Abstract
Background
Intra-uterine infusion treatments were reported to be beneficial to embryo implantation and pregnancy outcomes, and considered as potential therapies for infertile patients with recurrent implantation failure (RIF). Nevertheless, their efficiencies were controversial and there lack of consensus on which intrauterine treatment is the most effective.
Methods
All prospective trials (in Chinese or English) were searched in Databases PubMed, Cochrane, Web of Science, and CNKI from July 2013 to July 2023. We included studies that investigated various uterine infusions, including chorionic gonadotropin, granulocyte colony-stimulating factor, monocytes, platelet-rich plasma, etc. during IVF treatment and reported subsequent pregnancy outcomes.
Results
We finally included 56 researches, including 40 randomized controlled trials, 14 non-randomized controlled trials, and 3 prospective cohort studies. This study included a total of 11 uterine perfusion methods: Placebo, Human Chorionic Gonadotropin (HCG), Granulocyte Colony-Stimulating Factor (G-CSF), platelet-rich plasma (PRP), Peripheral Blood Mononuclear Cell (PBMC), Growth hormone (GH), dexamethasone (DEX), Embryo culture supernatant (ESC), PRP combined with G-CSF (PRP + G-CSF), RPR combined with subcutaneous injection of G-CSF (RPR + G-CSFsc), G-CSF combined with subcutaneous injection of AXaIU (G-CSF + AXaIUsc). Intrauterine infusion of HCG, PBMC, G-CSF, and PRP significantly improves pregnancy outcomes in patients with repeated implantation failure compared with blank controls or placebo, and PRP improved the clinical pregnancy and live birth most. GH and ESC infusion might improve the pregnancy outcomes, but uterine infusion of DEX was shown with high miscarriage. The combination therapy did not show a significant advantage over the mono-therapy.
Conclusions
Intrauterine infusion of HCG, PBMC, G-CSF, and PRP are promising strategies for improving pregnancy outcomes for infertile patients with recurrent implantation failure. Among these treatments, PRP may be the best. More researches are required to explore the effect of drug combinations and less commonly used drugs as well.
Trial registration
Our study was registered in PROSPERO and the ID was CRD42023467188.
Similar content being viewed by others
Synopsis
Intrauterine infusion seems a promising strategy for improving the pregnancy outcomes for infertile patients with recurrent implantation failure.
Introduction
Assisted reproductive technology (ART) has been widely used since the first baby was born in 1778s. Nowadays, more than 8 million babies are born with the help of ART worldwide [1]. However, live birth rates of this treatment need to be improved, for the clinical pregnancy rate is between 30 and 40% [2], and the live birth rate is estimated to be between 20 and 29% [3, 4]. Implantation failure is one of the tough bottlenecks of ART success [5, 6].
Successful embryo implantation is a complex process, requiring a competent embryo, a receptive endometrium, and an appropriate dialogue [7]. The competence of the embryo is influenced by various factors, such as age, environmental pollutant exposure, and unhealthy lifestyle. Endometrial receptivity can be altered by endometrial blood supply, immune state, the structural uterine malformation (such as polyps and adhesions). An appropriate dialogue includes embryo location, adhesion, and intrusion. Synchronous development between embryo and endometrium was reported to be involved. In infertile patients with recurrent implantation failure, despite the transfer of good-quality embryos, the implantation was aborted, indicating the significant difference in the endometrial receptivity and synchronization between the embryo and the endometrium [8,9,10].
Numerous strategies and interventions have been used as adjuvant treatments to enhance endometrial receptivity, including estradiol hormonal supplementations, angiogenesis regulators, antioxidants, immunomodulators, etc. [11, 12]. Meanwhile, intrauterine infusion treatments get the most attention for their safety, simplicity, and operation friendliness. Intra-uterine infusion of human chorionic gonadotropin (HCG), autologous peripheral blood mononuclear cells (PBMCs), granulocyte colony-stimulating factor (G-CSF) or Platelet-rich plasma (PRP) were reported to be beneficial to embryo implantation and pregnant outcomes, considered as potential therapies for RIF [13,14,15,16]. Nevertheless, their efficiencies were controversial, with some advocating improvement, and others showing no benefit [17,18,19]. Furthermore, most of the previous studies compared the effects of two treatments, and few studies compared multiple treatments at the same time. Besides, insufficient attention has been attached to new but less-applied infusion strategies, such as combination therapy. Consequently, there lack of consensus on which intrauterine treatment is the most effective, and unable to give good guidance for the clinical treatment.
Network meta-analysis (NMA) is probably an effective method to explore the most effective uterine-infusion treatment, for it can compare three or more treatments simultaneously, integrating the results of direct and indirect comparisons, summarizing the possibility values of the efficacy of each treatment, and reporting the best option. Network meta-analyses have been used to explore the best IUI protocol for unexplained infertility [20], compare the efficiency of diverse luteal phase support [21], and evaluate the role of different adjuvant treatment strategies on the probability of pregnancy achievement in poor responders undergoing IVF [22], etc.
It remains a challenge for clinicians to treat repeated IVF failures characterized by no anatomical pathologies, good response to treatment, and good embryo quality but no occurrence of pregnancy. Therefore, the study aimed to screen research that compared the effects of different intrauterine infusion treatments in women with RIF. Furthermore, a network meta-analysis is performed to comprehensively evaluate the improvement of pregnant outcomes. The research aims to rank the efficacy of each uterine infusion strategy in different pregnancy outcomes to guide clinical decision-making for women with repeated implantation failure.
Methods
Literature search and study selection
We reported and conducted a systematic review and network meta-analysis based on the guidelines of the preferred reporting items for systematic reviews and meta-analyses (PRISMA). The PubMed, Cochrane Library, CNKI, and Web of Science databases were systematically searched from July 2013 to July 2023. MeSH search combined with random word search was used in our study. The search items included “Chorionic Gonadotropin”, “Granulocyte Colony-Stimulating Factor”, “G-CSF”, “Monocytes”, “PBMC”, “Platelet-Rich Plasma”, “PRP”, “Lymphocytes”, “Uterine perfusion”, “RIF”, “ Repeated implantation failure”, “Recurrent implantation failure”. Meanwhile, we also checked references listed in the included studies and all related reviews and guidelines to supplement any previously ignored literature. Our study was registered in PROSPERO and the ID was CRD42023467188.
Prospective trials (in Chinese or English) were included if they met the following criteria: 1. Objectives of the study were RIF patients. 2. At least one of the uterine perfusion treatments was included. 3. Reported at least one of the pregnant outcomes: chemical pregnancy, clinical pregnancy, implantation, miscarriage, and live birth. Two independent investigators (Q.X. and XZ.Q.) identified and evaluated all eligible studies. Registration of all subjects or ethical approval is not applicable because the data used by this meta-analysis were from published studies.
Quality assessment of risk of bias
The methodology and categories described in the Cochrane Collaboration Handbook were used to assess the risk of bias in RCT studies. The risks of bias graphs were constructed with Review Manager 5.3 software.
