapy was not required to be considered IAAT. We also required that antibiotics had to be prescribed for at least 24 hours. All other regimens were classified as non-IAAT. Prior antibiotic exposure was any exposure to an antibiotic within the preceding 90 days. Combination antimicrobial treatment was not required for IAAT designation. This is supported by multiple studies indicating that while dual therapy is more likely than single therapy to result in appropriate coverage, it is not necessarily associated with better outcomes provided the organism is adequately covered by a single drug [18]. We utilized the same time frame (90 days prior to the onset of the current episode of bacteremia) to define prior hospitalization. In contrast, prior bacteremia was defined by a bacteremia that had occurred within 30 days of the current episode. Multidrug-resistant P. aeruginosa (MDR-PA) was defined as P. aeruginosa resistant to at least three of the following classes of antimicrobials: aminoglycosides, anti-pseudomonal penicillins, anti-pseudomonal cephalosporins, carbapenems, and fluoroquinolones. A case was classified as MDR if the blood culture was positive for a MDR-PA, an ESBL organism, or a CPE. Both ESBL and CPE status were established based on molecular laboratory testing.
Antimicrobial treatment algorithms
From January 2002 through to the present, Barnes-Jewish Hospital utilized an antibiotic control program to help guide antimicrobial therapy. During this time cefepime, gentamicin, vancomycin, or fluconazole use was unrestricted. However, initiation of ciprofloxacin, imipenem, meropenem, piperacillin/tazobactam, linezolid, daptomycin, or micafungin was restricted and required preauthorization from a clinical pharmacist or infectious diseases physician. Each ICU had a clinical pharmacist who reviewed antibiotic orders to ensure that dosing and the interval of administration were adequate for patients based on body size, renal function, and resuscitation status. After daytime hours the on-call clinical pharmacist reviewed and approved the antibiotic orders. The initial antibiotic dosages employed for treatment were as follows: cefepime, 1 to 2 g every 8 hours; piperacillin-tazobactam, 4.5 g every 6 hours; imipenem, 0.5 g every 6 hours; meropenem, 1 g every 8 hours; ciprofloxacin, 400 mg every 8 hours; gentamicin, 5 mg/kg once daily; vancomycin, 15 mg/kg every 12 hours; linezolid, 600 mg every 12 hours; daptomycin, 6 mg/kg every 24 hours; fluconazole, 800 mg on the first day followed by 400 mg daily; and micafungin, 100 mg daily.
Starting in June 2005, with regular updates, a sepsis order set was implemented in the emergency department, general wards, and the ICUs with the intent of standardizing empiric antibiotic selection for patients with sepsis based on the infection type (i.e. community-acquired pneumonia, healthcare-associated pneumonia, intra-abdominal infection, and so forth) and the hospital’s antibiogram. However, antimicrobial selection, dosing, and de-escalation of therapy were still optimized by clinical pharmacists in these clinical areas.
Antimicrobial susceptibility testing
The microbiology laboratory performed antimicrobial susceptibility of the Gram-negative blood isolates using the disk diffusion method according to guidelines and breakpoints established by the Clinical Laboratory and Standards Institute and published during the inclusive years of the study [19],[20].
Data elements
Patient-specific baseline characteristics and process of care variables were collected from the automated hospital medical record, microbiology database, and pharmacy database of Barnes-Jewish Hospital. Electronic inpatient and outpatient medical records available for all patients in the BJC Healthcare system were reviewed to determine prior antibiotic exposure. The baseline characteristics collected included: age, gender, race, past history of congestive heart failure, chronic obstructive pulmonary disease, diabetes mellitus, chronic liver disease, underlying malignancy, and end-stage renal disease requiring dialysis. The comorbidities were identified based on their corresponding ICD-9-CM codes. The Acute Physiology and Chronic Health Evaluation II and Charlson comorbidity scores were calculated based on clinical data present during the 24 hours after the positive blood cultures were obtained [21]. This was done to accommodate patients with community-acquired and healthcare-associated community-onset infections who only had clinical data available after blood cultures were drawn. Healthcare-associated infections were defined by the presence of at least one of the following risk factors: recent hospitalization (within 90 days of the current one); immune suppression; nursing home residence; hemodialysis; and prior antibiotics (within 90 days of the current hospitalization). The primary outcome variable was hospital mortality. Because we were interested in understanding the contribution of MDR pathogens to the risk of receiving non-IAAT, we examined this variable as a secondary endpoint in a logistic regression.
