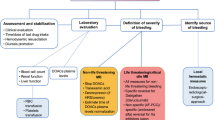
Abstract
Background
Patients with spontaneous intracerebral hemorrhage (ICH) have a higher risk of venous thromboembolism (VTE) and in-hospital VTE is independently associated with poor outcomes for this patient population.
Methods
A comprehensive literature search about patients with VTE after spontaneous ICH was conducted using databases MEDLINE and PubMed. We searched for the following terms and other related terms (in US and UK spelling) to identify relevant studies: intracerebral hemorrhage, ICH, intraparenchymal hemorrhage, IPH, venous thromboembolism, VTE, deep vein thrombosis, DVT, pulmonary embolism, and PE. The search was restricted to human subjects and limited to articles published in English. Abstracts were screened and data from potentially relevant articles was analyzed.
Results
The prophylaxis and treatment of VTE are of vital importance for patients with spontaneous ICH. Prophylaxis measures can be mainly categorized into mechanical prophylaxis and chemoprophylaxis. Treatment strategies include anticoagulation, vena cava filter, systemic thrombolytic therapy, catheter-based thrombus removal, and surgical embolectomy. We briefly summarized the state of knowledge regarding the prophylaxis measures and treatment strategies of VTE after spontaneous ICH in this review, especially on chemoprophylaxis and anticoagulation therapy. Early mechanical prophylaxis, especially with intermittent pneumatic compression, is recommended by recent guidelines for patients with spontaneous ICH. While decision-making on chemoprophylaxis and anticoagulation therapy evokes debate among clinicians, because of the concern that anticoagulants may increase the risk of recurrent ICH and hematoma expansion. Uncertainty still exists regarding optimal anticoagulants, the timing of initiation, and dosage.
Conclusion
Based on current evidence, we deem that initiating chemoprophylaxis with UFH/LMWH within 24–48 h of ICH onset could be safe; anticoagulation therapy should depend on individual clinical condition; the role of NOACs in this patient population could be promising.
Similar content being viewed by others
Introduction
Intracerebral hemorrhage (ICH) accounts for approximately 10–30% of all strokes, but is associated with over-proportionally high mortality and enormous health-care costs [1,2,3]. The most prevalent subtype of ICH is spontaneous ICH, which is principally caused by cerebral small vessel diseases; vascular malformations, tumors, anticoagulants, antiplatelet medication, and vasculitis are among the other reasons [4]. Venous thromboembolism (VTE), including deep venous thrombosis (DVT) and pulmonary embolism (PE), is a common complication in hospitalized patients and is associated with substantial morbidity, mortality, and health-care cost [5]. Patients with ICH have a higher risk of VTE, which can reach 2–4 times that of patients with acute ischemic stroke [6, 7]. In some large database retrospective studies, the incidence of VTE, symptomatic DVT, and clinically evident PE in hospitalized patients with spontaneous ICH has been estimated to be 2–4%, 1–2%, and 0.7–2%, respectively [6,7,8,9,10]. In two prospective studies, the incidence of DVT detected by ultrasonography during hospitalization reached 20–40% [11, 12].
VTE is one of the most common and preventable complications of spontaneous ICH. However, the treatment options of the two are full of contradictions. The focus of VTE treatment is anticoagulation and prevention of recurrent thrombosis, while the treatment of ICH is focused on hemostasis and averting hematoma expansion. For such patients, how should clinicians balance the treatment need of the two conditions and make appropriate treatment strategies is a question worthy of discussion.
In this review, we will briefly summarize the state of knowledge regarding the risk factors, prophylaxis measures, and treatment strategies of VTE after spontaneous ICH.
Risk factors
Virchow’s triad
Virchow’s triad describes the three key predisposing factors to VTE: abnormal flow, vessel wall abnormalities (endothelial injury), and coagulation state [13]. In patients with spontaneous ICH, there is venous stasis due to immobility and hemiplegia [14, 15], endothelial injury caused by invasive operations, and hypercoagulability as a result of dehydrating, hemostatic and antifibrinolytic agents [16].
Other factors
A large number of studies on the risk factors of VTE in patients with spontaneous ICH were conducted; nevertheless, no independent study on this aspect of PE has been performed. Advanced age, immobility, hemiplegia, high D-Dimer and CRP value, high NIHSS score, and previous VTE history as risk factors of VTE in patients with spontaneous ICH have basically reached a consensus [8, 9, 12, 17].
Melmed et al. [18] conducted a study on 414 patients with ICH to clarify the relationship between infection and VTE. They found that respiratory and bloodstream infections were associated with VTE after primary ICH, while urinary and other infections were not.
Whether gender is a relevant risk factor remains controversial. Retrospective studies published by Kim et al. [8] and Ding et al. [9], and the prospective cohort study published by Ogata et al. [12] all pointed out that gender was not a risk factor of VTE. While the prospective cohort studies published by Kawase et al. [11] for the Japanese population and Cheng et al. [17] for the Chinese population indicated that female sex was a risk factor. The discrepancy in the results could be due to differences in design, sample size and population.
Prophylaxis measures
Patients with spontaneous intracerebral hemorrhage are predisposed to VTE and in-hospital VTE is independently associated with poor outcomes at discharge, 3-month, and 1-year [19]. One study demonstrated that without prophylaxis, up to 75% of patients with residual hemiplegia following ICH developed DVT, and PE-related deaths occurred in approximately 5% of patients with ICH [20]. As a consequence, the prophylaxis of VTE is of vital importance. Prophylaxis measures can be categorized into active limb movement, mechanical prophylaxis, and chemoprophylaxis. Active limb movement, such as ankle pump exercise (APE) which rhythmically contracts and relaxes the calf muscles through the ankle joint movement, and squeezes the venous plexus to promote the venous blood return in lower limbs, is efficient and cost-effective in eliminating venous stasis to contribute to the prophylaxis of DVT [21,22,23]. We mainly focus on mechanical and chemical prophylaxis in this review.
Mechanical prophylaxis
For some hospitalized medical patients at high risk of thrombosis (e.g. patients with hemorrhagic stroke [7], lower extremity fractures [24], nephrotic syndrome [25], etc.) who are bleeding or at high risk of major bleeding, the guideline from American College of Chest Physicians (ACCP) has suggested the optimal use of mechanical prophylaxis with graduated compression stockings (GCS) or intermittent pneumatic compression (IPC), rather than no mechanical prophylaxis [26].
