Abstract
Objectives
To investigate the clinicopathological characteristics and prognosis of patients with primary sarcoma of the uterine cervix.
Methods
We identified all patients with primary cervical sarcomas treated at our institution from 2002 to 2020 and analyzed the clinicopathological characteristics and prognosis.
Results
34 patients were identified, 7 (20.6%) patients had leiomyosarcoma, 6 (17.6%) had carcinosarcoma, 5 (14.7%) had Ewing sarcoma, 4 (11.8%) had rhabdomyosarcoma, 4 (11.8%) had undifferentiated sarcoma, 2 (5.9%) had adenosarcoma, 2 (5.9%) had endometrial stromal sarcoma, 1 (2.9%) had dermatofibrosarcoma protuberans, 1 (2.9%) had alveolar soft tissue sarcoma and 2 (5.9%) had sarcoma not otherwise specified. The median age of the whole patients was 43.5 years (range, 13–63). The median age of patients with Ewing sarcoma or rhabdomyosarcoma was 22 years (range, 13–39) and 17 years (range, 13–36 years), respectively. The distribution by stage was: stage I in 21 (61.8%) patients, stage II in 4 (11.8%), stage III in 6 (17.6%) and stage IV in 3 (8.8%). Overall, 30 patients (88.2%) received surgical treatment. The median follow-up was 33.3 months (range 3.6–187.3 months). 11 patients died within 2 years after diagnosis, most of them were patients with carcinosarcoma or undifferentiated sarcoma (45.5%, 5/11). In the entire cohort, 2- and 5-year OS were 67.2% and 56.9%, respectively. 5-year OS was 25.0% for undifferentiated sarcoma, 50.0% for rhabdomyosarcoma, 50.0% for carcinosarcoma, 53.3% for Ewing sarcoma, 57.1% for leiomyosarcoma.
Conclusion
Cervical sarcomas are rare neoplasms with multiple histological subtypes and follow an aggressive course. Prognosis may be associated with tumor histology and stage.
Similar content being viewed by others
Introduction
Cervical cancer is the fourth most common female malignancy worldwide and represents a major global health challenge [1]. According to the WHO histological classification of tumours of the uterine cervix, squamous cell carcinomas account for 70–80% of cervical cancers and adenocarcinomas for 20–25% [2]. Cervical sarcomas are ultra-rare neoplasms with multiple histological subtypes and follow an aggressive course. The common subtype of cervical sarcomas includes leiomyosarcoma, carcinosarcoma or malignant mixed Müllerian tumor, Ewing sarcoma, rhabdomyosarcoma, undifferentiated sarcoma, adenosarcoma, and endometrial stromal sarcoma [3].
Because of the low incidence and the lack of prospective studies, it is very difficult to reach conclusions as to the best disease management recommendations for cervical sarcoma. Most clinicopathological data are derived from case reports and retrospective studies of a few cases.
We performed a retrospective study to investigate the clinicopathological characteristics and prognosis of patients diagnosed with primary sarcoma of the uterine cervix in our center.
Patients and methods
Patients
Following Institutional Review Board approval, we performed a retrospective analysis of all patients diagnosed with primary sarcoma of the uterine cervix from January 1, 2002 to January 1, 2020 who received treatment in the Department of Gynecological Oncology of Cancer Hospital, Chinese Academy of Medical Sciences, National Cancer Center.
Only patients with a diagnosis of cervical sarcoma confirmed by an experienced gynecologic pathologist in our hospital were included. The full medical records of these patients were included in this study. Clinical and pathologic variables, treatment modalities, and outcomes were assessed. Stage was retrospectively assigned using the International Federation of Gynecology and Obstetrics (FIGO) 2018 staging system for cancer of the cervix uteri [4].
Statistical analyses
For the survival analyses, Overall survival (OS) was defined as the time from the date of diagnosis to death for which cervical sarcoma was the primary or underlying cause. Survival was estimated using the Kaplan–Meier product-limit method, and differences were tested for statistical significance using the log-rank test. Two-sided P values less than 0.05 were considered to be statistically significant. All analyses were performed using the SPSS Statistics20.0 software.
