Abstract
Background
Despite common enthusiasm for cardiovascular magnetic resonance (CMR), its application in Europe is quite diverse. Restrictions are attributed to a number of factors, like limited access, deficits in training, and incomplete reimbursement. Aim of this study is to perform a systematic summary of the representation of CMR in the guidelines of the European Society of Cardiology (ESC).
Methods
Twenty-nine ESC guidelines were screened for the terms “magnetic”, “MRI”, “CMR”, “MR” and “imaging”. As 3 topics were published twice (endocarditis, pulmonary hypertension, NSTEMI), 26 guidelines were finally included. MRI in the context of non-cardiovascular examinations was not recognized. The main CMR-related conclusions and, if available, the level of evidence and the class of recommendation were extracted.
Results
Fourteen of the 26 guidelines (53.8 %) contain specific recommendations regarding the use of CMR. Nine guidelines (34.6 %) mention CMR in the text, and 3 (11.5 %) do not mention CMR. The 14 guidelines with recommendations regarding the use of CMR contain 39 class-I recommendations, 12 class-IIa recommendations, 10 class-IIb recommendations and 2 class-III recommendations. Most of the recommendations have evidence level C (41/63; 65.1 %), followed by level B (16/63; 25.4 %) and level A (6/63; 9.5 %). The four guidelines, which absolutely contained most recommendations for CMR, were stable coronary artery disease (n = 14), aortic diseases (n = 9), HCM (n = 7) and myocardial revascularization (n = 7).
Conclusions
CMR is represented in the majority of the ESC guidelines. They contain many recommendations in favour of the use of CMR in specific scenarios. Issues regarding access, training and reimbursement have to be solved to offer CMR to patients in accordance with the ESC guidelines.
Similar content being viewed by others
Background
Cardiovascular magnetic resonance (CMR) has been applied in a wide variety of indications in clinical cardiology. The most frequent indications are inflammatory and ischemic heart disease as well as cardiomyopathies. But also in rare diseases like amyloidosis, as well as in congenital heart disease, CMR has demonstrated its usefulness [1, 2]. CMR provides detailed information about cardiovascular anatomy and function by combining diverse techniques. In particular, the characterization of the myocardial tissue including the detection of oedema and the highly resolved determination of fibrosis is a unique feature of CMR [3]. Furthermore, with myocardial stress-perfusion imaging – free of ionizing radiation and with high diagnostic accuracy – the large patient group with (suspected) coronary artery disease is addressed [4]. Finally, the introduction of robust and fast imaging techniques as well as targeted examination protocols facilitated the clinical use [5].
Despite common enthusiasm for this modality, its use in Europe is quite diverse. This restriction is attributed to a number of factors, like missing skills both to run a CMR examination and to interpret the images under integration of profound cardiologic knowledge; relatively high costs and incomplete reimbursement; and limited access to scanners with cardiac dedication.
Aim of this study is to perform a systematic summary of the representation of CMR in the guidelines of the European Society of Cardiology (ESC) in order to stimulate the discussion about future plans for training, distribution and reimbursement of CMR in Europe.
Methods
All ESC guidelines, which are listed on the ESC website (http://www.escardio.org/Guidelines-&-Education/Clinical-Practice-Guidelines/ESC-Clinical-Practice-Guidelines-list/listing) were collected (Table 1). If more than one guideline for the same topic has been published in this period, both were analysed for changes, but only the most recent was included in the final analysis. The documents were screened for the terms “magnetic”, “MRI”, “CMR”, “MR” and “imaging”. MRI in the context of non-cardiovascular examinations like brain MRI was not recognized. The main conclusions were extracted and if available, the level of evidence and the class of recommendation were given (Tables 2 and 3). The number in parenthesis behind the citation provides the page of the fulltext guideline. If a recommendation refers to “imaging” in general, it was registered if the context included CMR. This process was done twice for every guideline to reassure that no relevant information was missed. The absolute number of recommendations is finally summarized, whereby equal recommendations that appeared in more than one guideline were only counted once. The order of the guidelines is chronologic beginning with the most recent. Guidelines other than by the ESC as well as ESC position statements were not included to guarantee one common level of guideline.
