Abstract
Introduction
Human papillomavirus (HPV) presents a potential threat to the onset of carcinogenesis in the cervix, anogenital regions, and oropharynx. HPV encompasses over 200 types, with at least 12 having the potential to cause cancer, impacting the majority of sexually active individuals. In this current research, we explore the occurrence and spread of HPV genotypes.
Material and methods
During this cross-sectional study conducted in Sanandaj, Iran from Feb 2022 to Aug 2023, diverse samples including oral, vaginal, and genital were collected from individuals referred to private laboratories in Sanandaj, Iran. After sample collection and DNA extraction (FAVORGEN, Taiwan), they were subjected to PCR and genotyping (MehrViru, Iran). The subsequent statistical analysis unveiled infection rates across different demographics and age groups. STATA (version 17) were used for statistical analysis. We examined infection rates across demographics using t-tests and Odds Ratio.
Results
Overall, 26% (249) out of 950 cases tested positive for HPV, with 69% of these classified as high-risk. Among the examined population, 98% (933) were female, and 2% (17) were male. Females aged 31–40 exhibited the highest percentage of HPV prevalence (115/460) in the study with the majority of positive cases belonging to HR genotypes. The overall most frequent genotypes identified were 6, 16, 52, 53, 51, 58, and 56. HPV-16 exhibited the highest frequency among HR genotypes, accounting for 42 (17%) occurrences, followed by HPV-52 with a frequency of 32 (13%).
Conclusion
Our findings emphasize the significant prevalence of HPV among females, particularly in the 21–30 age group. The identification of high-risk genotypes, underscores the importance of targeted interventions for specific age cohorts. The age-stratified analysis highlights a consistent predominance of high-risk HPV across age groups, indicating the need for age-specific preventive measures. These results contribute valuable information for designing effective screening and vaccination strategies, to alleviate the impact of diseases associated with HPV.
Similar content being viewed by others
Introduction
Human papillomavirus (HPV) is recognized as sexually transmitted infection (STI). It carries the potential to induce carcinogenesis in the cervix, anogenital areas, and oropharynx [1, 2]. HPV contributes to nearly 99.7% of cervical cancer cases, ranking among the most widespread cancers in women [3]. In 2020, cervical cancer had a global impact on more than 604,000 individuals (13·3 cases per 100,000 female) and 341,831 deaths (7·2 fatality per 100,000 female), with Asia experiencing the highest prevalence (58%) [4]. Cervical cancer incidence in Iran was reported to 1.90 per 100,000 individuals [5]. In the Iranian female population, prevalence of HPV was detected in 58% of low-grade squamous intraepithelial lesions, 69% of high-grade cases, and 81% of patients with invasive cervical cancer [6].
In addition, HPV is found in over 88% of anal cancers and over 95% of anal high-grade squamous intraepithelial lesions (HSIL), highlighting its major role in anal cancer. Globally, a total of 50,865 new instances of anal cancer were reported in 2020 [7]. In a cross-sectional study (2020–2021) conducted in Tehran, 23.1% of women tested positive for anal HPV [8].
Furthermore, beyond the usual dangers of smoking and alcohol consumption, HPV has appeared as a significant element in oropharyngeal cancer, constituting a distinct subset of head and neck cancers [9]. In a study conducted in Shiraz, Iran, 14% of 100 oral cancer cases were found to have HPV infection [10]. Research shows rise in HPV infection over the past decade in Iran, likely attributed to insufficient sex education for youth and inadequate access to the HPV vaccine [11].
Currently, Over 200 HPV genotypes have been detected [12]. In the context of cancer, HPV types are categorized into two major group which are low-risk HPV (LR-HPV: type 6, 11, 42, 43, 81 and 83) and high-risk HPV (HR-HPV: type 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59) [13]. Understanding HPV genotype distribution would be essential for malignancy screening strategies and the development of effective vaccination targeting HPV-related carcinoma [14]. The HPV vaccination program has the capacity to prevent 90% of cervical cancer [15]. To eliminate cervical cancer in lower-middle-income countries, Brisson et al. recommend achieving 90% coverage for girls-only HPV vaccination, undergoing screening twice in a lifetime, and ensuring sustained safeguard against HR-HPV (16, 18, 31, 33, 45, 52, and 58) [16]. Therefore, in this investigation, our objective was to evaluate the frequency of HPV infection divided by genotypes and identify the frequency of HR-HPV genotypes among Iranian females specifically, in Sanandaj, Iran. Our goal is to provide compelling evidence that supports the inclusion of the HPV vaccine in Iran’s national vaccination program.