Statistical analyses
Continuous variables were reported as means with standard deviations and as medians with 25th and 75th percentiles. Differences between mean values were tested via the Student’s t test, while those between medians were examined using the Mann–Whitney U test. Categorical data were summarized as proportions, and the chi-square test or Fisher’s exact test for small samples was used to examine differences between groups. We developed several multiple logistic regression models to identify clinical risk factors that were associated with hospital mortality. In the mortality models, all risk factors that were significant at ≤0.20 in the univariate analyses, as well as all biologically plausible factors even if they did not reach this level of significance, were included in the corresponding multivariable analyses. All variables entered into the models were examined to assess for colinearity, and interaction terms were tested. The most parsimonious models were derived using the backward manual elimination method, and the best-fitting model was chosen based on the area under the receiver operating characteristics curve (c statistic). The model’s calibration was assessed with the Hosmer–Lemeshow goodness-of-fit test. Similarly, the most parsimonious model for the predictors of inappropriate empiric antibiotic was computed and its fit was tested with the c statistic and the Hosmer–Lemeshow goodness of fit. All tests were two tailed, and P <0.05 was deemed a priori to represent statistical significance.
All computations were performed in Stata/SE, version 9 (StataCorp, College Station, TX, USA).
Results
In total, 1,076 patients with severe sepsis or septic shock due to a Gram-negative pathogen met the inclusion criteria. The distribution of the pathogens is presented in Table 1. Among these 1,076 culture-positive cases, there were 63 (5.9%) cultures that met the MDR criteria (Table 1). The most common MDR organism was MDR-PA, accounting for 15.0% of all P. aeruginosa isolates.
Among the 1,064 patients whose hospital disposition was known, 311 (29.2%) died in the hospital. Their baseline characteristics are presented in Table 2. Patients who died were older, less likely to be admitted from home, and had a higher comorbidity burden than those who survived their hospitalization, as signified by the Charlson comorbidity score. A higher proportion of those patients who died prior to discharge (95.7%) had a risk factor for a healthcare-associated infection than those who were discharged alive (91.4%, P = 0.014).
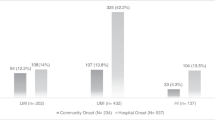
In the run-up to and at the time of sepsis onset, patients who did not survive had a slightly longer presepsis hospital length of stay, although this difference did not meet the predetermined level of statistical significance (Table 2). Several healthcare-associated factors (hemodialysis, prior hospitalization, and antibiotics) were more prevalent among nonsurvivors. However, the vast majority of the cohort (over 90%) had at least one healthcare-associated risk factor (Table 2). Additionally, survivors had a higher frequency of having had surgery during the index hospitalization than those who died. All markers of severity of acute illness were higher in patients who died compared with those who survived; the Acute Physiology and Chronic Health Evaluation II score was higher, and septic shock and the need for mechanical ventilation were significantly more prevalent among nonsurvivors than among survivors (Table 2, Figure 1). Urine and an infected line were less likely sources of infection and the lung was more likely as a source of infection among nonsurvivors compared with survivors. There were also striking differences between the two groups in terms of the likelihood of a MDR pathogen as the sepsis culprit (10.0% among nonsurvivors vs. 4.0% among survivors, P <0.001) (Figure 1). Additionally, nonsurvivors were approximately three times more likely to receive non-IAAT than those patients who survived their hospitalization (43.4% vs. 14.6%, P <0.001) (Figure 1). Among the 245 patients who received non-IAAT, resistance to instituted empiric therapy was far more prevalent as a reason (75.5%) than delay in treatment (24.5%). When stratified by hospital death, the relationship generally held, although delay in treatment was slightly more likely among those who died (28.9%) than those who survived their hospitalization (19.1%, P = 0.076). Similarly, delay in therapy accounted for a minority of non-IAAT among patients with a MDR pathogen (25.5%), with a nearly identical frequency of delay observed among those without a MDR pathogen (23.8%, P = 0.798).