Results
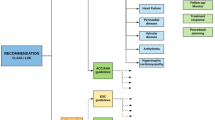
Of the 29 ESC guidelines we screened, three topics were covered twice (endocarditis 2015 and 2009, pulmonary hypertension 2015 and 2009, NSTEMI 2015 and 2011). Of the remaining 26 ESC guidelines, 14 (53.8 %) contain specific recommendations regarding the use of CMR (Fig. 1, Table 1). Nine guidelines (34.6 %; endocarditis, pulmonary hypertension, diabetes, pacing, heart valve disease, definition of infarction, prevention, congenital heart disease, syncope) principally mention scenarios in which CMR may be used, but without giving any specific recommendation. Three guidelines (11.5 %; atrial fibrillation, dyslipidaemias, device therapy in heart failure) do not mention CMR at all. The 14 guidelines with recommendations regarding the use of CMR contain 39 class-I recommendations, 12 class-IIa recommendations, 10 class-IIb recommendations and 2 class-III recommendations (Fig. 2). (The diverse recommendations for myocardial revascularization in dependency of the evidence of ischemia were only counted once as IA). Most of the recommendations have evidence level C (41/63; 65.1 %), followed by level B (16/63; 25.4 %) and level A (6/63; 9.5 %). The two class-III recommendations in the context of CMR are: i) In the guideline for pulmonary embolism, MR angiography should not be used to rule out pulmonary embolism. ii) In the guideline about assessment before non-cardiac surgery, imaging stress testing in general is not recommended before low-risk surgery. The four guidelines, which absolutely contained most recommendations with referral to CMR, were stable coronary artery disease from 2013 (n = 14), aortic diseases (n = 9), HCM (n = 7) as well as myocardial revascularization (n = 7) from 2014. Twenty-eight recommendations refer to stress-imaging, 17 recommendations refer to the vasculature, 7 to cardiomyopathies, 5 to left- and right-ventricular function assessment (in part including fibrosis imaging), 4 to the pericardium and 2 to myocarditis. A summary of clinical scenarios/diagnoses, where the ESC made recommendations regarding CMR, is provided in the appendix of this paper.
2015 ESC guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death [6]
Table 4 summarizes the recommendations for CMR in the context of patients with ventricular arrhythmias and the prevention of sudden cardiac death.
For family members of sudden unexplained death syndrome or sudden arrhythmic death syndrome victims, echocardiography and/or CMR is recommended (Appendix on page 11 of the guideline). In patients with sustained ventricular tachycardia or ventricular fibrillation, the recommended algorithm for further patient assessment includes CMR (Fig. 1 on page 14). For instance myocarditis should also be suspected and a CMR scan may reveal abnormal fibrotic myocardial tissue (page 54). In patients with non-ischaemic cardiomyopathy, CMR fibrosis imaging (using late gadolinium enhancement, LGE) is associated with increased risk of all-cause mortality, heart failure hospitalization and sudden cardiac death. The incremental value of LGE over other prognostic markers needs to be determined (page 36). Standardized evaluation of patients with HCM should include CMR in the case of inadequate echo window (page 39). LGE has been suggested to be used to guide ICD therapy in individuals with HCM with intermediate risk, however with few supportive data (page 38). Similarly, LGE on CMR of the right and left ventricle has been reported as risk factors for sudden cardiac death or appropriate ICD discharge in ARVC (page 40). In particular in ARVC, CMR provides excellent assessment of right ventricular size, function and regional wall motion. In paediatric patients with frequent premature ventricular complexes, cardiac evaluation including CMR is recommended (page 46). Non-invasive imaging of cardiac structure, best done by CMR, can be used to plan and guide ablation procedures for ventricular tachycardia (page 22).
2015 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension [7]
CMR is listed among the tests to contribute to the diagnosis of pulmonary hypertension, being accurate and reproducible in the assessment of right ventricular size, morphology and function and of blood flow, stroke volume, cardiac output, pulmonary arterial distensibility and right ventricular mass. The presence of LGE, reduced pulmonary arterial distensibility and retrograde flow have high predictive value for the identification of pulmonary hypertension. In patients with pulmonary hypertension, CMR may also be useful in cases of suspected congenital heart disease if echocardiography is not conclusive. MR angiography has a potential in patients with suspected chronic thromboembolic pulmonary hypertension. CMR provides useful prognostic information in patients with pulmonary artery hypertension (page 12). Specifically, right atrial size and the presence of pericardial effusion as assessed by CMR are used for risk assessment in pulmonary arterial hypertension (table 13 of the guideline). There is no specific recommendation regarding CMR and no significant change regarding the role of CMR in pulmonary hypertension between the present guideline and the 2009 version [8].
2015 ESC guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation [9]
CMR can assess both perfusion and wall motion abnormalities, and patients presenting with acute chest pain with a normal stress CMR have an excellent short- and midterm prognosis. CMR also permits detection of scar tissue and can differentiate this from recent infarction. CMR can facilitate the differential diagnosis between infarction and myocarditis or Tako-Tsubo cardiomyopathy (page 11). In subjects with no criteria for early invasive strategy, a non-invasive imaging stress test is recommended. No specific test is mentioned (Table 5). There is no significant change of the role of CMR compared to the 2011 NSTEMI guideline [10].
