Abstract
Background
Escherichia coli is an opportunistic pathogen which colonizes various host species. However, to what extent genetic lineages of E. coli are adapted or restricted to specific hosts and the genomic determinants of such adaptation or restriction is poorly understood.
Results
We randomly sampled E. coli isolates from four countries (Germany, UK, Spain, and Vietnam), obtained from five host species (human, pig, cattle, chicken, and wild boar) over 16 years, from both healthy and diseased hosts, to construct a collection of 1198 whole-genome sequenced E. coli isolates. We identified associations between specific E. coli lineages and the host from which they were isolated. A genome-wide association study (GWAS) identified several E. coli genes that were associated with human, cattle, or chicken hosts, whereas no genes associated with the pig host could be found. In silico characterization of nine contiguous genes (collectively designated as nan-9) associated with the human host indicated that these genes are involved in the metabolism of sialic acids (Sia). In contrast, the previously described sialic acid regulon known as sialoregulon (i.e. nanRATEK-yhcH, nanXY, and nanCMS) was not associated with any host species. In vitro growth experiments with a Δnan-9 E. coli mutant strain, using the sialic acids 5-N-acetylneuraminic acid (Neu5Ac) and N-glycolylneuraminic acid (Neu5Gc) as sole carbon source, showed impaired growth behaviour compared to the wild-type.
Conclusions
This study provides an extensive analysis of genetic determinants which may contribute to host specificity in E. coli. Our findings should inform risk analysis and epidemiological monitoring of (antimicrobial resistant) E. coli.
Similar content being viewed by others
Background
Escherichia coli is a Gram-negative bacterium that can colonize various hosts, including humans, cattle, chickens, and pigs [1]. Because of its host promiscuity, the E. coli species as a whole can act as a reservoir for genes encoding antimicrobial resistance (AMR) [2] that can be transmitted between different animal host species. The likelihood that E. coli and its AMR encoding genes persist in a new host after transmission depends on multiple factors [3, 4]. For example, small changes in metabolic pathways may enable particular E. coli clones within the species to colonize a certain host more efficiently [1]. Several studies have suggested that highly successful E. coli clones, such as those of sequence type 131 (ST131) [5, 6] or clones belonging to clonal complex 87 (ST58 and ST155), facilitate the spread of AMR in the human population [7], whilst other studies have shown that different lineages of AMR E. coli vary in their ability to spread [8]. These latter findings indicate that AMR genes, at least to some extent, can hitchhike on bacterial strains that are specifically equipped to colonize a given host. Beyond classical virulence or adhesion factors, genetic and functional traits defining different degrees of host adaptation [3, 9], thereby indirectly impacting on the spread of AMR between host species, have not been identified thus far.
Comparative genomics approaches have previously revealed the signatures of host-adaptation in bacterial genomes [10,11,12,13]. The emergence of large-scale bacterial genome-wide association studies (GWAS) allowed the detection of genes or genomic variants that are associated with antimicrobial resistance, pathogenicity, and host-adaptive traits [14,15,16]. Here, we have applied population-based bacterial GWAS to identify host-associated genomic determinants in a diverse panel of 1198 E. coli isolates, irrespective of their AMR profile. Isolates were recovered from five different host species, including healthy and diseased individuals from four different countries in two continents over 16 years. We analysed host colonization patterns associated with E. coli lineages and applied gene-based and k-mer-based bacterial GWAS to identify host-associated genomic determinants in the E. coli accessory genome.
Results
Data collection
After WGS quality control, sequences of 14 isolates were excluded because of poor quality. One additional isolate was excluded since this isolate was identified as Escherichia marmotae (formerly cryptic clade V) [17, 18], a species commonly mistaken for E. coli. Our final collection comprised 1198 E. coli whole-genome sequences with associated metadata (Additional file 2: Table S1), including 8 cryptic clade I isolates, which were included as E. coli based on the recommended species cut-off of 95–96% average nucleotide identity [17]. Our collection consisted of 22.1% (n = 265) cattle, 28.1% (n = 337) chicken, 27.3% (n = 327) human, 20.3% (n = 240) pigs, and 2.4% (n = 29) wild boar isolates (Additional file 1: Fig. S1A). Fifty-one percent (n = 612), 19.4% (n = 233), 14.5% (n = 174), and 14.9% (n = 179) of these isolates were from Germany, Spain, the UK, and Vietnam, respectively (Additional file 1: Fig. S1A). Chicken isolates were from all four countries, human isolates from Germany, the UK, and Vietnam; pig isolates from Germany, Spain, and Vietnam; and cattle isolates from Germany and Spain. Only Spain provided wild boar isolates. In total, 35.5% (n = 426) of the isolates were from hosts with reported disease, whereas 62.0% (n = 743) were from hosts without reported disease. Host health status was unknown for the wild boar isolates (2.4%, n = 29). Of the 1198 isolates analysed, 1140 were grouped into 358 different STs, and 58 could not be assigned to any known ST. The population structure of the collection closely resembles that of the ECOR collection [19], indicating that it represents most of the known diversity of E. coli sensu stricto (Additional file 1: Fig. S2).