Multiple logistic regression models were constructed and tested for fit, with the factors presented in Table 3 having the best discrimination. In this model, as in others that included this model, receiving non-IAAT was the strongest predictor of hospital death with an adjusted odds ratio of 3.87 (95% confidence interval = 2.77 to 5.41, P <0.001, c statistic = 0.777).
When focusing on the choice of empiric treatment among patients with a MDR pathogen versus those without, the unadjusted odds ratio of receiving non-IAAT was 11.79 (95% confidence interval = 6.55 to 21.23, P <0.001). In a logistic regression model to examine the factors that contribute to this inappropriate choice of therapy, a MDR pathogen as the etiology of sepsis was the strongest predictor of inappropriate treatment with an adjusted odds ratio of 13.05 (95% confidence interval = 7.00 to 24.31, P < 0.001, c statistic = 0.738) (Table 4). This parameter had by far the highest odds of any variable retained in the model of predictors of non-IAAT. (Tables 5, 6 and 7 present the details of characteristics based on appropriateness of treatment, as well as an alternative model for the predictors of non-IAAT. See Table 7 footnote for a brief discussion of that model.)
Discussion
This large retrospective analysis confirms that non-IAAT is a key determinant of short-term mortality among patients with severe sepsis and septic shock due to a Gram-negative organism. More importantly, our findings indicate that the presence of a MDR Gram-negative pathogen is strongly associated with non-IAAT. Despite the relatively low prevalence of a MDR phenotype among all subjects with Gram-negative bacteremia, these pathogens exert an excessive impact on mortality. In other words, MDR pathogens disproportionately affect outcomes through an intermediate step as it relates to antibiotic therapy. In light of the increasing frequency of multidrug resistance, our observations suggest that urgent action is needed to prevent potential escalation of mortality rates in severe sepsis and septic shock.
Because the co-occurrence of MDR pathogens and non-IAAT was relatively rare, it is important to consider the context of total non-IAAT exposure. The pool for the MDR pathogens as defined in our study comprises the vast majority of Gram-negative organisms responsible for serious infections in the ICU. That is, compared with Acinetobacter spp., for example, the relative prevalence of P. aeruginosa and Enterobacteriaceae was an order of magnitude higher. Epidemiologically, this imbalance makes it imperative for clinicians to consider these organisms first and foremost when choosing empiric treatment. We have demonstrated that multidrug resistance among these organisms comprises one important mechanism for errors in empiric coverage. At the same time, Acinetobacter spp. and Stenotrophomonas maltophilia infections, although a minority, were extremely likely to be subject to inappropriate empiric treatment (Table 6). Because the risk for drug resistance is very high among these organisms, the observed elevated rates of non-IAAT are probably not because the clinician did not consider their risk for resistance, but rather due to his/her determination that these were not likely pathogens. This approach therefore represents a slightly different mechanism for causing non-IAAT and implies a different solution. Rather than understanding the antibiogram of common pathogens, this requires a clinician to be aware of the rates of specific less common organisms at his/her institution. An additional important mechanism for receiving non-IAAT exists based on the timing of empiric therapy. Fully one-quarter of all non-IAAT fell into this category when there was no evidence of empiric treatment within 24 hours of obtaining the blood culture. This informs yet another corrective approach, one that requires simply to recognize the presence of a severe infection and to institute empiric treatment in a timely manner. These three mechanisms for exposure to non-IAAT and their corrective strategies are subtly yet importantly different from one another. In the current study we focus specifically on the impact of multidrug resistance on the risk of non-IAAT.
The prevalence of Gram-negative resistance has been mounting over the last decade [4]-[6]. However, most prior work describing the epidemiology of MDR Gram-negative pathogens has focused on the prevalence of resistance among specific species in specific infections. For example, a recent study demonstrated that between 2000 and 2009 nationwide in the United States there was a rise of MDR-PA from 10.7 to 13.5% in bloodstream infections, and from 19.2 to 21.7% in pneumonia [4]. The proportion of P. aeruginosa that met the MDR definition in the current study (15.0%) is consistent with these national estimates. The prevalence of carbapenem-resistant Enterobacteriaceae that we report here is also in line with national estimates [4]-[6]. In general, the similarity of the overall prevalence of multidrug resistance in our study to what has been reported nationally lends external validity to our observations. Moreover, our study is unique in its pragmatic perspective relevant to an ICU clinician and focuses on a common syndrome that represents a final common pathway for several infection types.