The risk of bias in non-random controlled court and prospective observe study was assessed by the methodological index for non-randomized studies (MINORS) and Newcastle–Ottawa Scale (NOS) respectively.
Two independent investigators (QX and XZY) assessed the risk of bias. The other investigators YLL and XZQ would attend if any discrepancy existed.
Data extraction
Extracted data included: 1. Characteristics of study (authors and publication year); 2. Patient characteristics (RIF definition, inclusion and exclusion criteria); 3. Trial design details (sample size, uterus perfusion methods, embryo transfer method, embryo grades, pregnant outcome). Two independent investigators (Q.X. and XZ. Q.) extracted the relevant data. If there existed any discrepancies, reviewer XZ. Y. would attend to and resolve them by consensus of the reviewers.
Data synthesis and statistical analysis
Traditional direct pairwise comparisons were performed by Review Manager 5.3 software if direct data were available. The pregnant outcomes would be analyzed if the amount of relevant research was not less than three. We synthesized data and calculated summary odds ratios (ORs) and 95% CIs. If heterogeneity index I2 ≥ 50%, and P < 0.05, a random effect model was applied, otherwise, a fixed effect model was used. Funnel plots were used to detect publication bias.
The network meta-analysis was conducted by the STATA software package (version 15.0, StataCorp, College Station, TX) and the GeMTC software package. STATA software package portrayed a network of eligible comparisons. All trials with recurrent implantation failure were included in the network meta-analysis. Before conducting the network meta-analysis, consistency and convergence were assessed. We verified inconsistencies by the node splitting method, which separated evidence for a particular comparison into direct and indirect evidence, and indicated a significant inconsistency. An inconsistency model was used if inconsistency P < 0.05 was identified, otherwise, the relative effects of the interventions were analyzed using a consistency model. We used the Brooks-Gelman-Rulin method to assess the Convergence: the potential scale reduction factors (PSRF) were close to 1 for all of the chains, and the results were considered to be well-converged. We summarized the possibility values of the efficacy of each treatment and reported the best option and surface under the cumulative ranking curve (SUCRA). The value of SUCRA ranges from 0 to 100%, and the higher the score is, the more likely it is that the treatment will be the best option.
Besides, publication bias was analyzed by the funnel plot, and sensitivity analysis was conducted according to the origin of the publication.
Results
Selection and characteristics of the included studies
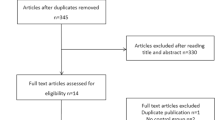
After electronic searches, 679 potential citations were identified. The selection process is shown in Fig. 1. Subsequently, a total of 56 studies were included: 40 were randomized controlled trials (RCTs) [19, 23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61], 13 were non-RCTs [48, 62,63,64,65,66,67,68,69,70,71,72,73], and 3 were prospective cohort studies [74,75,76]. Meanwhile, 31 were published in English and 25 in Chinese. The control group was blank without any uterine infusion. Comparative efficacies of 11 therapies were conducted: Placebo, Human Chorionic Gonadotropin (HCG), Granulocyte Colony-Stimulating Factor (G-CSF), Platelet-rich plasma (PRP), Peripheral Blood Mononuclear Cell (PBMC), Growth hormone (GH), Dexamethasone (DEX), Embryo culture supernatant (ESC), PRP combined with G-CSF (PRP + G-CSF), RPR combined with subcutaneous injection of G-CSF (RPR + G-CSFsc), G-CSF combined with subcutaneous injection of AXaIU (G-CSF + AXaIUsc). The main characteristics of the studies included are presented in Supplemental Table S1. The number of subjects ranges from 37 to 393. Among a total of 56 studies, 12, 14, 15, and 19 studies involved HCG, G-CSF, PRP, and PBMC, respectively. Only one study involved GH, DEX, ESC, PRP + G-CSFsc, PRP + G-CSF, or G-CSF + AXaIUsc.
Risk of bias assessment results
The risk of bias of 40 RCTs was assessed by the Cochrane Collaboration Handbook, as shown in Supplemental Figure S1. 31(76%) researches were with low risk in random sequence generation. 17(43%) researches showed a high risk of bias with "Blind of participants and personnel", 15(38%) researches with low risk, and 8(20%) researches with unclear. Allocation concealment was not referred to in 28(70%) researches and 11 (28%) researches with low risk in allocation concealment. Other terms were shown with low risk.
MINORS questionnaire was performed to evaluate the risk of bias in 13 non-randomized controlled trials, as shown in Supplemental Table S2a. All studies did not estimate the sample size, which might increase the risk of bias and decrease the incredibility.
We assessed the risk of bias in 3 prospective cohort studies by NOS questionnaire (Supplemental Table S2b). All these researches seemed of high quality.
Meta-analysis results
Primary outcome measure: clinical pregnancy rate
The outcome of the direct pairwise meta-analysis is shown in Fig. 2. Compared with the control group, PBMC, G-CSF, HCG, and PRP could significantly increase the clinical pregnancy rate (OR 2.87, 95% CI 2.27 to 3.63; 2.16, 1.59 to 2.95; 2.00, 1.51 to 2.64; 2.90, 2.15 to 3.90; respectively). Only HCG (OR 1.74, 95% CI 1.34 to 2.26), but not the PBMC or the G-CSF group (OR 1.36, 95% CI 0.92 to 2.01; 1.37, 0.95 to 1.98, respectively) with a higher clinical pregnancy rate than the placebo group with statistical significance. The clinical pregnancy rate between the HCG and the PBMC group, G-CSF and HCG group were comparable (OR 0.97, 95% CI 0.62 to 1.50; 0.71, 0.35 to 1.45, respectively).
The results of the network meta-analysis are shown in Table 1 and Fig. 3. When compared to the control group, except the ECS and PRP + GCSFsc group, almost all uterine infusions significantly improve the clinical pregnancy including DEX (OR 2.76, 95% CI 1.16 to 5.81)), G-CSF (OR 2.58, 95% CI 1.90 to 3.43)), GCSF + AXaIUsc (OR 3.34, 95% CI 1.38 to 8.19)), GH (OR 2.80, 95% CI 1.14 to 6.60)), HCG (OR 2.27, 95% CI 1.76 to 3.01)), PBMC (OR 3.03, 95% CI 2.42 to 4.07)), PRP (OR 3.07, 95% CI 2.30 to 3.88)), PRP + GCSF (OR 2.90, 95% CI 1.19 to 10.06)), and placebo group(OR 1.59, 95% CI 1.18 to 2.06)). Meanwhile, OR for clinical pregnancy rates in placebo was significantly inferior to PRP (OR 0.49, 95% CI 0.37 to 0.76), PBMC (OR 0.52, 95% CI 0.37 to 0.67), HCG (OR 0.69, 95% CI 0.51 to 0.89), G-CSF (OR 0.62, 95% CI 0.46 to 0.85) respectively. Furthermore, OR for clinical pregnancy rates comparing PBMC to ECS and HCG was 2.22 (95% CI 1.14 to 5.23) and 1.36 (95% CI 1.00 to 1.89). OR for clinical pregnancy rates comparing PRP to ECS was 2.40(95% CI 1.07 to 4.94).