Pan-genome analysis
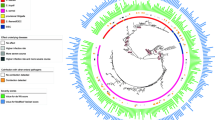
The pan-genome of the 1198 E. coli isolates consisted of 77,130 genes, of which 1956 genes belonged to the core genome (i.e. present in at least 99% of the isolates). Bayesian analysis of population structure (BAPS) revealed 14 BAPS clusters using core-genome alignment (Additional file 1: Fig. S3). Furthermore, most of the isolates were assigned to phylogroups B1 (n = 366, 30.55%), A (n = 313, 26.12%), and B2 (n = 213, 17.77%). The remaining isolates were distributed among phylogroups D (n = 97, 8.09%), E (n = 55, 4.59%), G (n = 49, 4.09%), F (n = 35, 2.92%), C (n = 60, 5.0%), and clade I (n = 8, 0.6%). The distribution of phylogroups, country, and host on a maximum likelihood (ML) tree based on 110,920 core-genome SNPs is shown in Fig. 1a. Clustering of isolates based on core gene alignment and accessory gene profile appeared to correlate with phylogroup (Fig. 1a, b) and BAPS cluster, as expected (Additional file 1: Fig S3). The χ2-tests for independence revealed a positive correlation between host species and phylogroups (p < 2.2e−16, df = 32). This indicates the enrichment of certain hosts among phylogroups, such as B1 (with cattle), A (with pigs), B2 (with humans and chickens), and G (with chickens) within our collection (Additional file 1: Fig. S1B).
Distribution of 1198 E. coli isolates with host species by a core-genome phylogeny and b clustering based on accessory gene content. Clades on the phylogeny represent phylogroups, the inner-ring represents host-species, and the outer ring indicates the geographical region. Each bubble in accessory gene clustering represents individual E. coli strains coloured according to phylogroups
A minimum spanning tree was built on the allelic profiles of 358 (n = 1140 isolates) known STs and 58 isolates belonging to unknown STs using GrapeTree [20] along with the host distribution (Additional file 1: Fig. S4). Several sequence types, of which at least ten isolates were available, appeared to be linked with certain host species. ST33 (n = 10/10, 10 human isolates out of all 10 isolates), ST73 (n = 11/17), ST131 (n = 37/42) and ST1193 (n = 12/12) were associated with a human host. ST131 was also found in chickens (n = 4/42) and a pig (n = 1/42) in this collection. ST23 (n = 18/22), ST95 (n = 25/31), ST115 (n = 11/11), ST117 (n = 30/33), ST140 (n = 19/20), and ST752 (n = 29/30) were associated with the chicken host.
GWAS
The genome-wide association analysis was performed on 1169 E. coli isolates from cattle, chickens, humans, and pigs using k-mers as well as a pan-genome matrix of gene presence. The 29 wild boar isolates were excluded because of their small group size. The thresholds used to filter k-mers or genes can be found in Additional file 1.
GWAS detected the association of 11,869, 3050, and 111,895 k-mers with a human, cattle, or chicken host, respectively. These k-mers accounted for 262, 69, and 1167 genes, respectively (Additional file 2: Table S2). The host-associated genes identified through the k-mer approach were further mapped back to their corresponding orthologous group in the pan-genome.
GWAS using the pan-genome gene presence matrix revealed a positive association of 28, 12, and 145 genes with a human, cattle, or chicken host.
There were 19, 9, and 137 accessory genes of E. coli which were reported to be associated with a human, cattle, or chicken host, respectively, by both GWAS methods (Fig. 2, Additional file 2: Table S2). These genes were subjected to/chosen for further downstream analysis. Neither GWAS approach identified any genes as being associated with the pig host.
Plots representing the E. coli genes or gene variants associated with the a Human host, b Cattle host, and c Chicken host. The bubble size represents the number of k-mers mapped to a specific gene. The bubbles in red indicate genes which came classified as significant by both GWAS approaches (using k-mers and pan-genome) and the bubbles in grey indicate significance by GWAS using k-mers only
Association of novel nan genes with human host
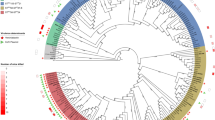
GWAS revealed a strong association of nine contiguous genes, assigned to the group of nan genes, with the human host (Fig. 2a). Seven of these genes were annotated in silico as nan genes (Fig. 3a), and the remaining two genes were annotated as being similar to axeA1 of Prevotella ruminicola ATCC 19189 (Uniprot accession D5EV35). However, translated amino acid sequences of these axeA1-like genes only shared 19–20% similarity with the AxeA1 reference sequence. Further investigation with EggNOG and CD-search revealed an acetylesterase/lipase-encoding region (COG0657) in both genes and confirmed the nan gene annotations. Previous evidence and the genomic location (i.e. between the nan genes; Fig. 3a) suggest that these genes encode potential acetyl-esterases and may be analogous to sialyl esterases (NanS) [21]. Hence, these nine novel nan genes are collectively termed “human-associated nan gene cluster (nan-9)” (Fig. 3a).
Distinct nan genes are present in E. coli and are also known as the sialoregulon (nanRATEK-yhcH, nanXY [yjhBC], and nanCMS; Fig. 3a) [22]. The sialoregulon is involved in metabolism of sialic acids [23,24,25], a diverse group of nine-carbon sugars, abundant in the glycocalyx of many animal tissues [26, 27]. Sialic acids present on mucin proteins in the human gut are an essential energy source for many intestinal bacteria [28]. The proteins encoded by the seven genes of nan-9 (i.e. nanAKTCMRS) share 45–64% similarity with the corresponding nan genes of the sialoregulon in E. coli or the recently described phage-encoded nanS-p genes of enterohemorrhagic E. coli [29]. Both the human-associated nan gene cluster and the sialoregulon are located on the bacterial chromosome. The human-associated nan gene cluster was found in 7% of our isolate collection, whereas the genes comprising the sialoregulon, i.e. nanXY was identified in ~15% of isolates, nanCMS in ~93% of isolates, whilst nanRATEK-yhcH was found in almost all (> 99%) isolates.