Much research from the last decade has highlighted the strong relationship between the choice of empiric antimicrobial treatment and the risk of death among patients hospitalized with serious infections. Most studies suggest that the risk of hospital death in association with non-IAAT goes up twofold to fourfold when compared with patients who receive appropriate coverage [7]-[9],[11]-[15]. Furthermore, switching from inappropriate to appropriate coverage once the culture results have become available does not reduce the mortality risk imparted by this early failure [10]. In this way our study adds to the understanding of the importance of choosing appropriate empiric treatment specifically to the outcomes of Gram-negative sepsis, and extends this understanding to suggest not only the mechanism for this finding, but also the contribution of multidrug resistance to the risk of making this important error in early management.
The potential policy and public health implications of our results are significant. Most attempts to improve rates of IAAT have relied on a strategy of prompt administration of broad empiric coverage informed by the local antibiogram, followed by de-escalation. In fact, this is the strategy advocated by the Surviving Sepsis Campaign [16]. To prevent antibiotic abuse, the broad regimen is tailored as culture data become available, and the shortest appropriate course of therapy is given. This paradigm suggests that the way to address low rates of IAAT is to shift to using broader spectrum agents such as anti-pseudomonal carbapenems or ESBLs and/or chephalosporins. Unfortunately, in the case of MDR Gram-negative organisms, this is simply not an option. Few agents currently available provide in vitro activity against MDR-PA and CPE. Those agents that are available, such as colistin, carry important, albeit somewhat controversial, safety concerns [22]-[25]. Simply selecting broader spectrum agents for the initial therapy is therefore not an option, because the current antibiotic armamentarium does not cover these MDR organisms. This highlights why new agents are urgently needed. As such, regulatory authorities and policy-makers need to develop expedited pathways for antibiotic development and approval. Such initiatives in the United States as the Generating Antibiotic Incentives Now Act, which provide incentives to support the development of newer antibiotics, are to be lauded [26]. These efforts must continue to be expanded and refined.
An additional point worth emphasizing is the relatively low prevalence of MDR pathogens in our study, and the implications of this for potential overuse of empiric broad-spectrum antibiotics, if such are available. Although certainly suboptimal with respect to both overuse and increased resource utilization, at the moment there is no way to tailor such therapies with any degree of precision. Yet not administering appropriate coverage results in a high penalty for the patient who is unlucky enough to harbor a MDR organism, with a fourfold increase in the risk of death. This situation underscores the urgency of the need for development of faster diagnostic tools, as well as risk stratification algorithms that may help clinicians to use broad-spectrum drugs appropriately. At the moment, however, the only viable solution appears to be to understand local resistance patterns in real time and make therapeutic choices based on them.
Our study has a number of limitations. As a retrospective cohort it is prone to several forms of bias, most notably selection bias. We attempted to mitigate this by enrolling consecutive patients fitting the pre-determined enrollment criteria. Although we dealt with confounders by adjusting for those that were available, it is possible that some residual confounding remains. One specific potential residual confounder is the type of surgery; that is, although we have data on whether each patient either had a surgical procedure or was cared for on a surgical service during his/her hospitalization, we do not know whether the surgery was related to the sepsis episode or was performed for infectious source control. However, based on prior experience at BJC, only a minority of patients is likely to have undergone source control surgery. The fact that this is a single-center study in a very specific population of patients (those with Gram-negative sepsis) may diminish the generalizability of our results to other centers and populations. One important point is that Clinical Laboratory and Standards Institute break-points for susceptibility changed for some of the antibiotics during the study time frame [19],[20]. The lowering of these values almost certainly resulted in an increase in the proportion of resistant organisms. This likely increase, however, would dilute rather than inflate the impact of multidrug resistance on the receipt of IAAT. Since we used only the susceptibility profile and the timing of antibiotic administration as surrogates for IAAT, our definition may have been overly liberal and included some cases that would have been deemed non-IAAT if other factors such as dosing and tissue penetration had been examined. Another source of possible misclassification is our use of ICD-9-CM codes to identify organ failures. While this identification may be less accurate than clinical data, this methodology has been validated and widely utilized in health services research [17]. The same situation arose for comorbidities, thus eliminating the possibility of examining whether or how their severity may impact the outcomes. Finally, because we examined hospital mortality rather than the more standard 28-day mortality as the primary outcome for our study, we may have underestimated the magnitude of this outcome.
