Abstract
Background
Treatment with regorafenib, a multiple-kinase inhibitor, to manage metastatic colorectal cancers (mCRCs) shows a modest improvement in overall survival but is associated with severe toxicities. Thus, to reduce regorafenib-induced toxicity, we used regorafenib at low concentration along with a dual JAK/HDAC small-molecule inhibitor (JAK/HDACi) to leverage the advantages of both JAK and HDAC inhibition to enhance antitumor activity. The therapeutic efficacy and safety of the combination treatment was evaluated with CRC models.
Methods
The cytotoxicity of JAK/HDACi, regorafenib, and their combination were tested with normal colonic and CRC cells exhibiting various genetic backgrounds. Kinomic, ATAC-seq, RNA-seq, cell cycle, and apoptosis analyses were performed to evaluate the cellular functions/molecular alterations affected by the combination. Efficacy of the combination was assessed using patient-derived xenograft (PDX) and experimental metastasis models of CRC. To evaluate the interplay between tumor, its microenvironment, and modulation of immune response, MC38 syngeneic mice were utilized.
Results
The combination therapy decreased cell viability; phosphorylation of JAKs, STAT3, EGFR, and other key kinases; and inhibited deacetylation of histone H3K9, H4K8, and alpha tubulin proteins. It induced cell cycle arrest at G0-G1 phase and apoptosis of CRC cells. Whole transcriptomic analysis showed that combination treatment modulated molecules involved in apoptosis, extracellular matrix-receptor interaction, and focal adhesion pathways. It synergistically reduces PDX tumor growth and experimental metastasis, and, in a syngeneic mouse model, the treatment enhances the antitumor immune response as evidenced by higher infiltration of CD45 and cytotoxic cells. Pharmacokinetic studies showed that combination increased the bioavailability of regorafenib.
Conclusions
The combination treatment was more effective than with regorafenib or JAK/HDACi alone, and had minimal toxicity. A clinical trial to evaluate this combination for treatment of mCRCs is warranted.
Similar content being viewed by others
Background
In the United States, colorectal cancer (CRC) is the second leading cause of cancer mortality; in 2023, an estimated 153,020 new cases and 52,550 deaths, were predicted [1]. The prognosis of metastatic CRC (mCRC) is poor. Despite all the advances in the treatment of CRC, the 5-years survival to date is still around 14% [1, 2]. Although treatment of early stages of CRC typically includes surgical resection, mCRCs are treated mainly with systemic therapy including, but not limited to, chemotherapy, targeted therapy and immune check point inhibitors [2]. In the last two decades, median overall survival (OS) for mCRC patients has increased from less than 1 year to around 30 months due to advancements in early and late line treatment options [3,4,5,6,7]. For CRC, early lines chemotherapy regimens include fluorouracil (FU)-based regimens, which include FOLFOX (folinic acid, FU, and oxaliplatin) and FOLFIRI (folinic acid, FU, and irinotecan), with or without targeted biologic agents such as cetuximab and panitumumab, which are anti-EGFR agents or bevacizumab (a VEGF inhibitor) [8, 9]. In 2012, the US FDA approved regorafenib, which targets several receptor tyrosine kinases (RTKs), including fibroblast growth factor receptors (FGFRs), platelet-derived growth factor receptor beta (PDGFRβ), epidermal growth factor receptor (EGFR), and extracellular signal-regulated kinase (ERK1/2) [10], for treatment of mCRC patients who fail FU-based chemotherapy regimens [11, 12].
Several clinical trials have evaluated regorafenib in previously treated mCRC patients. CORRECT and CONCUR phase III clinical trials have shown modest OS benefit compared to placebo. The most common grade 3 and 4 adverse events were hand-foot skin reaction, fatigue diarrhea, hypertension, and rash and that led to treatment delay, dose reduction or discontinuation [13, 14]. CONSIGN and REGARD single arm clinical trials results were consistent with the known safety profile of regorafenib [15, 16]. Though regorafenib has anti-angiogenic and anti-proliferative properties, it also exhibits high toxicity and poor tolerability for patients [17], effects that were evident in the first cycle of treatment [18]. Further, to optimize regorafenib treatment and to increase tolerability, a randomized phase II, ReDOS trial was conducted in previously treated mCRC. Fewer adverse effects were evident in the dose-escalation group [19, 20]. JAK and HDAC inhibitors have been explored individually as potential therapeutic agents for various solid tumors with limited efficacy [21, 22]. A bifunctional/hybrid small-molecule Janus kinase/Histone deacetylase inhibitor (JAK/HDACi) demonstrates co-inhibition of HDAC and JAK kinases, resulting in suppression of progression of hematologic and solid tumors [23, 24]. JAK/HDACi, a potent JAK1-3/HDAC2/6 dual inhibitor, exhibits antiproliferative and proapoptotic activities [23, 25]. Thus, we initiated a preclinical investigation to evaluate the effect of regorafenib in combination with JAK/HDACi and to assess whether this combination reduces regorafenib-induced toxicity in CRC and enhances its therapeutic efficacy in combination with JAK/HDACi at lower drug concentrations. Advantages/rationales to use this hybrid molecule are: a) concurrent inhibition of enzymes of JAK and HDAC families, which are activated in CRC [26,27,28,29,30], by JAK/HDACi could enhance the anti-tumor immunity, modulate expression profiles of genes involved in cell cycle regulation and apoptosis, and result in synergistic effects, leading to enhanced efficacy of regorafenib; b) the combination of regorafenib with JAK/HDACi may lead to changes in the epigenetic landscape of CRC, potentially reactivating silenced tumor suppressor genes and may inhibit additional kinases; and c) combining JAK/HDACi with regorafenib may inhibit other signaling pathways activated in CRC. In the current study, we evaluated the effects of this combination in enhancing the therapeutic efficacy and reducing regorafenib-induced toxicity, using in vitro and in vivo CRC preclinical models, which include patient-derived xenografts (PDXs), luciferase tagged HT29 experimental metastasis, and MC38 syngeneic mouse models. Our results indicate that the efficacy of regorafenib in combination with JAK/HDACi has a synergistic inhibitory effect on tumor growth, and metastasis, with no apparent toxicity.
Methods
CRC clinical tissue specimens
Frozen archival CRC tissues, along with their uninvolved adjacent noncancerous/tissue specimens, were procured through the Anatomic Pathology Division of the University of Alabama at Birmingham (UAB). Following guidelines described by the Declaration of Helsinki, Institutional Review Board (IRB#090513004) approval from UAB was obtained for experimental use of specimens. Lysates for tumor (T) and adjacent uninvolved noncancerous tissue (N) were used for protein analyses.
Cell lines and culture media
Normal primary colonic epithelium cells were obtained from Cell Biologics, and CRC cell lines (HCT116, SW480, HT29, and RKO) were procured from American Type Culture Collection (ATCC). The CRC cell lines exhibiting various statuses of TP53, KRAS, BRAF, PIK3CA, PTEN and Microsatellite status: stable (MSS)/Instable (MSI) summarized in Table S1, were grown in McCoy’s medium (Fisher Scientific, Hampton, NH), and normal primary colonic epithelium cells were grown in human epithelial cell medium (Cell Biologics, Chicago, IL). The cells were STR profiled and were tested for mycoplasma contamination. Culture media were supplemented with 10% fetal bovine serum and penicillin–streptomycin, and cells were incubated at 37ºC with 5% CO2.
Treatment with JAK/HDAC inhibitor and regorafenib
We tested JAK/HDACi (Cat. # HY-126141) and regorafenib (Cat. # HY-10331) obtained from MedChem Express (Monmouth Junction, NJ). Both drugs were dissolved in DMSO, and 5 mM stock was prepared for cell culture experiments. For animal experiments, drugs were prepared according to the vender’s suggestions.
Cell viability assay
The viability of normal primary colonic epithelium cells were determined after 72 h of treatment, following procedure described in our previous study [31]. Concentration-dependent dose titrations in normal primary colonic epithelium cells with a range of concentrations from 250 nM-5 µM were conducted, with JAK/HDACi, regorafenib and combination treatment. In addition, to evaluate the efficacy of drugs in CRC cells, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT) experiment were conducted at 500 nM concentration. Further in vitro experiments in CRC cells were conducted at 500 nM concentration.
Colony formation assay
To assess the colony formation after drug treatment, 1,000 CRC cells in triplicate were plated into 6-well plates, after treatment JAK/HDACi, regorafenib, and their combination. After 10 days, cell colonies were fixed with 5% glutaraldehyde and stained with crystal violet (Sigma-Aldrich). Quantification of colonies was performed by the 10% acetic acid extraction method described by Kueng and coworkers [32].
Kinome analysis
SW480 and RKO cells were treated with vehicle (DMSO), JAK/HDACi, regorafenib, and their combination for 45 min, and cell lysates were prepared. Tyrosine kinase (PTK) activity (kinomic) profiling was performed using the PamStation®12 platform (PamGene, BV, The Netherlands). As per standard kinomic protocol as described previously [33]. Whole-chip comparative analysis (BioNavigator Upkin PTK v8.0) was performed between groups generating Mean Kinase Statistics (direction of change).
Immunoblot analysis
Western blot analyses were performed using NuPAGE 4–12% Bis–Tris Midi Protein Gels, (Thermo Fisher Scientific, Waltham, MA), following procedure described in our previous study [31]. Details of antibodies used in the study is provided in Table S2. The Super Signal West Femto Maximum sensitivity substrate (EMD Millipore, Rockford, IL) was used to detect signals. The signal was detected with an Amersham Imager 600RGB (GE Healthcare Life Sciences).