When ranked from the best therapy, it was shown that uterine infusion with PRP + GCSF was the most effective (SUCRA 26%), followed by GCSF + AXaIUsc (SUCRA 20%), PBMC (SUCRA 22%), and PRP (SUCRA 20%), as shown in Table 2.
Secondary outcome measures: live birth
The direct pair-wise meta-analysis (Supplemental Figure S2) showed that G-CSF, PBMC, and PRP significantly increased the live birth compared with the control group (OR 2.90, 95% CI 1.68 to 5.00; 2.23, 1.25 to 4.00; 4.27, 1.25 to 14.66; respectively). However, no significant difference was found between the placebo and the G-CSF group (OR 1.28, 95% CI 0.65 to 2.51).
The outcome of the network meta-analysis showed that, compared to the control group (Supplemental Table S3 and Figure S3), the PRP group (OR 4.85, 95% CI 1.76 to 14.56) significantly improved the live birth rate. Others including G-CSF, GH, HCG, PBMC, PRP + GCSFsc, and Placebo group didn't significantly increase the live birth rate. There existed no significant difference in live births among all uterine treatments.
When ranked from the best therapy, it was shown that uterine infusion with GH was the most effective (SUCRA 13%), followed by HCG (SUCRA 21%), GCSF (SUCRA 22%), and Placebo (SUCRA 23%) (Table 2).
Secondary outcome measures: embryo implantation
The direct pair-wise meta-analysis (Supplemental Figure S4) showed that, compared with the control group, PBMC, HCG, G-CSF, and PRP significantly increased the implantation (OR 2.43, 95% CI 1.89 to 3.13; 1.47, 1.05 to 2.05; 2.20, 1.62 to 2.99; 2.64, 1.72 to 4.07; respectively). The effect persisted in PBMC and HCG even when compared with the placebo group (OR 1.52, 95% CI 1.03 to 12.22; 1.65, 1.29 to 2.12).
In the network meta-analysis, when compared to the control group (Supplemental Table S4 and Figure S3), G-CSF (OR 2.62, 95% CI 1.89 to 3.74), GCSFAIU (OR 2.81, 95% CI 1.20 to 6.46), GH (OR 3.57, 95% CI 1.53 to 8.25), HCG (OR 1.94, 95% CI 1.33 to 2.79), PBMC (OR 3.03, 95% CI 2.25 to 4.32), PRP (OR 2.77, 95% CI 1.55 to 4.83) significantly improved the clinical pregnancy. Meanwhile, OR for implantation rates comparing PBMC to HCG was 1.57 (95% CI 1.05 to 2.42).
When ranked from the best therapy, it was shown that uterine infusion with GH was the most effective (SUCRA 38%), followed by PBMC (SUCRA 21%), PRP (SUCRA 15%) and G-CSF (SUCRA 22%) (Table 2).
Secondary outcome measures: chemical pregnancy
The direct pair-wise meta-analysis (Supplemental Figure S5) showed that the chemical pregnancy in the PRP group was significantly higher than the control group (OR 2.41, 95% CI 1.92 to 3.03). However, no significant difference was found between the placebo and the G-CSF group in terms of chemical pregnancy.
In the network meta-analysis, when compared to the control group (Supplemental Table S5 and Figure S3), the PRP group (OR 1.90, 95% CI 1.00 to 3.54) significantly improved the chemical pregnancy. Others including G-CSF, GH, HCG, PBMC, PRP + GCSF, and Placebo group didn’t significantly increase the implantation rate. No significant difference in chemical pregnancy among all uterine treatments.
When ranked from the best therapy, it was shown that uterine infusion with PRP + GCSF was the most effective (SUCRA 47%), followed by PRP (SUCRA 18%), GCSF (SUCRA 22%) and HCG (SUCRA 18%) (Table 2).
Secondary outcome measures: miscarriage
In the direct pair-wise meta-analysis (Supplemental Figure S6), G-CSF (OR 0.28, 95% CI 0.13 to 0.59), but not the PBMC and HCG could significantly decrease the miscarriage rate compared with the control group. When compared with the placebo group, PBMC and G-CSF (OR 0.24, 95% CI 0.11 to 0.56; 0.24, 0.12 to 0.49), but not the HCG significantly decreased the miscarriage rate. Besides, there was no difference between the PBMC and HCG, HCG and G-CSF group in terms of miscarriage.
In the network meta-analysis, when compared to the control group (Supplemental Table S6 and Figure S3), the DEX group significantly increased the miscarriage rate, but the GCSF and PBMC group significantly decreased the miscarriage rate (OR 0.29, 95% CI 0.14 to 0.64; 0.38, 0.19 to 0.83). The miscarriage in the Placebo group was significantly higher than in the PBMC, HCG, and GCSF group (OR 2.82, 95% CI 1.40 to 6.10; 1.79, 1.03 to 3.50; 3.79, 1.92 to 8.25; respectively). Meanwhile, OR for miscarriage rates in PRP was significantly higher than G-CSF (OR 3.36, 95% CI 1.03 to 8.73).
When ranked, it was shown that uterine infusion with DEX had the highest probability of miscarriage (SUCRA 91%), followed by PRP + GCSFsc (SUCRA 63%), GH (SUCRA 27%), and Placebo (SUCRA 25%) (Table 2).
Heterogeneity and publication bias
During data merging, most studies showed low heterogeneity, except for the outcomes of clinical pregnancy and implantation between HCG and G-CSF, live birth between the PRP and control, and implantation and chemical pregnancy between G-CSF and placebo. The inadequate quantity (≤ 5) of research might be one of the reasons for the heterogeneity. On the other hand, the overall publication bias appears low as analyzed by the funnel plot, as shown in Supplemental Figure S7.
Sensitivity analysis
We did sensitivity analysis according to the origin of the publication: English and Chinese.
In the subgroup analysis by English researches, compared with the control group (Supplemental Figure S8), HCG (OR 2.20, 95% CI 1.51 to 3.21), G-CSF (OR 2.28, 95% CI 1.27 to 4.07), PBMC (OR 2.66, 95% CI 1.95 to 3.63), and PRP (OR 2.83, 95% CI 2.20 to 3.64) infusion could significantly increase the clinical pregnancy. HCG group and G-CSF group had a higher clinical pregnancy than the placebo group as well (OR 1.82, 95% CI 1.25 to 2.64; 1.68, 1.15 to 2.48). In the network meta-analysis (Supplemental Table S7), G-CSF, HCG, PBMC, PRP, PRP + G-CSFsc, and placebo could significantly increase the clinical pregnancy than the control group. The clinical pregnancy rate in the placebo group was significantly lower than in the G-CSF and PBMC groups.