2015 ESC guidelines for the diagnosis and management of pericardial diseases [11]
CMR has shifted towards a comprehensive imaging modality, allowing visualization and tissue characterization of the pericardium (and heart) in patients with pericardial disease and appraisal of the consequences of pericardial abnormalities on cardiac function and filling patterns (page 20). Table 12 of the guideline summarizes the contribution of different imaging modalities in various pericardial diseases and table 13 of the guideline compares non-invasive imaging modalities to study the pericardium. Thereby, CMR is predominantly ranked as good (“++”) or excellent (“+++”). Under the headline “what is new”, CMR is recommended for the detection of pericardial inflammation to identify forms of initial reversible constrictive pericarditis, allowing a trial of medical anti-inflammatory therapy (page 5). The evidence of pericardial inflammation by CMR is also mentioned as one diagnostic criterion for acute pericarditis (table 4 of the guideline). In patients with myocarditis, CMR is recommended for the confirmation of myocardial involvement (page 13). CMR may be helpful to detect loculated pericardial effusion and pericardial thickening and masses, as well as associated chest abnormalities. CMR can contribute to the differentiation of constrictive pericarditis and restrictive cardiomyopathy (table 10 of the guideline), e.g. by assessment of ventricular coupling with real-time cine magnetic resonance during free breathing (page 19). In some cases of pericardial cysts, CMR may be helpful (page 35). The recommendations made for CMR in pericardial diseases are summarized in Table 6.
2015 ESC guidelines for the management of infective endocarditis [12]
Within the subchapter about ‘complications of infective endocarditis’ dealing with ‘myocarditis and pericarditis’, CMR is mentioned (next to echocardiography) to assess myocardial involvement during infective endocarditis (page 30). This CMR indication is new compared to the 2009 guideline [13].
2014 ESC guidelines on diagnosis and management of hypertrophic cardiomyopathy [14]
HCM in adults is defined by a wall thickness ≥15 mm and in first-degree relatives ≥13 mm in one or more LV myocardial segments - as measured by any imaging technique, including CMR (page 7, 8). Some patients with apical or distal hypertrophy develop small apical aneurysms, sometimes associated with myocardial scarring. These may only be detectable on CMR, ventriculography or contrast echo (page 9). The prevalence of non-sustained ventricular tachycardia increases with age and correlates with LV wall thickness and the presence of LGE on CMR (page 36). However, even though the extent of LGE on CMR has some utility in predicting cardiovascular mortality, current data do not support the use of LGE in prediction of sudden cardiac death risk (page 14). LGE at the right ventricular insertion points or localized to segments of maximum LV thickening on CMR assists for differentiating the diagnosis of hypertensive heart disease and HCM (table 9 of the guideline). The specific recommendations made for CMR in HCM are summarized in Table 7.
2014 ESC guidelines on the diagnosis and treatment of aortic diseases [15]
CMR is regarded as a valuable tool to image the aorta. On a scale from “+” to “+++”, the ease of use is graded as “++”, diagnostic reliability as “+++”, serial examinations as “+++”, and aortic wall visualization as “+++” (page 11). CMR is considered the leading technique for diagnosis of aortic dissection, with a reported sensitivity and specificity of 98 %. However, several methodological and practical limitations preclude the use of this modality in the majority of cases and in unstable patients (page 21). Recommendations for the use of CMR in patients with aortic diseases are given Table 8.
2014 ESC/EACTS guidelines on myocardial revascularization [16]
This guideline contains recommendations for CMR both for determining myocardial ischemia and for follow-up patients after myocardial revascularization, as well as for preparation before surgical myocardial revascularization. Table 9 shows the specific recommendations. There is no clear recommendation for CMR viability testing. Even though CMR has a high diagnostic accuracy for assessing the transmural extent of myocardial scar tissue and contractile reserve, its ability to detect viability and predict recovery of wall motion is no better than other imaging techniques (page 15).
2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism [17]
MR angiography, although promising, is not yet ready for clinical practice due to its low sensitivity, high proportion of inconclusive MR angiography scans, and low availability in most emergency settings (Table 10).
2014 ESC/ESA guidelines on non-cardiac surgery: cardiovascular assessment and management [18]
Resting LV function can be evaluated before non-cardiac surgery in high-risk surgery (IIb, C). Following the guidelines, this can be done by radionuclide ventriculography, gated single photon emission computed tomography, echocardiography, CMR or multislice CT all with similar accuracy. The recommendations for non-invasive stress testing of ischemic heart disease are given in Table 11. As nuclear myocardial perfusion imaging and stress echocardiography were mainly used in clinical studies about preoperative ischemic testing, these modalities are pronounced. CMR (both perfusion and wall motion analysis) is mentioned as an accurate alternative method.
2013 ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD [19]
Patients with glucose perturbations are in need of early risk assessment to identify co-morbidities and factors that increase cardiovascular risk. This includes among other the evaluation of myocardial viability and LV function by means of echo-Doppler and/or CMR (page 28), and Duplex ultrasonography, computed tomography angiography and CMR to evaluate carotid artery stenosis (page 45). To evaluate inducible ischaemia, only exercise testing, stress echocardiography, or myocardial scintigraphy are mentioned (page 28).
2013 ESC guidelines on the management of stable coronary artery disease [20]
Table 12 summarizes the corresponding recommendations for CMR in the context of stable coronary artery disease. CMR may be used to define structural cardiac abnormalities and evaluate ventricular function. Use of CMR is recommended in patients, in whom, despite the use of echo contrast agents, transthoracic echocardiography is unable to answer the clinical question (usually because of a restricted acoustic window) and who have no contra-indications for CMR (page 13). In patients with suspected coronary artery disease and intermediate pretest probability, non-invasive testing is recommended. Among the modalities to perform stress imaging, CMR is mentioned on the same level as stress echocardiography, SPECT and PET. To stratify the risk for events, high risk is assumed in stress CMR if there are ≥2/16 segments with new perfusion defects or ≥3 dobutamine-induced dysfunctional segments (page 20). CMR coronary arteriography must still be regarded primarily as a research tool and is not recommended for routine clinical practice in the diagnostic evaluation of suspected coronary artery disease (page 19).