Conclusions
In summary, our study provides evidence that once the high risk of a serious infection has been recognized by a clinician and empiric treatment for common pathogens instituted, MDR organisms are an important factor in determining the risk of non-IAAT, and, by extension, hospital mortality in Gram-negative sepsis. Given the paucity of currently available antimicrobial options to cover this emerging threat, the key immediate solution is their prevention through various protocols to address ventilator and central venous catheter care, as well as through antibiotic stewardship programs [27]-[29].
Definitions
Septic shock: vasopressors (norepinephrine, dopamine, epinephrine, phenylephrine, or vasopressin) initiated within 24 hours of the blood culture collection date and time.
IAAT: initially prescribed antibiotic regimen active against the identified pathogen based on in vitro susceptibility testing and administered within 24 hours following blood culture collection.
Prior antibiotic exposure: any exposure to an antibiotic within the preceding 90 days.
Prior hospitalization: any hospitalization within the preceding 90 days.
Prior bacteremia: a bacteremia episode within 30 days of the current episode.
MDR-PA: a P. aeruginosa resistant to at least three of the following classes of antimicrobials: aminoglycosides, anti-pseudomonal penicillins, anti-pseudomonal cephalosporins, carbapenems, and fluoroquinolones.
MDR case: blood culture positive for a MDR-PA, an ESBL organism or a CPE.
Healthcare-associated infection: the presence of at least one of the following risk factors: recent hospitalization (within 90 days of the current one); immune suppression; nursing home residence; hemodialysis; and prior antibiotics (within 90 days of the current hospitalization).
Key messages
-
Among patients with severe sepsis/septic shock due to a Gram-negative organism, initially inappropriate antibiotic treatment is associated with a threefold increase in hospital mortality.
-
Multidrug resistance is strongly associated with inappropriate treatment.
Abbreviations
- CPE:
-
carbapenemase-producing Enterobacteriaceae
- ESBL:
-
extended-spectrum beta-lactamase
- IAAT:
-
initial appropriate antibiotic therapy
- ICD-9-CM:
-
International Classification of Diseases, Version 9, Clinical Modification
- MDR:
-
multidrug resistant
- MDR-PA:
-
multidrug-resistant Pseudomonas aeruginosa
References
Maragakis LL, Perencevich EN, Cosgrove SE: Clinical and economic burden of antimicrobial resistance. Expert Rev Anti Infect Ther. 2008, 6: 751-763. 10.1586/14787210.6.5.751.
National Nosocomial Infections Surveillance (NNIS) system report. Am J Infect Control 2004, 32:470.,
Obritsch MD, Fish DN, MacLaren R, Jung R: National surveillance of antimicrobial resistance in Pseudomonas aeruginosa isolates obtained from intensive care unit patients from 1993 to 2002. Antimicrob Agents Chemother. 2004, 48: 4606-4610. 10.1128/AAC.48.12.4606-4610.2004.
Zilberberg MD, Shorr AF: Prevalence of multidrug-resistant Pseudomonas aeruginosa and carbapenem-resistant Enterobacteriaceae among specimens from hospitalized patients with pneumonia and bloodstream infections in the United States from 2000 to 2009. J Hosp Med. 2013, 8: 559-563.
Zilberberg MD, Shorr AF: Secular trends in Gram-negative resistance among urinary tract infection hospitalizations in the United States, 2000–2009. Infect Control Hosp Epidemiol. 2013, 34: 940-946. 10.1086/671740.