GCS and IPC
For the prophylaxis of VTE, guidelines published in recent years recommended the use of IPC and/or GCS (Table 1). The Clots in Legs Or sTockings after Stroke (CLOTS) trial 1, a multicenter and randomized controlled trial in stroke patients who were enrolled between 2001 and 2008 (n = 2518, 232 with ICH), demonstrated that GCS alone was ineffective in preventing DVT (10.0% vs. 10.5%, OR 0.98, 95%CI 0.76–1.27) and caused more adverse effects, including skin breaks, ulcers, blisters, and necrosis of lower extremities (5.0% vs. 1.0%, OR 4.18, 95%CI 2.40–7.27) [32]. The CLOTS trial 3, which was also a multicenter and randomized controlled trial conducted between 2008 to 2012 in patients who were immobile after stroke (n = 2876, 376 with ICH) to assess the effectiveness of IPC in preventing DVT, addressed that the routine application of IPC in patients with ICH was associated with a significantly decreased risk of DVT (6.7% vs. 17.0%, OR 0.36, 95%CI 0.17–0.75) [33]. Therefore, recent guidelines have tended to recommend IPC alone.
Timing of initiation
Regarding the timing of intervention for mechanical prophylaxis in patients with spontaneous ICH, the recommendations of the current guidelines (Table 1) have tended to converge: start at the time of hospital admission and use as early as possible. In 2018, the guideline from NICE added recommendations that mechanical prophylaxis should be initiated within 3 days of ICH [30].
Chemoprophylaxis
The use of chemoprophylaxis with unfractionated heparin (UFH), low molecular weight heparin (LMWH), and direct oral anticoagulants (e.g. warfarin, rivaroxaban, apixaban, dabigatran, etc.) in patients with spontaneous ICH is an area of ongoing debate. Determining appropriate chemoprophylaxis is challenging in the presence of ICH. The concern of hematoma expansion and recurrent ICH limits the use of chemoprophylaxis in clinical practice. A large database retrospective study including 32,690 patients with spontaneous ICH from 2006 to 2010 demonstrated that 5395 patients (16.5%) received chemoprophylaxis [34]. Another multicenter observational cohort study demonstrated that among 74,283 patients with ICH, only 5929 received chemoprophylaxis while 66,444 received mechanical prophylaxis (7.9% vs. 89.4%) [35].
Timing of initiation
To date, the optimal timing for the initiation of chemoprophylaxis is still uncertain. A large database retrospective study demonstrated 44.8% (2416/5395) of patients with spontaneous ICH received prophylactic anticoagulation with UFH, enoxaparin, or dalteparin by day 2 of onset [34]. Hematoma expansion usually occurs in the early phase of ICH. Results from imaging studies have demonstrated that 70% of patients with spontaneous ICH have hematoma expansion within 24 h of onset and expansion after 24 h seems extremely rare [36, 37]. In contrast, patients with ICH develop DVT as early as the second day without prophylaxis, with the peak incidence occurring between days 2 and 7 [38]. In addition, anticoagulation is associated with recurrent ICH in patients with active bleeding, while no such association has been found in patients who have documentation of hematoma stabilization on neuroimaging [39]. Therefore, we speculate that initiating chemoprophylaxis within 24–48 h of onset, under the circumstance of no signs of hematoma expansion or active bleeding on neuroimaging, could be safe and effective. A number of studies (Table 2) demonstrated that initiating chemoprophylaxis with UFH/LMWH within 24–48 h was safe for being not associated with hematoma expansion or recurrent ICH, but differed in the efficiency. Initiating chemoprophylaxis within 24–48 h seems not to be more effective in reducing the risk of VTE than other timing. Recommendations in relevant guidelines vary as well (Table 3). Hence, more high-quality studies are needed on the timing of initiating chemoprophylaxis among patients with spontaneous ICH.
Anticoagulants
Anticoagulants mostly used in relevant studies are UFH or LMWH. A large database retrospective study on patients with spontaneous ICH from 2006 to 2010 (n = 32,690) showed that the most commonly used agents for VTE prophylaxis were heparin (71.1%), enoxaparin (27.5%), and dalteparin(1.4%) [34]. The efficacy and safety of most direct oral anticoagulants (DOACs) in preventing VTE in patients with spontaneous ICH have not been evaluated because they have been excluded from randomized trials on VTE chemoprophylaxis with DOACs. The majority of studies (Table 2) have demonstrated that UFH or LMWH is not associated with recurrent ICH, hematoma expansion, and increased mortality. A meta-analysis, including 9 studies and 4055 patients with spontaneous ICH, also showed that chemoprophylaxis with UFH/LMWH was not associated with a significant increase in hematoma expansion (6.6% vs. 3.2%, P = 0.14) or mortality (12.0% vs. 11.8%, P = 0.29) in comparison with non-heparin prophylaxis [48]. However, the efficacy of VTE chemoprophylaxis has not reached a consensus (Table 2).
Dosage
The appropriate anticoagulant dosage is still inconclusive. For VTE prophylaxis with UFH, the suggested dosage is 5000 units subcutaneously two or three times daily. No consensus has been established regarding the optimal frequency of dosing (two vs. three times daily). While with LMWH, Tetri et al. retrospectively evaluated the safety and efficacy of enoxaparin in VTE prophylaxis among 407 patients with spontaneous ICH and demonstrated that the appropriate dosage could be 2000 or 4000 IU once daily [49]. Wu et al. [50] also pointed out that subcutaneous administration of 4000 IU of enoxaparin once daily can prevent DVT in patients with ICH without hematoma expansion or recurrent ICH.
In conclusion, controversies exist regarding the efficacy, optimal anticoagulants, timing, and dosage of VTE chemoprophylaxis for patients with spontaneous ICH. Recommendations vary among relevant guidelines (Table 3). We expect more high-quality studies to provide direction for clinical decision-making.
Treatment strategies
The treatment strategies for VTE include anticoagulation, vena cava filter, systemic thrombolytic therapy, catheter-based thrombus removal, and surgical embolectomy. Controversies and uncertainties remain in the treatment of VTE after spontaneous ICH, especially in the acute phase of ICH (usually within 2 weeks of onset) which has a high risk of hematoma expansion and recurrence [51]. We briefly summarize the evidence of VTE treatment after acute spontaneous ICH as follows.
Anticoagulation
Anticoagulation therapy is the cornerstone of VTE treatment. However, active bleeding is considered a contraindication to anticoagulation therapy. Decision-making on anticoagulation therapy after spontaneous ICH evokes debate among clinicians and the inadequacies in the evidence, for the reason that such patients have been excluded from randomized trials of anticoagulation therapy for VTE, makes the decision-making challenging. The three questions that need to be answered are as follows: first, whether anticoagulation therapy should be applied on such patients; second, when is the appropriate timing to initiate anticoagulation; and third, which anticoagulant is the optimal choice.