2013 ESC guidelines on cardiac pacing and cardiac resynchronization therapy [21]
Regarding patient selection for cardiac resynchronization therapy, it is mentioned that CMR and other imaging techniques were evaluated. However, the real value of these novel technologies remains to be determined in randomized trials (page 23). Furthermore, general safety-based recommendations for MR imaging in patients with implanted cardiac devices are given, according to conventional or MR-conditional devices (page 44).
2013 ESH/ESC guidelines for the management of arterial hypertension [22]
When searching for asymptomatic organ damage in patients with arterial hypertension, CMR should be considered for assessment of LV size and mass when echocardiography is technically not feasible and when LGE imaging would have therapeutic consequences (page 17). On a scale from “+” to “++++”, CMR was graded with “++” regarding cardiovascular predictive value, “+” regarding availability, “+++” regarding reproducibility” and “++” regarding cost-effectiveness (page 20). CMR is rated as highly sensitive to detect changes of LV hypertrophy, superior to echocardiography and ECG (page 51). Concerning renal artery stenosis as cause of secondary hypertension, CMR is named as additional/confirmatory test after renal ultrasonography (page 22). The recommendation for CMR in suspected ischemic heart disease in the context of hypertension-induced organ damage is given in Table 13.
2012 guidelines on the management of valvular heart disease [23]
In patients with inadequate echocardiographic quality or discrepant results, CMR should be used to assess the severity of valvular lesions - particularly regurgitant lesions - and to assess ventricular volumes and systolic function, as CMR assesses these parameters with higher reproducibility than echocardiography. CMR is the reference method for the evaluation of right ventricular volumes and function and is therefore useful to evaluate the consequences of tricuspid regurgitation. In practice, the routine use of CMR is limited because of its limited availability, compared with echocardiography (page 7).
In aortic regurgitation, CMR (or CT) is recommended for the evaluation of the aorta in patients with Marfan syndrome, or if an enlarged aorta is detected by echocardiography, particularly in patients with bicuspid aortic valves (page 10). Furthermore, CT or preferably CMR are advisable when the distal ascending aorta is not well visualized and/or when the surgical indication may be based on aortic enlargement, rather than LV size or function.
In aortic stenosis with paradoxical low flow, the diagnosis of severe AS requires careful exclusion of diverse reasons for the echo constellation before making the decision to intervene. In addition to more detailed echocardiographic measurements, this may require CMR and catheterization (page 14). (CT and) CMR provide additional information on the assessment of the ascending aorta when it is enlarged (page 14). Furthermore, CMR may also be useful for the detection and quantification of myocardial fibrosis, providing additional prognostic information in symptomatic patients without coronary artery disease (page 14).
In secondary mitral regurgitation and low LVEF, it is also mandatory to assess the absence, or presence and extent, of myocardial viability by one of the available imaging techniques (dobutamine echocardiography, SPECT, PET or CMR) (page 23). There are no specific recommendations for CMR in this guideline.
2012 focused update of the ESC Guidelines for the management of atrial fibrillation [24]
CMR is not mentioned in this guideline.
2012 third universal definition of myocardial infarction [25]
Imaging evidence of new loss of viable myocardium or new regional wall motion abnormality is listed among the criteria for acute myocardial infarction. Among the criteria for prior myocardial infarction, imaging evidence of a region of loss of viable myocardium that is thinned and fails to contract, in the absence of a non-ischaemic cause, is listed (page 3). CMR is mentioned next to other imaging tests (page 9) for assessing myocardial viability, perfusion, and function. Furthermore, its value in detecting myocardial disease states that can mimic myocardial infarct, such as myocarditis, is emphasized (page 10). There are no specific recommendations for CMR in this guideline.
2012 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation [26]
Contrast-enhanced CMR is mentioned as one of several techniques to make the diagnosis of no-reflow. If, in spite of the angiography performed in the acute phase, there are concerns about inducible ischaemia, an outpatient exercise-testing or stress-imaging test (using scintigraphy, echocardiography or CMR) is appropriate. Regarding the assessment of viability, the same statement as within the revascularization guidelines from 2014 is given: CMR has a high diagnostic accuracy for assessing transmural extent of myocardial scar tissue, but its ability to detect viability and predict recovery of wall motion is not superior to other imaging techniques (page 27). Table 14 shows the recommendations for CMR in patients with STEMI.
