Abstract
Background
Due to high fertility rates in some low and lower-middle income countries, the interval between pregnancies can be short, which may lead to adverse maternal and neonatal outcomes.
Methods
We analyzed data from women enrolled in the NICHD Global Network Maternal Newborn Health Registry (MNHR) from 2013 through 2018. We report maternal characteristics and outcomes in relationship to the inter-delivery interval (IDI, time from previous delivery [live or stillborn] to the delivery of the index birth), by category of 6–17 months (short), 18–36 months (reference), 37–60 months, and 61–180 months (long). We used non-parametric tests for maternal characteristics, and multivariable logistic regression models for outcomes, controlling for differences in baseline characteristics.
Results
We evaluated 181,782 women from sites in the Democratic Republic of Congo, Zambia, Kenya, Guatemala, India, and Pakistan. Women with short IDI varied by site, from 3% in the Zambia site to 20% in the Pakistan site. Relative to a 18–36 month IDI, women with short IDI had increased risk of neonatal death (RR = 1.89 [1.74, 2.05]), stillbirth (RR = 1.70 [1.56, 1.86]), low birth weight (RR = 1.38 [1.32, 1.44]), and very low birth weight (RR = 2.35 [2.10, 2.62]). Relative to a 18–36 month IDI, women with IDI of 37–60 months had an increased risk of maternal death (RR 1.40 [1.05, 1.88]), stillbirth (RR 1.14 [1.08, 1.22]), and very low birth weight (RR 1.10 [1.01, 1.21]). Relative to a 18–36 month IDI, women with long IDI had increased risk of maternal death (RR 1.54 [1.10, 2.16]), neonatal death (RR = 1.25 [1.14, 1.38]), stillbirth (RR = 1.50 [1.38, 1.62]), low birth weight (RR = 1.22 [1.17, 1.27]), and very low birth weight (RR = 1.47 [1.32,1.64]). Short and long IDIs were also associated with increased risk of obstructed labor, hemorrhage, hypertensive disorders, fetal malposition, infection, hospitalization, preterm delivery, and neonatal hospitalization.
Conclusions
IDI varies by site. When compared to 18–36 month IDI, women with both short IDI and long IDI had increased risk of adverse maternal and neonatal outcomes.
Trial registration
The MNHR is registered at NCT01073475.
Similar content being viewed by others
Plain English summary
Due to high fertility rates in some low and lower-middle income countries, the interval between pregnancies can be short, which may lead to poor maternal and neonatal health outcomes. We measured the time between the delivery of one child to the delivery of the next child in six low and lower-middle income countries. We highlight differences, by country, in the number of women who have a short delivery interval from 4% of women in the Zambia site to 20% of women in the Pakistan site. We also highlight differences, by country, in the number of women who have long delivery intervals, from 4% of women in the Democratic Republic of Congo site to 24% of women in the Zambia site. Women with both a short and long delivery interval have higher risk of poor outcomes related to childbirth (obstructed labor, hemorrhage, disorders of high blood pressure, fetal malposition, infection and hospitalization), and poor outcomes for their babies (neonatal death, stillbirth, preterm delivery, low birth weight and hospitalization). Women with long delivery intervals also experience higher risk of maternal death.
Background
High fertility rates are common in low and lower-middle income countries (LMICs). Among the 6 LMICs included in the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Global Network for Women’s and Children’s Health Research (GN), fertility rates varied from 2.9 to 6.0 births per woman [1]. High fertility rates lead to shortened time between pregnancies, without allowing the mother to fully recover to baseline health status prior to a subsequent gestation [2, 3]. Short intervals between pregnancies are associated with many adverse health outcomes for the mother, including anemia, placental abruption, placenta previa and uterine rupture [4]. Short birth intervals are also associated with adverse newborn health outcomes such as infant mortality, preterm birth, low birth weight (LBW) and congenital malformations [4,5,6,7,8]. Conversely, long birth intervals can also be associated with adverse maternal and neonatal health outcomes, such as increased risk for induction of labor, chorioamnionitis, Caesarean delivery, preterm birth, LBW, and small for gestational age infants [4, 7, 9]. The ideal timing between pregnancies associated with optimal maternal and neonatal health outcomes has not been definitively established.