Results
Patient characteristics
Thirty-four patients were identified. Of these patients, 7 (20.6%) patients had leiomyosarcoma, 6 (17.6%) patients had carcinosarcoma, 5 (14.7%) patients had Ewing sarcoma, 4 (11.8%) patients had rhabdomyosarcoma, 4 (11.8%) patients had undifferentiated sarcoma, 2 (5.9%) patients had adenosarcoma, 2 (5.9%) patients had endometrial stromal sarcoma, 1 (2.9%) patient had alveolar soft tissue sarcoma, 1 (2.9%) patient had dermatofibrosarcoma protuberans, and 2 patients (5.9%) had sarcoma not otherwise specified (Table 1). Immunohistochemistry results for patients with different sarcoma subtypes were listed in Supplementary Table 1. Photographs (10X, 20X) with haematoxylin eosin staining of representative histomorphology from different sarcoma subtypes were showed in Supplementary Fig. 1.
The median age of the whole cohort was 43.5 years (range, 13–63 years). The median age of patients with Ewing sarcoma or rhabdomyosarcoma was 22 years (range, 13–39) and 17 years (range, 13–36), respectively. All patients with Ewing sarcoma or rhabdomyosarcoma or endometrial stromal sarcoma were diagnosed before 40 years. But the median age of patients with carcinosarcoma or leiomyosarcoma was older, which was 54 years (range, 28–61) and 48 years (range, 38–59), respectively. Twenty-four (70.6%) patients presented with vaginal bleeding which was the most common primary symptoms. 7 (20.6%) patients were diagnosed by physical examination.
The proportion of patients with smaller tumor (<5 cm) was 41.2%, compared to 38.2% for 5 to 10 cm, 20.6% for ≥ 10 cm (Table 1). All patients with Ewing sarcoma had lager tumors (≥ 5 cm). The 2018 distribution by stage was: stage I in 21 (61.8%) patients, stage II in 4 (11.8%) patients, stage III in 6 (17.6%) patients and stage IV in 3 (8.8%) patients (Table 1).
Treatment
Overall, 30 patients (88.2%) received surgical treatment. Other 4 patients (11.8%) with advanced disease received chemotherapy with (n = 3) or without (n = 1) radiotherapy.
Surgical treatment
7 (23.3%) patients received neoadjuvant chemotherapy because of larger tumors. The neoadjuvant chemotherapy regimens included: paclitaxel/platinum (3, 42.9%), doxorubicin/ifosfamide (2, 28.6%), paclitaxel/ifosfamide/cisplatin (1, 14.3%). doxorubicin/ifosfamide/cisplatin (1, 14.3%). For patients who underwent surgical treatment, 15 patients (50.0%) underwent total hysterectomy, 14 patients (46.7%) underwent radical hysterectomy, 1 patient (3.3%) underwent primary tumor resection (Table 1). Of these patients, 17 patients (60.7%) had outer third cervical stromal invasion and 18 patients (62.1%) had lymphvascular space invasion.
14 (46.7%) patients underwent pelvic lymphadenectomy, 6 (20.0%) underwent pre-aortic lymphadenectomy, as part of the initial surgical treatment. 21.4% (3/14) and 16.7% (1/6) patients had pelvic or pre-aortic lymph node metastasis, respectively (Table 1).
Adjuvant therapy
After surgical treatment, 66.7% (20/30) of them received adjuvant chemotherapy. 13.3% (4/30) of them received pelvic external beam radiotherapy (EBRT), and 13.3% (4/30) patients received vaginal brachytherapy.
The adjuvant chemotherapy regimens included: doxorubicin/ifosfamide/cisplatin (5, 25.0%), gemcitabine/docetaxel (2, 10.0%), doxorubicin/ifosfamide (4, 20.0%), paclitaxel/platinum (3, 15.0%), paclitaxel/ifosfamide (3, 15.0%), doxorubicin/cisplatin (1, 5.0%), etoposide/cisplatin (2, 10.0%).
Survival analysis
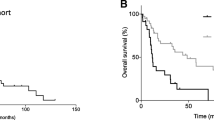
The median follow-up was 33.3 months (range 3.6–187.3 months). 15 patients died during follow up, and 11 patients died within 2 years after diagnosis, most of them were patients with carcinosarcoma or undifferentiated sarcoma (45.5%, 5/11). 4 patients with advanced stage or metastatic cervical sarcoma received systematic therapy without surgical treatment. All of them died within 1 year after diagnosis. In the entire cohort, 2-year overall survival (OS) and 5-year OS were 67.2% and 56.9%, respectively (Fig. 1).