Methods and materials
Study population
The present cross-sectional study was conducted on various samples, including anal swab, anal wart, biopsy of skin lesions, cervical swab, genital swab, paraffin-embedded block of tissue, skin scraping, throat swab, and vaginal swab. The samples were collected from individuals who visited the laboratories of Sanandaj city in collaboration with the Clinical Virology Research Center (RCCV) at Tehran University of Medical Sciences. The research was conducted from Feb 2022 to Aug 2023. Individuals who were included in the survey were invited to complete informed consent. The participating patients did not undergo any medical interventions.
In the standard cytology assessment, physicians forwarded samples, including Pap-Preps (liquid-based cytology) and secretions related to the cervix, to laboratories following established cervical cancer screening procedures. However, for individuals not adhering to the regular screening protocols, such as those who are virgins, samples were obtained with shallower insertion from cervical and vaginal secretions. This collection was performed by either physicians or trained laboratory staff for HPV identification. In the case of males, samples were acquired through anal and penile biopsies and skin scrape sample. The testing procedures on these samples are conducted on the same day as collection, with the samples kept at a temperature of 4 °C until testing is initiated which was a day after sample collection.
The criteria involved women who, following a Pop smear test, chose to undergo additional tests based on the advice of a specialist physician. Furthermore, for women, the criteria extended to those recommended by their healthcare provider to undergo testing due to the presence of malignances including cervical cancer (CC), breast cancer, and Head & Neck Tumor. In the case of men, the criteria included individuals engaging in high-risk sexual behavior, having a sexual partner infected with the HPV, prompting them to seek HPV testing for reassurance about their health status. All demographic information of the patients extracted from laboratory records and subsequently transferred to the RCCV for further analysis.
DNA extraction and PCR
The pre-amplification of the samples, DNA extraction, and genotyping of HPV were carried out in the Molecular Genetics Department of the laboratory, following the protocol established by the quality control supervisor and under the supervision of RCCV. The extraction of HPV-DNA was carried out using the extraction kit from Favorgen Biotech Corp (FAVORGEN, Taiwan) in accordance with the manufacturer’s instructions. PCR was conducted on the extracted genome by HPV detection kit, and genotyping was conducted by MehrViru’s kit (MehrViru, Iran), equipped to recognize HR-HPV genotypes, including 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59. Additionally, it designates 7 genotypes (26, 53, 66, 67, 68, 73, and 82) as potentially high risk. Moreover, the kit identifies LR-HPV genotypes including 6, 11, 40, 42, 43, 44, 54, 61, 62, 89, and 90.
Statistical analysis
In this research, we utilized STATA (version 17) for our data analysis. Our approach involved assessing the positive response ratio to the infection test across the entire population, as well as within distinct sex and age categories. Subsequently, we conducted a comparative analysis using the independent t-test. Additionally, Odds Ratio (OR) was considered to evaluate the comparative impact of different groups, maintaining a significance level of 0.05. The individuals who sought medical attention at the specified centers underwent testing for 30 different HPV genotypes. A positive response to the test for one or more types was indicative of infection. The study population was stratified into six age groups, encompassing patients across various age brackets. Following the statistical description of the recorded data, we rigorously examined the reliability and accuracy of our findings through appropriate statistical methodologies.
Results
Demographic data
In the examination of HPV prevalence within various demographic subgroups, the data in Table 1 reveals the frequency and percentage of positive and negative cases. Out of 950 cases, 933 (98%) pertain to females, and 17 (2%) to males. The participants were further divided into six age groups for analysis. Within the female subgroup, consisting of 933 cases, 236 (25%) tested positive for HPV, while 697 (75%) tested negative. Among the male participants, 13 out of 17, or 76%, tested positive for HPV. Examining specific age groups, 238 individuals (25%) in the 21–30 age range were tested, with 83 (35%) testing positive and 155 (65%) testing negative. Details for other age groups can be found in Table 2. The highest percentage of positive tests was observed in those under 20 years old (40%) and the 21–30 years old (35%), respectively. However, in the context of numbers of infected individuals, age range between 31 and 40 had more HPV positive cases.