Sievert DM, Ricks P, Edwards JR, Schneider A, Patel J, Srinivasan A, Kallen A, Limbago B, Fridkin S: Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the national healthcare safety network at the centers for disease control and prevention, 2009–2010. Infect Control Hosp Epidemiol. 2013, 34: 1-14. 10.1086/668770.
Micek ST, Kollef KE, Reichley RM, Roubinian N, Kollef MH: Health care-associated pneumonia and community-acquired pneumonia: a single-center experience. Antimicrob Agents Chemother. 2007, 51: 3568-3573. 10.1128/AAC.00851-07.
Iregui M, Ward S, Sherman G, Fraser VJ, Kollef MH: Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator-associated pneumonia. Chest. 2002, 122: 262-268. 10.1378/chest.122.1.262.
Alvarez-Lerma F: ICU-acquired pneumonia study group: modification of empiric antibiotic treatment in patients with pneumonia acquired in the intensive care unit. Intensive Care Med. 1996, 22: 387-394. 10.1007/BF01712153.
Zilberberg MD, Shorr AF, Micek MT, Mody SH, Kollef MH: Antimicrobial therapy escalation and hospital mortality among patients with HCAP: a single center experience. Chest. 2008, 134: 963-968. 10.1378/chest.08-0842.
Shorr AF, Micek ST, Welch EC, Doherty JA, Reichley RM, Kollef MH: Inappropriate antibiotic therapy in Gram-negative sepsis increases hospital length of stay. Crit Care Med. 2011, 39: 46-51. 10.1097/CCM.0b013e3181fa41a7.
Kollef MH, Sherman G, Ward S, Fraser VJ: Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest. 1999, 115: 462-474. 10.1378/chest.115.2.462.
Garnacho-Montero J, Garcia-Garmendia JL, Barrero-Almodovar A, Jimenez-Jimenez FJ, Perez-Paredes C, Ortiz-Leyba C: Impact of adequate empirical antibiotic therapy on the outcome of patients admitted to the intensive care unit with sepsis. Crit Care Med. 2003, 31: 2742-2751. 10.1097/01.CCM.0000098031.24329.10.
Harbarth S, Garbino J, Pugin J, Romand JA, Lew D, Pittet D: Inappropriate initial antimicrobial therapy and its effect on survival in a clinical trial of immunomodulating therapy for severe sepsis. Am J Med. 2003, 115: 529-535. 10.1016/j.amjmed.2003.07.005.
Ferrer R, Artigas A, Suarez D, Palencia E, Levy MM, Arenzana A, Pérez XL, Sirvent JM: Effectiveness of treatments for severe sepsis: a prospective, multicenter, observational study. Am J Respir Crit Care Med. 2009, 180: 861-866. 10.1164/rccm.200812-1912OC.
Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart K, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb SA, Beale RJ, Vincent JL, Moreno R: Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013, 41: 580-637. 10.1097/CCM.0b013e31827e83af.
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR: Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001, 29: 1303-1310. 10.1097/00003246-200107000-00002.
Safdar N, Handelsman J, Maki DG: Does combination antimicrobial therapy reduce mortality in Gram-negative bacteraemia? A meta-analysis. Lancet Infect Dis. 2004, 4: 519-527. 10.1016/S1473-3099(04)01108-9.
Performance Standards for Antimicrobial Susceptibility Testing: Twelfth Informational Supplement. M100-S12. 2002, National Committee for Clinical Laboratory Standards, Wayne, PA
Performance Standards for Antimicrobial Susceptibility Testing: Seventeenth Informational Supplement. M100-S17. 2007, Wayne, PA, Clinical Laboratory Standards Institute
Knaus WA, Draper EA, Wagner DP, Zimmerman JE: APACHE II: a severity of disease classification system. Crit Care Med. 1985, 13: 818-829. 10.1097/00003246-198510000-00009.
Falagas M, Fragoulis K, Kasiakou S, Sermaidis G, Michalopoulos A: Nephrotoxicity of intravenous colistin: a prospective evaluation. Int J Antimicrob Agents. 2005, 26: 504-507. 10.1016/j.ijantimicag.2005.09.004.