Whole transcriptomic sequencing and Assay for Transposase-Accessible Chromatin with sequencing (ATAC-seq)
A total of 1 × 106 cells/dish were seeded for RNA extraction from CRC cells after 12 h of treatment with JAK/HDACi, regorafenib, and their combination with Trizol, following the manufacturer’s instructions (Invitrogen). Poly A enrichment from total RNA was performed, and library were prepared using cDNA synthesis, cDNA library preparation kits from Agilent Technologies. Final library were run on Illumina Nextseq 550 sequencer using Illumina NextSeq 500/550 High Output Kit, as 75-bp single-end reads. For RNA sequencing data analyses approaches were followed as described in our prior studies [34, 35]. For ATAC-seq, 100,000 cells were counted after 6 h drug treatment, and treated with DNase solution following the vendor’s protocol, and submitted to Active Motif (Carlsbad, CA) for library preparation, sequencing, data processing, analyses, and visualization.
cDNA synthsis and Quantitative PCR (qPCR)
High-capacity cDNA reverse transcription kit with RNase inhibitor (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA) was used to reverse transcribed 2 µg of total RNA. Quantification of gene targets was done with10 ng cDNA, for validation of RNA-seq targets using PowerUp SYBR green master mix (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA) on an ABI real-time PCR machine and analyzed using Quant-studio Real-Time PCR software (Applied Biosystems). List of primers are provided in Table S3.
Immunohistochemical analysis
The immunophenotypic expressions of Ki67, in sections from PDX tissues, and CD8, CD45, pSTAT3Y705, pERK1/2 in tumor sections collected from immunocompetent mice were assessed by immunohistochemical (IHC) assays as described earlier [36].
TUNEL staining
Apoptosis induced in a PDX model by JAK/HDACi, regorafenib and their combination treatments was analyzed by staining FFPE sections using TACS-2 TdT-DAB In Situ Apoptosis Detection Kit (R&D Biotechne, Minneapolis, MN), following the manufacturer’s instructions.
Cell cycle analysis
To evaluate the effect of drugs on cell cycle arrest, SW480 and RKO cells were seeded, 1 × 106/dish in triplicates in four groups: vehicle, JAK/HDACi, regorafenib, and their combination (for 24 h, following procedure described in our previous study [31]. Cell cycle analysis was performed using BD LSR FortessaTM Flow Cytometer (BD Biosciences, Mountain View, CA).
Flow cytometry for quantification of apoptosis
To evaluate the effect of drugs on apoptosis, cells were stained by use of TACS Annexin V-FITC apoptosis detection kit obtained from R&D Systems (Minneapolis, MN), following the manufacturer’s protocol. CRC cells (HCT116, RKO, HT29, and SW480) were seeded at 1 × 105/dish in triplicates in four groups: vehicle, JAK/HDACi, regorafenib, and the combination for 72 h. The cells were analyzed with a BD LSR FortessaTM Flow Cytometer (BD Biosciences, Mountain View, CA).
Drug efficacy on patient-derived xenografts (PDXs)
To assess the effect of individual and combination drug treatments on tumor growth, a PDX that was developed from histologically confirmed colon cancer tissue, obtained from the Department of Surgery at UAB. It was used after approval of the UAB Institutional Animal Care and Use Committee of the University of Alabama at Birmingham (Animal Project Number (APN): IACUC-20207). The details of the PDX model development is described in our publication [36]. Animals were treated with vehicle (n = 4), JAK/HDACi (n = 4; 30 mg/kg body wt), regorafenib (n = 4; 6 mg/kg body wt), or the combination (n = 4; JAK/HDACi: 30 mg/kg body wt + regorafenib: 6 mg/kg body wt) every third day and tumor were measured weekly. Mice were sacrificed upon completion of experiment and tumors were harvested, photographed, weighed, and processed for FFPE block preparation.
Efficacy of combination therapy in an experimental metastasis model
To assess the effect of JAK/HDACi, regorafenib and their combination drug treatments on metastasis, an experiment was performed with 6-week-old NOD/SCID/IL2γ receptor-null (NSG) mice (male and female) as described [37]. All procedures and experimental protocols were approved and conducted in compliance (IACUC-21501). For metastasis experiments, as described previously [31], tail vein injections with HT29 cells (0.5 × 106) carrying a luciferase expression construct were conducted. Mice were imaged at 0, 10, and 30 days post injection, and treated with vehicle or drugs (with JAK/HDACi, regorafenib and their combination). Drug treatments were performed as described in PDX experiment. Bioluminescence imaging was accomplished using an IVIS Lumina III instrument (Perkin Elmer, Waltham, MA), and mice were injected with luciferin 10 min prior to imaging. After 30 days, mouse lungs and kidneys were dissected, and ex vivo analyses were performed.
Syngeneic mouse model to evaluate the efficacy of combination therapy
To examine the effect of individual and combination treatments on immunomodulation, MC38 (0.25 × 106) were injected into male and female immunocompetent C57BL/6 mice that were obtained from the Jackson Laboratory. Drug treatments were done in similar manner as described in PDX experiment. All procedures and experimental protocols were approved and conducted in compliance (IACUC-21501). The animals were sacrificed upon completion of the experiment, and tumor tissue was harvested and processed for, RNA, protein isolation and FFPE block preparation.
NanoString nCounter differential gene expression analysis
Gene expression was quantified digitally using the nCounter Gene Expression Assay (NanoString Technologies, Seattle, WA) by mouse PanCancer IO 360. Assays were performed according to the manufacturer’s instructions. The hybridized samples were loaded onto the nCounter PrepStation with automatic processing. The raw counts of expression data files were analyzed using the Rosalind web platform.
Toxicity analysis in serum samples
Upon completion of the experiment for 30 days in HT29 metastasis model, before mice were sacrificed, blood was collected and serum was isolated to evaluate liver and kidney functions to assess toxicity with JAK/HDACi and regorafenib alone, and their combination treatment, using a comprehensive Panel EWrap (Cat# 6330-COMP) by a DRY CHEM 4000 Analyzer.
Cytokine analysis in serum samples of C57BL/6 mice
Cytokines were measured in serum samples of syngeneic mouse, treated with JAK/HDACi and regorafenib, and their combination along with vehicle control, using MesoScale Discovery (Rockville, MD) mouse V-Plex Proinflammatory Panel I kit with a SIRRUS Stanbio automated chemical analyzer.
Pharmacokinetic (PK) profiles of regorafenib and JAK-HDACi
Blood samples were collected from C57BL/6 mice treated with regorafenib and JAK-HDACi at 1, 7, 21, or 48 h after treatment, and plasma samples were assayed. The quantifications of regorafenib and JAK-HDACi in plasma were conducted using a Shimadzu LC-20AD LC (Shimadzu, OR) and an API5000 MS (Sciex, MA). For sample preparation, a one-step protein precipitation with acetonitrile was used. Plasma (10 µL) was added into 250 µL of acetonitrile. Chromatographic separation was performed using an ACE Excel 3 C18 column. The mobile phase was (A) 0.1% formic acid in water and (B) 0.1% formic acid in acetonitrile utilizing a gradient from 20% to 75%. The injection volume was 10 µL. The run time was 5 min, and the retention time was 2.4 min. Standard curves for regorafenib and JAK-HDACi in plasma were prepared in the ranges of 5–15,000 ng/ml.
Statistical analysis
Data were presented as means ± SD for replicates. Number of each replicate (n) are mentioned in figure legends. Student t test (one-tailed, unequal variance) was used to perform comparisons between the groups, as described in figure legends. P-values ≤ 0.05 were considered statistically significant. Combination index (CI) values (for synergy), in in vivo models for JAK/HDACi and regorafenib were calculated by Bliss-independent equations as described by Liu et al., [38].
Results
Combination of regorafenib with the dual JAK/HDAC inhibitor reduces CRC cell proliferation
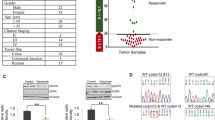
Regorafenib inhibits multiple kinases which are activated in key signaling pathways, such as stromal RTKs (FGFRs, PDGFRβ), angiogenic receptor tyrosine kinases (EGFR), and intracellular signaling kinase (ERK1/2) [17]. Several studies [39], have evaluated the association of increased HDACs expression with CRC, additionally inhibition of activated JAK/STAT pathway holds good promise in CRC regression [40]. We therefore used a hybrid molecule JAK/HDACi in combination with regorafenib in our study, with a rationale to inhibit multiple pathways which are activated in CRCs. We first as proof of concept evaluated expression of JAK/HDACi targets (pJAK2, pSTAT3Y705, and HDAC2/6) and regorafenib targets (PDGFRβ, pEGFR, and pERK1/2) by western blots in tissue lysates of CRCs (T) and their corresponding normal/uninvolved tissues (N), wherein increased expression was observed in tumor tissue compared to their adjacent normal (Fig. 1A-B), indicating that targeting CRC with this combination could be promising. Since regorafenib toxicity is an issue to be addressed, we evaluated cell proliferation in normal primary colonic cells at varying concentrations (250 nM- 5 µM) of JAK/HDACi and regorafenib individually and in combination and found that, at 500 nM, these normal cells, were unaffected (Fig. 1C). Thus, this concentration was used to assess the effect of drugs individually and in combination on cell proliferation employing four CRC cell lines, i.e., HCT116, RKO, HT29, and SW480, which exhibit different molecular backgrounds (p53, KRAS, BRAF, PIK3CA, JAK, STAT, HDAC mutations, and MSI status) summarized in Table S1. With individual and combination treatments, there was reduced proliferation of SW480, HT29, and HCT116 cells (Fig. 1Di-iii), the effect was pronounced in SW480 and HT29 CRC cells (Fig. 1Di-ii), which exhibit no genetic alterations in JAK/HDAC target molecules (see Table S1). At this dose the effect on cell viability was not noted in RKO cells (Fig. 1Div) At 500 nM, there was a similar pattern of response for colony formation in all four cell lines (Fig. 1E). Significant reductions in the number of colonies were noted for SW480, HT29, and HCT116 cells (Fig. 1Fi-iii), but marginal response was evident for RKO cells (Fig. 1Fiv). HCT116 cells exhibit mutations in all HDACs and JAK1 but are wild-type for STAT3, whereas RKO cells harbor mutations in STAT3, FGFR1, and HDAC6 (see Table S1). These results demonstrate, for CRC cell lines, the efficacy of the combination treatment in the nanomolar range compared to previous studies with higher treatment concentrations of regorafenib [41]. There was a marginal effect on cell viability of normal primary colonic cells at 500 nM concentration in combination treatment (Fig. 1C). At this concentration, the combination treatment in SW480, HT29, and HCT116 has demonstrated a significant decrease in cell viability and colony formation but these effects were not observed in RKO cells. The differential response on cell proliferation may be due to the molecular/genetic background of CRC cells.