Distribution of HPV genotypes
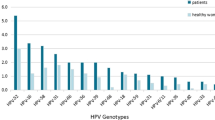
The frequency distribution of each genotype can be seen in Fig. 1. The most frequent genotypes are 6, 16, 52, 53, 51, 58, and 56 respectively. The frequency diagram in whole samples and divided by age is shown in Fig. 2. Genotypes 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68 are high-risk types, whose frequency distribution is listed in Fig. 3. Out of total of 236 samples, 164 (69%) HR-HPV and 72 (31%) for LR-HPV detected. The highest frequency in HR genotypes belonged to type 16 with a frequency of 42 (17%), 52 with a frequency of 32 (13%) and 51 with a frequency of 22 (9%), and the lowest frequency is related to types 35 and 59 with a frequency of 8 (3%). In our research, no positive cases of HPV-33 were seen.
Diagram of the most frequent genotypes including type 6, 16, 51, 52, 53, 56, and 58. a In the whole sample, b By gender. In part b of this figure, the number of the most frequent genotypes in men and women is shown. Besides, two genotypes of HPV in infected men, which are 11 and 44 genotypes, are not being displayed
The frequency and distribution of high-risk genotypes. The frequency and percentage for HR genotypes are as follows: HPV-16 with 42 occurrences (17%), HPV-18 with 16 occurrences (6%), HPV-31 with 18 occurrences (7%), HPV-33 with no occurrences (0%), HPV-35 with 8 occurrences (3%), HPV-39 with 17 occurrences (7%), HPV-45 with 15 occurrences (6%), HPV-51 with 22 occurrences (9%), HPV-52 with 32 occurrences (13%), HPV-56 with 20 occurrences (8%), HPV-58 with 21 occurrences (8%), HPV-59 with 8 occurrences (3%), HPV-66 with 17 occurrences (7%), and HPV-68 with 9 occurrences (4%)
Positive HPV across across sampling types
This section investigates the proportion of positive HPV tests in various sampling categories, including anal, genital, paraffin-based, vaginal, throat and cervical samples. The HPV testing was conducted using different sampling methods, with Table 3 illustrating the frequency distribution. Among the samples studied, cervical samples exhibited the highest number of positive cases (62/393), followed by genital samples (45/68) and anal samples (34/184). Paraffin and skin biopsy samples displayed no negative cases. Vaginal samples had the highest total count (209), comprising 30 positive and 179 negative cases. Statistical analysis revealed highly significant disparities between positive and negative cases across all sample types, with p-value ranging from 0.000 to 0.021.
Discussion
HPV-related disease rates, can differ based on factors like HPV type, geographical situation and sampled body site [17]. Examining the occurrence of HPV infection is essential as it aids in the formulation of strategies for preventing and managing cancer, with a specific focus on cervical cancer [13]. Our study in Sanandaj, located in west of Iran, revealed a 26% prevalence of HPV infection among 950 participants. A 25% prevalence of HPV infection, was identified among 933 females in our research. Our finding aligns with 24% HPV prevalence in 571 healthy women in Iran (2021) [18]. Similarly, a systematic review and meta-analysis conducted between 2004 and 2017 reported a 24.2% pooled prevalence of HPV in Asia, which was lower than the 41.8% prevalence observed in Africa [19]. It’s noteworthy that Iran’s neighboring countries reported varying prevalence rates, with Iraq (17.96%), showing a lower prevalence, while Turkey reported a higher prevalence at 36.3% [20, 21]. Various aspects associated with lifestyle, including sexual behavior, financial status, alertness of disease prevention and screening, as well as the efficacy of laboratory methods and sensitivity of HPV diagnostic techniques, can impact the prevalence rate of HPV in diverse areas [22].