2012 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure [27]
CMR is particularly valuable in identifying inflammatory and infiltrative conditions as well as in the work-up of patients with suspected cardiomyopathy, arrhythmias, suspected cardiac tumours, or pericardial diseases, and is the imaging method of choice in patients with complex congenital heart disease (pages 16–17). Table 7 of the guideline summarizes possible applications of various imaging techniques in the diagnosis of heart failure. Thereby, the value of CMR is rated - on a scale from “+” to “+++” - with “+++” regarding coronary artery disease, myocarditis, sarcoidosis, amyloidosis, eosinophilic syndromes, iron overload, arrhythmogenic right ventricular cardiomyopathy, endomyocardial fibrosis; with “++” regarding valvular regurgitation, HCM, pericarditis, Takotsubo-cardiomyopathy, and with “+” regarding valvular stenosis and Anderson-Fabry-Disease. In patients presenting with heart failure and ECG signs of LV hypertrophy or low QRS voltage, CMR is recommended for further work-up. Table 15 provides recommendations for CMR in ambulatory patients suspected of having heart failure.
2012 European guidelines on cardiovascular disease prevention in clinical practice [28]
This guideline dedicates a chapter to the early detection of cardiovascular disease in asymptomatic subjects by CMR. It concludes that at present, CMR is a promising research tool, but its routine use remains limited and it is not yet appropriate for identifying patients at high risk for cardiovascular disease (page 23).
2011 ESC/EAS guidelines for the management of dyslipidaemias [29]
This guideline does not contain relevant paragraphs regarding CMR.
2011 ESC guidelines on the management of cardiovascular diseases during pregnancy [30]
CMR may be useful in diagnosing complex heart disease or pathology of the aorta. Limited data during organogenesis are available, but CMR is probably safe, especially after the first trimester. Gadolinium can be assumed to cross the fetal blood-placental barrier, but data are limited. The long-term risks of exposure of the developing fetus to free gadolinium ions are not known, and therefore gadolinium should be avoided (page 8). In bicuspid aortic valve disease, dilatation is often maximal in the distal part of the ascending aorta, which cannot be adequately visualized echocardiographically; therefore, CMR or CT should be performed before pre-pregnancy (page 21). Table 16 summarizes recommendations for CMR during pregnancy.
2011 ESC guidelines on the diagnosis and treatment of peripheral artery diseases [31]
MR angiography (MRA) is regarded as one of the main diagnostic modalities to assess peripheral artery disease. Table 17 summarizes the recommendations for MRA to assess peripheral artery disease.
2010 ESC guidelines for the management of grown-up congenital heart disease [32]
CMR has become increasingly important in grown-up with congenital heart disease (GUCH) and is an essential facility in the specialist unit. ESC recommendations for the use of CMR in GUCH patients have been published separately [33]. There are several groups of indications for CMR when assessing adult congenital heart disease in clinical practice:
-
1.
CMR as an alternative to echocardiography, when both techniques can provide similar information but echocardiography cannot be obtained with sufficient quality.
-
2.
CMR as a second method when echocardiography measurements are borderline or ambiguous.
-
3.
Indications where CMR is considered superior to echocardiography and should be regularly used when the information is essential for patient management. These indications include the quantification of right ventricular (RV) volumes and ejection fraction, RV and LV mass, evaluation of RV outflow tract and conduits, quantification of pulmonary regurgitation, evaluation of pulmonary arteries, aorta, systemic and pulmonary veins, collaterals and arteriovenous malformations, coronary anomalies and coronary artery disease, evaluation of intra- and extracardiac masses, and myocardial tissue characterization (fibrosis, fat, iron).
2010 focused update of ESC Guidelines on device therapy in heart failure [34]
CMR is not mentioned in this guideline.
2009 guidelines for the diagnosis and management of syncope [35]
In the diagnostic work-up of syncope, CMR - along with other imaging modalities - may be performed in selected cases (e.g. aortic dissection and haematoma, pulmonary embolism, cardiac masses, pericardial and myocardial diseases, congenital anomalies of coronary arteries) (page 23).
Discussion
This is the first systematic summary of the representation of CMR in the ESC guidelines. It shows that CMR is mentioned in the majority of guidelines (89 %) and that more than 50 % of the guidelines contain specific recommendations, when and how to use CMR. Almost all recommendations are in favour of the use of CMR.
The majority of recommendations refer to stress imaging to assess coronary artery disease in general. Even though CMR is not listed as the only recommended modality, it is ranked equally to nuclear studies and stress-echocardiography. Recently, large and important studies like CE-Marc have promoted this favourable position of CMR [4]. Accordingly, the evaluation of suspected coronary artery disease or ischemia in known coronary artery disease makes up the largest indication group for CMR in the EuroCMR registry [2].
Interestingly, in the context of ischemic heart disease, the ESC guidelines are relatively conservative in the evaluation of CMR viability testing. They rate its ability to detect viability and predict recovery of wall motion no better than with other imaging techniques and do not word a specific recommendation [16]. By way of contrast, viability testing makes up the third largest indication group in the EuroCMR registry [2].