The limited existing evidence on the optimal timing between pregnancies is complicated by varying methodologies used to calculate birth spacing. Birth spacing can be defined in several ways, such as the birth-to-pregnancy interval (the period from the prior live birth to the conception of the index pregnancy), the inter-pregnancy interval (the period from the prior birth, regardless of whether the pregnancy resulted in miscarriage/stillbirth/live birth, to the conception of the index pregnancy) or the inter-delivery interval (IDI; the period from the delivery of the prior live birth to the delivery of the index pregnancy) [10]. In the 2005 World Health Organization (WHO) Technical Report, an expert panel preferred birth-to-pregnancy interval to measure birth spacing [10]. Birth-to-pregnancy interval is challenging to measure in low-resource settings where pregnancy dating is inaccurate and therefore length of gestation is difficult to determine [11]. In order to calculate birth-to-pregnancy interval, the expert panel used delivery to delivery interval minus 9 months, thus assuming the index pregnancy resulted in a term gestation. This methodology underestimates the time between births and negates the opportunity to evaluate the effect of birth spacing on the risk of prematurity. The use of IDI might be more appropriate in low-resource settings to investigate associations between birth spacing and neonatal outcomes, without introducing the bias of unknown gestational age.
Based on limited evidence, the WHO recommends a birth-to-pregnancy interval of 24 months, corresponding to an IDI of approximately 33 months, for optimal maternal and neonatal outcomes [10]. After the WHO 2005 Technical Meeting on birth spacing, there was a call for further research to better understand the effect of birth spacing on maternal morbidity and mortality using large datasets. In this paper, we describe IDI in a prospective, multi-country pregnancy registry from 7 research sites in 6 LMICs. We examine maternal characteristics associated with varying lengths of IDI as well as the relationship between adverse delivery and neonatal outcomes and IDI.
Methods
We analyzed data from women who were enrolled in the NICHD GN’s Maternal Newborn Health Registry (MNHR) from November 2013 through December 2018. The MNHR is a multi-country, population-based, prospectively collected record of pregnancy characteristics as well as maternal and infant outcomes [12]. The MNHR includes research sites in North and South Ubangi, Democratic Republic of Congo (DRC); Kafue and Chongwe (located south and east of the capital city of Lusaka), Zambia; Busia, Bungoma and Kakamega (within the western region), Kenya; Chimaltenango (in the Western Highlands), Guatemala; Belagavi and Bagalkot (within the northern part of the southern state of Karnataka), India; Nagpur (within the state of Maharashtra), India; and Thatta (two of the five sub-districts in the southern Sindh province, near the city of Karachi), Pakistan. The sites represent study clusters from both semi-urban and rural environments.
A detailed description of the MNHR methods are described elsewhere, but briefly, MNHR data were collected from abstraction of medical records as well as a series of interviews conducted by trained study staff [13, 14]. Maternal characteristics, including demographic information, were collected at the time a woman was screened and consented. We also gathered information about the prior pregnancy by maternal report at the time of enrollment. Antenatal and delivery characteristics were recorded within 3–7 days after delivery. Postpartum characteristics were collected at a clinic or home visit 6 weeks after delivery. Maternal anthropometry was not routinely collected at all sites throughout the study period. Maternal weight was collected at the time of enrollment, however enrollment could occur at any time during pregnancy, so these measurements do not provide a consistent reflection of nutritional status. Maternal height was collected at most sites, but this measurement was not collected until 2017 in Kenya. Body mass index (BMI) was calculated from maternal height and weight, when available. Due to these methodological limitations, maternal anthropometry is presented as descriptive data only.
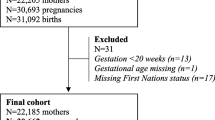
In this analysis, we included all women in the MNHR with an index pregnancy during the specified time period with the following exclusions: women who were lost to follow up prior to delivery, primiparous women, women without a previous pregnancy lasting greater than or equal to 20 weeks, women with unknown parity, multiparous women who had a missing or unknown delivery date for the previous pregnancy, women whose index pregnancy resulted in a miscarriage or medically terminated pregnancy (MTP), and women who had an extreme IDI (< 6 months or > 180 months (15 years)). We defined miscarriage or MTP as a pregnancy that ended prior to 20 weeks gestation. As we could not reliably collect data on pregnancies resulting in a miscarriage or MTP across all sites, these pregnancies were also excluded.