The nan-9 cluster was detected in 86 isolates, mainly from phylogroups B2 and D (Fig. 3b) and predominantly in isolates belonging to ST131, ST73, and ST69, both in our collection as well as across 17,994 E. coli genomes present in the RefSeq database (Table 1). The order and orientation of genes in the human-associated nan gene cluster were found to be identical in 82 out of 86 isolates (Additional file 1: Fig. S5). In 63 isolates, insertion sequence (IS) 682 was found upstream, and in 23 isolates, IS2 was found downstream of this novel gene cluster (Additional file 1: Fig. S5).
To further explore the function of the human-associated nan-9 gene cluster, the entire cluster was knocked-out from strain IMT12185 (ST131), yielding strain IMT12185Δnan-9. Correct gene deletion was confirmed through WGS. No significant differences in carbon utilization and chemical sensitivity were observed between wild-type strain IMT12185 and its mutant IMT12185Δnan-9 in Biolog phenotyping array experiments (PM1 and Gen III MicroPlates).
Deletion mutant IMT12185Δnan-9 was grown in minimal medium (MM) with 0.2% 5-N-acetylneuraminic acid (Neu5Ac) or with 0.1% N-glycolylneuraminic acid (Neu5Gc) as sole carbon and energy source. Neu5Ac is the most common sialic acid of the glycocalyx of both humans and other mammals, whereas Neu5Gc is absent in humans. In the presence of Neu5Ac, mutant IMT12185Δnan-9 grew to a maximal optical density at 600nm (OD600) of 1.34 comparable to that of the parental strain IMT12185 (OD600 = 1.37). However, mutant IMT12185Δnan-9 exhibited a delayed growth start of approximately three hours (Fig. 4a). When Neu5Gc was offered as substrate, mutant IMT12185Δnan-9 not only showed a similar growth start retardation, but also a slower growth rate and a lower maximal OD600 (1.31) in comparison with strain IMT12185 (OD600 = 1.43) (Fig. 4b). Both Neu5Ac and Neu5Gc are degraded by the enzymatic activities of the enzymes NanRATEK, of which four, namely NanRATK, are encoded by redundant genes which are located on both the loci nanRATEK and nan-9. For comparison, a mutant, which lacked the nanRATEK locus from the sialoregulon (IMT12185ΔnanRATEK), was also constructed from wild-type IMT12185. Deletion mutant IMT12185ΔnanRATEK was unable to grow with Neu5Ac as sole carbon and energy source (Fig. 4c), demonstrating that nan-9 alone is not sufficient for sialic acid degradation, probably due to a lack of nanE in the nan-9 gene cluster. To exclude a pleiotropic effect of the nan-9 deletion, parental strain IMT12185 and its mutant IMT12185Δnan-9 were grown in LB medium. No significant difference was observed between the two growth curves (Fig. 4d). These data demonstrate that the nan-9 determinant of strain IMT12185 is biologically functional and contributes to the degradation of the sialic acids Neu5Ac and Neu5Gc.
Growth curves of E. coli IMT12185 and its mutants derivatives in various media. a Growth of IMT12185 and IMT12185∆nan-9 in M9 minimal medium with 0.2% 5-N-acetylneuraminic acid (Neu5Ac). b Growth of IM12185 and IMT12185∆nan-9 in M9 minimal medium with 0.1% 5-N-Glycolylneuraminic acid (Neu5Gc). c Growth of IMT12185 and IMT12185∆nanRATEK in M9 minimal medium with 0.2% 5-N-acetylneuraminic acid (Neu5Ac), d Growth of IMT12185 and IMT12185∆nan-9 in lysogeny broth (LB)
Association of distinct omptins with the cattle and chicken hosts
We detected multiple homologues of the ompT (encoding outer-membrane protease VII) gene, a member of the omptin family of proteases, in our dataset (Fig. 2b, c). Two homologues, ompP (UniProt accession P34210, sharing 70% amino acid identity with OmpT) and arlC (also referred to as ompTp, UniProt accession Q3L7I1, sharing 74% amino acid identity with OmpT), were found to be associated with the cattle and the chicken host, respectively (Fig. 5). In our collection, ompP was predominant in phylogroup B1 (n = 68), whereas arlC was found in distinct phylogroups, including B2, B1, and G (Fig. 5), and in isolates belonging to ST95 and ST117 (Additional file 2: Table S3). A similar association of arlC with these STs was observed in 17,994 E. coli genomes in the RefSeq database (Additional file 2: Table S3). Previous studies have reported an increased prevalence of arlC (erroneously reported there as ompT) in a cluster of uropathogenic E. coli (UPEC) and avian pathogenic E. coli (APEC) classified as ST95 [30]. Notably, arlC is associated with increased degradation of antimicrobial peptides (AMPs) in UPEC isolates [31]. OmpP is also able to degrade AMPs but displays an AMP cleavage specificity different from that of OmpT [32].