The use of CMR in HCM is also well represented in the corresponding guideline. Thereby, CMR is mainly recommended to describe the phenotype and make the diagnosis, while its value for risk stratification for sudden cardiac death is still under debate [36]. Other cardiomyopathies (e.g. DCM, ARVC) and myocarditis are less well expressed in specific ESC recommendations. This can be attributed to the lack of large-scale data, as well as the absence of specific ESC-guidelines dedicated to cardiomyopathies (other than HCM) or inflammatory heart disease. The significance of CMR in these indication groups is underlined by several ESC position statements: A recent document about myocarditis stated that CMR may be considered in clinically stable patients with myocarditis [37]. A recent document about cardiomyopathies stated that the incremental contribution of CMR to the diagnosis of cardiomyopathies derives from accurate assessment of the morphology and function of the heart and tissue characterization [38]. Finally, a document about the role of endomyocardial biopsy in the management of cardiovascular disease mentions CMR repeatedly as a valuable tool in patients scheduled for biopsy either to assist or to replace biopsy [39]. In those centers taking part in the EuroCMR registry, cardiomyopathies and myocarditis make up the second largest CMR indication group [2].
Other well-established indications for CMR are completely unmentioned in the ESC guidelines, like CMR in the context of sarcoid disease. A recent consensus statement by the Heart Rhythm Society from 2014 on the diagnosis and management of arrhythmias associated with cardiac sarcoid defined the presence of LGE on CMR as one criteria for the diagnosis of cardiac sarcoid [40]. Screening for cardiac involvement in patients with biopsy-proven extracardiac sarcoidosis should include advanced cardiac imaging like CMR under certain circumstances. Planning the ablation procedure based on the predominant location of scarring as detected by LGE-CMR may be helpful and CMR may support sudden death risk stratification. Nevertheless, for the present analysis we decided to stick only to the ESC guidelines to warrant a consistent level of guideline standard.
Regarding valvular and congenital heart disease, the ESC guidelines contain extensive text passages about the value of CMR, reflecting current practice, where these indications make up a substantial part of all examinations [2]. In future guideline versions, the translation of these paragraphs into specific recommendations is needed to clarify the position of CMR.
This study touches several aspects: First, the frequent representation of CMR in the ESC guidelines demonstrates that the cardiology society has accepted CMR as an integral part of the armamentarium of cardiovascular diagnostic modalities (e.g. stress testing). As a next step, studies are needed that analyse the adherence to the ESC guidelines and how it impacts patients’ management [41]. Second, there are several clinical scenarios, where CMR is already used at dedicated centres, but which are not well represented in the ESC guidelines (e.g. myocarditis). Here, further studies are needed to provide the required evidence. Third, CMR has not yet arrived in the clinical reality in many regions of Europe. Hence, not everywhere in Europe can the patients be managed according to the ESC guidelines. The reasons are certainly multifactorial, with economic issues playing a central role: i) in some diseases alternative techniques are often readily available that provide similar information as CMR does. This is especially true for testing for myocardial ischemia, where SPECT and stress echocardiography are still the dominant modalities. ii) CMR is recognized as expensive and reimbursement not aspired by the medical insurances in many countries. iii) knowledge both to run a CMR examination and to interpret the images with profound cardiologic knowledge is often limited and structures for systematic training are needed, including the establishment of cooperation between radiologists and cardiologists.
Already now, there are attempts how to overcome the latter obstacles and to enable the use of CMR in accordance with the guidelines: Recent large-scale studies demonstrated the diagnostic accuracy of CMR and its superiority in some indications [4]; prognostic data are available that demonstrate the benefit of CMR [42]; there are studies that demonstrate the potential for saving resources by using CMR [43]; structures for acquiring CMR skills including e-based learning are evolving [44]; and CMR imaging became faster and the user interfaces easier to handle.
Limitations of the study
This summary is not intended to provide a balanced comparison of the various imaging modalities in the ESC guidelines, but aimed at describing only the role of CMR.
Conclusions
CMR is represented in the majority of the ESC guidelines. They contain many recommendations in favour of the use of CMR in specific scenarios. Issues regarding training, costs and reimbursement have to be solved to provide CMR to the patients in accordance with the ESC recommendations.
Abbreviations
- CMR:
-
cardiovascular magnetic resonance
- CT:
-
computed tomography
- ECG:
-
electrocardiography
- ESC:
-
European society for cardiology
- LAD:
-
left anterior descending coronary artery
- LGE:
-
late gadolinium enhancement
- LV:
-
left ventricle
- MRA:
-
magnetic resonance angiography
- PET:
-
positron emission tomography
- RV:
-
right ventricle
- SPECT:
-
single photon emission computed tomography
- STEMI:
-
ST elevation myocardial infarct
- TTE:
-
transthoracic echocardiography
References
von Knobelsdorff-Brenkenhoff F, Bublak A, El-Mahmoud S, Wassmuth R, Opitz C, Schulz-Menger J. Single-centre survey of the application of cardiovascular magnetic resonance in clinical routine. Eur Heart J Cardiovasc Imaging. 2013;14:62–8.
Bruder O, Wagner A, Lombardi M, Schwitter J, van Rossum A, Pilz G, et al. European Cardiovascular Magnetic Resonance (EuroCMR) registry--multi national results from 57 centers in 15 countries. J Cardiovasc Magn Reson. 2013;15:9.