In the subgroup analysis by Chinese researches, compared with the control group (Supplemental Figure S9), HCG (OR 1.78, 95% CI 1.17 to 2.69), G-CSF (OR 2.12, 95% CI 1.47 to 3.06), PBMC (OR 3.17, 95% CI 2.22 to 4.52), and PRP (OR 3.47, 95% CI 2.20 to 5.47) infusion could significantly increase the clinical pregnancy. HCG (OR 1.67, 95% CI 1.15 to 2.40) but not the G-CSF and the PBMC (OR 1.05, 95% CI 0.67 to 1.63; 1.48, 0.96 to 2.27) was with a higher clinical pregnancy than the placebo group. In the network meta-analysis (Supplemental Table S8), DEX, G-CSF, G-CSF + AIU, GH, HCG PBMC, PRP, PRP + G-CSF and Placebo but not the ESC could significantly increase the clinical pregnancy than the control group. The clinical pregnancy in the PBMC and PRP groups was significantly higher than in the ESC group. The placebo group had a lower clinical pregnancy than the HCG, PBMC, and PRP groups.
Discussion
In the traditional pairwise meta-analysis, HCG, PBMC, G-CSF, and PRP treatments improved pregnancy outcomes no matter compared with the control group or the placebo group. Meanwhile, there existed no significant difference between PBMC and HCG in terms of clinical pregnancy and miscarriage, and between HCG and G-CSF in terms of implantation, clinical pregnancy, and miscarriage. In the network meta-analysis, no one treatment showed absolute dominance among pregnancy outcomes but the PRP improved the clinical pregnancy and live birth most. In terms of clinical pregnancy, PBMC was superior to HCG and ECS, and PRP was superior to ECS. PBMC was superior to HCG at implantation. DEX treatment showed high risk of miscarriage, and the risk of miscarriage of PRP is higher than G-CSF treatment. No significant difference was found among intrauterine infusion treatments in chemical pregnancy and live birth. When it comes to the outcomes rank, intrauterine infusion with PRP + GCSF ranked highest in clinical pregnancy, GH ranked highest in embryo implantation, PRP + GCSF ranked highest in chemical pregnancy, DEX ranked highest in miscarriage, GH ranked highest in live birth. The probability of rank for pregnant outcomes is rather low except for the miscarriage, it should be interpreted with caution. In subgroup analysis by the origin of the publication, the outcome was similar.
Several uterine infusion strategies have been proposed for improving pregnancy outcomes for RIF patients, especially HCG, G-CSF, PBMC, and PRP. These treatments were reported to mediate the balance of immune cells and the abnormal cytokine secretion, anti-apoptosis, promote angiogenesis, and so on [28, 77,78,79,80].
In our study, the number of research about HCG, PBMC, G-CSF, and PRP were the most, indicating they were possibly effective treatments from researchers' perspectives. The pair-wise comparison showed that pregnant outcomes were significantly improved by HCG, PBMC, G-CSF, and PRP treatments compared with the control group or the placebo group, which were consistent with other studies.
The study of Xie, H. et al. included six trials, of 1432 women to explore the efficiency of intrauterine perfusion of hCG [14]. They found HCG infusion could significantly improve the clinical pregnancy rate and live birth rate compared with no perfusion of HCG. Implantation, pregnancy, and live birth rates were significantly increased in the group of PBMCs treatment. But Pourmoghadam, Z. et al. also found that the miscarriage rate was significantly decreased in the PBMC-treated group than the non-treated group, which did not reach significance in our study [16]. A meta-analysis consisting of 20 RCTs found that G-CSF increased the biochemical pregnancy rate, embryo implantation rate, and clinical pregnancy rate [81]. Another meta-analysis found G-CSF administration may improve clinical pregnancy rate, but it concluded that they were uncertain whether the administration of G-CSF improves ongoing pregnancy or overall clinical pregnancy rates or reduces miscarriage rate compared to no treatment or placebo for the included studies had unclear allocation concealment or were at high risk of performance bias [18]. As regards the efficiency of PRP, the rates of clinical pregnancy, chemical pregnancy, live births, and implantation were significantly higher than in the control group [82,83,84]. A network analysis published recently compared the effect of PRP, G-CSF, PBMC, and HCG intrauterine infusion on the pregnant outcomes of RIF patients [85]. Its outcomes confirmed that all four intrauterine infusion drugs can improve pregnancy outcomes in RIF patients to varying degrees, with PRP being the most effective, which was similar to our study.
GH, DEX, and ECS intrauterine infusions were also included in our study, but the amount of research was few, and no direct comparisons with other infusion treatments exist. Zihua Wang et al. indicated that DEX could significantly improve embryo implantation and clinical pregnancy, and have no significant effect on miscarriage [23]. However, outcomes of the network meta-analysis combining all the direct and indirect comparisons showed that DEX could significantly increase the possibility of miscarriage, so the use of DEX needs more consideration. Embryos are reported to secrete certain factors during development. These factors might affect the process of implantation and predict the successful embryo implantation [86,87,88]. The implantation rate may be improved by the performance of intrauterine perfusion of embryo culture supernatant before embryo transfer [65]. There was only one study about the effect of GH as well. GH treatment was shown to significantly improve the implantation compared with the control group and the effect seems to be superior to the G-CSF treatment group [60]. Outcomes of network meta-analysis showed that implantation and clinical pregnancy were significantly improved by GH infusion. Meanwhile, GH infusion performed the best in the efficient ranking of the embryo implantation rate and live birth rate. However, the probabilities of the ranking were universally low, requiring further verifications.
In addition, the combination treatments did not show obvious advantages. No one combination of treatments significantly improved the pregnant outcomes with comparisons to the HCG, PBMC, G-CSF, or PRP treatment. In terms of the effectiveness ranking, the biochemical pregnancy rate and clinical pregnancy rate in the G-CSF + PRP were the highest. However, the ranking outcomes were with low probabilities, which should be interpreted with caution. More high-quality researches are required to further verify the efficiency of combination treatments.
There are some strengths in our study. First, we showed the overview of the various uterine infusion treatments, which also included new but less-applied infusion strategies, such as combination therapy. Meanwhile, main pregnancy outcomes including implantation, chemical pregnancy, clinical pregnancy, miscarriage, and live birth were explored. Second, a network meta-analysis was performed, which can apply indirect comparison to draw comparison outcomes and provide the opportunity to rank the different treatment strategies in order of effectiveness, facilitating clinical decision-making. Third, the number of researches included was relatively large for we included both English and Chinese articles, which might contribute to accurate results. Furthermore, subgroup analysis by the publication origin was conducted.
Some limitations must be noted. First, there was no universe definition of RIF, 25 researches (45%) in our study considered RIF patients to be women under the age of 40 years who have failed to achieve a clinical pregnancy after the transfer of at least four good-quality embryos in a minimum of three IVF fresh or frozen cycles. 19 researches (34%) defined RIF as patients failed transfer of good-quality embryos at least once, more than twice, or twice to more than three times. 12 researches (21%) did not clearly describe the definition. Besides, we included RCT, non-RCT trials, and prospective cohort trials, which might lead to some heterogeneity. Second, variations existed in terms of infusion time point and times of infusion, ET cycle, endometrium preparation, etc. Meanwhile, no subgroup analysis was further conducted to decrease the bias of confounding factors such as age, which is an essential factor in deciding the pregnant outcomes of patients with recurrent implantation failure. Third, meta-analyses for safety were not able to be performed due to inadequate research. The development and health of children delivered under the related treatment should also be focused.