We evaluated IDI in 4 categories based on distinctions in the medical literature: 6–17 months, 18–36 months, 37–60 months, > 60 months [9]. We defined short IDI as the interval from 6 to 17 months. We chose 6 months as the lower limit for analysis to account for at least a 1-month period for return to fecundability and an additional 5 months gestation, since we excluded index pregnancies that resulted in miscarriage or MTP before 20 weeks / 5 months gestation. The category 18–36 months includes the WHO recommended 33 months for optimal birth spacing and therefore was used as the referent category. We defined long IDI as > 60 months. We calculated IDI as the number of months from the date of delivery of the previous pregnancy (resulting in a liveborn or stillborn infant), as reported by the mother, to the date of delivery of the index pregnancy.
To determine if the distribution of IDI differed across sites, we performed a non-parametric Kruskal Wallis test for overall site difference and non-parametric Wilcoxon rank sum tests for all pairwise site comparisons. To determine the relationship between IDI and maternal characteristics, we performed Cochran-Mantel-Haenszel tests of each maternal factor and IDI category stratified by cluster. Risk of maternal outcomes and fetal/neonatal outcomes associated with IDI categories were determined from multivariable generalized linear models with general estimating equations to control for cluster level effects. Models were adjusted for maternal age, education, parity, antenatal care (ANC) visits and iron supplementation. In the maternal risk factors model, the mode of delivery was not included because it is not solely a maternal risk factor and is often influenced by the occurrence of several of the other outcomes. Maternal height, weight, and BMI were not included due to poor data consistency. We used a Poisson distribution for the low-prevalence outcomes of stillbirth and very low birth weight (VLBW), all other outcomes were modeled with a binomial distribution. We report the relative risks (RR) and 95% confidence intervals (CIs) for each outcome by IDI categories with the referent category of 18–36 months. Fetal/neonatal outcomes are reported at the maternal level if at least one fetus/neonate from a multiple birth pregnancy had that outcome.
At each site, institutional review boards or research ethics committees and Ministries of Health approved the collection of data included in the MNHR. We used sensitization meetings to achieve approval within local communities prior to the initiation of the study. All study participants were enrolled with informed consent. A data monitoring committee appointed by the NICHD oversaw and reviewed the MNHR annually.
Results
We screened 314,313 pregnant women in 7 research sites for inclusion in the MNHR from November 2013 through 2018 (Table 1). We included 312,885 (99.5%) who were eligible and consented. After exclusion of women with unknown parity and nulliparous women, we retained 213,198 (68.1%) women. After exclusions for loss to follow-up prior to delivery, MTP, unreliable prior delivery date and IDI outlying the desired range, we included 181,782 (58.1%) women for analysis. Sites differed in the number of nulliparous mothers (17.1% in the Pakistan site to 49.2% in the Nagpur site). Of the 181,782 subjects, each site contributed between 20,148 and 34,342 women for analysis (Table 2). The distribution of IDI differed across sites overall as well as for each pairwise site comparison (p < 0.0001 for all comparisons). The percentage of women with short IDI varied from 3.4% of women in the Zambia site to 19.8% of women in the Pakistan site. The percentage of women with long IDI varied from 4.1% of women in the DRC site to 23.9% of women in the Zambia site. The overall median IDI was 32 (24, 45) months, ranging from 27 months in the Belagavi and Pakistan sites to 43 months in the Zambia site.
All maternal characteristics showed a statistically significant difference when evaluated by IDI (Table 3, p < 0.001 for all comparisons). Generally, as IDI lengthened, women were more likely to be older, more educated, receive more ANC and receive more iron supplements. Descriptive data for maternal anthropometry is included in the supplemental material. In the multivariable models, short and long IDI had significantly greater risk for nearly all adverse maternal outcomes when compared to an 18–36-month IDI (Table 4). Women with a short IDI had an increased risk of obstructed labor (RR = 1.17 [1.07, 1.28]), maternal hemorrhage (RR = 1.17 [1.04, 1.33]), hypertensive disorders (RR = 1.38 [1.19,1.61]), fetal malposition (RR = 1.27 [1.11, 1.46]), maternal infection (RR = 1.35 [1.17, 1.56]) and maternal hospitalization (RR = 1.31 [1.22, 1.41]). Women with a long IDI also had increased risk of the same adverse delivery outcomes: obstructed labor (RR = 1.54 [1.43, 1.65]), maternal hemorrhage (RR = 1.19 [1.06, 1.32]), hypertensive disorders (RR = 2.10 [1.87, 2.36]), fetal malposition (RR = 1.34 [1.22, 1.48]), maternal infection (RR = 1.33 [1.21, 1.46]), and maternal hospitalization (RR = 1.55 [1.43, 1.67]). Additionally, women with a long IDI had an increased risk of maternal mortality (RR = 1.54 [1.10, 2.16]). Overall, the adjusted RR for maternal mortality was increased for all IDI categories relative to the 18–36-month referent group, although only statistically significant for 37–60 months and long IDI (Fig. 1).