The JAK/HDACi and regorafenib combination reduces cell proliferation and colony formation of CRC cells. A Western blotting demonstrating the key targets of JAK/HDACi and regorafenib in tissue lysates of CRCs (T) along with their adjacent, uninvolved normal tissues (N). B Figure represents relative protein expression (normalized to their respective β-actin) of drug targets illustrated in Fig A. C MTT assay analyzing the effect on cell proliferation of JAK/HDACi (0.25 – 5.0 µM) and regorafenib (0.25 – 5.0 µM) in single and their combination drug treatments in normal primary colonic epithelium cells. Results are presented as percents of vehicle control (DMSO). D MTT assay evaluating the efficacy of JAK/HDACi (500nM) and regorafenib (500nM) alone and in combination drug treatments on proliferation of CRC cell lines. Results are presented as percents of vehicle control (DMSO) in each set (i) SW480, (ii) HT29, (iii) HCT116, and (iv) RKO. E Colony assay for four CRC cell lines (SW480, HT29, HCT116, and RKO) showing the efficacy on colony formation of JAK/HDACi (500nM) and regorafenib (500nM) alone and in combination drug treatments. F Quantification of colonies was performed by a 10% acetic acid extraction method. Results are presented as averages of triplicates from each treatment group and as percents of vehicle control (DMSO) (i) SW480, (ii) HT29, (iii) HCT116, and (iv) RKO. P-values were labeled as follows: * p ≤ 0.05, ** p ≤ 0.01, and *** p ≤ 0.001
Combination treatment increases cell cycle arrest at the G0-G1 phase and induces apoptosis
To assess the role of these drugs, individually and in combination treatment, on the cell cycle, we conducted experiments with SW480 and RKO cells. For SW480 cells, there were higher numbers of cells arrested at the G0-G1 phase by the combination treatment (52%) as compared to DMSO (31.7%), JAK/HDACi (41%), and regorafenib (39.1%). There was also a delay in the synthesis (S) phase for the combination treatment (41%), as compared to DMSO (51%), JAK/HDACi (48%), and regorafenib (50%) (Fig. 2A-B). However, for RKO cells, there was no change in cell arrest with either individually or with combination treatment (Supplementary Fig. S1A-B). Additionally, in four CRC cell lines, we analyzed the effect of drug treatments on apoptosis by propidium iodide (PI) and annexin V staining. In SW480 cells, there was increased apoptosis in the combination treatment (Fig. 2C), HT29 (Fig. 2D) and HCT116 (Fig. 2E), cells but not observed in RKO cells (Fig. 2F). These results correlate with cell proliferation and colony formation results. For each cell line, quantification is presented as an average of three experiments and as the percentage of apoptotic cells, which includes the sum of early and late apoptotic cells from each treatment group. For SW480 cells, the percentages of apoptotic cells were, for combination treatment (79.91%), JAK/HDACi (54.46%), regorafenib (11.17%), and DMSO (4.04%) (Fig. 2C). For HT29 cells, the percentages of apoptotic cells were, for combination treatment (61.83%), JAK/HDACi (32.28%), regorafenib (5.16%), and DMSO (3.51%) (Fig. 2D). For HCT116 cells, the percentages of apoptotic cells were, for combination treatment (53.03%), JAK/HDACi (39.13%), regorafenib (13.62%), and DMSO (7.04%) (Fig. 2E). For RKO cells, the percentages of apoptotic cells were, for combination treatment (2.8%), JAK/HDACi (2.44%), regorafenib (1.18%), and DMSO (0.73%) (Fig. 2F). These results show that the combination treatment causes G0-G1 arrest, delays the S phase, and induces apoptosis.
The JAK/HDACi and regorafenib combination induces cell arrest at the G0-G1 phase, delays the S phase, and induces apoptosis. A Cell cycle arrest was analyzed by flow cytometry of SW480 cells exposed to DMSO (control), JAK/HDACi, regorafenib, or the combination. B The average of three experiments was quantified for each treatment group and plotted as percent cell arrest. C-F Apoptosis was analyzed by flow cytometry quantifying FITC-conjugated annexin and propidium iodide (PI) positive cells. Average of three experiments (sum of % early and late apoptosis) were quantified for each treatment group and plotted as percent apoptotic cells. C SW480 cells, D HT29 cells, E HCT116 cells, F RKO cells. P-values were labeled as follows: * p ≤ 0.05, ** p ≤ 0.01, and *** p ≤ 0.001
Combination treatment inactivates kinases, inhibits deacetylation of key histone proteins, and modulates DNA accessibility
Since regorafenib is a multiple-kinase inhibitor [17], and JAK-HDACi also inhibits important kinases [25], we conducted kinome analysis on lysates of SW480 and RKO cells to derive a holistic picture of kinases altered by individual and combination treatment. The rationale to select SW480 and RKO cells was to gain a better understanding of modulation in kinase activities, as these cell lines exhibited distinct responses to treatments (SW480-responsive, RKO-nonresponsive/partial responsive). SW480 cells treated with JAK-HDACi showed decrease in the activity of various kinases, including, JAK2, Ephrin A receptors, EGFR, and others in a signaling network that was JAK2-centric (Fig. 3Ai). SW480 cells treated with regorafenib showed decrease in the kinase activity of Met, VEGFR-3, ErbB4, ErbB2, FLT3, and other receptor tyrosine kinases, and mapped to a network that was IGF1R-centric (Fig. 3Aii). Marked decreases in kinase activity, including SYK, SFK, FGFR1, and FGFR4, were evident after combination treatment, as compared to individual drug treatments (Fig. 3Aiii). These altered kinases mapped to an FGFR1 centric network (Fig. 3Aiii). However, for RKO cells, several kinases were unaltered/activated by drug treatments (Fig. 3Bi-iii).
Combination treatment of JAK/HDACi and regorafenib decreases kinase activity in CRC cells. A-B Network models of altered kinase activity is displayed. A (i) JAK/HDACi, (ii) Regorafenib, and (iii) Combination altered kinases in SW480 cells. B (i) JAK/HDACi, (ii) Regorafenib, and (iii) Combination, altered kinases in RKO cells. Networks were generated using MetaCore (portal.genego.com) with an auto expand network set with 75 maximum nodes, with metabolites, and orphan nodes were excluded. Input nodes (kinases) have large blue circles around them. The smaller blue circles represent that kinase activity decreased, while smaller red circles represent that kinase activity increased/unaltered. Arrowheads denote the direction of interactions, and color of the lines indicates type of interaction (green; positive, red; inhibitory, grey; complex). The key for symbols is provided in Table S5. C Table summarizes Mean Kinase Statistics (MKS) of kinases modulated by treatment of JAK/HDACi and regorafenib and their combination in SW480 and RKO cells. Mean Kinase Statistic for each treatment groups values are represented as change from DMSO control. D Western blotting demonstrating inactivation of kinases. In lysates of four CRC cell lines, JAK/HDACi targets pJAK1, pJAK2, pJAK3, and pSTAT3Y705, and regorafenib targets pEGFR and pERK 1/2. β-Actin was used as a loading control. E Western blots demonstrating that JAK/HDACi and the combination treatment inhibit deacetylation of histone H3K9, H4K8 and alpha tubulin, as illustrated by increased proteins of acetylated-histone H3K9, H4K8, and α-tubulin. β-Actin was used as a loading control. P-values were labeled as follows: * p ≤ 0.05, ** p ≤ 0.01, and *** p ≤ 0.001
The inhibition of established target kinases for JAK/HDACi (JAK1/2 and FLT3/4) and regorafenib (FGFR 1–4, EGFR, Src, Fgr, and PDGFRα/β) are shown in Fig. 3C. Additionally, inhibition of unreported kinases for these drugs, which included EphA 1–5, and 8, HER 2–4, and FAK 1–2, was more pronounced in the combination treated SW480 cells as compared to JAK/HDACi or regorafenib alone, as evidenced by reduced kinase activity (Fig. 3C and supplemental Table S4). However, these kinases were not inhibited in RKO cells (Fig. 3C, supplemental Table S4). For four CRC cell lines, we validated the kinome results by western blots, wherein combination treatment showed marked reductions in phosphorylation of JAK1/2/3 and STAT3 (targets for JAK/HDACi) and pEGFR and pERK1/2 kinases (targets for regorafenib) (Fig. 3D). Among the four CRC cell lines, the response to combination treatment differed at varying levels; the higher response was observed for SW480, HT29, and HCT116 cells, and the least response was observed for RKO cells (Fig. 3D).