Association of genes involved in metal acquisition with the chicken host
GWAS analysis revealed an association of the iroBCDEN gene cluster with the chicken host, but not with other host species included in this study (Additional file 2: Table S2). The prevalence of the iro gene cluster was 24.3% (n = 291/1,198) in our collection, of which 61.5% (n = 179/291) were from the chicken host. The gene cluster was found in different STs and with higher prevalence in STs such as ST117, ST95, ST23, and ST140 (Additional file 2: Table S4). The chromosomal iroBCDEN gene cluster was first described in Salmonella enterica and is involved in uptake of catecholate-type siderophores, high-affinity iron-chelating molecules contributing to bacterial survival during infection by sequestering iron [33]. In E. coli, this gene cluster has mainly been described in uropathogenic (UPEC) and avian pathogenic E. coli (APEC) and is regarded as a virulence factor [34]. The cluster has been reported on a chromosomal pathogenicity island, although in ExPEC, the cluster can also be located on ColV or ColBM virulence plasmids [35, 36].
Discussion
Different clones comprising the bacterial species Escherichia coli can colonize various ecological niches in a diverse range of host species, with consequences ranging from a commensal lifestyle to causing intra- or extra-intestinal infectious diseases. The presence of certain adhesin and other virulence-associated genes is well known to correlate with the relative ability of E. coli clones to colonize the intestinal tract of certain hosts (e.g. ecp for humans [37], F9 fimbriae, and H7 flagellae for cattle [38, 39] or Stg fimbriae for chickens [40]). Variations in host adaptation levels and their molecular basis in E. coli clones presumptively realizing a commensal-like lifestyle in the reservoir host are rarely described and poorly understood as of yet [41]. Commensal E. coli strains may act like a carrier of AMR and a source of mobile genetic elements conferring AMR to other bacteria including pathogenic strains in a shared microbiome, e.g. in the intestinal tract of animals including humans. We therefore collated an extensive and diverse dataset to identify genetic determinants of E. coli host adaptation. We observed significant enrichment of specific hosts within some phylogroups and STs in our collection. Furthermore, we unveiled correlations between the likelihood of genetically related isolates having been isolated from a certain host with the possession of distinctive genetic traits. Some of these traits, e.g. the iroBCDEN gene cluster, have been linked to E. coli and Salmonella virulence before, whilst others, in particular the human-associated nan gene cluster, are novel traits and have not been implicated in the colonization and infection process of E. coli. Of note, the latter gene cluster encodes metabolic properties which have received little attention in bacterial infectious disease research thus far. Specific metabolic properties have been linked to the relative ability of Shiga toxin-encoding E. coli (STEC) to asymptomatically colonize cattle, their reservoir host [42]. Unravelling the nutrient and energy flows in the complex interplay of intestinal bacteria, the surrounding microbiome and the host may open novel avenues to control the persistence and transmission of pathogenic and/or antimicrobial resistant bacteria [43].
We employed a k-mer as well as gene-based bacterial GWAS, applied in previous studies to associate multiple types of genetic variation with phenotypes [44, 45]. Our approach focused on accessory genes associated with particular host species, detected through either whole gene or k-mer associations. Although in theory k-mer-based bacterial GWAS would be able to also associate SNP variation to phenotypes, this is difficult in diverse datasets such as these. If multiple sites within a gene vary in sequence, the number of possible k-mers from that sequence inflates quickly. If we tried to link mutations in this region to a phenotype, this would be very complicated based on k-mers alone which is why we focused on variation in the accessory genome. By doing so, we were able to confirm previously published host associations, indicating the validity of our approach. For example, carriage of the salmochelin operon encoded by iroBCDEN was previously identified as associated with increased ability of E. coli strains to colonize chickens [34, 46].
In addition to iroBCDEN, we found an association of omptin proteins (OmpP and ArlC) with chickens and cattle as hosts, respectively. Earlier studies using UPEC strains demonstrated that these proteins are associated with cleavage and inactivation of cationic antimicrobial peptides (AMPs) [31]. Because AMPs are secreted as part of the host’s innate immune response [47,48,49], these proteins may play a vital role in colonization. Because AMPs are also considered as alternatives to antimicrobial agents in animal farming [50,51,52], further investigation into the contribution of these Omp variants to host colonization as well as to resistance to exogenous AMPs is warranted.
We did not identify any significant association of E. coli genes with strains isolated from the pig host. Bacterial colonization of the porcine intestine by edema-disease E. coli (EDEC) is mediated by the ability of these bacteria to adhere to villous epithelial cells via their cytoadhesive F18 fimbriae [53]. The expression of receptors for these fimbriae on the apical enterocyte surface is inherited as a dominant trait among pigs and determines susceptibility to diseases caused by F18-fimbriated pathogenic E. coli [54]. Enterotoxigenic E. coli (ETEC) express F4 or F5 fimbriae with similar consequences [55]. However, we found only three, four, and six isolates harbouring genes for F4, F5, and F18 fimbriae, respectively. Thus, we might not have had all E. coli pathovars associated with pig host sufficiently present in our collection, although we did observe an association between phylogroup A and pig colonization. On the other hand, we did found many genes associated with chicken colonization. This might be due to several reasons. First, E. coli lineages that are typically isolated from chicken are abundant in our dataset, such as ST95 [56] and ST117 [57]. The salmochelin operon, associated with chicken in our analysis, is also prevalent in these lineages. Second, the comparison of chicken (avian species) versus human, cattle, and pig (mammalian species) may provide the best resolution of all GWAS comparisons. Due to the large evolutionary difference between mammalian and avian host species, which might result in diverse host adaptive mechanisms in E. coli, it is likely that certain traits of the gastrointestinal tract or other niches are shared among mammalian species but are different in avian species.