For patients with different pathologic subtype, 5-year OS was 25.0% for undifferentiated sarcoma, 50.0% for rhabdomyosarcoma, 50.0% for carcinosarcoma, 53.3% for Ewing sarcoma, 57.1% for leiomyosarcoma, and 75.0% for others, in the entire cohort (Fig. 2). The 5-year OS for stage I-II and III-IV patients were 62.3% and 41.7%, respectively (Fig. 3). The difference was not significant since the small sample size. For patients who received surgical treatment (n = 30), patients with outer third cervical stromal invasion, larger tumor size, and stage III-IV had a non-significantly worse survival (Table 2).
Discussion
Primary sarcomas of the uterine cervix are rare group of tumors with an aggressive behavior and poor outcomes. In the present study, 34 patients with cervical sarcoma treated at our institution from 2001 to 2020 were analyzed. Our series is one of the largest to report on surgical and treatment outcomes for patients with cervical sarcoma in a single center.
Cervical sarcomas are ultra-rare tumors including multiple histological subtypes. Carcinosarcoma is the most common histologic variant and account for 40% of the cervical sarcomas [4]. In our present study, the most common histology of cervical sarcoma was leiomyosarcoma, followed by carcinosarcoma, Ewing sarcoma, rhabdomyosarcoma, undifferentiated sarcoma. The present histology distribution is consistent with previous reported studies.
The age at diagnosis varied according the pathological subtype for cervical sarcoma. Most patients were diagnosed at an early age, especially for patients with Ewing sarcoma or rhabdomyosarcoma or endometrial stromal sarcoma, which were diagnosed before 40 years in our study. Other studies have demonstrated that among cervical cancer patients aged ≤ 25 years, 16 (26.7%) were diagnosed with cervical sarcoma [5]. However, among patients with cervical carcinosarcomas or leiomyosarcoma, the median age at diagnosis was older in the present study. Patients with cervical sarcoma have larger tumors compared with squamous cell carcinomas and adenocarcinomas [6]. We found that more than half of patients had a tumor ≥ 5 cm. Patients diagnosed before 40 years and patients with Ewing sarcoma or rhabdomyosarcoma were more likely to have a large tumor.
Because of their low incidence and the lack of prospective studies, it is very difficult to reach conclusions as to the best disease management recommendations for cervical sarcomas. The treatment of cervical sarcomas is not within the scope of National Comprehensive Cancer Network (NCCN) Guidelines. Multidisciplinary team (MDT) should be recommended for these patients [7]. Cervical sarcoma should be treated with a multimodality management protocol.
Total abdominal hysterectomy/ radical hysterectomy with bilateral salpingo-oophorectomy is the main treatment for patients having disease confined to the cervix. Radical hysterectomy is the important treatment for cervical cancer, but it may not be easily implemented in patients with cervical sarcoma because of the larger tumor size. Emerging evidence suggested that the type of hysterectomy does not have an effect on oncologic outcome in cervical leiomyosarcoma [8]. We found no association between the type of hysterectomy and survival in our cohort. Since most patients with cervical sarcoma are diagnosed at an early age and ovarian involvement is rare, the role of oophorectomy is controversial. For patients diagnosed before 40 years, only 20% of them received oophorectomy in our study.
Neoadjuvant chemotherapy may have impact on down-staging of the tumor to improve the radical curability and safety of surgery; and inhibiting of micrometastasis and distant metastasis [9]. Patients with cervical sarcoma have larger tumors. Neoadjuvant may be a suitable choice for younger patients with a larger tumor. In the present study, 23.3% patients received neoadjuvant chemotherapy before surgical treatment. All these patients achieved a partial response after neoadjuvant chemotherapy and received a surgical treatment. The effect of postoperative treatment among patients who have undergone surgical resection is a subject for debate. Most patients in our cohort received adjuvant chemotherapy, especially for patients with aggressive subtype. Whether the adjuvant chemotherapy should be implemented or not may accord to the pathologic subtype.