In our study, 13 out of the 17 men suspected of HPV infection tested positive, yielding an infection rate of 76%. However, the worldwide prevalence rate of HPV infection in men was stated to be 31% in a systematic review study [23]. The discrepancy results from the small representation of men (2% of our study population), and the limited sample size may not accurately reflect their prevalence in larger populations. Moreover, higher mortality rates in women attributed to HPV have led to established screening programs, while men’s screening has been less emphasized [24]. Consequently, the notable prevalence of men in our study can be ascribed to their tendency for seek clinical care solely when they manifest clinical symptoms, rather than for routine screening. Nevertheless, in line with our findings, other studies have also reported elevated prevalence rates of HPV in males compared to females [11, 25]. We analyzed HPV prevalence across age groups and found that the greatest incidence of infection occurred in individuals < 20 years (40%), followed by the 21–30 age group at 35%. We observed that the occurrence of HPV infection tends to decrease with age, ranging from under 20 years to 70 years, but it doesn’t completely disappear. In Zhou et al.’s investigation, based on an analysis of age-specified HPV infection, the observed trend revealed a decline in prevalence up to 30 years, followed by an increase until the age of 60 years [26]. Almost mirroring our findings, a study in Malaysia detected the highest HPV prevalence (11.9%) in the 18–24 age range [27], while some other studies reported a different trend with the highest prevalence (7.29%) observed in the 40–49 age group [28]. The elevated prevalence of HPV in female under 20 in our research could be attributed to the lower vaccination percentage between adolescent girls compared to adults. Globally, the HPV vaccination for teenage girls was only 12% in 2021, and it was even lower in South East Asia at 3% [29]. Moreover, among the women aged 19 to 23, there is a higher level of awareness regarding the risk factors of cervical cancer (98.8%), with a corresponding desire for vaccination at 92.5% [30].
Research indicates that testing for high-risk HPV genotypes can serve as an effective initial screening process for cervical cancer, boasting a sensitivity of 93.97% [31]. Therefore, we examined distribution and frequency of different HPV genotype and found that the most commonly encountered genotypes were HPV-6 (54%), then types 16, 52, 53, 51, 58, 56, 31, 44, 90, 39, 66 and 18 in descending order. Likewise, HPV-6, classified as a LR-HPV, demonstrated the highest prevalence in various regions of Iran, registering rates of 50% in the northeast [32], 14.7% in the southeast [33] and 32% in Tehran (among female) [11]. Moreover, our findings revealed that 69% of the participants carried HR-HPV, while 31% had LR-HPV. Shalchimanesh et al. validated our findings with a HR-HPV occurrence rate of 67.12% in Tehran [34], whereas in China out of 414,540 women, HR-HPV was 17.8% [35].
We observed that the predominant LR-HPV genotypes were 6, 44, 90, 62, and 11, while the least prevalent LR genotype was HPV-42. A study in South-Eastern of Iran revealed HPV prevalence of 47% in the HR-group, 34% in the LR-group, and 19% in the potential high-risk group, with HPV-6 being the most predominant LR-HPV (14.7%) [33]. In another study in Eastern of Iran, the most prevalent LR-HPV types were HPV-6, 89, 11, and 42 [36].
In our investigation, the distribution of HR-HPV genotypes revealed varying frequency, with HPV-16 at 17%, followed by HPV-52 at 13%, HPV-51 at 9%, HPV-58 and HPV-56 both at 8%, HPV-31, HPV-39, and HPV-66 each at 7%, HPV-18 and HPV-45 at 6%, HPV-68 at 4%. The lowest frequency of HR-HP was related to HPV-33 (0, 35%), followed HPV-59 and HPV-35 with a frequency of 8 (3%). Global results indicate that HPV-16 (15.56–83.78%) is the most frequently identified HR strain in cancer [37]. In a study across different Iranian provinces, HPV-52 (3.2 and 9.6%) ranked as the second most common HR genotype, consistent with our findings [38]. However, in a distinct region, a different genotype emerging as the second dominant HR types such as HPV-16 in Shanghai [31] and HPV-31 in Hungary [39]. Moreover, HPV-51, a non-vaccine genotype, ranks as the third most frequent high-risk genotype in our study. Hence, recognizing the significant role of HR-HPV types in causing cancer, particularly contributing to 94.1% of squamous cell carcinomas and 83.3% of cervical adenocarcinomas [40], emphasizes the crucial need to prioritize preventive and control measures for this infection. It is essential to acknowledge certain limitations in present study. The absence of random sampling across diverse laboratories may have hindered the ability to draw unequivocal conclusions. The reported HPV prevalence in outpatients may not accurately reflect the actual HPV epidemiology within the entire population. Moreover, we had a limited number of male participants, and given the observed high prevalence of HPV among males in our study, further investigation in a larger target population is warranted. This current research highlighting the clinical development of cervical lesions and the significance of this matter, there is a pressing need for more extensive and comprehensive investigations in the forthcoming.