Conclusion
Intrauterine infusion of human chorionic gonadotropin, peripheral blood mononuclear cells, granulocyte-colony-stimulating factor, and platelet-rich plasma seems a promising strategy for improving the pregnant outcomes for infertile patients with recurrent implantation failure. Among these treatments, PRP may be the best. More researches are required to explore the effect of drug combinations and less commonly used drugs as well.
Availability of data and materials
No datasets were generated or analysed during the current study.
References
Adamson GD, de Mouzon J, Chambers GM, Zegers-Hochschild F, Mansour R, Ishihara O, Banker M, Dyer S. International committee for monitoring assisted reproductive technology: world report on assisted reproductive technology, 2011. Fertil Steril. 2018;110(6):1067–80.
Coughlan C, Ledger W, Wang Q, Liu F, Demirol A, Gurgan T, Cutting R, Ong K, Sallam H, Li TC. Recurrent implantation failure: definition and management. Reprod Biomed Online. 2014;28(1):14–38.
Kushnir VA, Barad DH, Albertini DF, Darmon SK, Gleicher N. Systematic review of worldwide trends in assisted reproductive technology 2004–2013. Reprod Biol Endocrinol. 2017;15(1):6.
Dyer S, Chambers GM, de Mouzon J, Nygren KG, Zegers-Hochschild F, Mansour R, Ishihara O, Banker M, Adamson GD. International committee for monitoring assisted reproductive technologies world report: assisted reproductive technology 2008, 2009 and 2010. Hum Reprod. 2016;31(7):1588–609.
Gnainsky Y, Dekel N, Granot I. Implantation: mutual activity of sex steroid hormones and the immune system guarantee the maternal-embryo interaction. Semin Reprod Med. 2014;32(5):337–45.
Cha J, Sun X, Dey SK. Mechanisms of implantation: strategies for successful pregnancy. Nat Med. 2012;18(12):1754–67.
Nimbkar-Joshi S, Rosario G, Katkam RR, Manjramkar DD, Metkari SM, Puri CP, Sachdeva G. Embryo-induced alterations in the molecular phenotype of primate endometrium. J Reprod Immunol. 2009;83(1–2):65–71.
Valdes CT, Schutt A, Simon C. Implantation failure of endometrial origin: it is not pathology, but our failure to synchronize the developing embryo with a receptive endometrium. Fertil Steril. 2017;108(1):15–8.
Macklon N. Recurrent implantation failure is a pathology with a specific transcriptomic signature. Fertil Steril. 2017;108(1):9–14.
Saxtorph MH, Hallager T, Persson G, Petersen KB, Eriksen JO, Larsen LG, Hviid TV, Macklon N. Assessing endometrial receptivity after recurrent implantation failure: a prospective controlled cohort study. Reprod Biomed Online. 2020;41(6):998–1006.
Wang L, Huang X, Li X, Lv F, He X, Pan Y, Wang L, Zhang X. Efficacy evaluation of low-dose aspirin in IVF/ICSI patients evidence from 13 RCTs: A systematic review and meta-analysis. Medicine (Baltimore). 2017;96(37):e7720.
Davar R, Janati S, Mohseni F, Khabazkhoob M, Asgari S. A comparison of the effects of transdermal estradiol and estradiol valerate on endometrial receptivity in frozen-thawed embryo transfer cycles: a randomized clinical trial. J Reprod Infertil. 2016;17(2):97–103.
Zhang L, Xu WH, Fu XH, Huang QX, Guo XY, Zhang L, Li SS, Zhu J, Shu J. Therapeutic role of granulocyte colony-stimulating factor (G-CSF) for infertile women under in vitro fertilization and embryo transfer (IVF-ET) treatment: a meta-analysis. Arch Gynecol Obstet. 2018;298(5):861–71.
Xie H, Zeng H, He D, Liu N. Effect of intrauterine perfusion of human chorionic gonadotropin before embryo transfer after two or more implantation failures: A systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2019;243:133–8.
Benkhalifa M, Joao F, Duval C, Montjean D, Bouricha M, Cabry R, Belanger M-C, Bahri H, Miron P, Benkhalifa M. Endometrium Immunomodulation to Prevent Recurrent Implantation Failure in Assisted Reproductive Technology. Int J Mol Sci. 2022;23(21):12787.
Pourmoghadam Z, Abdolmohammadi-Vahid S, Pashazadeh F, Aghebati-Maleki L, Ansari F, Yousefi M. Efficacy of intrauterine administration of autologous peripheral blood mononuclear cells on the pregnancy outcomes in patients with recurrent implantation failure: A systematic review and meta-analysis. J Reprod Immunol. 2020;137:103077.
Aleyasin A, Abediasl Z, Nazari A, Sheikh M. Granulocyte colony-stimulating factor in repeated IVF failure, a randomized trial. Reproduction (Cambridge, England). 2016;151(6):637–42.
Kamath MS, Kirubakaran R, Sunkara SK. Granulocyte-colony stimulating factor administration for subfertile women undergoing assisted reproduction. Cochrane Database Syst Rev. 2020;1(1):CD013226.
Kalem Z, Namli Kalem M, Bakirarar B, Kent E, Makrigiannakis A, Gurgan T. Intrauterine G-CSF administration in recurrent implantation failure (RIF): an Rct. Sci Rep. 2020;10(1):5139.
Danhof NA, Wang R, van Wely M, van der Veen F, Mol BWJ, Mochtar MH. IUI for unexplained infertility-a network meta-analysis. Hum Reprod Update. 2020;26(1):1–15.
Wu H, Zhang S, Lin X, Wang S, Zhou P. Luteal phase support for in vitro fertilization/intracytoplasmic sperm injection fresh cycles: a systematic review and network meta-analysis. Reprod Biol Endocrinol. 2021;19(1):103.
Zhang Y, Zhang C, Shu J, Guo J, Chang HM, Leung PCK, Sheng JZ, Huang H. Adjuvant treatment strategies in ovarian stimulation for poor responders undergoing IVF: a systematic review and network meta-analysis. Hum Reprod Update. 2020;26(2):247–63.
Wang Z, Ren CE, Ren W, Shi H. The frozen⁃thawed embryo transfer(FET) outcomes of the patients with repeated embryo implantationfailure after intrauterine injection of dexamethasone sodium phosphate. J Prac Med. 2021;37(08):1064–7. %@ 1006-5725 %L 1044-1193/R %W CNKI.
Santibanez A, Garcia J, Pashkova O, Colin O, Castellanos G, Sanchez AP, De la Jara JF. Effect of intrauterine injection of human chorionic gonadotropin before embryo transfer on clinical pregnancy rates from in vitro fertilisation cycles: a prospective study. Reprod Biol Endocrinol. 2014;12:9.
Huang P, Wei L, Li X. A study of intrauterine infusion of human chorionic gonadotropin (hCG) before frozen-thawed embryo transfer after two or more implantation failures. Gynecol Endocrinol. 2017;33(1):67–9.