We identified a novel human host-associated nan gene cluster, distinct from the previously reported sialic acid (Sia) metabolic operon (nanRATEK-yhcH, nanXY, and nanCMS) [22]. This novel cluster was found to be conserved and abundant in ExPEC lineages, such as ST131, ST73, and ST69. The gene cluster is flanked by insertion sequences which might play a role in the horizontal exchange between different E. coli lineages. Knock-out in vitro studies indicated that this novel nan-9 gene cluster contributes to catabolism of the sialic acids Neu5Ac and Neu5Gc, although it cannot replace the function of the nanRATEK locus which is encoded in the core genome of E. coli. Hence, we hypothesize that E. coli harbouring the nan-9 gene cluster have an evolutionary advantage through either more efficient access to sialic acids or through access to more diverse sialic acids. The genes annotated as acetylxylan esterases are expected to represent novel sialyl esterases, as known sialyl esterases (nanS variants) have previously been mistaken for acetylxylan esterases [21]. Additional sialyl esterases—possibly with alternative deacetylation specificity—might provide a more efficient catabolism of acetylated sialic acids. Future studies should investigate the role of the human-associated nan-9 gene cluster in the catabolism of differentially acetylated sialic acids and their relevance for the human host.
Approximately one third of the isolates in our dataset were obtained from diseased hosts, whilst the remaining isolates were from healthy hosts. Many of the isolates in our dataset that originate from healthy hosts belong to ExPEC lineages which are typically considered to be pathogenic. In fact, the locus most strongly associated with the human host, the nan-9 gene cluster, is abundant in ExPEC lineages. This does not necessarily mean that the nan-9 gene cluster is associated with pathogenicity. In fact, this observation may support the notion that these pathogenic E. coli are highly efficient colonizers of the human intestine [41]. Based on our results, we hypothesize that the human-associated nan-9 gene cluster is one of the factors driving the adaptation of ExPEC to the human intestine.
Conclusions
Our study identified several distinct genetic determinants that may promote E. coli adaptation to different host species and provide an adaptive advantage. These findings are important as they aid to better understand the potential outcome of transmission events of E. coli between host species. This is particularly relevant for controlling the spread of antimicrobial resistant commensal and zoonotic E. coli strains within and across human and animal populations. The data generated here can also pave the way for the improvement of risk analysis and diagnostic and monitoring efforts. More importantly, our study identified biological processes, including sialic acid catabolism, that should be investigated in more detail to better understand E. coli host adaptation.
Methods
Sampling strategy
A panel of 1213 E. coli isolates from four countries (Germany, UK, Spain, and Vietnam), obtained from five host species (human, pig, cattle, chicken, and wild boar) during three time periods (2003–2007, 2008–2012, and 2013–2018) from both healthy and diseased hosts were selected randomly from existing strain collections and newly collected isolates. Out of 120 possible strata (defined as a unique combination of country, host, time-period, and host health status), isolates were available for 42 strata. We included all isolates available per stratum if there were fewer than 30 isolates and performed a random selection of up to 30 isolates if more were available for a particular stratum. Potentially duplicate isolates that were part of an outbreak, isolated at a single location within a short timeframe, or from a single farm or a single individual were excluded. Only one isolate per individual was included in the analyses. Isolates included per stratum are shown in Additional file 2: Table S1.
E. coli isolates from Vietnam (n = 179) were retrieved from multiple collaborative studies at the Oxford University Clinical Research Unit, including the antimicrobial resistance surveillance in healthy humans (n = 62), chickens (n = 57), and pigs (n = 29) in Tien Giang province. E. coli from animal hosts were isolated from faecal material (faecal/rectal swabs or boot-swabs/hand-held swabs), whereas E. coli from diseased humans (n = 31) was isolated from blood and urine samples submitted to the Microbiology Laboratory at the Hospital for Tropical Diseases in Ho Chi Minh, Vietnam for routine diagnostic purposes.
Isolates from Spain (n = 233) were cultured from caecal samples collected in the frame of different research and surveillance projects. Samples from livestock were collected from the intestine of healthy animals at the abattoir immediately after slaughter with each sample, formed by specimens from one or more animals, representing an independent epidemiological unit (farm/flock). Samples from wild boar were collected from hunter-harvested wild boars during regular hunting seasons. All samples were refrigerated and sent to the VISAVET Health Surveillance Centre for the isolation and identification of E. coli.
Isolates from the UK (n = 174) were received from multiple sources including human hospitals (n = 64), university collections (n = 15), and private veterinary practices (n = 95). Human isolates (n = 64) were cultured from urine samples (n = 32) and faecal samples (n =32). Chicken isolates (n = 110) were obtained from diseased birds at post-mortem examination (n = 80) and faeces/cloacal swabs from healthy chickens (n = 30). Isolates were cultured as for routine diagnostic purposes.