Conclusion
Our investigation explored a 26% occurrence of HPV infection and identified its genotypic distribution, revealing that 69% of HR-HPV genotypes were present in Iranian females in Sanandaj, the central city of Kurdistan province in western Iran. The high prevalence of HR subtype of HPV in our findings, underscore the pressing need for proactive measures, advocating for the inclusion of the HPV vaccine in Iran’s national vaccination program. Implementing the HPV vaccination and awareness program, particularly targeting girls under 20 years old who demonstrated a significant prevalence of HPV infection in our study, is a crucial preventive measure. This strategic intervention holds the potential to alleviate the burden of HPV-related diseases such as cancer, fostering improved public health outcomes within the region.
Availability of data and materials
No datasets were generated or analysed during the current study.
References
Szymonowicz KA, Chen J. Biological and clinical aspects of HPV-related cancers. Cancer biol med. 2020;17(4):864.
Martinelli M, Musumeci R, Sechi I, Sotgiu G, Piana A, Perdoni F, et al. Prevalence of human papillomavirus (HPV) and other sexually transmitted infections (STIs) among Italian women referred for a colposcopy. Int J Environ Res Public Health. 2019;16(24):5000.
Okunade KS. Human papillomavirus and cervical cancer. J Obstet Gynaecol. 2020;40(5):602–8.
Singh D, Vignat J, Lorenzoni V, Eslahi M, Ginsburg O, Lauby-Secretan B, et al. Global estimates of incidence and mortality of cervical cancer in 2020: a baseline analysis of the WHO global cervical Cancer elimination initiative. Lancet Glob Health. 2023;11(2):e197–206.
Anjam Majoumerd A, Hosseini S, Hosseini SM, Roshandel G, Rahmanian V, Hazar N. Epidemiology of cervical cancer in Iran in 2016: A nationwide study of incidence and regional variation. Cancer Rep. 2024;7(2):e1973.
Salavatiha Z, Farahmand M, Shoja Z, Jalilvand S. A meta-analysis of human papillomavirus prevalence and types among Iranian women with normal cervical cytology, premalignant lesions, and cervical cancer. J Med Virol. 2021;93(8):4647–58.
Macedo AC, Grande AJ, Figueiredo T, Colonetti T, Gonçalves JC, Testoni E, et al. DNA high-risk HPV, mRNA HPV and P16 tests for diagnosis of anal cancer and precursor lesions: a systematic review and meta-analysis. EClin Med. 2023:62.
Ashrafganjoei T, Hosseini MS, Pirastehfar Z, Farzaneh F, Arab M, Moghaddam NA, et al. Evaluation of anal cytology and human papillomavirus infection in high-risk women: a cross-sectional study. Med J Indonesia. 2022;31(4):245–9.
Sabatini ME, Chiocca S. Human papillomavirus as a driver of head and neck cancers. Br J Cancer. 2020;122(3):306–14.
Habibi L, Farzi R, Pirbonyeh N, Edalat F, Letafati A, Moattari A. Correlation between sampling site and prevalence of human papilloma virus (HPV) in patients with oral cancer referred to Shiraz, Iran. Oral Science International.
Rezaee Azhar I, Yaghoobi M, Mossalaeie MM, Kollaee Darabi A, Nejadeh AH, Jamshidi M, et al. Prevalence of human papilloma virus (HPV) genotypes between outpatients males and females referred to seven laboratories in Tehran. Iran Infect Agents Cancer. 2022;17(1):7.
Seyoum A, Seyoum B, Gure T, Alemu A, Alemayehu DH, Alemu A, et al. High rate of non-vaccine targeted high-risk HPV genotypes circulate among women in eastern Ethiopia. Sci Rep. 2024;14(1):958.