Anticoagulation or not
Limited data are available on the pros and cons of anticoagulation therapy for VTE following spontaneous ICH. Whether anticoagulation should be used depends on the individual risk-benefit ratio of anticoagulation therapy, that is, the risk of recurrent ICH and hematoma expansion versus the risk of VTE progression.
No reliable methods have been established to predict the risk of recurrent ICH and hematoma expansion. The pathophysiological mechanism of hematoma expansion remains unclear. A number of studies have demonstrated that previous use of anticoagulants or antiplatelet agents, advanced age, systolic hypertension, hyperglycemia, high NIHSS score or Glasgow Coma Scale score are related clinical risk factors for hematoma expansion [52,53,54,55,56,57,58,59,60,61]; CTA spot sign, blend sign, black hole sign, island sign, and iodine sign are the radiological risk factors [62,63,64,65,66,67,68]. The annualized rate of recurrent spontaneous ICH is approximately 2% [1, 69]. Spontaneous ICH principally results from small vessel diseases that are mainly composed of hypertensive arteriopathy and cerebral amyloid angiopathy (CAA). Hypertension has been identified as one of the most crucial and modifiable risk factors for recurrent ICH. Lowering blood pressure is associated with a reduced recurrence of ICH [70,71,72]. Amyloid angiopathy has a predilection for the cortical arteries, hence CAA is a major contributor of spontaneous lobar ICH, which has a significantly higher risk for recurrence compared to deep ICH [73,74,75]. In addition, a number of studies have demonstrated that older age, cerebral microbleeds (CMB) and cortical superficial siderosis (cSS) on MRI, and Apolipoprotein E (APOE) genotype (ε2 or ε4) are associated with a higher recurrence risk [76,77,78]. In conclusion, anticoagulation therapy should be more careful in patients with the risk factors mentioned above. Besides, the modifiable risk factors, such as hypertension and hyperglycemia, should be minimized if anticoagulants are to be used in this patient population.
Patients with spontaneous ICH have been excluded from prospective studies and randomized trials on anticoagulation therapy for VTE. The available literature is almost exclusively case reports. Ajmeri et al. [79] presented a case of a 68-year-old male who suffered from recent ICH and recurrent PE. The anticoagulation therapy was initiated with UFH after a CT scan was done to eliminate active bleeding, and then altered to enoxaparin. The patient was discharged from the hospital in a stable condition without neurological deficits. Similar cases were also reported by Becattini et al. [80] and Lee et al. [81]
A Danish large-database retrospective study (n = 2978) which was focusing on patients with spontaneous ICH, demonstrated that oral anticoagulant (OAC) resumption was associated with decreased risk of thrombotic events and not increasing the risk of recurrent ICH [82]. Current studies on anticoagulation therapy are mostly focused on the OAC resumption after anticoagulation-related ICH. In addition to VTE, the indications for anticoagulation resumption include atrial fibrillation, mechanical valves, myocardial infarction, etc. The majority of studies have tended to reach a consensus that anticoagulation resumption results in a clinical benefit in terms of thromboembolic event reduction without increasing the risk of recurrent ICH or hematoma expansion (Table 4). These results were confirmed by a meta-analysis (8 studies, 5306 patients with ICH) that demonstrated restarting OAC decreased the risk of thrombotic events (6.7% vs. 17.6%, RR 0.34, 95%CI 0.25–0.45) without significantly increased risk of recurrent ICH (8.7% vs. 7.8%, RR 1.01, 95%CI 0.58–1.77) [87]. What is noteworthy is that only the patients with smaller hemorrhage volumes and mild functional changes have been eligible for anticoagulation resumption in many studies, consequently decreasing the risk of recurrent ICH.
Nevertheless, some opposite results exist. A retrospective study with 79 patients who had brain tumors and following anticoagulation-related ICH showed that anticoagulation resumption with LMWH or DOACs was associated with a significantly lower risk of recurrent VTE (8.1% vs. 35.3%, P = 0.003) but a higher risk of recurrent ICH (6.1% vs. 4.2%), especially in patients with primary brain tumors and major ICH [88]. Different study designs and research objects may explain the opposite results. Several retrospective studies and meta-analysis studies have shown that anticoagulation is associated with an increased risk of ICH in patients with primary brain tumors, however, the association seems not to be found in patients with metastatic brain tumors [89,90,91]. Hence, anticoagulation resumption in patients with ICH and primary brain tumors must be cautious.
Timing of initiation
No consensus exists on optimal timing for initiation of anticoagulation therapy following spontaneous ICH. The timing ranged from 2.5 days to 18 weeks in retrospective studies on anticoagulation resumption after ICH [92]. No randomized trial data are available to guide the decision. Hence, the timing of initiating anticoagulation therapy should depend on the individual clinical conditions.
Anticoagulant choice
No comparative studies on different anticoagulants in patients with VTE after spontaneous ICH have been published. Existing studies mostly focus on the safety of anticoagulants and ICH is one of the crucial indicators. The relevant studies almost exclusively referred to vitamin-K antagonists used for anticoagulation resumption after ICH. However, the role of novel oral anticoagulants (NOACs), including dabigatran (inhibitor of factor IIa), rivaroxaban, apixaban, and edoxaban (inhibitors of factor Xa), may be more promising than vitamin-K antagonists for the reasons as follows: (1) NOACs are more convenient to use for lack of monitoring requirements and less interaction with food and other drugs; (2) a number of large-scale retrospective studies and randomized trials have proved that NOACs are associated with a reduction in the risk of ICH compared with vitamin K antagonists (Table 5); (3) NOACs-related ICH is less severe than warfarin-related ICH, with smaller hematoma volume, lower rate of hematoma expansion, favorable functional and vital outcomes, and lower mortality [98,99,100]; (4) the availability of antidotes (idarucizumab and andexanet) that allow an immediate and complete reversal of the anticoagulant effect of NOACs [101,102,103]. We expect high-quality and large-scale studies on patients with spontaneous ICH to provide support.
Inferior vena cava filter
The efficacy of inferior vena cava (IVC) filter in preventing formation or aggravation of PE caused by shedding of deep vein thrombosis of lower extremities has been confirmed in general patients. However, none of the studies have focused solely on patients with spontaneous ICH.