Cancer progression is governed by genetic mutations and interplay of epigenetic alterations, wherein histones have been considered as potential biomarkers to predict prognosis and patient outcomes [42]. Since ATAC-seq illustrates the chromatin accessibility (hyper-accessible/open or hypo-accessible/closed) of transcription machinery [43, 44], we performed this technique to identify changes in accessible chromatin regions in responsive (SW480) and non-responsive (RKO) CRC cell lines. Our findings have demonstrated striking differences in the patterns of accessible chromatin to regulate the transcription machinery and this change was more pronounced in the combination treatment, specifically in SW480 cells (Supplementary Fig. 2A). For SW480 cells, the open and closed chromatin regions in JAK/HDACi vs. DMSO (hyper-accessible regions: 10,298; hypo-accessible regions: 4932), regorafenib vs. DMSO (hyper-accessible regions: 1408; hypo-accessible regions: 1552), and combination vs. DMSO (hyper-accessible regions: 10,405; hypo-accessible regions: 5137) as shown in Supplementary Fig. S2A. However, RKO was least responsive to drug treatment, which reflects modest changes in accessible chromatin landscape as determined by ATAC-seq. The values were JAK/HDACi vs. DMSO (hyper-accessible regions: 2719; hypo-accessible regions: 906), regorafenib vs. DMSO (hyper-accessible regions: 277; hypo-accessible regions: 419), and combination vs. DMSO (hyper-accessible regions: 2925; hypo-accessible regions: 1253) (Supplementary Fig. S2B).
The results shown in Supplementary Figs. 2A-B demonstrate the effect of chromatin modulation on gene transcription by drug treatments in SW480 and RKO cells. In SW480 cells, Integrated Genome Viewer (IGV) plots used for data visualization, shows an increase in hyper-accessible chromatin in the transcriptional start-site (TSS) of CYP4F12 (Supplementary Fig. 2C), resulting in its increased gene expression (Fig. 4E). Since overexpression of CYP4F12 is involved in inhibition of tumor progression and metastasis and its low expression has been associated with poor patient survival [45]; increased expression of CYP4F12 in the combination therapy demonstrates its therapeutic advantage. Similarly, hypo-accessible chromatin in the TSS of ITFB4 (Supplementary Fig. 2D) led to reduced gene expression (Fig. 4E). Increased ITGB4 expression is involved with tumor growth, progression, and metastasis in CRC, and unfavorable overall survival [46]. Thus, decrease in ITGB4 expression observed in our study indicate the efficacy of combination treatment.
Whole transcriptomic analysis shows that combination treatment of JAK/HDACi and regorafenib modulates important pathways. Results are presented from of duplicates (n = 2) from each treatment group. A, B Venn diagram illustrating number of genes up-regulated/down-regulated by JAK/HDACi (500 nM), regorafenib (500 nM), and in their combination treatment. C Volcano plots illustrating statistically significant genes in three comparisons (i) JAK/HDACi (JH) vs. DMSO, (ii) regorafenib (Reg) vs. DMSO, and (iii) combination (combo) vs. DMSO, for SW480 cells. D significantly enriched KEGG pathways in genes uniquely altered by combination treatment. E Heatmap of genes uniquely modulated in combination treatment. Genes that are differentially expressed with log2 fold change ≥ 1 or ≤ -1 and p-value < 0.05 in only combination treatment are referred as uniquely modulated genes. F, G Quantitative PCR showing expression of gene targets (normalized to GAPDH). F The upregulated gene targets are shown (i) PCDHA10, (ii) ITIH5, (iii) PCDHB10, (iv) MYO18B, and (iv) CYP4F12, and G the down-regulated gene targets are shown (i) HSPH1 (ii) HSPD1 (iii) LYAR and (iv) EPHA2. H, I Western blots showing HSPH1 and cyclin D1 protein expression in cell lysates of H SW480, and I RKO cells. β-Actin was used as a loading control. The values above western blots represent protein expression (normalized to their respective β-actin) compared to their DMSO controls. P-values were labeled as follows: * p ≤ 0.05, ** p ≤ 0.01, and *** p ≤ 0.001
Since HDACs, tumor-promoting enzymes causing deacetylation of acetyl-lysine (KAc) residues on their substrates (histones and non-histone proteins), have emerged as candidates for epigenetic effects, we evaluated how JAK/HDACi, regorafenib, and their combination modulate acetylation of histones. Combination treatment increased intracellular levels of acetyl-histone H4 (Ac-HH4) and acetyl-α-tubulin (Ac-Tub), which are substrates for HDAC1/2/3 and HDAC6, respectively, more dramatically in SW480, HT29, HCT116 as compared to RKO (Fig. 3E).
These results indicate that treatment inhibits drug targets and their respected over-activated kinase pathways, more predominately by combination treatment than by JAK/HDACi and regorafenib agents alone, thus illustrating the higher efficacy of combination treatment. The effect can vary, however, depending on the mutational background of CRC cells.
Combination treatment modulates signaling pathways
Our transcriptomic profiling results with SW480 cells show that combination treatment up-regulated 601 genes, and down-regulated 146 genes as shown in a Venn diagram (Fig. 4A-B). Volcano plots highlighted the difference in gene expression for individual and combination treatment (Fig. 4C), for which there were a higher number of genes modulated by treatment with the combination. Upregulated protein-coding genes in JAK/HDACi vs. DMSO, regorafenib vs. DMSO, and the combination vs. DMSO were 212, 53, and 822, respectively and downregulated protein-coding genes were 28, 24, and 166, respectively (Fig. 4C).
We further investigated which pathways are altered by these modulated genes in combination treatment. Using Kyoto Encyclopedia of Genes and Genomes (KEGG), which groups genes to similar biological processes at the cellular level, we found that the extracellular matrix-receptor interaction, focal adhesion, and apoptosis pathways were modulated (Fig. 4D), and the representative genes are shown in the heat map (Fig. 4E). Interestingly, increased expression of several tumor suppressor genes (PCDHA10, ITIH5, PCDHB10, and MYO18B) [47,48,49,50,51], and CYP4F12 gene [45] involved in inhibition of cell migration and epithelial-mesenchymal transition (EMT) was observed (Fig. 4Fi-v). In addition, we found genes associated with tumor progression were downregulated; among these were heat shock proteins (HSP), such as HSPH1, HSPD1, LYAR and EPHA2 [52,53,54]. We further validated these findings by performing qPCR (Fig. 4Gi-iv). These genes are well-established in modulating tumor growth and metastasis.
HSPH1 is associated with CRC progression and is involved in regulation of Wnt/β-catenin pathway. Inhibition of HSPH1 inhibits expression of STAT3 and its downstream targets, including cyclin D1 [55]. We verified decreased expression of HSPH1 observed in transcriptomic profiling further by western blotting and found decrease in HSPH1 protein levels with concomitant reduction in cyclin D1 expression in combination treatment of SW480 cells (Fig. 4H), which was not evident in least responsive RKO cells (Fig. 4I). These results indicate the advantage of combination treatment in differentially modulating the genes involved in key cellular pathways.
Combination treatment causes a synergistic reduction in CRC PDX growth
To evaluate the therapeutic potential of the JAK/HDACi, regorafenib and combination drug treatments in a CRC PDX model, CRC PDX tissues were implanted into flanks of NSG mice. Tumors were routinely measured, and mice were sacrificed after 28 days of treatment. The combination treatment synergistically reduced tumor sizes, tumor volumes, and tumor weights as compared to vehicle control, JAK/HDACi and regorafenib alone (Fig. 5 A-C). Hematoxylin and eosin (H&E) staining of excised tumors showed more necrotic lesions in combination treatment (Fig. 5D). IHC staining showed reduced Ki67-positive staining, indicating that proliferation was reduced by the combination treatment as compared to single drugs and vehicle control treatment groups (Fig. 5E-upper panel). Additionally, combination treatment induced apoptosis as demonstrated by higher numbers of TUNEL-positive cells as compared to individual drug treatments (Fig. 5E-lower panel). These results suggest that combination treatment reduces tumor growth in CRC PDX model by decreasing proliferation and inducing apoptosis.
Combination treatment leads to reductions in tumor size, volume, and weight in CRC-PDXs. A Image illustrating harvested CRC-PDXs were smaller in the combination treated groups compared to the vehicle control, JAK/HDACi and regorafenib treated groups. B Tumor progression kinetics illustrating the efficacy of treatment. The statistical significance is calculated at 28th day of treatment. P-values were labeled as follows: * p ≤ 0.05, ** p ≤ 0.01, and *** p ≤ 0.001. D H&E staining, illustrating necrotic regions as shown in dotted line. E IHC analysis showing decreased numbers of Ki67-positive cells in tissues treated with the combination, indicating fewer proliferating cells (upper panel). Increased TUNEL-positive cells present in the combination-treated group, indicating induced apoptosis (bottom panel)
Combination treatment reduces experimental metastasis, with no evident toxicity
We evaluated the effects of individual and combination drug treatment on metastasis using an HT29 experimental model. There was significant reduction in the distant spread of HT29 luciferase tagged cells as shown by non-invasive bioluminescence imaging in combination treatment (Fig. 6A). The bioluminescence intensity was quantified as total flux on the 30th day of treatment. Efficacy of the combination treatment was reflected by reduced luciferase activity as compared to vehicle and individual drug treatments (Fig. 6B). H&E staining of lung and kidney sections corroborated well with bioluminescence imaging of smaller tumors (shown with dotted lines), indicating fewer metastases to distant organs (Fig. 6C). To evaluate the effect of drug treatment on kidney and liver functions, serum analysis was performed on the last day of the experiment. All parameters were within the reference range, indicating no signs of toxicity after drug administration (Fig. 6D). These results suggest that combination treatment decreases metastasis of luciferase-tagged HT29 cells, and has no evident toxicity.