Isolates from Germany collected at the Friedrich-Loeffler-Institut (FLI) (n = 30) were cultured from samples obtained in the context of different research and surveillance projects. Faecal samples from cattle were collected either from healthy animals present at the FLI facilities in Braunschweig or Jena or from the intestines of healthy animals at the abattoir with each sample taken from one animal from an independent farm. Human isolates (n = 170) were obtained from diseased patients with suspicion of infection with pathogenic E. coli (mainly STEC), sent to National Reference Centre at the Robert Koch Institute (RKI) from laboratories of primary diagnostics for confirmation and subtyping. The strains were chosen randomly within the predicted timeslot and the two categories of diseased with or healthy persons without symptoms. Other animals (pig, cattle and chickens) isolates (n = 202) were obtained from diagnostic labs, isolated from samples sent by vets because the corresponding animals were ill, whereas, for the healthy animals (pig, cattle and chickens) (n = 210), faecal samples were collected for routine diagnostic purposes.
DNA extraction and sequencing
The DNA of the E. coli isolates from Germany was extracted using the QIAamp DNA Mini Kit (Qiagen) following the manufacturer’s instructions. The DNA concentration was evaluated fluorometrically by using QubitTM 2.0 fluorometer (Invitrogen, USA) and the associated QubitTM dsDNA HS Assay Kit (0.2–100 ng) and QubitTM BR Assay Kit (2–1000 ng), respectively. The libraries were generated using Nextera DNA library preparation (Illumina, https://www.illumina.com). The sequencing was performed using the Illumina MiSeq and HiSeq systems, generating 2 × 250 bp and 2 × 150 bp reads, respectively.
The DNA of the E. coli isolates from the UK was purified using a Promega DNA Wizard® genomic purification kit and quantified using Nanodrop. Libraries were generated using Nextera XT technology (Illumina), and DNA sequencing of isolates was performed at the Animal and Plant Health Agency (APHA, Surrey, UK, https://www.gov.uk/government/-organisations/animal-and-plant-healthagency) using an Illumina MiSeq system generating 2 × 150 bp reads.
For E. coli isolates from Spain, DNA was extracted using the DNA blood and tissue Qiagen kit according to the manufacturer’s instructions. The total amount of DNA was quantified using a Qubit fluorometer and frozen at – 20 °C until further analysis. Libraries were prepared using Nextera XT DNA Library preparation (Illumina), and DNA samples were sequenced using a MiSeq platform (2 × 300 cycle V3 Kit).
The DNA of the E. coli isolates from Vietnam was extracted using the Wizard Genomic DNA purification kit (Promega, Madison, WI, USA) following the manufacturer’s instructions. The concentration of the DNA was measured fluorometrically by using picogreen (Invitrogen). The sequencing was performed using an Illumina HiSeq 4000 system, which generates 2 × 150 bp reads.
Quality control
Adapter sequences were removed from raw reads using flexbar v3.0.3 [58, 59] with trimming mode (-ae) ANY. Low-quality bases within raw reads (Phred score value < 20) were trimmed using a sliding window approach (-q WIN). FastQC v0.11.7 [60] and MultiQC v1.6 [61] were used for quality control before and after processing steps.
Genome assembly and annotation
Adapter-trimmed reads were assembled using SPAdes v3.13.1 [62] using read correction. Scaffolds smaller than 500bp were discarded. QUAST v5.0.0 [63] was used to assess assembly quality using default parameters. Draft assemblies were excluded if the N50 was below an arbitrary value of 30 kbp or consisted of more than 900 contigs. Draft genomes were annotated using prokka v1.13 [64] with a genus-specific blast for Escherichia. Phylogroups were predicted using ClermonTyper v1.4.1 [65], and sequence types (STs) of the isolates were identified in silico using the Achtman seven gene MLST scheme using mlst (https://github.com/tseemann/mlst).
Pan-genome and phylogenetic analysis
Roary v3.12.0 [66] was used to define the pan-genome of the population, using paralog splitting. The core genes were aligned using prank [67] on default parameters. The core gene alignment was used to construct the phylogenetic tree using RaxML 8.2.4 [68] with 100 bootstraps under a General Time Reversible (GTR) substitution model with the Gamma model of rate heterogeneity and Lewis ascertainment bias correction [69]. The core gene phylogeny was corrected for recombination using ClonalFrameML [70] using default parameters. BAPS clusters within the dataset were defined using hierBAPS [71, 72] based on the core gene alignment. The accessory gene clustering was performed using package Rtsne v0.15 [73, 74] with 5000 iterations and perplexity 15 in R v3.6.1. iTOL [75] and Microreact [76] were used to visualize the population structure in the context of available metadata. The function chisq.test from the MASS library [77] (v7.3-51.1) was used in R [78] (v3.5.2) to perform χ2-tests of independence between phylogenetic clusters and host species. Tests were carried out on the full dataset (14 phylogenetic clusters vs. five hosts and nine phylogroups vs. five host species).
Genome-wide association study (GWAS)
We excluded the wild boar E. coli isolates from the GWAS analysis, because of their low number (n = 29). The GWAS was performed on k-mers as well as on the pan-genome matrix (i.e. gene presence and absence) generated from Roary.
First, assemblies were shredded into k-mers of 9–100 bases using FSM-lite (https://github.com/nvalimak/fsm-lite). The association of k-mers and gene presence/absence with host phenotype (i.e. pig, human, chicken, and cattle) was carried out using FaST-LMM, a linear mixed model implemented in pyseer whilst correcting for population structure and sampling bias [79]. To correct for population structure, a pairwise similarity matrix (based on patristic distances) generated from core-genome phylogeny (recombination free) was used with a FaST-LMM mixed model [79]. To account for sampling bias, a covariate matrix consisting of geographical location, year of isolation, and host health status was used.