A retrospective study included 371 stroke patients (including 105 patients with hemorrhagic stroke) who received IVC filters and demonstrated that IVC filters were effective in preventing life-threatening PE; however, the filter-related complications, including filter migration, filter fracture, and filter thrombosis, were also worthy of attention [104]. Meritxell et al. [105] used the data of the RIETE registry to figure out the association between the use of IVC filters and the outcome of patients presenting with major bleeding during anticoagulation for VTE. Among 1065 patients (including 124 patients with intracranial hemorrhage) in this study, 11% received IVC filters; the result suggested that patients receiving IVC filters had a lower risk for all-cause death (HR 0.55, 95%CI 0.23–1.40) and a similar risk for PE recurrence (HR 1.57, 95%CI 0.38–6.36). However, the study by Kare et al. [106] on 1068 patients with acute brain injury (the proportion of ICH was not mentioned) who received IVC filters demonstrated that IVC filters were ineffective in preventing PE (HR 3.19, 95%CI 1.3–3.3) and reducing mortality (HR 1.0, 95%CI 0.8–1.3). Besides, once IVC filters were placed, few were removed. Jan et al. [107] also supported this result. Among 204 retrievable IVC filters inserted in neurosurgical patients in their study, only 19% were retrieved and 55% converted to permanent devices. We deem that the difference in results is partly due to the different patient groups included in each study. Therefore, whether the IVC filters are beneficial to patients with VTE after spontaneous ICH still needs targeted studies.
Other treatments
The application of systemic thrombolytic therapy, catheter-based thrombus removal, and surgical embolectomy in patients with VTE after spontaneous ICH is only seen in a few case reports and still needs further exploration.
Current guidelines have recommended the immediate administration of thrombolytic therapy in patients with massive PE and without contraindications [26]. Thrombolytic therapy is said to be contraindicated in the presence of ICH. However, some case reports revealed that thrombolysis could be safe and effective in patients with the two conditions. Wendy et al. [108] presented a case of a 60-year-old woman with massive PE (led to cardiac arrest with pulseless electrical activity) and a recent hemorrhagic cerebrovascular accident who was administered tissue plasminogen activator (t-PA). No hematoma expansion occurred, and the patient was eventually discharged from the hospital. In another report, systemic thrombolysis with recombinant tissue plasminogen activator (rt-PA) for massive PE was used with success in a 53-year-old male with acute spontaneous ICH [109]. Therefore, in patients who are at high risk of death from massive pulmonary embolism, contraindications to thrombolysis should be weighed against the potential benefit.
The ACCP guideline recommends catheter-based thrombus removal or surgical embolectomy in patients with acute PE who have hypotension and contraindications to thrombolysis if appropriate expertise and resources are available [26]. No studies or case reports have been reported in the application of catheter-based thrombus removal in patients with VTE after spontaneous ICH as yet. For surgical methods, Endo et al. [110] reported a 69-year-old presented with acute PE 11 days after ICH and underwent surgical embolectomy with cardiopulmonary bypass. No recurrence of ICH was observed in this patient.
Patients with preexisting VTE and ICH
Although this review mainly focuses on the prophylaxis and treatment of VTE after spontaneous ICH, another crucial and overlapping clinical scenario that happens in patients with preexisting VTE and following ICH is also noteworthy. Anticoagulation-related ICH is more common in this patient population due to the need for long-term anticoagulation therapy of VTE. Anticoagulation-related ICH is more severe and associated with a higher risk of hematoma expansion and higher mortality, compared to other spontaneous ICH [56, 85, 111]. Managements of anticoagulation-related ICH include halting anticoagulation and anticoagulants reversal, which has protamine sulfate, vitamin K, fresh frozen plasma (FFP), prothrombin complex concentrate (PCC), rFVIIa, idarucizumab, and andexanet as options for different anticoagulants [112]. In addition, anticoagulation resumption should also be considered in patients with preexisting VTE. Anticoagulation should be restarted when the risk of VTE progression outweighs recurrent ICH or hematoma expansion. However, as the description in Section 4.1, the optimal timing and anticoagulants remain unclear.
Conclusion
-
(1)
Early mechanical prophylaxis with IPC and/or GCS is recommended, having a preference for IPC. Controversies exist regarding the effectiveness, optimal anticoagulants, timing, and dosage of VTE chemoprophylaxis. Initiating chemoprophylaxis with UFH/LMWH within 24–48 h of spontaneous ICH onset could be safe.
-
(2)
Given the limited evidence and the observational nature of the studies regarding anticoagulation therapy, uncertainty exists regarding anticoagulation, timing, and anticoagulants. Anticoagulation treatment should depend on individual clinical condition. The role of NOACs in this patient population could be promising.
-
(3)
More targeted and high-quality studies on vena cava filter, systemic thrombolytic therapy, catheter-based thrombus removal, and surgical embolectomy are needed.
Availability of data and materials
Not applicable.
Abbreviations
- ACCP:
-
the American College of Chest Physicians
- AF:
-
Atrial fibrillation
- AHS/ASA:
-
the American Heart Association/American Stroke Association
- DOACs:
-
Direct oral anticoagulants
- DVT:
-
Deep venous thrombosis
- ESO:
-
European Stroke Organization
- GCS:
-
Graduated compression stockings
- HE:
-
Hematoma expansion
- HR:
-
Hazard ratio
- HSFC:
-
Heart and Stroke Foundation of Canada
- ICH:
-
Intracerebral hemorrhage
- IPC:
-
Intermittent pneumatic compression
- IVC:
-
Inferior vena cava
- LMWH:
-
Low molecular weight heparin
- MI:
-
Myocardial infarction
- MV:
-
Mechanical valves
- NCS:
-
The Neurocritical Care Society
- NICE:
-
National Institute for Health and Care Excellence
- OAC:
-
Oral anticoagulants
- PAD:
-
Peripheral vascular disease
- PE:
-
Pulmonary embolism
- UFH:
-
Unfractionated heparin
- VTE:
-
Venous thromboembolism
References
Poon MTC, Fonville AF, Al-Shahi SR. Long-term prognosis after intracerebral haemorrhage: systematic review and meta-analysis. J Neurol Neurosurg Psychiatry. 2014;85(6):660–7.
Feigin VL, Norrving B, Mensah GA. Global burden of stroke. Circ Res. 2017;120(3):439–48.
van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol. 2010;9(2):167–76.
Dastur CK, Yu W. Current management of spontaneous intracerebral haemorrhage. Stroke Vasc Neurol. 2017;2(1):21–9.
Di Nisio M, van Es N, Büller HR. Deep vein thrombosis and pulmonary embolism. Lancet. 2016;388(10063):3060–73.
Skaf E, Stein PD, Beemath A, Sanchez J, Bustamante MA, Olson RE. Venous thromboembolism in patients with ischemic and hemorrhagic stroke. Am J Cardiol. 2005;96(12):1731–3.