Similarly, short and long IDI had significantly greater risk for nearly all adverse fetal/neonatal outcomes when compared to an 18–36-month IDI (Table 5). Women with a short IDI had an increased risk of LBW (RR = 1.38 [1.32, 1.44]) and VLBW (RR = 2.35 [2.10, 2.62]), stillbirth (RR = 1.70 [1.56, 1.86]), neonatal mortality (RR = 1.89 [1.74, 2.05]), preterm delivery (RR = 1.44 [1.39, 1.50]), and neonatal hospitalization (RR = 1.24 [1.11, 1.38]). Women with a long IDI also had increased risk of the same adverse neonatal outcomes: LBW (RR = 1.22 [1.17, 1.27]) and VLBW (RR = 1.47 [1.32,1.64]), stillbirth (RR = 1.50 [1.38, 1.62]), neonatal mortality (RR = 1.25 [1.14, 1.38]), preterm delivery (RR = 1.06 [1.02, 1.10]), and neonatal hospitalization (RR = 1.28 [1.15, 1.43]). Overall, the adjusted RR for neonatal mortality was increased for all IDI categories relative to the 18–36-month referent group, although the increase at 37–60 months was very small and not statistically significant (Fig. 2). In contrast, the adjusted RR for stillbirth and very low birth weight was statistically significantly increased for all IDI categories relative to the 18–36-month referent group (Figs. 3 and 4).
Discussion
Our results indicate that IDI is associated with a number of adverse maternal and neonatal health consequences. We highlight differences by country in the number of women who have a short IDI, from 3% of women in the Zambia site to 20% of women in the Pakistan site. We also highlight differences by country in the number of women who have a long IDI, from 4% of women in the Pakistan and DRC sites to 24% of women in the Zambia site. Women with both a short and long IDI have higher risk of adverse delivery outcomes (obstructed labor, hemorrhage, hypertensive disorders, fetal malposition, infection and hospitalization) and adverse neonatal outcomes (neonatal death, stillbirth, preterm delivery, LBW, VLBW and hospitalization). Women with a long IDI also experience higher risk of maternal death.
Similar to other studies, we demonstrated a bimodal distribution of adverse birth outcomes, with increased risk among women with short and long IDI [15,16,17]. Of particular note, we demonstrated an increased risk of LBW and VLBW infants at the extremes of IDI. For short birth intervals, this might be explained by a nutritional depletion hypothesis in which the short birth interval results from maternal nutrient deficiency after depletion from the previous gestational period, particularly folate deficiency, which results in impaired fetal growth [2, 18]. However, long IDIs were also associated with LBW infants. This finding indicates that the nutritional depletion hypothesis is insufficient to explain LBW among this group. Among women with a long IDI, we observed a higher RR of hypertensive disorders. The higher prevalence of hypertensive disorders, such as pre-eclampsia, might be a potential mechanism leading to LBW infants, given the established causal relationship in which pre-eclampsia leads to prematurity and growth restriction [19].
The findings of our study support the WHO recommendations for an optimal IDI of 33 months. When compared to short and long intervals, the interval around 33 months was associated with the best maternal and neonatal outcomes. We evaluated IDI categorically in order to compare with previous studies and evaluate current recommendations. Analysis by categories is advantageous given the nonlinear relationship with delivery intervals and birth outcomes [20]. While this approach did not allow for prediction of the optimal number of months for IDI, our robust data support the WHO recommendations of IDI of approximately 33 months.
We noted important differences in maternal characteristics by IDI. Women who had longer IDIs also had characteristics that are usually associated with better delivery outcomes, for example older age, more education, the receipt of more ANC, and receipt of iron supplements. Before we adjusted for these associations, it appeared that neonatal outcomes improved with increasing IDI. However, when we adjusted for maternal characteristics in our models, the models showed an increased risk of adverse neonatal outcomes at both lower and higher IDI categories. This indicates that women with long IDIs do not return to baseline risk for adverse perinatal outcomes even with improvements in health seeking behaviors.