GWAS analysis was carried out by comparing human hosts vs other hosts (i.e. pig, cattle, and chicken), cattle vs other hosts (i.e. human, chicken, and pig), chicken vs other hosts (i.e. human, pig, and cattle), and pig vs other hosts (i.e. cattle, chicken, and human) within the collection. To further reduce the false-positive associations and account for unbalanced groups, we employed a bootstrapping approach as implemented by Epping et al. (2021) [80]. Further details can be found in the additional file 1. K-mers and genes (presence-absence), which were significantly associated with the hosts in 90% of the runs, were retained. These k-mers were mapped to reference genomes (Additional file 2: Table S5) using a fastmap algorithm in bwa [79, 81]. The genes significantly associated with a certain host in both GWAS approaches were considered further for downstream analysis.
Prevalence of a human-associated nan gene cluster
All available E. coli genome assemblies in NCBI RefSeq were downloaded on November 29, 2019, using NCBI-genome-download (https://github.com/kblin/ncbi-genome-download). Using a custom ABRicate (https://github.com/tseemann/abricate) database, consisting of the nine genes of the novel human-associated nan gene cluster, all downloaded genomes (n = 17994) were scanned. STs for all the genomes were assigned as described above.
Construction of mutants and phenotypic experiments
Mutants Δnan-9 (AmpR) and ΔnanRATEK of extra-intestinal pathogenic E. coli (ExPEC) strain IMT12185 (ST131; RKI 20-00501; AmpR) were constructed using the Datsenko-Wanner method [82]. The genomic DNA of the wild-type and the mutant strains was isolated using a QIAamp DNA Mini Kit (QIAGEN). Short read sequencing libraries were prepared using the Nextera XT DNA Library preparation kit (Illumina) and paired-end sequencing was performed on an Illumina MiSeq machine. The absence of the desired genes was confirmed by mapping sequencing reads on the most closely related complete reference genome (GCF_900520325.1) using Snippy 4.6.0.
Carbon utilization and chemical sensitivity of the deletion mutants and their parental strain were tested using a Biolog Phenotypic Array system, using the PM1 MicroPlate and the Gen III MicroPlate according to the manufacturer’s instructions.
Growth curve analysis
E. coli strains were grown at 37 °C aerobically in lysogeny broth (LB) (10 g/l tryptone, 5 g/l yeast extract, 5 g/l NaCl, pH 7.5) or in minimal medium (MM). MM is M9 mineral medium (33.7 mM Na2HPO4, 22.0 mM KH2PO4, 8.55 mM NaCl, 9.35 mM NH4Cl) supplemented with 2 mM MgSO4 and 0.1 mM CaCl2. As carbon and energy source, either 27.8 mM [0.5% w/v] glucose, 6.47 mM [0.2% w/v] 5-N-acetylneuraminic acid (Neu5Ac) or 6.15 mM [0.1% w/v] N-glycolylneuraminic acid (Neu5Gc) (all purchased from Sigma-Aldrich, Taufkirchen, Germany) was added. If appropriate, the following antibiotics were used: ampicillin sodium salt (150 μg/ml) or kanamycin (50 μg/ml). For solid media, 1.5% agar (w/v) was added. For all growth experiments, bacterial strains were grown in LB medium overnight at 37 °C aerobically, washed twice in PBS and then adjusted to an optical density at 600 nm (OD600) of 0.005 in the desired liquid growth medium, or streaked on agar plates. Growth curves were obtained from bacterial cultures incubated at 37 °C with gentle agitation in 96-well microtitre plates containing 200 μl medium. The OD600 was measured by an automatic reader (Epoch2T; BioTek, Bad Friedrichshall, Germany) at appropriate time intervals as indicated.
Availability of data and materials
Abbreviations
- AMP:
-
Antimicrobial peptide
- AMR:
-
Antimicrobial resistance
- APEC:
-
Avian pathogenic Escherichia coli
- BAPS:
-
Bayesian analysis of population structure
- EDEC:
-
Edema-disease Escherichia coli
- ETEC:
-
Enterotoxigenic Escherichia coli
- ExPEC:
-
Extraintestinal pathogenic Escherichia coli
- GWAS:
-
Genome-wide association study
- LB:
-
Lysogeny broth
- ML:
-
Maximum-likelihood
- MM:
-
Minimal medium
- NCBI:
-
National Center for Biotechnology Information
- Neu5Ac:
-
5-N-acetylneuraminic acid
- Neu5GC:
-
5-N-glycolylneuraminic acid
- Sia:
-
Sialic acids
- ST:
-
Sequence type
- STEC:
-
Shiga toxin-encoding Escherichia coli
- UPEC:
-
Uropathogenic Escherichia coli
References
Alteri CJ, Mobley HLT. Escherichia coli physiology and metabolism dictates adaptation to diverse host microenvironments. Curr Opin Microbiol. 2012;15(1):3–9.
Ewers C, Bethe A, Semmler T, Guenther S, Wieler LH. Extended-spectrum β-lactamase-producing and AmpC-producing Escherichia coli from livestock and companion animals, and their putative impact on public health: A global perspective. Clin Microbiol Infect. 2012;18(7):646–55.
Bonnet R, Beyrouthy R, Haenni M, Nicolas-Chanoine M-H, Dalmasso G, Madec J-Y. Host colonization as a major evolutionary force favoring the diversity and the emergence of the worldwide multidrug-resistant Escherichia coli ST131. MBio. 2021;12(4): e0145121.