Gregory PC, Kuhlemeier KV. Prevalence of venous thromboembolism in acute hemorrhagic and thromboembolic stroke. Am J Phys Med Rehabil. 2003;82(5):364–9.
Kim KS, Brophy GM. Symptomatic venous thromboembolism: incidence and risk factors in patients with spontaneous or traumatic intracranial hemorrhage. Neurocrit Care. 2009;11(1):28–33.
Ding D, Sekar P, Moomaw CJ, Comeau ME, James ML, Testai F, et al. Venous thromboembolism in patients with spontaneous Intracerebral hemorrhage: a multicenter study. Neurosurgery. 2019;84(6):E304–E10.
Goldstein JN, Fazen LE, Wendell L, Chang Y, Rost NS, Snider R, et al. Risk of thromboembolism following acute intracerebral hemorrhage. Neurocrit Care. 2009;10(1):28–34.
Kawase K, Okazaki S, Toyoda K, Toratani N, Yoshimura S, Kawano H, et al. Sex difference in the prevalence of deep-vein thrombosis in Japanese patients with acute intracerebral hemorrhage. Cerebrovasc Dis. 2009;27(4):313–9.
Ogata T, Yasaka M, Wakugawa Y, Inoue T, Ibayashi S, Okada Y. Deep venous thrombosis after acute intracerebral hemorrhage. J Neurol Sci. 2008;272(1–2):83–6.
Malone PC, Agutter PS. The aetiology of deep venous thrombosis. QJM : monthly journal of the Association of Physicians. 2006;99(9):581–93.
Sartori M, Favaretto E, Cosmi B. Relevance of immobility as a risk factor for symptomatic proximal and isolated distal deep vein thrombosis in acutely ill medical inpatients. Vascular medicine (London, England). 2021;26(5):542–8.
Pottier P, Hardouin JB, Lejeune S, Jolliet P, Gillet B, Planchon B. Immobilization and the risk of venous thromboembolism. A meta-analysis on epidemiological studies. Thromb Res. 2009;124(4):468–76.
Kelly J, Hunt BJ, Lewis RR, Swaminathan R, Moody A, Seed PT, et al. Dehydration and venous thromboembolism after acute stroke. QJM. 2004;97(5):293–6.
Cheng X, Zhang L, Xie NC, Ma YQ, Lian YJ. High plasma levels of D-dimer are independently associated with a heightened risk of deep vein thrombosis in patients with Intracerebral hemorrhage. Mol Neurobiol. 2016;53(8):5671–8.
Melmed KR, Boehme A, Ironside N, Murthy S, Park S, Agarwal S, et al. Respiratory and blood stream infections are associated with subsequent venous thromboembolism after primary Intracerebral hemorrhage. Neurocrit Care. 2020;34(1):85–91.
Li J, Wang D, Wang W, Jia J, Kang K, Zhang J, et al. In-hospital venous thromboembolism is associated with poor outcome in patients with spontaneous intracerebral hemorrhage: a multicenter, prospective study. J Stroke Cerebrovasc Dis. 2020;29(8):104958.
Wijdicks EF, Scott JP. Pulmonary embolism associated with acute stroke. Mayo Clin Proc. 1997;72(4):297–300.
Sochart DH, Hardinge K. The relationship of foot and ankle movements to venous return in the lower limb. J Bone Joint Surg Br. 1999;81(4):700–4.
Li Y, Guan X-H, Wang R, Li B, Ning B, Su W, et al. Active ankle movements prevent formation of lower-extremity deep venous thrombosis after orthopedic surgery. Med Sci Monit. 2016;22:3169–76.
Palamone J, Brunovsky S, Groth M, Morris L, Kwasny M. "Tap and twist": preventing deep vein thrombosis in neuroscience patients through foot and ankle range-of-motion exercises. J Neurosci Nurs. 2011;43(6).
Whiting PS, Jahangir AA. Thromboembolic disease after orthopedic trauma. Orthop Clin North Am. 2016;47(2):335–44.
Singhal R, Brimble KS. Thromboembolic complications in the nephrotic syndrome: pathophysiology and clinical management. Thromb Res. 2006;118(3):397–407.
Guyatt GH, Akl EA, Crowther M, Gutterman DD, Schuunemann HJ. American College of Chest Physicians Antithrombotic T, et al. executive summary: antithrombotic therapy and prevention of thrombosis, 9th ed: American college of chest physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):7S–47S.
Steiner T, Al-Shahi Salman R, Beer R, Christensen H, Cordonnier C, Csiba L, et al. European stroke organisation (ESO) guidelines for the management of spontaneous intracerebral hemorrhage. International journal of stroke : official journal of the International Stroke Society. 2014;9(7):840–55.
Hemphill JC 3rd, Greenberg SM, Anderson CS, Becker K, Bendok BR, Cushman M, et al. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46(7):2032–60.
Nyquist P, Bautista C, Jichici D, Burns J, Chhangani S, DeFilippis M, et al. Prophylaxis of venous thrombosis in Neurocritical care patients: an evidence-based guideline: a statement for healthcare professionals from the Neurocritical care society. Neurocrit Care. 2015;24(1):47–60.
Dawoud D, Lewis S, Glen J, Sharpin C, Committee NG. Venous thromboembolism in over 16s: reducing the risk of hospital-acquired deep vein thrombosis or pulmonary embolism. NICE guideline [NG89]. 2018.
Shoamanesh Co-Chair A, Patrice Lindsay M, Castellucci LA, Cayley A, Crowther M, de Wit K, et al. Canadian stroke best practice recommendations: Management of Spontaneous Intracerebral Hemorrhage, 7th edition update 2020. Int J Stroke. 2021;16(3):321–41.
Dennis M, Sandercock PAG, Reid J, Graham C, Murray G, Venables G, et al. Effectiveness of thigh-length graduated compression stockings to reduce the risk of deep vein thrombosis after stroke (CLOTS trial 1): a multicentre, randomised controlled trial. Lancet. 2009;373(9679):1958–65.
Dennis M, Sandercock P, Reid J, Graham C, Forbes J, Murray G. Effectiveness of intermittent pneumatic compression in reduction of risk of deep vein thrombosis in patients who have had a stroke (CLOTS 3): a multicentre randomised controlled trial. Lancet. 2013;382(9891):516–24.
Prabhakaran S, Herbers P, Khoury J, Adeoye O, Khatri P, Ferioli S, et al. Is prophylactic anticoagulation for deep venous thrombosis common practice after intracerebral hemorrhage? Stroke. 2015;46(2):369–75.