Combination treatment decreases metastasis of luciferase-tagged HT-29 cells injected into NSG mice. A Bioluminescence imaging of vehicle- and drug-treated groups at 0, 10, and 30-day intervals, highlighting efficacy of the treatment for the combination group, as evidenced by reduced bioluminescence intensity of luciferase-tagged HT29 cells. B Quantification of bioluminescence intensity on day 30th of vehicle and drug-treated groups, showing reduced intensities in the regorafenib- and combination-treated groups. P-values were labeled as follows: * p ≤ 0.05, ** p ≤ 0.01, and *** p ≤ 0.001. C H&E staining of FFPE sections from lung at 10X magnification (top panel) and kidney at 2X magnification (bottom panel) showing reduced tumor burdens in these organs in combination treatment. D Toxicity analysis in serum samples of mice with the vehicle- and drug-treated HT29 metastatic model. Values were within the reference range, illustrating no evident toxicity was observed by either by JAK/HDACi and regorafenib alone or their combination treatment
The antitumor immune response is enhanced by the combination treatment showing increased infiltration of CD45 and cytotoxic CD8 T cells
To evaluate the effect of drug treatment on immunomodulation, we employed a syngeneic mouse model. Murine CRC MC38 cells were injected into flanks of C57BL/6 mice and were treated with test agents alone or in combination every third day. The combination treatment synergistically reduced tumor sizes (Fig. 7A), tumor progressions (Fig. 7B), and tumor weights (Fig. 7C), as compared to vehicle control and individual treatments of JAK/HDACi and regorafenib (Fig. 7 A-C). To investigate the association of drug treatment on the immune response, we analyzed gene expression using the Immuno Oncology (IO360 Mouse) panel of Nanostring (Fig. 7D). and found that the combination treatment had higher Cd45 and cytotoxic cells abundance scores (Fig. 7Ei-ii). Interestingly, we observed marked reduction in CX3C motif chemokine receptor 1(Cx3cr1) and significant increased expression of Gzmb an Gzme in combination treatment (Fig. 7Fi-iii). We confirmed the finding with IHC staining of FFPE sections of harvested tumors. There was higher CD45 staining in tissue sections following combination treatment, corroborating Nanostring gene expression results. CD45 has phosphatase activity and dephosphorylates JAK2 [56], which modules STAT3 [56, 57]; this finding corroborates with our results, where we observed, decreased pJAK2 (Fig. 7H), and decreased pSTAT3Y705 expression (Fig. 7G) in the combination treatment. Thus, concomitant increased CD45 expression (Fig. 7E(i) and G), corelating well with inactivation of pJAK2 and pSTAT3 Y705 levels. Serum analysis showed the inflammatory cytokines, tumor necrosis factor alpha (TNFα) and keratinocyte chemoattractant/human growth-regulated oncogene KC/GRO, were reduced after treatment with the combination (Fig. 7Ii-ii). Previous study [58] has reported regorafenib induces the immune response by CD8 infiltration, interestingly our results show pronounced infiltration of CD8 T cells, after combination treatment, relative to regorafenib alone (Fig. 7G). The results suggest that, therefore combination treatment increases immune response of regorafenib. Further, to evaluate the bioavailability of JAK/HDACi, regorafenib, and their combination, plasma samples from C57BL/6 mice were used for pharmacokinetic analysis. When given in combination with JAK/HDACi, the bioavailability of regorafenib increased, indicating the efficacy of the treatment could be due prolonged bioavailability (Fig. 7J). In sum, our results demonstrate that the combination treatment enhances antitumor immunity.
Combination treatment reduces tumor growth in an MC38 syngeneic mouse model, and increased infiltration of CD45 and cytotoxic CD8 T cells. A Image illustrating that harvested syngeneic tumors were smaller in treated groups compared to vehicle control B Tumor progression curve illustrating efficacy of the treatment; in the combination-treated group the tumors regressed, and statistical significance is calculated at 28th day of treatment. P-values were labeled as follows: * p ≤ 0.05, ** p ≤ 0.01, and *** p ≤ 0.001. C Tumor weights (in grams) were reduced in the combination-treated group. D Table summarizing the functional aspects of modulated genes in the combination-treated group. E Higher Log2 abundance scores of Cd45 and cytotoxic cells were evident for the combination-treated group. F Gene expression of targets (i) Cx3cr1, (ii) Gzmb, and (iii) Gzme were altered in JAK/HDACi, regorafenib and the combination-treated group. G H&E and IHC staining for CD8, CD45, pSTAT3Y705 and pERK1/2 are shown. H Western blots illustrating expressions of pJAK2, pERK1/2, and granzyme B. I Plasma analysis for cytokines released after drug treatment (i)TNF-Alpha and (ii) KC/GRO. J Pharmacokinetics in plasma samples of C57BL/6 mice after JAK/HDACi and regorafenib alone and in their combination treatment at 1, 7, 21, and 48 h
Discussion
Our study evaluated combination therapy in reducing the drug-induced toxicity by regorafenib, a multi-kinase inhibitor used to treat mCRC patients, refractory to first-line FU- or irinotecan-based therapy [59]. We assessed the efficacy of a combination, JAK/HDACi with regorafenib at nanomolar concentrations, to reduce the drug-induced toxicity of regorafenib by lowering its dosage and evaluating the efficacy of JAK/HDACi and regorafenib alone and their combination treatments. JAK/HDACi is a dual/hybrid molecule, showing promise in treatment of other cancers [25]. Preclinical studies have evaluated the anticancer activities of HDACi, due to their role in cell cycle arrest and apoptosis [60] HDACi modulate tumor vasculature to reduce angiogenesis and regulate host immune responses [61, 62]. To date, four HDACi, belinostat (PXD101), romidepsin (FK228), panobinostat (LBH589), and vorinostat (SAHA) have been approved by the US Food and Drug Administration (FDA). Although HDACi alone shows promising effects, and the above-mentioned HDACi are in clinical use, these, like regorafenib, have side effects, which include, but are not limited to, fatigue, diarrhea, nausea, vomiting, and cardiotoxicity [61,62,63,64]. In addition, they have limited efficacy in treatment of solid tumors [65]. To address these shortcomings, several groups have designed isoform selective HDACi and dual/bifunctional/hybrid/multitarget inhibitors [66]. These hybrid inhibitors modulate various pathways, as several bioactive groups are assembled into one compact molecule, thereby circumventing pharmacokinetic limitations [67,68,69].
One of the drawbacks of SAHA treatment, is activation of the JAK-STAT pathway as reported by Liang et al. [25], by upregulating phosphorylation of STAT3 at Tyr705 residue. However, it is interesting to note, that this JAK/HDACi suppresses the JAK-STAT pathway, as evidenced by reduced phosphorylation of STAT3 at Tyr705 with JAK/HDACi alone and its combination with regorafenib. Additionally increased stabilization of acetylation was observed in Ac-HH3, Ac-HH4 and Ac-Tub, which potentiates the fact, that this inhibitor concurrently inhibits both JAKs and HDACs, and combination with regorafenib is further advantageous as multiple oncogenic kinases are inhibited.
The results of our study demonstrate that the combination of JAK/HDACi and regorafenib, which inhibits key targets that are upregulated in CRC, is promising. Treatment with 500 nM concentration, which has marginal toxicity to normal primary colonic epithelium cells, is effective in reducing CRC cell proliferation, as key targets of both drugs are activated in fast-dividing CRC cells. The inhibition of activated kinases is evidenced by kinome analyses of SW480 cells, which illustrates effectiveness of the drug treatment with JAK/HDACi and regorafenib, and combination drug treatment. The combination therapy showed varied responses for various CRC cell lines, as observed in analyses of cell proliferation, colony formation, inactivation of key kinases, inhibition of kinase activity, cell cycle, and apoptosis.
Cancer cells modulate cellular metabolism and heavily rely on chaperones like HSPH1, as this protein has anti-aggregation properties and can modify the misfolded proteins, and thus cell can sustain cellular stress and survive [55]. HSPH1 has been previously reported to promote cell growth by activation of STAT3 [55]. Thus, dramatic decrease in HSPH1 expression observed by whole transcriptomic analysis in SW480, by combination treatment was a promising observation, which leads to inactivation of STAT3 and decrease in its downstream target cyclin D1. We speculate that the partial response for RKO cells could be due to its constitutively activated STAT3, since RKO cells harbor a p.E616del mutation, present in the SH2 domain of the protein. OncoKB annotates [70] that the STAT3 E616del alteration as likely oncogenic [71]. It is a gain-of-function variant [72], as STAT3 activation is accomplished without a stimulus, it is due to higher positive electrostatic potential, which increases interaction with the DNA phosphate carrying a negative charge. This electrostatic interaction increases the binding affinity to DNA and leads to increased transcriptional activity of STAT3 and to its longer nuclear retention [72]. Molecular dynamics simulations conducted by Husby et al. [71] showed that E616 residue of STAT3 a key amino acid of monomer-B involved in the STAT3 protein-DNA interaction. Using cBioportal [73], we evaluated 18 studies of colon and rectal cancers with 7162 patients to investigate the mutational frequency of E616del; we found that only 4 patients had this STAT3 mutation. Thus, our findings indicate that treatment with the combination would benefit a wide spectrum of CRC patients.