Shanfei Z, Wenling Z, Xinru G. Effects of intrauterine infusion of HCG on pregnancy outcome in patients with repeated implantion failure in freeze-thaw embryo transfer cycles. J Chin Prescription Drug. 2020;18(11):8–10. %@ 1671-1945X %L 1644-1549/T %W CNKI.
Wang M, Deng H, Hong Y. Intrauterine injection of human chorionic gonadotropin improves pregnancy outcome in patients with repeated implantation failure in frozen-thawed embryo transfer. J Cent South Univ (Med Sci). 2019;44(11):1247–51. %@ 1672-7347 %L 1243-1427/R %W CNKI.
Nobijari FF, Arefi SS, Moini A, Taheripanah R, Fazeli E, Kharazi H, Hosseini SZ, Hosseini A, Valojerdi MR, Copin H, et al. Endometrium immunomodulation by intrauterine insemination administration of treated peripheral blood mononuclear cell prior frozen/thawed embryos in patients with repeated implantation failure. Zygote. 2019;27(4):214–8.
Yu N, Zhang B, Xu M, Wang S, Liu R, Wu J, Yang J, Feng L. Intrauterine administration of autologous peripheral blood mononuclear cells (PBMCs) activated by HCG improves the implantation and pregnancy rates in patients with repeated implantation failure: a prospective randomized study. Am J Reprod Immunol. 2016;76(3):212–6.
Madkour A, Bouamoud N, Louanjli N, Kaarouch I, Copin H, Benkhalifa M, Sefrioui O. Intrauterine insemination of cultured peripheral blood mononuclear cells prior to embryo transfer improves clinical outcome for patients with repeated implantation failures. Zygote. 2016;24(1):58–69.
Xu H, Tan X-J, Huang X-H. Effect of intrauterine perfusion of peripheral blood mononuclear cells on patients with repeated implant failure. J Chin Prescription Drug. 2021;19(06):172–4.
Lei L, Zhang Y, Lu L. Effect of intrauterine perfusion of autologous peripheral blood mononuclear cell of women with repeated implantation failure in IVF-ET on their pregnancy outcomes. Chin J Fam Plan. 2021;29(02):251–4. %@ 1004-8189 %L 1011-4550/R %W CNKI.
Zhao H. Analysis of the effect of lymphocyte on pregnancy outcome in patients with repeated implant failure. Clin Res. 2020;28(10):1–2. %@ 2096-1278 %L 2061-1502/R %W CNKI.
Xiang Y, Yu G, Song Y, Li Y, Wan L, Ma L, Tan L. Effects of autologous peripheral blood mononuclear cells on endometrial receptivity marker molecules and pregnancy outcome after freeze-thaw embryo transfer in patients with repeated implantation failure. J Pract Obstet Gynecol. 2019;35(03):347–51. %@ 1005-2216 %L 1021-1332/R.
Lu X, Qiao P, Peng Y, Shi H, Ren C. Pregnancy outcomes and serum TNF-αlevels in patients withrepeated implantation failure treated with intrauterine perfusion of PBMCs. Acta Acad Med Weifang. 2019;41(03):25–227. %@ 1004-3101 %L 1037-1195/R.
Lin Q, Lian R, Chen X, Liu C, Diao L, Huang C, Chen X, Zeng Y, Liang P. Effect of intrauterine administration of peripheral blood mononuclear cells on the pregnancy outcome of patients with recurrent implantation failure. J Reprod Med. 2013;22(12):922–6. %@ 1004-3845 %L 1011-4645/R %W CNKI.
Davari-Tanha F, Shahrokh Tehraninejad E, Ghazi M, Shahraki Z. The role of G-CSF in recurrent implantation failure: A randomized double blind placebo control trial. Int J Reprod Biomed. 2016;14(12):737–42.
Pourmoghadam Z, Soltani-Zangbar MS, Sheikhansari G, Azizi R, Eghbal-Fard S, Mohammadi H, Siahmansouri H, Aghebati-Maleki L, Danaii S, Mehdizadeh A, et al. Intrauterine administration of autologous hCG- activated peripheral blood mononuclear cells improves pregnancy outcomes in patients with recurrent implantation failure; A double-blind, randomized control trial study. J Reprod Immunol. 2020;142:103182.
Li J, Yang L, Liu J, Huang Y, Xu D, Hu S, Xue J. Effect of intrauterine infusion of human autologous PBMC on the pregnancy outcome of FET in patients with recurrent implantation failure. Mod Diagn Treat. 2022;33(05):636–8. %@ 1001-8174 %L 1036-1160/R %W CNKI.
Huang P, Yao C, Wei L, Lin Z. The intrauterine perfusion of granulocyte-colony stimulating factor (G-CSF) before frozen-thawed embryo transfer in patients with two or more implantation failures. Hum Fertil. 2022;25(2):301–5.
Eftekhar M, Miraj S, Mojtahedi MF, Neghab N. Efficacy of Intrauterine infusion of granulocyte colony stimulating factor on patients with history of implantation failure: A randomized control trial. Int J Reprod BioMed. 2016;14:687–90.
Karimi A, Mokhtar S, Sadeghi MR, Zafardoust S, Ataei M, Ghoodjani A, Mohazzab A, Nazari F, Ansaripour S. May intrauterine granulocyte colony stimulating factor improve clinical & ongoing pregnancy & live birth rates in unexplained repeated implantation failure patients? a randomized clinical trial. EJMCM. 2020;7(2):82–7.
Xia T, Shao X, Wei H, Liu Y, Li D, Yin X, Wang L. Evaluation of effect of intrauterine infusion of granulocyte colony stimulating factor in patients with unexplained repeated implantation failure. J Reprod Med. 2022;31(01):38–44.
Zhang X, Xu H, Luo Y, Zhu X, Dong M, Liu F. Effect of intrauterine perfusion of granulocyte stimulating factor on the pregnancy outcomes and immunestate in patients with recurrent implantation failure. J Pract Med. 2020;36(13):1770–3.
He J, Chen C, Zhou H. Granulocyte colony-stimulating factor for Repeated Implantation Failure after Embryo Transfer. Pract Clin Med. 2015;16(02):64–7. %@ 1009-8194 %L 1036-1242/R.
Zamaniyan M, Peyvandi S, HeidaryanGorji H, Moradi S, Jamal J, Yahya Poor Aghmashhadi F, Hossein Mohammadi M. Effect of platelet-rich plasma on pregnancy outcomes in infertile women with recurrent implantation failure: a randomized controlled trial. Gynecol Endocrinol. 2021;37(2):141–5.
Bakhsh AS, Maleki N, Sadeghi MR, SadeghiTabar A, Tavakoli M, Zafardoust S, Karimi A, Askari S, Jouhari S, Mohammadzadeh A. Effects of autologous platelet-rich plasma in women with repeated implantation failure undergoing assisted reproduction. JBRA Assist Reprod. 2022;26(1):84–7.
Kusumi M, Ihana T, Kurosawa T, Ohashi Y, Tsutsumi O. Intrauterine administration of platelet-rich plasma improves embryo implantation by increasing the endometrial thickness in women with repeated implantation failure: A single-arm self-controlled trial. Reprod Med Biol. 2020;19(4):350–6.