Cherian LJ, Smith EE, Schwamm LH, Fonarow GC, Schulte PJ, Xian Y, et al. Current practice trends for use of early venous thromboembolism prophylaxis after Intracerebral hemorrhage. Neurosurgery. 2018;82(1):85–92.
Brott T, Broderick J, Kothari R, Barsan W, Tomsick T, Sauerbeck L, et al. Early hemorrhage growth in patients with intracerebral hemorrhage. Stroke. 1997;28(1):1–5.
Kazui S, Naritomi H, Yamamoto H, Sawada T, Yamaguchi T. Enlargement of spontaneous intracerebral hemorrhage. Incidence and time course. Stroke. 1996;27(10):1783–7.
Kelly J, Rudd A, Lewis R, Hunt BJ. Venous thromboembolism after acute stroke. Stroke. 2001;32(1):262–7.
Burchell SR, Tang J, Zhang JH. Hematoma expansion following Intracerebral hemorrhage: mechanisms targeting the coagulation Cascade and platelet activation. Curr Drug Targets. 2017;18(12):1329–44.
Boeer A, Voth E, Henze T, Prange HW. Early heparin therapy in patients with spontaneous intracerebral haemorrhage. J Neurol Neurosurg Psychiatry. 1991;54(5):466–7.
Wasay M, Khan S, Zaki KS, Khealani BA, Kamal A, Azam I, et al. A non-randomized study of safety and efficacy of heparin for DVT prophylaxis in intracerebral haemorrhage. J Pak Med Assoc. 2008;58(7):362–4.
Orken DN, Kenangil G, Ozkurt H, Guner C, Gundogdu L, Basak M, et al. Prevention of deep venous thrombosis and pulmonary embolism in patients with acute intracerebral hemorrhage. Neurologist. 2009;15(6):329–31.
Kiphuth IC, Staykov D, Köhrmann M, Struffert T, Richter G, Bardutzky J, et al. Early administration of low molecular weight heparin after spontaneous intracerebral hemorrhage. A safety analysis. Cerebrovascular diseases (Basel, Switzerland). 2009;27(2):146–50.
Faust AC, Finch CK, Hurdle AC, Elijovich L. Early versus delayed initiation of pharmacological venous thromboembolism prophylaxis after an intracranial hemorrhage. Neurologist. 2017;22(5):166–70.
Ianosi B, Gaasch M, Rass V, Huber L, Hackl W, Kofler M, et al. Early thrombosis prophylaxis with enoxaparin is not associated with hematoma expansion in patients with spontaneous intracerebral hemorrhage. Eur J Neurol. 2019;26(2):333–41.
Kananeh MF, Fonseca-Paricio MJ, Liang JW, Sullivan LT, Sharma K, Shah SO, et al. Ultra-early venous thromboembolism (VTE) prophylaxis in spontaneous Intracerebral hemorrhage (sICH). J Stroke Cerebrovasc Dis. 2021;30(2):105476.
Qian C, Huhtakangas J, Juvela S, Bode MK, Tatlisumak T, Savolainen M, et al. Early vs late enoxaparin for the prevention of venous thromboembolism in patients with ICH: A double blind placebo controlled multicenter study Clin Neurol Neurosurg 2021;202:106534.
Pan X, Li J, Xu L, Deng S, Wang Z. Safety of prophylactic heparin in the prevention of venous thromboembolism after spontaneous Intracerebral hemorrhage: a Meta-analysis. J Neurol Surg A Cent Eur Neurosurg. 2020;81(3):253–60.
Tetri S, Hakala J, Juvela S, Saloheimo P, Pyhtinen J, Rusanen H, et al. Safety of low-dose subcutaneous enoxaparin for the prevention of venous thromboembolism after primary intracerebral haemorrhage. Thromb Res. 2008;123(2):206–12.
Wu TC, Kasam M, Harun N, Hallevi H, Bektas H, Acosta I, et al. Pharmacological deep vein thrombosis prophylaxis does not lead to hematoma expansion in intracerebral hemorrhage with intraventricular extension. Stroke. 2011;42(3):705–9.
Godoy DA, Piñero GR, Koller P, Masotti L, Di Napoli M. Steps to consider in the approach and management of critically ill patient with spontaneous intracerebral hemorrhage. World J Crit Care Med. 2015;4(3):213–29.
Ohwaki K, Yano E, Nagashima H, Hirata M, Nakagomi T, Tamura A. Blood pressure management in acute intracerebral hemorrhage: relationship between elevated blood pressure and hematoma enlargement. Stroke. 2004;35(6):1364–7.
Rodriguez-Luna D, Piñeiro S, Rubiera M, Ribo M, Coscojuela P, Pagola J, et al. Impact of blood pressure changes and course on hematoma growth in acute intracerebral hemorrhage. Eur J Neurol. 2013;20(9):1277–83.
Toyoda K, Okada Y, Minematsu K, Kamouchi M, Fujimoto S, Ibayashi S, et al. Antiplatelet therapy contributes to acute deterioration of intracerebral hemorrhage. Neurology. 2005;65(7):1000–4.
Camps-Renom P, Alejaldre-Monforte A, Delgado-Mederos R, Martínez-Domeño A, Prats-Sánchez L, Pascual-Goñi E, et al. Does prior antiplatelet therapy influence hematoma volume and hematoma growth following intracerebral hemorrhage? Results from a prospective study and a meta-analysis. Eur J Neurol. 2017;24(2):302–8.
Flibotte JJ, Hagan N, O'Donnell J, Greenberg SM, Rosand J. Warfarin, hematoma expansion, and outcome of intracerebral hemorrhage. Neurology. 2004;63(6):1059–64.
Huynh TJ, Aviv RI, Dowlatshahi D, Gladstone DJ, Laupacis A, Kiss A, et al. Validation of the 9-point and 24-point hematoma expansion prediction scores and derivation of the PREDICT a/B scores. Stroke. 2015;46(11):3105–10.
Parikh NS, Kamel H, Navi BB, Iadecola C, Merkler AE, Jesudian A, et al. Liver fibrosis indices and outcomes after primary Intracerebral hemorrhage. Stroke. 2020;51(3):830–7.
Marini S, Morotti A, Ayres AM, Crawford K, Kourkoulis CE, Lena UK, et al. Sex differences in intracerebral hemorrhage expansion and mortality. J Neurol Sci. 2017;379:112–6.
Forti P, Maioli F, Domenico Spampinato M, Barbara C, Nativio V, Coveri M, et al. The Effect of Age on Characteristics and Mortality of Intracerebral Hemorrhage in the Oldest-Old. Cerebrovascular diseases (Basel, Switzerland). 2016;42(5–6):485–92.