Our investigation showed promising results for the CRC-PDX model, the luciferase-tagged HT29 metastasis model, and the MC38 syngeneic mouse model. The Nanostring nCounter PanCancer IO 360™ Panel revealed an increase in abundance of Cd45 and cytotoxic cells. The elevated numbers of CD45 cells after combination treatment is relevant, as CD45 dephosphorylates JAK and negatively regulates JAK-STAT signaling [57]. CD45 also negatively impacts cytokine receptor signaling by suppressing JAK kinases [56]. It acts as protein phosphatase and binds to JAKs. Targeted cd45 disruption, through increased cytokine and interferon-receptor signaling, activates JAKs and STAT [56]. The results from our syngeneic mouse model demonstrate that, higher the number of infiltrating CD45 lymphocytes, more pronounced is the dephosphorylation of JAK2, which is in corroboration with Sasaki et al. [56] findings, illustrating dephosphorylation of JAK2 is due to protein-tyrosine phosphatase activity of CD45. Further, there are increased abundance scores of cytotoxic cells, which are reflected by elevated Gzmb and Gzme expression, further demonstrating the treatment efficacy. We observed reduced expression of Cx3cr1, in treatment groups compared to the vehicle control; the effect was more pronounced for the combination treatment. These results are in line with the suggestion [74] that CX3CR1 is involved in the tumor microenvironment by contributing to angiogenic macrophage survival, thus promoting metastasis. Zheng et al. [74] found that, in human colon carcinomas, CX3CR1 is expressed in a stage- and histologic grade-dependent manner, where a poor prognosis was associated with CX3CR1 upregulation in tumor-associated macrophages. Their findings further showed that the metastasis of CRC cells was lowered when CX3CR1 was absent in the tumor microenvironment.
Our studies suggested reduced TNFα levels in serum samples of mice treated with the combination. TNFα, a pro-inflammatory mediator involved in apoptosis, is negatively regulated by CD45 [75]. This is consistent with low TNFα levels, as combination-treated mice had high numbers of CD45-positive cells. Thus, our investigations show that the therapeutic combination enhances the immune response at the tumor site and the marked regression of tumors after combination treatment. Pharmacokinetic studies with C57BL/6 mice showed that, after i.p. administration, the bioavailability of regorafenib increased in the combination treatment compared to single treatments. As reported in the literature, several factors contribute to enhanced bioavailability of the drugs due to beneficial pharmacokinetic interactions, for which one drug alters distribution of another drug by increasing absorption, inhibiting metabolism, and/or decreasing excretion, leading to prolonged drug plasma levels [76,77,78].
The preclinical findings reported in this study provide a basis to investigate further benefits of combining regorafenib with the JAK/HDACi hybrid molecule. Future studies should focus on a) investigating the role of the tumor microenvironment, including stromal and immune cells, in the combination treatment of the MC38 syngeneic mouse model; b) expanding the preclinical studies to assess the use of immune checkpoint inhibitors by combination of regorafenib with JAK/HDACi; and c) assessing the effect of epigenetic modifications of the genes identified by ATAC-seq.
Conclusion
In summary, our preclinical study shows efficacy of the combination therapy as compared with JAK/HDACi and regorafenib alone. Moreover, the combination therapy is promising, as it has no evident toxicity. These findings lend support to a clinical trial to assess this combination for treatment of patients with advanced CRC.
Availability of data and materials
The RNA-seq data provided in this manuscript are publicly available from the NCBI Gene Expression Omnibus (GEO) with accession number GSE252554. Other data generated in this study will be available upon request.
Abbreviations
- ATAC:
-
Assay for transposase-accessible chromatin
- ATCC:
-
American type culture collection
- CRC:
-
Colorectal cancer
- CX3CR1:
-
CX3C motif chemokine receptor 1
- EGFR:
-
Epidermal growth factor receptor
- ERK:
-
Extracellular signal-regulated kinase
- FGFR:
-
Fibroblast growth factor receptor
- FU:
-
Fluorouracil
- HDAC:
-
Histone deacetylase
- HSP:
-
Heat shock proteins
- JAK:
-
Janus kinase
- KC/GRO:
-
Keratinocyte chemoattractant/human growth-regulated oncogene
- mCRCs:
-
Metastatic colorectal cancers
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide
- NSG:
-
NOD/SCID/IL2γ receptor-null
- OS:
-
Overall survival
- PDGFR:
-
Platelet-derived growth factor receptor
- PDX:
-
Patient-derived xenograft
- RTK:
-
Receptor tyrosine kinases
- STAT3:
-
Signal transducer and activator of transcription 3
- TNF:
-
Tumor necrosis factor
References
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73(1):17–48.
Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014;383(9927):1490–502.
Van Cutsem E, Cervantes A, Adam R, Sobrero A, Van Krieken JH, Aderka D, Aranda Aguilar E, Bardelli A, Benson A, Bodoky G, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016;27(8):1386–422.
Vogel A, Hofheinz RD, Kubicka S, Arnold D. Treatment decisions in metastatic colorectal cancer - Beyond first and second line combination therapies. Cancer Treat Rev. 2017;59:54–60.
Tampellini M, Di Maio M, Baratelli C, Anania L, Brizzi MP, Sonetto C, La Salvia A, Scagliotti GV. Treatment of patients with metastatic colorectal cancer in a real-world scenario: probability of receiving second and further lines of therapy and description of clinical benefit. Clin Colorectal Cancer. 2017;16(4):372–6.
Yoshino T, Arnold D, Taniguchi H, Pentheroudakis G, Yamazaki K, Xu RH, Kim TW, Ismail F, Tan IB, Yeh KH, et al. Pan-Asian adapted ESMO consensus guidelines for the management of patients with metastatic colorectal cancer: a JSMO-ESMO initiative endorsed by CSCO, KACO, MOS, SSO and TOS. Ann Oncol. 2018;29(1):44–70.
Bekaii-Saab T, Kim R, Kim TW, O’Connor JM, Strickler JH, Malka D, Sartore-Bianchi A, Bi F, Yamaguchi K, Yoshino T, et al. Third- or Later-line therapy for metastatic colorectal cancer: reviewing best practice. Clin Colorectal Cancer. 2019;18(1):e117–29.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350(23):2335–42.
Qin S, Li J, Wang L, Xu J, Cheng Y, Bai Y, Li W, Xu N, Lin LZ, Wu Q, et al. Efficacy and tolerability of first-line Cetuximab plus Leucovorin, Fluorouracil, and Oxaliplatin (FOLFOX-4) versus FOLFOX-4 in patients with RAS wild-type metastatic colorectal cancer: the open-label, randomized, Phase III TAILOR trial. J Clin Oncol. 2018;36(30):3031–9.
Xu X, Yu Y, Liu M, Liang L, Liu T. Efficacy and safety of regorafenib and fruquintinib as third-line treatment for colorectal cancer: a narrative review. Transl Cancer Res. 2022;11(1):276–87.
Miura K, Satoh M, Kinouchi M, Yamamoto K, Hasegawa Y, Philchenkov A, Kakugawa Y, Fujiya T. The preclinical development of regorafenib for the treatment of colorectal cancer. Expert Opin Drug Discov. 2014;9(9):1087–101.
Shimizu T, Tolcher AW, Patnaik A, Papadopoulos K, Christensen O, Lin T, Blumenschein GR. Phase I dose-escalation study of continuously administered regorafenib (BAY 73–4506), an inhibitor of oncogenic and angiogenic kinases, in patients with advanced solid tumors. J Clin Oncol. 2010;28(15_suppl):3035–3035.
Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouche O, Mineur L, Barone C, et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381(9863):303–12.
Li J, Qin S, Xu R, Yau TC, Ma B, Pan H, Xu J, Bai Y, Chi Y, Wang L, et al. Regorafenib plus best supportive care versus placebo plus best supportive care in Asian patients with previously treated metastatic colorectal cancer (CONCUR): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2015;16(6):619–29.
Dane F, Ozgurdal K, Yalcin S, Benekli M, Aykan NF, Yucel I, Ozkan M, Evrensel T, Sevinc A, Coskun HS, et al. Safety and efficacy of regorafenib in patients with treatment-refractory metastatic colorectal cancer in Turkey: the single-arm, open-label REGARD study. BMJ Open. 2020;10(3):e027665.
Van Cutsem E, Martinelli E, Cascinu S, Sobrero A, Banzi M, Seitz JF, Barone C, Ychou M, Peeters M, Brenner B, et al. Regorafenib for Patients with Metastatic Colorectal Cancer Who Progressed After Standard Therapy: Results of the Large, Single-Arm. Open-Label Phase IIIb CONSIGN Study Oncologist. 2019;24(2):185–92.
Goel G. Evolution of regorafenib from bench to bedside in colorectal cancer: Is it an attractive option or merely a “me too” drug? Cancer Manag Res. 2018;10:425–37.
Grothey A, George S, van Cutsem E, Blay JY, Sobrero A, Demetri GD. Optimizing treatment outcomes with regorafenib: personalized dosing and other strategies to support patient care. Oncologist. 2014;19(6):669–80.
Benson AB, Venook AP, Al-Hawary MM, Arain MA, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Farkas L, et al. Colon cancer, version 2.2021, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2021;19(3):329–59.