Markou N, Apostolakos H, Koumoudiou C, Athanasiou M, Koutsoukou A, Alamanos I, Gregorakos L: Intravenous colistin in the treatment of sepsis from multiresistant Gram-negative bacilli in critically ill patients. Crit Care. 2003, 7: R78-R83. 10.1186/cc2358.
Falagas E, Kasiakou S: Toxicity of polymyxins: a systematic review of the evidence from old and recent studies. Crit Care. 2006, 10: R27-10.1186/cc3995.
Florescu DF, Qiu F, McCartan MA, Mindru C, Fey PD, Kalil AC: What is the efficacy and safety of colistin for the treatment of ventilator-associated pneumonia? A systematic review and meta-regression. Clin Infect Dis. 2012, 54: 670-680. 10.1093/cid/cir934.
GAIN: How a New Law is Stimulating the Development of Antibiotics. [], [http://www.pewhealth.org/other-resource/gain-how-a-new-law-is-stimulating-the-development-of-antibiotics-85899518481]
Guidelines for the management of adults with hospital-acquired pneumonia, ventilator-associated pneumonia, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005, 171: 388-416. 10.1164/rccm.200405-644ST.
O’Grady NP, Alexander M, Burns LA, Dellinger EP, Garland J, Heard SO, Lipsett PA, Masur H, Mermel LA, Pearson ML, Raad II, Randolph AG, Rupp ME, Saint S: Guidelines for prevention of intravascular catheter-related infections. Clin Infect Dis. 2011, 52: 1087-1099. 10.1093/cid/cir138.
Dellit TH, Owens RC, McGowan JE, Gerding DN, Weinstein RA, Burke JP, Huskins WC, Paterson DL, Fishman NO, Carpenter CF, Brennan PJ, Billeter M, Hooton TM: Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America Guidelines for Developing an Institutional Program to Enhance Antimicrobial Stewardship. Clin Infect Dis. 2007, 44: 159-177. 10.1086/510393.
Disclosure
This study was supported by a grant from Cubist Pharmaceuticals, Lexington, MA, USA. MHK’s time was in part supported by the Barnes-Jewish Hospital Foundation. These data in part have been accepted for presentation at the 24th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID), 10 to 13 May 2014, Barcelona, Spain.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
This study was supported by a grant from Cubist Pharmaceuticals, Lexington, MA, USA. The funder had no role in the design, collection, analysis, or interpretation of the data; in the writing of the manuscript; or in the decision to submit the manuscript for publication. MDZ has served as a consultant to and/or received research funding from Cubist, Astellas, Pfizer, and CareFusion. AFS has served as a consultant to and/or received research funding from Cubist, Astellas, Pfizer, Forest, Theravance, and CareFusion. STM has served as a consultant to and/or received research funding from Cubist, Astellas, and Pfizer. CV-G has no financial competing interests to declare. MHK has served as a consultant to and/or received research funding from Cubist, Astellas, Pfizer, Forest, and Theravance.
Authors’ contributions
MDZ participated in the conception, design, analysis, and interpretation of the data, drafted the manuscript, and has given final approval for the version to be published. She takes responsibility for data accuracy and analysis and reporting integrity of the study. AFS participated in the conception, design, analysis, and interpretation of the data. He was involved in revising the manuscript critically for important intellectual content, and has given final approval for the version to be published. STM participated in the conception, design, acquisition, and interpretation of the data. He was involved in revising the manuscript critically for important intellectual content, and has given final approval for the version to be published. CV-G participated in the conception, design, acquisition, and interpretation of the data. She was involved in revising the manuscript critically for important intellectual content, and has given final approval for the version to be published. MHK participated in conception, design, acquisition, and interpretation of the data. He was involved in revising the manuscript critically for important intellectual content, and has given final approval for the version to be published.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zilberberg, M.D., Shorr, A.F., Micek, S.T. et al. Multi-drug resistance, inappropriate initial antibiotic therapy and mortality in Gram-negative severe sepsis and septic shock: a retrospective cohort study. Crit Care 18, 596 (2014). https://doi.org/10.1186/s13054-014-0596-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13054-014-0596-8