The prognosis for women with cervical sarcomas is inferior to that of squamous cell and adenocarcinomas [3, 6]. Cervical sarcomas have an aggressive course. In our study, more than 60% patients had outer third cervical stromal invasion and lymphvascular space invasion. Cervical sarcomas have multiple histological subtypes and the survival rates tend to vary widely according to histological subtypes. We found patients with carcinosarcoma, rhabdomyosarcoma, or undifferentiated sarcoma, had a worsen survival compared with other subtypes, which was in accordance with other studies.
Primary cervical leiomyosarcomas constitute 21% of all cervical sarcomas [8, 10]. Hysterectomy should be the first choice of treatment in these patients according to other studies. Multimodality management may suitable for cervical leiomyosarcoma [11]. Undifferentiated sarcomas are a rare group of tumors with an aggressive behavior and poor outcomes. Survival of cervical undifferentiated sarcoma is extremely poor. We found 3 of 4 patients died within 6 months after diagnosis.
Grayson W et al. firstly report infection with human papillomaviruses (HPV) play an important role in the evolution of cervical carcinosarcoma [12]. Several studies suggest a metaplastic theory of histogenesis of cervical carcinosarcoma [12,13,14,15]. In the present study, 33.3% (2/6) patients with cervical carcinosarcoma had HPV infection (HPV type 16 and 55, respectively) and a low-grade squamous intraepithelial lesion. The pathogenesis of cervical carcinosarcoma should be explored in more studies. Furthermore, carcinosarcomas of the uterus are no longer considered sarcomas and may be treated more like a carcinoma [16]. Whether cervical carcinosarcoma should be treated like a carcinoma was unclear.
Ewing sarcomas are small round cell tumors presenting with different degrees of neuroectodermal differentiation. Most patients present with a larger cervical tumor and are diagnosed in children or young adults. Patients with Ewing sarcoma in our study also had a young age onset and a larger tumor. More than half of them had pelvic lymph node metastasis. Rhabdomyosarcomas also tend to occur in children and young women, but appear to have a better prognosis than sarcoma botryoides of the vagina and uterus [17]. Rhabdomyosarcomas comprise 50% of cervical sarcomas in very young women (≤ 25 years) [5]. Many of these started as polypoidal tumours and could be managed surgically. Therapy has recently inclined towards conservative treatment [17]. Adenosarcomas are composed of benign epithelial elements in combination with a malignant stromal component [18]. Patients with adenosarcomas may have a good survival.
There are two limitations to our study. Because of the rarity of cervical sarcoma, the sample size of our study was small. The current study was retrospective, and the primary treatment was not assigned at randomized. Therefore, caution is required when interpreting our results.
Conclusion
Primary sarcomas of the uterine cervix are rare and aggressive neoplasms. Multimodality therapy is the main treatment for cervical sarcoma. Neoadjuvant chemotherapy may be suitable for these patients. Prognosis vary widely according to histological subtypes.
Data availability
No datasets were generated or analysed during the current study.
Abbreviations
- FIGO:
-
International Federation of Gynecology and Obstetrics
- OS:
-
Overall survival
- NCCN:
-
National Comprehensive Cancer Network
- MDT:
-
Multidisciplinary team
- HPV:
-
Human papillomaviruses
- EBRT:
-
External beam radiotherapy
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Marth C, Landoni F, Mahner S, McCormack M, Gonzalez-Martin A, Colombo N, et al. Cervical cancer: ESMO Clinical Practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv72–83.
Albert A, Lee A, Allbright R, Vijayakumar S. Primary sarcoma of the cervix: an analysis of patient and tumor characteristics, treatment patterns, and outcomes. J Gynecol Oncol. 2020;31:e25.
Wright JD, Rosenblum K, Huettner PC, Mutch DG, Rader JS, Powell MA, et al. Cervical sarcomas: an analysis of incidence and outcome. Gynecol Oncol. 2005;99:348–51.
Kong Y, Zong L, Yang J, Wu M, Xiang Y. Cervical cancer in women aged 25 years or younger: a retrospective study. Cancer Manag Res. 2019;11:2051–8.