Our study had a number of strengths. The MNHR pregnancy cohort includes a large and multi-national cohort of women from Africa, Asia and Central America. Our data were collected prospectively and included all pregnant women within a study community, allowing population level conclusions within those communities. Our dataset allowed us to describe associated maternal risk factors in addition to both maternal and neonatal outcomes within this population. However, we were also limited by some of the characteristics of the MNHR. We were limited in our ability to assess maternal nutritional status, therefore we are limited in our ability to address the nutrition hypotheses that might contribute to LBW associated with IDI. Because our studies occurred in low-resource settings, there might have been some variability in the reliable assignment of gestational age that could have introduced bias in our results if small for gestational age infants born at term were assigned to the premature category. To improve interpretation of birth weight, we chose to also evaluate LBW and VLBW separately. We chose to evaluate IDI rather than inter-pregnancy interval, so pregnancies that did not last more than 20 weeks are not included. Therefore, our analyses are limited since these pregnancies contribute to adverse maternal and neonatal outcomes.
While the social factors that determine birth spacing are complex and include familial influences and community level influences, there have been some intervention strategies that have been successful in lengthening birth intervals and mitigating some of the risk of adverse maternal and neonatal health outcomes [21, 22]. For example, in Bangladesh, a package of family planning interventions integrated into maternal and newborn health visits decreased the number of women who had a subsequent short birth interval and lowered the risk of preterm birth [23].
Conclusions
Our data increase the body of literature describing optimal birth intervals in relationship to maternal and newborn health outcomes in LMICs. We describe increased health risk at extremes of birth intervals and support the WHO recommendations for optimal birth spacing. Our data illustrate geographical differences in IDI which underpin the need for programmatic public health efforts to improve birth spacing in certain areas to achieve optimal maternal and neonatal outcomes. These data can inform communities with high rates of sub-optimal birth spacing to direct public health strategies to the regions in most need.
Availability of data and materials
The datasets generated and analysed during the current study are not yet publicly available due to ongoing data analyses, but they will be available in the NICHD Data and Specimen Hub. Requests for data prior to the public release will be handled by the authors.
Abbreviations
- IDI:
-
Interval delivery interval
- LMIC:
-
Low and lower-middle income countries
- NICHD:
-
National Institute of Health Eunice Kennedy Shriver National Institute of Child Health and Human Development
- GN:
-
NICHD Global Network for Women’s and Children’s Health Research
- MNHR:
-
Maternal Newborn Health Registry
- LBW:
-
Low birth weight
- VLBW:
-
Very low birth weight
- WHO:
-
World Health Organization
- DRC:
-
Democratic Republic of Congo
- BMI:
-
Body Mass Index
- MTP:
-
Medical termination of pregnancy
- RR:
-
Relative Risks
- CI:
-
Confidence Intervals
- ANC:
-
Antenatal care
References
Fertility rate, total (births per woman) [https://data.worldbank.org/indicator/sp.dyn.tfrt.in], Accessed 12, June 2019.
Dewey KG, Cohen RJ. Does birth spacing affect maternal or child nutritional status? A systematic literature review. Matern Child Nutr. 2007;3:151–73.
Rutstein SO. Effects of preceding birth intervals on neonatal, infant and under-five years mortality and nutritional status in developing countries: evidence from the demographic and health surveys. Int J Gynaecol Obstet. 2005;89(Suppl 1):S7–24.
Conde-Agudelo A, Rosas-Bermudez A, Kafury-Goeta AC. Birth spacing and risk of adverse perinatal outcomes: a meta-analysis. JAMA. 2006;295:1809–23.
Conde-Agudelo A, Rosas-Bermudez A, Kafury-Goeta AC. Effects of birth spacing on maternal health: a systematic review. Am J Obstet Gynecol. 2007;196:297–308.
Zhu BP, Rolfs RT, Nangle BE, Horan JM. Effect of the interval between pregnancies on perinatal outcomes. N Engl J Med. 1999;340:589–94.
DaVanzo J, Hale L, Razzaque A, Rahman M. Effects of interpregnancy interval and outcome of the preceding pregnancy on pregnancy outcomes in Matlab, Bangladesh. BJOG. 2007;114:1079–87.
Arafa MA, Alkhouly A, Youssef ME. Influence of inter-pregnancy interval on preterm delivery. Paediatr Perinat Epidemiol. 2004;18:248–52.
Yee LM, Truong YN, Caughey AB, Cheng YW. The association between interdelivery interval and adverse perinatal outcomes in a diverse US population. J Perinatol. 2016;36:593–7.
WHO. Report of a WHO Technical Consultation on Birth Spacing. (Department of Making Pregnancy Safer DoRHaR ed). Geneva: WHO; 2007.