Nazari L, Salehpour S, Hosseini MS, Hashemi Moghanjoughi P. The effects of autologous platelet-rich plasma in repeated implantation failure: a randomized controlled trial. Hum Fertil (Camb). 2020;23(3):209–13.
Nazari L, Salehpour S, Hosseini S, Sheibani S, Hosseinirad H. The effects of autologous platelet-rich plasma on pregnancy outcomes in repeated implantation failure patients undergoing frozen embryo transfer: a randomized controlled trial. Reprod Sci. 2021;29(3):993–1000.
Baybordi E, Mohseni J, Mosapour P. The effect of platelet-rich plasma on the improvement of pregnancy results in repeated implantation failure: A randomized controlled trial. Int J Reprod Biomed. 2022;20(9):753–60.
Salehpour LNS, Hosseini MS, Moghanjoughi PH. The effects of autologous platelet-rich plasma in repeated implantation failure: a randomized controlled trial. Hum Fertil. 2020;23(3):209–13.
Zargar M, Pazhouhanfar R, Najafian M, Choghakabodi PM. Effects of intrauterine autologous platelet-rich plasma infusions on outcomes in women with repetitive in vitro fertilization failures: a prospective randomized study. Clin Exp Obstet Gynecol. 2021;48(1):179–84.
Liu K, Gao S, Li L, Zhao X, Guo Y, Liu Y, Zhang X. Role of platelet-rich plasma in improving pregnancy outcomes in patients with repeated implantation failure. Prog Obstet Gynecol. 2021;30(01):42–44+49 %@ 1004–7379 %L 1037–1211/R %U. https://kns.cnki.net/kcms/detail/1037.1211.R.20201225.20201753.20201013.html.
Hu X, Ma H, Wang W. Application value of autologous platelet-enriched plasma intrauterine perfusion before freeze-thaw embryo transplantation in patients with repeated implantation failure. Electron J Pract Gynecol Endocrinol. 2020;7(24):43–4. %@ 2095-8803 %L 2011-9356/R.
Hu X, Ma H, Wang W. Application of autologous platelet-enriched plasma intrauterine perfusion in patients with repeated implantation failure. Electron J Pract Gynecol Endocrinol. 2019;6(32):42–3. %@ 2095-8803 %L 2011-9356/R.
Bakry MS, Eldesouky E, Alghazaly MM, Farag E, Sultan EEK, Elazzazy H, Mohamed A, Ali SMS, Anwar A, Elrashedy AA, et al. Granulocyte colony stimulating factor versus human chorionic gonadotropin for recurrent implantation failure in intra cytoplasmic sperm injection: a randomized clinical trial. BMC Pregnancy Childbirth. 2022;22(1):881.
Torky H, El-Desouky E-S, El-Baz A, Aly R, El-Taher O, Shata A, Hussein A, Marie H, Deif O, Eldemery A, et al. Effect of intra uterine granulocyte colony stimulating factor vs. human chorionic gonadotropin at ovum pick up day on pregnancy rate in IVF/ICSI cases with recurrent implantation failure. J Bra De Reprod Assist. 2022;26(2):274–9.
Xu D, Li L, Wang A, Yang L, Li J, Xiang Y. Effect of different intrauterine perfusion schemes on pregnancy outcome of frozen thawed embryo transfer in patients with repeated implantation failure. Chin Med Innov. 2023;20(18):146–51. %@ 1674-4985 %L 1611-5784/R %W CNKI.
Hou Z, Chen L, Chen L, Song J, Meng Y. A Study of Intrauterine Infusion of Growth Hormone and Granulocyte Colony-Stimulating Factor in the Treatment of Repeated Implantation Failure. J Pract Obstet Gynecol. 2022;38(06):460–5.
Pin-xiu H, Li-hong W, Mei Q, Fan Z, Xinling L. Different trauterine perfusion in patients with frozen-thawed embryo transfer after implantation failure. Chin J Eugen Genet. 2018;26(04):107–11.
Li S, Wang J, Cheng Y, Zhou D, Yin T, Xu W, Yu N, Yang J. Intrauterine administration of hCG-activated autologous human peripheral blood mononuclear cells (PBMC) promotes live birth rates in frozen/thawed embryo transfer cycles of patients with repeated implantation failure. J Reprod Immunol. 2017;119:15–22.
Makrigiannakis A, Vrekoussis T, Makrygiannakis F, Ruso H, Kalantaridou SN, Gurgan T. Intrauterine CRH-treated PBMC in repeated implantation failure. Eur J Clin Invest. 2019;49(5):e13084.
Liu N, Qi J, Wang Y. Effect of intrauterine perfusion with rH CG during FET after RIF on pregnancy outcome. Chin J Hum Sex. 2022;31(04):71–5. %@ 1672-1993 %L 1611-4982/R %W CNKI.
Bing L, Jieheng L, Jiaxin H, Jinzhuang Z, Yuling L. The impact of intrauterine perfusion of embryo culture supernatant on frozen-thawed embryo tansfer outcome. Guangzhou Med. 2020;51(05):90–2. %@ 1000-8535 %L 1044-1199/R %W CNKI.
Cao F, Huang X, Yu Chunmei, Haiyan Yang , Yufeng Wang , Wei Zhou , Juanfen Wu, Chen L. Effect of intrauterine infusion of lymphocytes on the outcome of pregnancy in patients with recurrent implantation failure. Chin Gen Pract. 2015;18(35):4317–4319+4329 %@ 1007–9572 %L 4313–1222/R %W CNKI.
Tehraninejad ES, Kashani NG, Hosseini A, Tarafdari A. Autologous platelet-rich plasma infusion does not improve pregnancy outcomes in frozen embryo transfer cycles in women with history of repeated implantation failure without thin endometrium. J Obstet Gynaecol Res. 2021;47(1):147–51.
Dieamant F, Vagnini LD, Petersen CG, Mauri AL, Renzi A, Petersen B, Mattila MC, Nicoletti A, Oliveira JBA, Baruffi R, et al. New therapeutic protocol for improvement of endometrial receptivity (PRIMER) for patients with recurrent implantation failure (RIF) - A pilot study. JBRA Assist Reprod. 2019;23(3):250–4.
Ma N, Zhou J, Zhou Z, Lin F, Li Y, Lu W. Effect of autologous platelet-enriched plasma intrauterine perfusion on pregnancy outcome of thawed embryo transfer in aged patients with endometritis. Chin J Hum Sex. 2023;23(02):158–161+168 %@ 1672–2124 %L 1611–4975/R.
Aghajanzadeh F, Esmaeilzadeh S, Basirat Z, Mahouti T, Heidari FN, Golsorkhtabaramiri M. Using autologous intrauterine platelet-rich plasma to improve the reproductive outcomes of women with recurrent implantation failure. JBRA Assist Reprod. 2020;24(1):30–3.
Mei J, Yan Y, Jiang R, Zhu Y-C, Ding L, Sun H. Clinical outcome of intrauterine administration of peripheral mononuclear cells or human chorionic gonadotropin in unexplained implantation failure. Am J Reprod Immunol. 2022;87(5):e13529.