Friedrich MG. There is more than shape and function. J Am Coll Cardiol. 2008;52:1581–3.
Greenwood JP, Maredia N, Younger JF, Brown JM, Nixon J, Everett CC, et al. Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): a prospective trial. Lancet. 2012;379:453–60.
Kramer CM, Barkhausen J, Flamm SD, Kim RJ, Nagel E. Society for Cardiovascular Magnetic Resonance Board of Trustees Task Force on Standardized P. Standardized cardiovascular magnetic resonance (CMR) protocols 2013 update. J Cardiovasc Magn Reson. 2013;15:91.
Authors/Task Force M, Priori SG, Blomstrom-Lundqvist C, Mazzanti A, Blom N, Borggrefe M et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: The Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC) Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J. 2015. pii: ehv316. [Epub ahead of print]
Authors/Task Force M, Galie N, Humbert M, Vachiery JL, Gibbs S, Lang I, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS) Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J. 2016;37:67–119.
Galie N, Hoeper MM, Humbert M, Torbicki A, Vachiery JL, Barbera JA, et al. Guidelines for the diagnosis and treatment of pulmonary hypertension: the Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS), endorsed by the International Society of Heart and Lung Transplantation (ISHLT). Eur Heart J. 2009;30:2493–537.
Authors/Task Force M, Roffi M, Patrono C, Collet JP, Mueller C, Valgimigli M et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur Heart J. 2015 pii: ehv320. [Epub ahead of print].
Hamm CW, Bassand JP, Agewall S, Bax J, Boersma E, Bueno H, et al. ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: The Task Force for the management of acute coronary syndromes (ACS) in patients presenting without persistent ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2011;32:2999–3054.
Authors/Task Force M, Adler Y, Charron P, Imazio M, Badano L, Baron-Esquivias G, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2015;36:2921–64.
Authors/Task Force M, Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta JP, et al. 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC)Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015;36:3075–128.
Habib G, Hoen B, Tornos P, Thuny F, Prendergast B, Vilacosta I, et al. Guidelines on the prevention, diagnosis, and treatment of infective endocarditis (new version 2009): the Task Force on the Prevention, Diagnosis, and Treatment of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and the International Society of Chemotherapy (ISC) for Infection and Cancer. Eur Heart J. 2009;30:2369–413.
Authors/Task Force M, Elliott PM, Anastasakis A, Borger MA, Borggrefe M, Cecchi F, et al. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: the Task Force for the Diagnosis and Management of Hypertrophic Cardiomyopathy of the European Society of Cardiology (ESC). Eur Heart J. 2014;35:2733–79.
Erbel R, Aboyans V, Boileau C, Bossone E, Bartolomeo RD, Eggebrecht H, et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2014;35:2873–926.
Authors/Task Force m, Windecker S, Kolh P, Alfonso F, Collet JP, Cremer J, et al. 2014 ESC/EACTS Guidelines on myocardial revascularization: The Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS) Developed with the special contribution of the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J. 2014;35:2541–619.
Konstantinides SV, Torbicki A, Agnelli G, Danchin N, Fitzmaurice D, Galie N, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2014;35:3033–69. 69a-69k.
Kristensen SD, Knuuti J, Saraste A, Anker S, Botker HE, Hert SD, et al. 2014 ESC/ESA Guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Eur Heart J. 2014;35:2383–431.
Authors/Task Force M, Ryden L, Grant PJ, Anker SD, Berne C, Cosentino F, et al. ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: the Task Force on diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and developed in collaboration with the European Association for the Study of Diabetes (EASD). Eur Heart J. 2013;34:3035–87.
Task Force M, Montalescot G, Sechtem U, Achenbach S, Andreotti F, Arden C, et al. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013;34:2949–3003.
Brignole M, Auricchio A, Baron-Esquivias G, Bordachar P, Boriani G, Breithardt OA, et al. 2013 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy: the Task Force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Eur Heart J. 2013;34:2281–329.
Mancia G, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Bohm M, et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J. 2013;34:2159–219.
Joint Task Force on the Management of Valvular Heart Disease of the European Society of C, European Association for Cardio-Thoracic S, Vahanian A, Alfieri O, Andreotti F, Antunes MJ, et al. Guidelines on the management of valvular heart disease (version 2012). Eur Heart J. 2012;33:2451–96.
Camm AJ, Lip GY, De Caterina R, Savelieva I, Atar D, Hohnloser SH, et al. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation. Developed with the special contribution of the European Heart Rhythm Association. Eur Heart J. 2012;33:2719–47.
Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD, et al. Third universal definition of myocardial infarction. Eur Heart J. 2012;33:2551–67.
Task Force on the management of STseamiotESoC, Steg PG, James SK, Atar D, Badano LP, Blomstrom-Lundqvist C, et al. ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2012;33:2569–619.
McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Bohm M, Dickstein K, et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2012;33:1787–847.
Perk J, De Backer G, Gohlke H, Graham I, Reiner Z, Verschuren M, et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur Heart J. 2012;33:1635–701.