Han C, Huang W, Ye M, Zou R, Lan J, Chen J, et al. HPV prevalence and genotype distribution in 2,306 patients with cervical squamous cell carcinoma in central and eastern China. Front Public Health. 2023:11.
Derbie A, Mekonnen D, Nibret E, Maier M, Woldeamanuel Y, Abebe T. Human papillomavirus genotype distribution in Ethiopia: an updated systematic review. Virol J. 2022;19(1):13.
Dionne M, Sauvageau C, Kiely M, Dahhou M, Hamel D, Rathwell M, et al. School-based vaccination program against HPV and hepatitis B: a longitudinal analysis of vaccine coverage between 2015 and 2021 in Quebec. Vaccine. 2024;42(1):17–23.
Brisson M, Kim JJ, Canfell K, Drolet M, Gingras G, Burger EA, et al. Impact of HPV vaccination and cervical screening on cervical cancer elimination: a comparative modelling analysis in 78 low-income and lower-middle-income countries. Lancet. 2020;395(10224):575–90.
Kombe Kombe AJ, Li B, Zahid A, Mengist HM, Bounda G-A, Zhou Y, et al. Epidemiology and burden of human papillomavirus and related diseases, molecular pathogenesis, and vaccine evaluation. Front Public Health. 2021;8:552028.
Farahmand Z, Soleimanjahi H, Garshasbi M, Hasanzadeh M, Zafari E. Distribution of the most common types of HPV in Iranian women with and without cervical cancer. Women Health. 2021;61(1):73–82.
Okoye JO, Chukwukelu CF, Okekpa SI, Ogenyi SI, Onyekachi-Umah IN, Ngokere AA. Racial disparities associated with the prevalence of vaccine and non-vaccine HPV types and multiple HPV infections between Asia and Africa: a systematic review and meta-analysis. Asian Pacific J cancer prevent: APJCP. 2021;22(9):2729.
Yousif MG, Al-Amran FG, Sadeq AM, Yousif NG. Prevalence and Associated Factors of Human Papillomavirus Infection among Iraqi Women. arXiv preprint arXiv:2307.14806. 2023.
Alacam S, Bakir A. Human papillomavirus prevalence and genotype distribution in cervical swab samples in Istanbul, Turkey. J Infect Develop Count. 2021;15(08):1190–6.
Li X, Xiang F, Dai J, Zhang T, Chen Z, Zhang M, et al. Prevalence of cervicovaginal human papillomavirus infection and genotype distribution in Shanghai, China. Virol J. 2022;19(1):146.
Bruni L, Albero G, Rowley J, Alemany L, Arbyn M, Giuliano AR, et al. Global and regional estimates of genital human papillomavirus prevalence among men: a systematic review and meta-analysis. Lancet Glob Health. 2023;11(9):e1345–e62.
Garolla A, Graziani A, Grande G, Ortolani C, Ferlin A. HPV-related diseases in male patients: An underestimated conundrum. J Endocrinol Invest. 2024;47(2):261–74.
Lewis RM, Laprise J-F, Gargano JW, Unger ER, Querec TD, Chesson HW, et al. Estimated prevalence and incidence of disease-associated human papillomavirus types among 15-to 59-year-olds in the United States. Sex Transm Dis. 2021;48(4):273–7.
Zhou Y-X, Ma X-H, Wang T-T, Qu X-L, Zhang X-Q. Analysis of age-specified and genotype distribution of HPV multiple infections in the Chinese population. Sci Rep. 2024;14(1):2678.
Khoo SP, Bhoo-Pathy N, Yap SH, Shafii MK, Nasir NH, Belinson J, Subramaniam S, Goh PP, Zeng M, Tan HD, Gravitt P. Prevalence and sociodemographic correlates of cervicovaginal human papillomavirus (HPV) carriage in a cross-sectional, multiethnic, community-based female Asian population. Sex Transm Infect. 2018;94(4):277–83.
Huang W, Xu H, Hu H, Zhang D, Liu Y, Guo Y, et al. The prevalence of human papillomavirus among women in northern Guangdong Province of China. Sci Rep. 2022;12(1):13353.