Lee AC, Panchal P, Folger L, Whelan H, Whelan R, Rosner B, et al. Diagnostic accuracy of neonatal assessment for gestational age determination: a systematic review. Pediatrics. 2017;140:1–26.
Bose CL, Bauserman M, Goldenberg RL, Goudar SS, McClure EM, Pasha O, et al. The Global Network Maternal Newborn Health Registry: a multi-national, community-based registry of pregnancy outcomes. Reprod Health. 2015;12(Suppl 2):S1.
Goudar SS, Carlo WA, McClure EM, Pasha O, Patel A, Esamai F, et al. The maternal and newborn health registry study of the global network for Women's and Children's Health Research. Int J Gynaecol Obstet. 2012;118:190–3.
Goudar SS, Stolka KB, Koso-Thomas M, Honnungar NV, Mastiholi SC, Ramadurg UY, et al. Data quality monitoring and performance metrics of a prospective, population-based observational study of maternal and newborn health in low resource settings. Reprod Health. 2015;12(Suppl 2):S2.
Conde-Agudelo A, Belizan JM. Maternal morbidity and mortality associated with interpregnancy interval: cross sectional study. BMJ. 2000;321:1255–9.
Conde-Agudelo A, Belizan JM, Norton MH, Rosas-Bermudez A. Effect of the interpregnancy interval on perinatal outcomes in Latin America. Obstet Gynecol. 2005;106:359–66.
Mahande MJ, Obure J. Effect of interpregnancy interval on adverse pregnancy outcomes in northern Tanzania: a registry-based retrospective cohort study. BMC Pregnancy Childbirth. 2016;16:140.
Fotso JC, Cleland J, Mberu B, Mutua M, Elungata P. Birth spacing and child mortality: an analysis of prospective data from the Nairobi urban health and demographic surveillance system. J Biosoc Sci. 2013;45:779–98.
Mol BWJ, Roberts CT, Thangaratinam S, Magee LA, de Groot CJM, Hofmeyr GJ. Pre-eclampsia. Lancet. 2016;387:999–1011.
Hutcheon JA, Moskosky S, Ananth CV, Basso O, Briss PA, Ferre CD, et al. Good practices for the design, analysis, and interpretation of observational studies on birth spacing and perinatal health outcomes. Paediatr Perinat Epidemiol. 2019;33:O15–24.
McGuire C, Stephenson R. Community factors influencing birth spacing among married women in Uganda and Zimbabwe. Afr J Reprod Health. 2015;19:14–24.
Class QA, Rickert ME, Oberg AS, Sujan AC, Almqvist C, Larsson H, et al. Within-family analysis of Interpregnancy interval and adverse birth outcomes. Obstet Gynecol. 2017;130:1304–11.
Baqui AH, Ahmed S, Begum N, Khanam R, Mohan D, Harrison M, et al. Impact of integrating a postpartum family planning program into a community-based maternal and newborn health program on birth spacing and preterm birth in rural Bangladesh. J Glob Health. 2018;8:020406.
Acknowledgements
Not applicable.
About this supplement
This article has been published as part of Reproductive Health, Volume 17 Supplement 2, 2020: Global Network MNH. The full contents of the supplement are available at https://reproductive-health-journal.biomedcentral.com/articles/supplements/volume-17-supplement-2
Funding
Publication of this supplement is funded by grants from Eunice Kennedy Shriver National Institute of Child Health and Human Development. The funders had no input in the design of the study, collection, analysis, interpretation of data or in the writing of the manuscript, with the exception of the Program Officer of NICHD, whose contributions are outlined.
Author information
Authors and Affiliations
Contributions
MB, TN, JM, KN, SS, RG, RD, WC and CB conceived of the idea for this paper. AL, AT, AP, PH, AG, NK, FE, EL, WC, EC, SG, RD, SS, RG, and EM carried out the research. JM, KN, TN performed the analyses. MB prepared the first draft of the manuscript. JP, MK and all authors reviewed and edited subsequent manuscript drafts. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
At each site, institutional review boards or ethics committess approved the study. All women provided written informed consent before the start.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Bauserman, M., Nowak, K., Nolen, T.L. et al. The relationship between birth intervals and adverse maternal and neonatal outcomes in six low and lower-middle income countries. Reprod Health 17 (Suppl 2), 157 (2020). https://doi.org/10.1186/s12978-020-01008-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12978-020-01008-4