Kato T, Kudo T, Kagawa Y, Murata K, Ota H, Noura S, Hasegawa J, Tamagawa H, Ohta K, Ikenaga M, et al. Phase II dose titration study of regorafenib in progressive unresectable metastatic colorectal cancer. Sci Rep. 2023;13(1):2331.
Slingerland M, Guchelaar HJ, Gelderblom H. Histone deacetylase inhibitors: an overview of the clinical studies in solid tumors. Anticancer Drugs. 2014;25(2):140–9.
Qureshy Z, Johnson DE, Grandis JR: Targeting the JAK/STAT pathway in solid tumors. J Cancer Metastasis Treat. 2020;6:27.
Liang X, Tang S, Liu X, Liu Y, Xu Q, Wang X, Saidahmatov A, Li C, Wang J, Zhou Y, et al. Discovery of novel pyrrolo[2,3-d]pyrimidine-based derivatives as potent JAK/HDAC dual inhibitors for the treatment of refractory solid tumors. J Med Chem. 2022;65(2):1243–64.
Qiu Q, Chi F, Zhou D, Xie Z, Liu Y, Wu H, Yin Z, Shi W, Qian H. Exploration of Janus Kinase (JAK) and Histone Deacetylase (HDAC) bispecific inhibitors based on the moiety of fedratinib for treatment of both hematologic malignancies and solid cancers. J Med Chem. 2023;66(8):5753–73.
Liang X, Zang J, Li X, Tang S, Huang M, Geng M, Chou CJ, Li C, Cao Y, Xu W, et al. Discovery of Novel Janus Kinase (JAK) and Histone Deacetylase (HDAC) dual inhibitors for the treatment of hematological malignancies. J Med Chem. 2019;62(8):3898–923.
Li N, Grivennikov SI, Karin M. The unholy trinity: inflammation, cytokines, and STAT3 shape the cancer microenvironment. Cancer Cell. 2011;19(4):429–31.
Morikawa T, Baba Y, Yamauchi M, Kuchiba A, Nosho K, Shima K, Tanaka N, Huttenhower C, Frank DA, Fuchs CS, et al. STAT3 expression, molecular features, inflammation patterns, and prognosis in a database of 724 colorectal cancers. Clin Cancer Res. 2011;17(6):1452–62.
Tsai KS, Yang SH, Lei YP, Tsai CC, Chen HW, Hsu CY, Chen LL, Wang HW, Miller SA, Chiou SH, et al. Mesenchymal stem cells promote formation of colorectal tumors in mice. Gastroenterology. 2011;141(3):1046–56.
Zhang X, Hu F, Li G, Li G, Yang X, Liu L, Zhang R, Zhang B, Feng Y. Human colorectal cancer-derived mesenchymal stem cells promote colorectal cancer progression through IL-6/JAK2/STAT3 signaling. Cell Death Dis. 2018;9(2):25.
Stempelj M, Kedinger M, Augenlicht L, Klampfer L. Essential role of the JAK/STAT1 signaling pathway in the expression of inducible nitric-oxide synthase in intestinal epithelial cells and its regulation by butyrate. J Biol Chem. 2007;282(13):9797–804.
Agarwal S, Afaq F, Bajpai P, Kim HG, Elkholy A, Behring M, Chandrashekar DS, Diffalha SA, Khushman M, Sugandha SP, et al. DCZ0415, a small-molecule inhibitor targeting TRIP13, inhibits EMT and metastasis via inactivation of the FGFR4/STAT3 axis and the Wnt/β-catenin pathway in colorectal cancer. Mol Oncol. 2022;16(8):1728–45.
Kueng W, Silber E, Eppenberger U. Quantification of cells cultured on 96-well plates. Anal Biochem. 1989;182(1):16–9.
Anderson JC, Taylor RB, Fiveash JB, de Wijn R, Gillespie GY, Willey CD: Kinomic Alterations in Atypical Meningioma. Med Res Arch. 2015;2015(3):10.18103.
Martini R, Delpe P, Chu TR, Arora K, Lord B, Verma A, Bedi D, Karanam B, Elhussin I, Chen Y, et al. African ancestry-associated gene expression profiles in triple-negative breast cancer underlie altered tumor biology and clinical outcome in women of African descent. Cancer Discov. 2022;12(11):2530–51.
Saleh M, Chandrashekar DS, Shahin S, Agarwal S, Kim HG, Behring M, Shaikh AJ, Moloo Z, Eltoum IA, Yates C, et al. Comparative analysis of triple-negative breast cancer transcriptomics of Kenyan, African American and Caucasian Women. Transl Oncol. 2021;14(7):101086.
Bajpai P, Banerjee NS, Moore DW, Kim HG, Afaq F, Contreras CM, Heslin MJ, Reddy VB, Peter S, Varambally S, et al. Developing 3D organoid raft cultures from patient-derived xenografts as rapid models to screen efficacy of experimental therapeutics. Int J Mol Sci. 2022;23(22):14392.
Agarwal S, Afaq F, Bajpai P, Behring M, Kim HG, Varambally A, Chandrashekar DS, Peter S, Diffalha SA, Khushman M, et al. BZW2 inhibition reduces colorectal cancer growth and metastasis. Mol Cancer Res. 2023;21(7):698–712.
Liu Q, Yin X, Languino LR, Altieri DC. Evaluation of drug combination effect using a Bliss independence dose-response surface model. Stat Biopharm Res. 2018;10(2):112–22.
Mariadason JM. HDACs and HDAC inhibitors in colon cancer. Epigenetics. 2008;3(1):28–37.
Spano JP, Milano G, Rixe C, Fagard R. JAK/STAT signalling pathway in colorectal cancer: a new biological target with therapeutic implications. Eur J Cancer. 2006;42(16):2668–70.
Wang YJ, Zhang YK, Zhang GN, Al Rihani SB, Wei MN, Gupta P, Zhang XY, Shukla S, Ambudkar SV, Kaddoumi A, et al. Regorafenib overcomes chemotherapeutic multidrug resistance mediated by ABCB1 transporter in colorectal cancer: in vitro and in vivo study. Cancer Lett. 2017;396:145–54.
Chervona Y, Costa M. Histone modifications and cancer: biomarkers of prognosis?. Am J Cancer Res. 2012;2(5):589–97.
Buenrostro JD, Giresi PG, Zaba LC, Chang HY, Greenleaf WJ. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat Methods. 2013;10(12):1213–8.
Klemm SL, Shipony Z, Greenleaf WJ. Chromatin accessibility and the regulatory epigenome. Nat Rev Genet. 2019;20(4):207–20.
Jia W, Chen S, Wei R, Yang X, Zhang M, Qian Y, Liu H, Lei D. CYP4F12 is a potential biomarker and inhibits cell migration of head and neck squamous cell carcinoma via EMT pathway. Sci Rep. 2023;13(1):10956.
Li M, Jiang X, Wang G, Zhai C, Liu Y, Li H, Zhang Y, Yu W, Zhao Z. ITGB4 is a novel prognostic factor in colon cancer. J Cancer. 2019;10(21):5223–33.
Hamm A, Veeck J, Bektas N, Wild PJ, Hartmann A, Heindrichs U, Kristiansen G, Werbowetski-Ogilvie T, Del Maestro R, Knuechel R, et al. Frequent expression loss of Inter-alpha-trypsin inhibitor heavy chain (ITIH) genes in multiple human solid tumors: a systematic expression analysis. BMC Cancer. 2008;8:25.
Jao TM, Tsai MH, Lio HY, Weng WT, Chen CC, Tzeng ST, Chang CY, Lai YC, Yen SJ, Yu SL, et al. Protocadherin 10 suppresses tumorigenesis and metastasis in colorectal cancer and its genetic loss predicts adverse prognosis. Int J Cancer. 2014;135(11):2593–603.
Kleinberger I, Sanders E, Staes K, Van Troys M, Hirano S, Hochepied T, Lemeire K, Martens L, Ampe C, van Roy F. Innovative mouse models for the tumor suppressor activity of Protocadherin-10 isoforms. BMC Cancer. 2022;22(1):451.
Kloten V, Rose M, Kaspar S, von Stillfried S, Knuchel R, Dahl E. Epigenetic inactivation of the novel candidate tumor suppressor gene ITIH5 in colon cancer predicts unfavorable overall survival in the CpG island methylator phenotype. Epigenetics. 2014;9(9):1290–301.
Nakano T, Tani M, Nishioka M, Kohno T, Otsuka A, Ohwada S, Yokota J. Genetic and epigenetic alterations of the candidate tumor-suppressor gene MYO18B, on chromosome arm 22q, in colorectal cancer. Genes Chromosomes Cancer. 2005;43(2):162–71.
Martini G, Cardone C, Vitiello PP, Belli V, Napolitano S, Troiani T, Ciardiello D, Della Corte CM, Morgillo F, Matrone N, et al. EPHA2 is a predictive biomarker of resistance and a potential therapeutic target for improving antiepidermal growth factor receptor therapy in colorectal cancer. Mol Cancer Ther. 2019;18(4):845–55.
Slaby O, Sobkova K, Svoboda M, Garajova I, Fabian P, Hrstka R, Nenutil R, Sachlova M, Kocakova I, Michalek J, et al. Significant overexpression of Hsp110 gene during colorectal cancer progression. Oncol Rep. 2009;21(5):1235–41.
Wu Y, Zhou Y, Gao H, Wang Y, Cheng Q, Jian S, Ding Q, Gu W, Yao Y, Ma J, et al. LYAR promotes colorectal cancer progression by upregulating FSCN1 expression and fatty acid metabolism. Oxid Med Cell Longev. 2021;2021:9979707.