Ershadi S, Noori N, Dashipoor A, Ghasemi M, Shamsa N. Evaluation of the effect of intrauterine injection of platelet-rich plasma on the pregnancy rate of patients with a history of implantation failure in the in vitro fertilization cycle. J Fam Med Prim Care. 2022;11(5):2162–6.
Zhang H, Fu Z, Liu Q, Zhao T, Wang P, Li X. Effect of hCG-activated autologous PBMCs intrauterine administration for repeated implantation failure. J Pract Obstet Gynecol. 2019;35(05):357–61. %@ 1003-6946 %L 1051-1145/R %W CNKI.
Liu X, Ma D, Wang W, Qu Q, Zhang N, Wang X, Fang J, Ma Z, Hao C. Intrauterine administration of human chorionic gonadotropin improves the live birth rates of patients with repeated implantation failure in frozen-thawed blastocyst transfer cycles by increasing the percentage of peripheral regulatory T cells. Arch Gynecol Obstet. 2019;299(4):1165–72.
Noushin MA, Ashraf M, Thunga C, Singh S, Singh S, Basheer R, Ashraf R, Jayaprakasan K. A comparative evaluation of subendometrial and intrauterine platelet-rich plasma treatment for women with recurrent implantation failure. F&S Sci. 2021;2(3):295–302.
Okitsu O, Kiyokawa M, Oda T, Miyake K, Sato Y, Fujiwara H. Intrauterine administration of autologous peripheral blood mononuclear cells increases clinical pregnancy rates in frozen/thawed embryo transfer cycles of patients with repeated implantation failure. J Reprod Immunol. 2013;92(1–2):82–7.
Rahmati M, Petitbarat M, Dubanchet S, Bensussan A, Chaouat G, Ledee N. Granulocyte-colony stimulating factor related pathways tested on an endometrial ex-vivo model. PLoS One. 2014;9(9):e102286.
Rutella S, Zavala F, Danese S, Kared H, Leone G. Granulocyte colony-stimulating factor: a novel mediator of T cell tolerance. J Immunol. 2005;175(11):7085–91.
Licht P, Russu V, Lehmeyer S, Wildt L. Molecular aspects of direct LH/hCG effects on human endometrium–lessons from intrauterine microdialysis in the human female in vivo. Reprod Biol. 2001;1(1):10–9.
Sharara FI, Lelea LL, Rahman S, Klebanoff JS, Moawad GN. A narrative review of platelet-rich plasma (PRP) in reproductive medicine. J Assist Reprod Genet. 2021;38(5):1003–12.
Fu L-L, Xu Y, Yan J, Zhang X-Y, Li D-D, Zheng L-W. Efficacy of granulocyte colony-stimulating factor for infertility undergoing IVF: a systematic review and meta-analysis. Reprod Biol Endocrinol. 2023;21(1):34.
Li M, Kang Y, Wang Q, Yan L. Efficacy of autologous intrauterine infusion of platelet-rich plasma in patients with unexplained repeated implantation failures in embryo transfer: a systematic review and meta-analysis. J Clin Med. 2022;11(22):6753.
Hu S, Jin Z, Tang Q. Effects of intrauterine infusion of autologous platelet-rich plasma in women undergoing treatment with assisted reproductive technology: a meta-analysis of randomized controlled trials. Geburtshilfe Und Frauenheilkunde. 2022;83(4):453–62.
Soliman A, Elsonbaty S, Saleh Y, Hegazy D, Faragallah HM. How autologous platelet-rich plasma affects pregnancy and birth outcomes in women with repeated embryo implantation failure: A prisma-compliant meta-analysis. Turk J Obstet Gynecol. 2023;20(2):154–63.
Kong X, Tang G, Liu Y, Zheng Z, Li Y, Yan F. Efficacy of intrauterine infusion therapy before embryo transfer in recurrent implantation failure: A systematic review and network meta-analysis. J Reprod Immunol. 2023;156:103819.
Jones KP, Warnock SH, Urry RL, Edwin SS, Mitchell MD. Immunosuppressive activity and alpha interferon concentrations in human embryo culture media as an index of potential for successful implantation. Fertil Steril. 1992;57(3):637–40.
Sheth KV, Roca GL, Al-Sedairy ST, Parhar RS, Hamilton CJ, Al-Abdul Jabbar F. Prediction of successful embryo implantation by measuring interleukin-1-alpha and immunosuppressive factor(s) in preimplantation embryo culture fluid. Fertil Steril. 1991;55(5):952–7.
Inés Barañao R, Piazza A, Rumi LS. Polak de Fried E: Interleukin-1 beta levels in human embryo culture supernatants and their predictive value for pregnancy. Early Human Dev. 1997;48(1–2):71–80.
Funding
This study was supported by the Hubei Provincial Health Commission (WJ2023M157).
Author information
Authors and Affiliations
Contributions
Study design: XZY, QX, XZQ. Data collection: QX, XZQ, YLL. Data analysis: QX, XZQ, YLL. Manuscript writing: QX, XZQ, YLL, XZY. Approval for publication of final version: XZY, QX, XZQ, YLL.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1.
Risk of bias assessment. a. Risk of bias summary; b. Risk of bias graph. Figure S2. Forest plot of the live birth in direct pair-wise meta-analysis. Figure S3. Network plots of eligible comparisons for secondary outcomes: clinical pregnancy rate. a. Live birth; b. Embryo implantation; c. Chemical pregnancy; d. Miscarriage. Figure S4. Forest plot of the embryo implantation in direct pair-wise meta-analysis. Figure S5. Forest plot of the chemical pregnancy in direct pair-wise meta-analysis. Figure S6. Forest plot of the miscarriage in direct pair-wise meta-analysis. Figure S7. Funnel plot of the pregnancy outcomes. Figure S8. Subgroup analysis of forest plot of the clinical pregnancy in the direct pair-wise meta-analysis by English researches. Figure S9. Subgroup analysis of forest plot of the clinical pregnancy in the direct pair-wise meta-analysis by Chinese researches. Supplemental Table S1. Characteristics of studies included in meta-analyses. Supplemental Table S2. Risk of bias assessment of the other prospective studies. Supplemental Table S3. Network meta-analysis for live birth comparing diverse uterine infusion strategies. Supplemental Table S4. Network meta-analysis for implantation comparing diverse uterine infusion strategies. Supplemental Table S5. Network meta-analysis for chemical pregnancy comparing diverse uterine infusion strategies. Supplemental Table S6. Network meta-analysis for miscarriage comparing diverse uterine infusion strategies. Supplemental Table S7. Subgroup analysis of network meta-analysis for clinical pregnancy by English researches. Supplemental Table S8. Subgroup analysis of network meta-analysis for clinical pregnancy by Chinese researches.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Xie, Q., Quan, X., Lan, Y. et al. Uterine infusion strategies for infertile patients with recurrent implantation failure: a systematic review and network meta-analysis. Reprod Biol Endocrinol 22, 44 (2024). https://doi.org/10.1186/s12958-024-01221-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12958-024-01221-x