Sakuta K, Yaguchi H, Sato T, Mukai T, Komatsu T, Sakai K, et al. The NAG scale can screen for hematoma expansion in acute intracerebral hemorrhage-a multi-institutional validation. J Neurol Sci. 2020;414:116834.
Fu F, Sun S, Liu L, Gu H, Su Y, Li Y. Iodine sign as a novel predictor of hematoma expansion and poor outcomes in primary Intracerebral hemorrhage patients. Stroke. 2018;49(9):2074–80.
Wada R, Aviv RI, Fox AJ, Sahlas DJ, Gladstone DJ, Tomlinson G, et al. CT angiography "spot sign" predicts hematoma expansion in acute intracerebral hemorrhage. Stroke. 2007;38(4):1257–62.
Tan CO, Lam S, Kuppens D, Bergmans RHJ, Parameswaran BK, Forghani R, et al. Spot and diffuse signs: quantitative markers of intracranial hematoma expansion at dual-energy CT. Radiology. 2019;290(1):179–86.
Zhang M, Chen J, Zhan C, Liu J, Chen Q, Xia T, et al. Blend sign is a strong predictor of the extent of early hematoma expansion in spontaneous Intracerebral hemorrhage. Front Neurol. 2020;11:334.
Li Q, Zhang G, Huang Y-J, Dong M-X, Lv F-J, Wei X, et al. Blend sign on computed tomography: novel and reliable predictor for early hematoma growth in patients with Intracerebral hemorrhage. Stroke. 2015;46(8):2119–23.
Li Q, Zhang G, Xiong X, Wang X-C, Yang W-S, Li K-W, et al. Black hole sign: novel imaging marker that predicts hematoma growth in patients with Intracerebral hemorrhage. Stroke. 2016;47(7):1777–81.
Li Q, Liu Q-J, Yang W-S, Wang X-C, Zhao L-B, Xiong X, et al. Island sign: an imaging predictor for early hematoma expansion and poor outcome in patients with Intracerebral hemorrhage. Stroke. 2017;48(11):3019–25.
Vermeer SE, Algra A, Franke CL, Koudstaal PJ, Rinkel GJE. Long-term prognosis after recovery from primary intracerebral hemorrhage. Neurology. 2002;59(2):205–9.
Arakawa S, Saku Y, Ibayashi S, Nagao T, Fujishima M. Blood pressure control and recurrence of hypertensive brain hemorrhage. Stroke. 1998;29(9):1806–9.
Arima H, Tzourio C, Anderson C, Woodward M, Bousser MG, MacMahon S, et al. Effects of perindopril-based lowering of blood pressure on intracerebral hemorrhage related to amyloid angiopathy: the PROGRESS trial. Stroke. 2010;41(2):394–6.
Biffi A, Anderson CD, Battey TWK, Ayres AM, Greenberg SM, Viswanathan A, et al. Association between blood pressure control and risk of recurrent Intracerebral hemorrhage. JAMA. 2015;314(9):904–12.
Vinters HV, Gilbert JJ. Cerebral amyloid angiopathy: incidence and complications in the aging brain. II. The distribution of amyloid vascular changes. Stroke. 1983;14(6):924–8.
Charidimou A, Imaizumi T, Moulin S, Biffi A, Samarasekera N, Yakushiji Y, et al. Brain hemorrhage recurrence, small vessel disease type, and cerebral microbleeds: a meta-analysis. Neurology. 2017;89(8):820–9.
Weimar C, Benemann J, Terborg C, Walter U, Weber R, Diener HC, et al. Recurrent stroke after lobar and deep intracerebral hemorrhage: a hospital-based cohort study. Cerebrovasc Dis. 2011;32(3):283–8.
O'Donnell HC, Rosand J, Knudsen KA, Furie KL, Segal AZ, Chiu RI, et al. Apolipoprotein E genotype and the risk of recurrent lobar intracerebral hemorrhage. N Engl J Med. 2000;342(4):240–5.
Charidimou A, Boulouis G, Roongpiboonsopit D, Xiong L, Pasi M, Schwab KM, et al. Cortical superficial siderosis and recurrent intracerebral hemorrhage risk in cerebral amyloid angiopathy: large prospective cohort and preliminary meta-analysis. Int J Stroke. 2019;14(7):723–33.
Li L, Poon MTC, Samarasekera NE, Perry LA, Moullaali TJ, Rodrigues MA, et al. Risks of recurrent stroke and all serious vascular events after spontaneous intracerebral haemorrhage: pooled analyses of two population-based studies. Lancet Neurol. 2021;20(6):437–47.
Ajmeri AN, Zaheer K, McCorkle C, Amro A, Mustafa B. Treating venous thromboembolism post intracranial hemorrhage: a case report. Cureus. 2020;12(1):e6746.
Becattini C, Cimini LA, Carrier M. Challenging anticoagulation cases: a case of pulmonary embolism shortly after spontaneous brain bleeding. Thromb Res. 2021;200:41–7.
Lee WC, Fang HY. Management of pulmonary embolism after recent intracranial hemorrhage: a case report. Medicine (Baltimore). 2018;97(15):e0479.
Ottosen TP, Grijota M, Hansen ML, Brandes A, Damgaard D, Husted SE, et al. Use of antithrombotic therapy and long-term clinical outcome among patients surviving Intracerebral hemorrhage. Stroke. 2016;47(7):1837–43.
Majeed A, Kim Y-K, Roberts RS, Holmström M, Schulman S. Optimal timing of resumption of warfarin after intracranial hemorrhage. Stroke. 2010;41(12):2860–6.
Yung D, Kapral MK, Asllani E, Fang J, Lee DS. Reinitiation of anticoagulation after warfarin-associated intracranial hemorrhage and mortality risk: the best practice for reinitiating anticoagulation therapy after intracranial bleeding (BRAIN) study. Can J Cardiol. 2012;28(1):33–9.
Kuramatsu JB, Gerner ST, Schellinger PD, Glahn J, Endres M, Sobesky J, et al. Anticoagulant Reversal, Blood Pressure Levels, and Anticoagulant Resumption in Patients With Anticoagulation-Related Intracerebral Hemorrhage. Jama. 2015;313(8).
Witt DM, Clark NP, Martinez K, Schroeder A, Garcia D, Crowther MA, et al. Risk of thromboembolism, recurrent hemorrhage, and death after warfarin therapy interruption for intracranial hemorrhage. Thromb Res. 2015;136(5):1040–4.