European Association for Cardiovascular P, Rehabilitation, Reiner Z, Catapano AL, De Backer G, Graham I, et al. ESC/EAS Guidelines for the management of dyslipidaemias: the Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Eur Heart J. 2011;32:1769–818.
European Society of G, Association for European Paediatric C, German Society for Gender M, Regitz-Zagrosek V, Blomstrom Lundqvist C, Borghi C, et al. ESC Guidelines on the management of cardiovascular diseases during pregnancy: the Task Force on the Management of Cardiovascular Diseases during Pregnancy of the European Society of Cardiology (ESC). Eur Heart J. 2011;32:3147–97.
European Stroke O, Tendera M, Aboyans V, Bartelink ML, Baumgartner I, Clement D, et al. ESC Guidelines on the diagnosis and treatment of peripheral artery diseases: Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries: the Task Force on the Diagnosis and Treatment of Peripheral Artery Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2011;32:2851–906.
Baumgartner H, Bonhoeffer P, De Groot NM, de Haan F, Deanfield JE, Galie N, et al. ESC Guidelines for the management of grown-up congenital heart disease (new version 2010). Eur Heart J. 2010;31:2915–57.
Kilner PJ, Geva T, Kaemmerer H, Trindade PT, Schwitter J, Webb GD. Recommendations for cardiovascular magnetic resonance in adults with congenital heart disease from the respective working groups of the European Society of Cardiology. Eur Heart J. 2010;31:794–805.
Dickstein K, Vardas PE, Auricchio A, Daubert JC, Linde C, McMurray J, et al. 2010 Focused Update of ESC Guidelines on device therapy in heart failure: an update of the 2008 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure and the 2007 ESC guidelines for cardiac and resynchronization therapy. Developed with the special contribution of the Heart Failure Association and the European Heart Rhythm Association. Eur Heart J. 2010;31:2677–87.
Task Force for the D, Management of S, European Society of C, European Heart Rhythm A, Heart Failure A, Heart Rhythm S et al. Guidelines for the diagnosis and management of syncope (version 2009). Eur Heart J. 2009;30:2631–71.
Nicholls M. The 2014 ESC Guidelines on the Diagnosis and Management of Hypertrophic Cardiomyopathy have been published. Eur Heart J. 2014;35:2849–50.
Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34:2636–48. 48a-48d.
Rapezzi C, Arbustini E, Caforio AL, Charron P, Gimeno-Blanes J, Helio T, et al. Diagnostic work-up in cardiomyopathies: bridging the gap between clinical phenotypes and final diagnosis. A position statement from the ESC Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34:1448–58.
Cooper LT, Baughman KL, Feldman AM, Frustaci A, Jessup M, Kuhl U, et al. The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology Endorsed by the Heart Failure Society of America and the Heart Failure Association of the European Society of Cardiology. Eur Heart J. 2007;28:3076–93.
Birnie DH, Sauer WH, Bogun F, Cooper JM, Culver DA, Duvernoy CS, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014;11:1305–23.
Maggioni AP, Anker SD, Dahlstrom U, Filippatos G, Ponikowski P, Zannad F, et al. Are hospitalized or ambulatory patients with heart failure treated in accordance with European Society of Cardiology guidelines? Evidence from 12,440 patients of the ESC Heart Failure Long-Term Registry. Eur J Heart Fail. 2013;15:1173–84.
Anderson LJ, Holden S, Davis B, Prescott E, Charrier CC, Bunce NH, et al. Cardiovascular T2-star (T2*) magnetic resonance for the early diagnosis of myocardial iron overload. Eur Heart J. 2001;22:2171–9.
Pilz G, Patel PA, Fell U, Ladapo JA, Rizzo JA, Fang H, et al. Adenosine-stress cardiac magnetic resonance imaging in suspected coronary artery disease: a net cost analysis and reimbursement implications. Int J Cardiovasc Imaging. 2010;27:113–21.
Muehlberg F, Neumann D, von Knobelsdorff-Brenkenhoff F, Traber J, Alwardt N, Schulz-Menger J. A Multicenter Cardiovascular MR Network for Tele-Training and Beyond: Setup and Initial Experiences. J Am Coll Radiol. 2015;12:876–83.
Acknowledgements
We thank Kai Philipp Hasemann for assisting during the analysis of the guideline fulltextes.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
FvKB was responsible for conception and design, acquisition of data, analysis and interpretation of data and drafted the manuscript. JSM revised the manuscript critically for important intellectual content and has given final approval of the version to be published. Both authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors read and approved the final manuscript.
Appendix: Summary of clinical scenarios/diagnoses, where the ESC made recommendations regarding CMR
Appendix: Summary of clinical scenarios/diagnoses, where the ESC made recommendations regarding CMR
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
von Knobelsdorff-Brenkenhoff, F., Schulz-Menger, J. Role of cardiovascular magnetic resonance in the guidelines of the European Society of Cardiology. J Cardiovasc Magn Reson 18, 6 (2015). https://doi.org/10.1186/s12968-016-0225-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12968-016-0225-6