Berthenet K, Bokhari A, Lagrange A, Marcion G, Boudesco C, Causse S, De Thonel A, Svrcek M, Goloudina AR, Dumont S, et al. HSP110 promotes colorectal cancer growth through STAT3 activation. Oncogene. 2017;36(16):2328–36.
Irie-Sasaki J, Sasaki T, Matsumoto W, Opavsky A, Cheng M, Welstead G, Griffiths E, Krawczyk C, Richardson CD, Aitken K, et al. CD45 is a JAK phosphatase and negatively regulates cytokine receptor signalling. Nature. 2001;409(6818):349–54.
Wu L, Bijian K, Shen SH. CD45 recruits adapter protein DOK-1 and negatively regulates JAK-STAT signaling in hematopoietic cells. Mol Immunol. 2009;46(11–12):2167–77.
Ou DL, Chen CW, Hsu CL, Chung CH, Feng ZR, Lee BS, Cheng AL, Yang MH, Hsu C. Regorafenib enhances antitumor immunity via inhibition of p38 kinase/Creb1/Klf4 axis in tumor-associated macrophages. J Immunother Cancer. 2021;9(3):e001657.
Ettrich TJ, Seufferlein T. Regorafenib. Recent Results Cancer Res. 2018;211:45–56.
Marks PA. The clinical development of histone deacetylase inhibitors as targeted anticancer drugs. Expert Opin Investig Drugs. 2010;19(9):1049–66.
Bolden JE, Peart MJ, Johnstone RW. Anticancer activities of histone deacetylase inhibitors. Nat Rev. 2006;5(9):769–84.
Lane AA, Chabner BA. Histone deacetylase inhibitors in cancer therapy. J Clin Oncol. 2009;27(32):5459–68.
Fu RG, Sun Y, Sheng WB, Liao DF. Designing multi-targeted agents: An emerging anticancer drug discovery paradigm. Eur J Med Chem. 2017;136:195–211.
Thurn KT, Thomas S, Moore A, Munster PN. Rational therapeutic combinations with histone deacetylase inhibitors for the treatment of cancer. Future oncology (London, England). 2011;7(2):263–83.
Rosik L, Niegisch G, Fischer U, Jung M, Schulz WA, Hoffmann MJ. Limited efficacy of specific HDAC6 inhibition in urothelial cancer cells. Cancer Biol Ther. 2014;15(6):742–57.
Luan Y, Li J, Bernatchez JA, Li R. Kinase and Histone Deacetylase Hybrid Inhibitors for Cancer Therapy. J Med Chem. 2019;62(7):3171–83.
Berube G. An overview of molecular hybrids in drug discovery. Expert Opin Drug Discov. 2016;11(3):281–305.
Fortin S, Berube G. Advances in the development of hybrid anticancer drugs. Expert Opin Drug Discov. 2013;8(8):1029–47.
Morphy R, Rankovic Z. Designed multiple ligands. An emerging drug discovery paradigm. J Med Chem. 2005;48(21):6523–43.
Chakravarty D, Gao J, Phillips SM, Kundra R, Zhang H, Wang J, Rudolph JE, Yaeger R, Soumerai T, Nissan MH, et al. OncoKB: A Precision Oncology Knowledge Base. JCO Precis Oncol. 2017;2017:PO.17.00011.
Husby J, Todd AK, Haider SM, Zinzalla G, Thurston DE, Neidle S. Molecular dynamics studies of the STAT3 homodimer:DNA complex: relationships between STAT3 mutations and protein-DNA recognition. J Chem Inf Model. 2012;52(5):1179–92.
Gutierrez M, Scaglia P, Keselman A, Martucci L, Karabatas L, Domene S, Martin A, Pennisi P, Blanco M, Sanguineti N, et al. Partial growth hormone insensitivity and dysregulatory immune disease associated with de novo germline activating STAT3 mutations. Mol Cell Endocrinol. 2018;473:166–77.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2(5):401–4.
Zheng J, Yang M, Shao J, Miao Y, Han J, Du J. Chemokine receptor CX3CR1 contributes to macrophage survival in tumor metastasis. Mol Cancer. 2013;12(1):141.
Piercy J, Petrova S, Tchilian EZ, Beverley PC. CD45 negatively regulates tumour necrosis factor and interleukin-6 production in dendritic cells. Immunology. 2006;118(2):250–6.
Caldwell J, Gardner I, Swales N. An introduction to drug disposition: the basic principles of absorption, distribution, metabolism, and excretion. Toxicol Pathol. 1995;23(2):102–14.
Martinez MN, Amidon GL. A mechanistic approach to understanding the factors affecting drug absorption: a review of fundamentals. J Clin Pharmacol. 2002;42(6):620–43.
Stielow M, Witczynska A, Kubryn N, Fijalkowski L, Nowaczyk J, Nowaczyk A. The bioavailability of drugs-the current state of knowledge. Molecules. 2023;28(24):8038.
Acknowledgements
We thank Dr Donald Hill, in the Department of Pathology, for his editorial assistance. We acknowledge the preclinical imaging shared facility of the O'Neal Comprehensive Cancer Center (OCCC), and the toxicity assessment study by the UAB Metabolism Core. We thank Dr Edward P Acosta, Dr Kevin J Ryan, and Alexander E. Dowell, (Department of Clinical Pharmacology/Toxicology) for PK analysis, Dr Christopher Willey and Dr Joshua Anderson for kinomic analysis, (Department of Radiation Oncology, Kinome Core). We thank Dr Harish Chandra Pal for FACS analysis (UAB Flow cytometry Core). We appreciate Dezhi Wang, and John Ness, of the UAB Pathology Core Research Lab for their help in tissue embedding, sectioning, and H&E staining. We appreciate Heather Lynn Hunter for conducting cytokine analysis (Department of Nutrition Sciences, UAB).
Funding
The work presented in this study was supported, in part, by grant 5U54CA118948 and by institutional funds (Department of Pathology and School of Medicine of UAB) awarded to UM. We also acknowledge the grants (P30CA013148) funded for the Tissue Biorepository and (1S10OD021697) Imaging Facilities of the UAB OCCC. We acknowledge UAB's Metabolism Core which is supported by the UAB Diabetes Research Center (P30DK079626) and the UAB Center for Clinical and Translational Science (UL1TR003096).
Author information
Authors and Affiliations
Contributions
PB and UM conceived and designed the study; PB, SA, FA, DSC, HGK, SKS, and UM acquisition of data; PB, SA, FA, DSC, HGK, AS, CRM, and UM analysis of data; PB, SA, FA, SAD, DSC, HGK, RS, SV, PNG, AM, RP, MK, and UM interpretation of data; PB, FA and UM writing original draft, PB, FA, SV, AM, RP, MK, and UM reviewed and edited the manuscript. The manuscript was read and approved by all the authors.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Frozen archival CRC tissues, and their uninvolved adjacent noncancerous/tissue specimens, were procured from the tissue biorepository through the Anatomic Pathology Division of the University of Alabama at Birmingham (UAB). Following guidelines described by the Declaration of Helsinki, Institutional Review Board (IRB#090513004) approval from UAB was obtained for experimental use of specimens. For generation of PDX, histologically confirmed colon cancer tissue, obtained from the Department of Surgery at UAB, were procured, and was used after approval of the UAB Institutional Animal Care and Use Committee (Animal Project Number (APN): IACUC-20207). The HT-29 luciferase metastasis experiment in NSG mice, all procedures and experimental protocols were approved and conducted in compliance (IACUC-21501). Syngeneic mouse model where MC38 cells were injected in C57BL/6 mice, all procedures and experimental protocols were approved and conducted in compliance (IACUC-21501).
Consent for publication
Not applicable.
Competing interests
All authors state that they have no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
13046_2024_3106_MOESM1_ESM.pdf
Additional file 1: Supplementary Fig. 1 The JAK/HDACi and regorafenib combination does not alter cell cycle in RKO cells. A Cell cycle arrest was analyzed by flow cytometry of RKO cells exposed to DMSO (control), JAK/HDACi, regorafenib, or the combination. B The average of three experiments was quantified for each treatment group and plotted as percent cell arrest. The NS, represent non-significant results. Supplementary Fig. 2 Treatment with the JAK/HDACi and regorafenib combination modulates the chromatin accessibility landscape of SW480 cells more prominently than that of RKO cells. A-B Distribution of differentially accessible regions (DARs) over chromosomes in A SW480 and B RKO cells. The hyper- and hypo-accessible regions are indicated in red and blue colors, respectively. C, D IGV plots demonstrating individual tracks of DMSO, JAK/HDACi, regorafenib and their combination treatment in SW480 and RKO cells. C Hyper-accessible regions (shaded in red) for CYP4F12 D Hypo-accessible regions (shaded in blue) for ITGB4. The TSS and direction of transcription is indicated with blue arrow.
13046_2024_3106_MOESM2_ESM.pdf
Additional file 2: Supplementary Tables: Table S1. Genes status in CRC cell lines, Table S2. List of antibodies used in this study, Table S3. Primer Sequences for validation of gene targets Table S4. Mean Kinase Statistic in SW480 and RKO cell lines treated with JAK/HDACi and regorafenib and their combination compared their respective control (DMSO treatment), Table S5. Symbol key for network modeling of altered kinases with MetaCore.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Bajpai, P., Agarwal, S., Afaq, F. et al. Combination of dual JAK/HDAC inhibitor with regorafenib synergistically reduces tumor growth, metastasis, and regorafenib-induced toxicity in colorectal cancer. J Exp Clin Cancer Res 43, 192 (2024). https://doi.org/10.1186/s13046-024-03106-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13046-024-03106-8