Murthy SB, Gupta A, Merkler AE, Navi BB, Mandava P, Iadecola C, et al. Restarting anticoagulant therapy after intracranial hemorrhage: a systematic review and Meta-analysis. Stroke. 2017;48(6):1594–600.
Carney BJ, Uhlmann EJ, Puligandla M, Mantia C, Weber GM, Neuberg DS, et al. Anticoagulation after intracranial hemorrhage in brain tumors: risk of recurrent hemorrhage and venous thromboembolism. Res Pract Thromb Haemost. 2020;4(5):860–5.
Mantia C, Zwicker JI. Anticoagulation in the setting of primary and metastatic brain tumors. Cancer Treat Res. 2019;179:179–89.
Donato J, Campigotto F, Uhlmann EJ, Coletti E, Neuberg D, Weber GM, et al. Intracranial hemorrhage in patients with brain metastases treated with therapeutic enoxaparin: a matched cohort study. Blood. 2015;126(4):494–9.
Porfidia A, Giordano M, Sturiale CL, D'Arrigo S, Donadini MP, Olivi A, et al. Risk of intracranial bleeding in patients with primary brain cancer receiving therapeutic anticoagulation for venous thromboembolism: a meta-analysis. Brain Behav. 2020;10(6):e01638.
Zhou Z, Yu J, Carcel C, Delcourt C, Shan J, Lindley RI, et al. Resuming anticoagulants after anticoagulation-associated intracranial haemorrhage: systematic review and meta-analysis. BMJ Open. 2018;8(5):e019672.
Schulman S, Kearon C, Kakkar AK, Mismetti P, Schellong S, Eriksson H, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361(24):2342–52.
Büller HR, Prins MH, Lensin AWA, Decousus H, Jacobson BF, Minar E, et al. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 2012;366(14):1287–97.
Agnelli G, Buller HR, Cohen A, Curto M, Gallus AS, Johnson M, et al. Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med. 2013;369(9):799–808.
Büller HR, Décousus H, Grosso MA, Mercuri M, Middeldorp S, Prins MH, et al. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N Engl J Med. 2013;369(15):1406–15.
Lamsam L, Sussman ES, Iyer AK, Bhambhvani HP, Han SS, Skirboll S, et al. Intracranial hemorrhage in deep vein thrombosis/pulmonary Embolus patients without atrial fibrillation. Stroke. 2018;49(8):1866–71.
Inohara T, Xian Y, Liang L, Matsouaka RA, Saver JL, Smith EE, et al. Association of Intracerebral Hemorrhage among Patients Taking non-Vitamin K Antagonist vs vitamin K antagonist Oral anticoagulants with in-hospital mortality. JAMA. 2018;319(5):463–73.
Hagii J, Tomita H, Metoki N, Saito S, Shiroto H, Hitomi H, et al. Characteristics of intracerebral hemorrhage during rivaroxaban treatment: comparison with those during warfarin. Stroke. 2014;45(9):2805–7.
Wilson D, Charidimou A, Shakeshaft C, Ambler G, White M, Cohen H, et al. Volume and functional outcome of intracerebral hemorrhage according to oral anticoagulant type. Neurology. 2016;86(4):360–6.
Pollack CV, Reilly PA, van Ryn J, Eikelboom JW, Glund S, Bernstein RA, et al. Idarucizumab for Dabigatran reversal - full cohort analysis. N Engl J Med. 2017;377(5):431–41.
Connolly SJ, Crowther M, Eikelboom JW, Gibson CM, Curnutte JT, Lawrence JH, et al. Full study report of Andexanet Alfa for bleeding associated with factor Xa inhibitors. N Engl J Med. 2019;380(14):1326–35.
Siegal DM, Curnutte JT, Connolly SJ, Lu G, Conley PB, Wiens BL, et al. Andexanet Alfa for the reversal of factor Xa inhibitor activity. N Engl J Med. 2015;373(25):2413–24.
Somarouthu B, Yeddula K, Wicky S, Hirsch JA, Kalva SP. Long-term safety and effectiveness of inferior vena cava filters in patients with stroke. J Neurointerv Surg. 2011;3(2):141–6.
Mellado M, Trujillo-Santos J, Bikdeli B, Jiménez D, Núñez MJ, Ellis M, et al. Vena cava filters in patients presenting with major bleeding during anticoagulation for venous thromboembolism. Intern Emerg Med. 2019;14(7):1101–12.
Melmed K, Chen ML, Al-Kawaz M, Kirsch HL, Bauerschmidt A, Kamel H. Use and removal of inferior vena cava filters in patients with acute brain injury. The Neurohospitalist. 2020;10(3):188–92.
Hansmann J, Sheybani A, Minocha J, Bui JT, Lipnik AJ, Shah KY, et al. Retrievable inferior vena cava filters in neurosurgical patients: retrieval rates and clinical outcomes. Clin Neurol Neurosurg. 2019;179:30–4.
Bottinor W, Turlington J, Raza S, Roberts CS, Malhotra R, Jovin IS, et al. Life-saving systemic thrombolysis in a patient with massive pulmonary embolism and a recent hemorrhagic cerebrovascular accident. Tex Heart Inst J. 2014;41(2):174–6.
Koroneos A, Koutsoukou A, Zervakis D, Politis P, Sourlas S, Pagoni E, et al. Successful resuscitation with thrombolysis of a patient suffering fulminant pulmonary embolism after recent intracerebral haemorrhage. Resuscitation. 2007;72(1):154–7.
Endo H, Kubota H, Sato M, Sudo K. Surgical treatment of pulmonary embolism with recent intracranial hemorrhage. Ann Thorac Cardiovasc Surg. 2005;11(4):256–9.
Hanger HC, Fletcher VJ, Wilkinson TJ, Brown AJ, Frampton CM, Sainsbury R. Effect of aspirin and warfarin on early survival after intracerebral haemorrhage. J Neurol. 2008;255(3):347–52.
Mittal MK, Rabinstein AA. Anticoagulation-related intracranial hemorrhages. Curr Atheroscler Rep. 2012;14(4):351–9.
Acknowledgements
Not applicable.
Funding
Health Commission of Chongqing, Chongqing Municipal People's Government, Grant Number: 2017jstg31, 2018jstg012, 2019jstg023.
Author information
Authors and Affiliations
Contributions
QC wrote the manuscript, XZ designed the tables and HC reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
All authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Cai, Q., Zhang, X. & Chen, H. Patients with venous thromboembolism after spontaneous intracerebral hemorrhage: a review. Thrombosis J 19, 93 (2021). https://doi.org/10.1186/s12959-021-00345-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12959-021-00345-z