Bansal S, Lewin SN, Burke WM, Deutsch I, Sun X, Herzog TJ, et al. Sarcoma of the cervix: natural history and outcomes. Gynecol Oncol. 2010;118:134–8.
Reed N, Balega J, Barwick T, Buckley L, Burton K, Eminowicz G, et al. British Gynaecological Cancer Society (BGCS) cervical cancer guidelines: recommendations for practice. Eur J Obstet Gynecol Reprod Biol. 2021;256:433–65.
Kiliç C, Yuksel D, Cakir C, Turkmen O, Kimyon Comert G, Başaran D, et al. Primary leiomyosarcoma of the uterine cervix: report of 4 cases, systematic review, and meta-analysis. Tumori. 2020;106:413–23.
Bhatla N, Aoki D, Sharma DN, Sankaranarayanan R. Cancer of the cervix uteri: 2021 update. Int J Gynaecol Obstet. 2021;155(Suppl 1):28–44.
Toyoshima M, Okamura C, Niikura H, Ito K, Yaegashi N. Epithelioid leiomyosarcoma of the uterine cervix: a case report and review of the literature. Gynecol Oncol. 2005;97:957–60.
Jayaram VK, Parikshith J, Narayanan GS, Tiwari R, Veena R, Prathima S, et al. Multimodality management of leiomyosarcoma of the cervix. Ecancermedicalscience. 2018;12:830.
Grayson W, Taylor LF, Cooper K. Carcinosarcoma of the uterine cervix: a report of eight cases with immunohistochemical analysis and evaluation of human papillomavirus status. Am J Surg Pathol. 2001;25:338–47.
Lin C-P, Ho C-L, Shen M-R, Huang L-H, Chou C-Y. Evidence of human papillomavirus infection, enhanced phosphorylation of retinoblastoma protein, and decreased apoptosis in sarcomatoid squamous cell carcinoma of uterine cervix. Int J Gynecol Cancer. 2006;16:336–40.
Kumar M, Bahl A, Sharma DN, Agarwal S, Halanaik D, Kumar R, et al. Sarcomatoid squamous cell carcinoma of uterine cervix: pathology, imaging, and treatment. J Cancer Res Ther. 2008;4:39–41.
Hrudka J, Rosová B, Halaška MJ. Squamous cell carcinoma with sarcomatoid differentiation or carcinosarcoma of the uterine cervix associated with HPV33 infection: report of a rare case. Diagn Pathol. 2020;15:12.
Pezzicoli G, Moscaritolo F, Silvestris E, Silvestris F, Cormio G, Porta C, et al. Uterine carcinosarcoma: an overview. Crit Rev Oncol Hematol. 2021;163:103369.
Bernal KL, Fahmy L, Remmenga S, Bridge J, Baker J. Embryonal rhabdomyosarcoma (sarcoma botryoides) of the cervix presenting as a cervical polyp treated with fertility-sparing surgery and adjuvant chemotherapy. Gynecol Oncol. 2004;95:243–6.
Togami S, Kawamura T, Fukuda M, Yanazume S, Kamio M, Kobayashi H. Clinical management of uterine cervical mullerian adenosarcoma: a clinicopathological study of six cases and review of the literature. Taiwan J Obstet Gynecol. 2018;57:479–82.
Acknowledgements
We are indebted to all gynaecologists and others of Department of Gynecologic Oncology in our center for their support and their help in data collection.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Conception and design: Yuan, Yao; Development of methodology: Yuan, Yao; Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): Yuan, Li1, Li2, Yao; Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): Yuan, Li1, Li2, Yao; Writing, review, and/or revision of the manuscript: Yuan, Li1, Yao; Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): Li, Yao; Study supervision: Li2, Yao.Notes: Li1, Lihong Li; Li2, Ning Li.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was conducted in accordance with the regulations on patient confidentiality and the ethical standards of Declaration of Helsinki. Written informed consent was obtained from individual or guardian participants. This research was approved by the institutional review board of the Cancer Hospital, Chinese Academy of Medical Sciences.
Consent for publication
All authors made the final approval of the version to be published.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yuan, H., Li, L., Li, N. et al. Sarcoma of the uterine cervix: experience of a single center. World J Surg Onc 22, 104 (2024). https://doi.org/10.1186/s12957-024-03376-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12957-024-03376-8