Ong SK, Abe SK, Thilagaratnam S, Haruyama R, Pathak R, Jayasekara H, Togawa K, Bhandari AK, Shankar A, Nessa A, Jugder U. Towards elimination of cervical cancer–human papillomavirus (HPV) vaccination and cervical cancer screening in Asian National Cancer Centers Alliance (ANCCA) member countries. Lancet Reg Health West Pac. 2023;39.
Gollu AN, Gore CA. Knowledge, awareness and attitude of medical students regarding HPV infection and HPV vaccination. Asian Pacific J Cancer Care. 2021;6(1):41–6.
Ruan Y, Li H, Liu M, Cao G, Xu X, Han L, et al. A retrospective analysis of human papillomavirus (HPV) prevalence and genotype distribution among 25,238 women in Shanghai, China revealed the limitations of current HPV-based screening and HPV vaccine. Cancer Epidemiol. 2023;84:102372.
Sabet F, Mosavat A, Ghezeldasht SA, Basharkhah S, Shamsian SAA, Abbasnia S, et al. Prevalence, genotypes and phylogenetic analysis of human papillomaviruses (HPV) in Northeast Iran. Int J Infect Dis. 2021;103:480–8.
Bahramabadi R, Honarvar Z, Iranpour M, Arababadi MK, Dehesh T, Dabiri B, et al. Epidemiological study of various HPV strains in cervical fluid samples in south-eastern Iran, 2018–2020. Archiv Iran Med. 2021;24(9):678–83.
Shalchimanesh Z, Ghane M, Kalantar E. Prevalence of human papillomavirus genotypes in Tehran. Iran J Res Health Sci. 2022;22(3)
Wang Y, Meng Y, Li W, Zhang X, Deng Z, Hu M, et al. Prevalence and characteristics of hrHPV infection among 414,540 women: a multicenter study in central and eastern China. J Cancer. 2019;10(8):1902.
Moossavi M, Fereidouni M, Zardast M, Khazaei Z, Ghanbarzadeh N. Genotype distribution of human papilloma virus among women with genital warts biopsies in southern Khorasan, eastern Iran. Meta Gene. 2020;25:100720.
Xia C, Li S, Long T, Chen Z, Chan PK, Boon SS. Current updates on cancer-causing types of human papillomaviruses (HPVs) in east, southeast, and South Asia. Cancers. 2021;13(11):2691.
Kesheh MM, Keyvani H. The prevalence of HPV genotypes in Iranian population: an update. Iran J Pathol. 2019;14(3):197.
Fogarasi AI, Benczik M, Moravcsik-Kornyicki Á, Kocsis A, Gyulai A, Kósa Z. The prevalence of high-risk human papillomavirus in Hungary—a geographically representative. Cross-Section Study Pathol Oncol Res. 2022;28:1610424.
Tang X, Jones TE, Jiang W, Austin M, He Y, Li L, et al. Extended human papillomavirus genotype distribution in cervical intraepithelial neoplasia and cancer: analysis of 40 352 cases from a large academic gynecologic center in China. J Med Virol. 2023;95(1):e28302.
Acknowledgements
The authors acknowledge the help of the Research Center for Clinical Virology (RCCV) at the Tehran University of Medical Science in Tehran, Iran.
Funding
No Funding.
Author information
Authors and Affiliations
Contributions
M.H and L.AM: Methodology, Investigation, Writing original draft. S.M, F.H, F.S.E, N.Z, Z.T, A.A, H.Y, P.M, M.M.R. M.A.S.N. A.S: Investigation, writing original draft. A.D: Formal analysis. A.M, M.N, F.H: Validation. A.M, A.L: Supervision, Review and editing, Investigation, Validation, Methodology.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The research adhered to the principles of the Declaration of Helsinki and received approval from the Ethics Committee of Tehran University of Medical Sciences in Tehran, Iran (IR.TUMS.SPH.REC.1402.317).
Consent for publication
Informed consent for publication of identifiable information/ images in open access journal was obtained from all study participants.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Haddadi, M., Atefmehr, L., Motlaghzadeh, S. et al. Prevailing of HPV-16 and 52 genotype in 2022–2023 in Sanandaj, Iran. Virol J 21, 106 (2024). https://doi.org/10.1186/s12985-024-02373-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12985-024-02373-3