Lopatkin AJ, Meredith HR, Srimani JK, Pfeiffer C, Durrett R, You L. Persistence and reversal of plasmid-mediated antibiotic resistance. Nat Commun. 2017;8(1):1689.
Pitout JDD, DeVinney R. Escherichia coli ST131: a multidrug-resistant clone primed for global domination. F1000Research. 2017;6.
Nicolas-Chanoine MH, Bertrand X, Madec JY. Escherichia coli ST131, an intriguing clonal group. Clin Microbiol Rev. 2014;27(3):543–74.
Skurnik D, Clermont O, Guillard T, Launay A, Danilchanka O, Pons S, et al. Emergence of antimicrobial-resistant Escherichia coli of animal origin spreading in humans. Mol Biol Evol. 2016;33(4):898–914.
Riley LW. Pandemic lineages of extraintestinal pathogenic Escherichia coli. Clin Microbiol Infect. 2014;20(5):380–90.
Cohen E, Azriel S, Austeri O, Gal A, Zitronblat C, Mikhlin S, et al. Pathoadaptation of the passerine-associated Salmonella enterica serovar Typhimurium lineage to the avian host. PLoS Pathog. 2021;17(3): e1009451.
Toft C, Andersson SGE. Evolutionary microbial genomics: Insights into bacterial host adaptation. Nat Rev Genet. 2010;11(7):465–75.
Silva WM, Dorella FA, Soares SC, Souza GHMF, Castro TLP, Seyffert N, et al. A shift in the virulence potential of Corynebacterium pseudotuberculosis biovar ovis after passage in a murine host demonstrated through comparative proteomics. BMC Microbiol. 2017;17(1):55.
Koskiniemi S, Gibbons HS, Sandegren L, Anwar N, Ouellette G, Broomall S, et al. Pathoadaptive mutations in Salmonella enterica isolated after serial passage in mice. PLoS One. 2013;8: e70147.
Liu X, Lu L, Liu X, Pan C, Feng E, Wang D, et al. Comparative proteomics of Shigella flexneri 2a strain 301 using a rabbit ileal loop model reveals key proteins for bacterial adaptation in host niches. Int J Infect Dis. 2015;40:28–33.
Sheppard SK, Didelot X, Meric G, Torralbo A, Jolley KA, Kelly DJ, et al. Genome-wide association study identifies vitamin B5 biosynthesis as a host specificity factor in Campylobacter. Proc Natl Acad Sci U S A. 2013;110(29):11923–7.
Mageiros L, Méric G, Bayliss SC, Pensar J, Pascoe B, Mourkas E, et al. Genome evolution and the emergence of pathogenicity in avian Escherichia coli. Nat Commun. 2021;12(1):765.
Salipante SJ, Roach DJ, Kitzman JO, Snyder MW, Stackhouse B, Butler-Wu SM, et al. Large-scale genomic sequencing of extraintestinal pathogenic Escherichia coli strains. Genome Res. 2015;25(1):119–28.
Chun J, Oren A, Ventosa A, Christensen H, Arahal DR, da Costa MS, et al. Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes. Int J Syst Evol Microbiol. 2018;68(1):461–6.
Liu S, Jin D, Lan R, Wang Y, Meng Q, Dai H, et al. Escherichia marmotae sp. nov., isolated from faeces of Marmota himalayana. Int J Syst Evol Microbiol. 2015;65(7):2130–4.
Ochman H, Selander RK. Standard reference strains of Escherichia coli from natural populations. J Bacteriol. 1984;157(2):690–3.
Zhou Z, Alikhan NF, Sergeant MJ, Luhmann N, Vaz C, Francisco AP, et al. GrapeTree: visualization of core genomic relationships among 100,000 bacterial pathogens. Genome Res. 2018;28(9):1395-404.
Steenbergen SM, Jirik JL, Vimr ER. YjhS (NanS) is required for Escherichia coli to grow on 9-O-acetylated N-acetylneuraminic acid. J Bacteriol. 2009;191(22):7134–9.
Kalivoda KA, Steenbergen SM, Vimr ER. Control of the Escherichia coli sialoregulon by transcriptional repressor NanR. J Bacteriol. 2013;195(20):4689–701.
Vimr ER, Kalivoda KA, Deszo EL, Steenbergen SM. Diversity of microbial sialic acid metabolism. Microbiol Mol Biol Rev. 2004;68(1):132–53.
Vimr ER, Troy FA. Identification of an inducible catabolic system for sialic acids (nan) in Escherichia coli. J Bacteriol. 1985;164(2):845–53.
Bell A, Severi E, Lee M, Monaco S, Latousakis D, Angulo J, et al. Uncovering a novel molecular mechanism for scavenging sialic acids in bacteria. J Biol Chem. 2020;295(40):13724–36.
Vimr ER. Unified theory of bacterial sialometabolism: how and why bacteria metabolize host sialic acids. ISRN Microbiol. 2013;2013: 816713.
Severi E, Hood DW, Thomas GH. Sialic acid utilization by bacterial pathogens. Microbiology. 2007;153(Pt 9):2817–22.
L. H-MB, Brian WW, J.B. L, Fidelma BE. Host Sialic Acids: A Delicacy for the Pathogen with Discerning Taste. Microbiol Spectr [Internet]. American Society for Microbiology; 2015;3:3.4.07. Available from: https://doi.org/10.1128/microbiolspec.MBP-0005-2014.
Saile N, Voigt A, Kessler S, Stressler T, Klumpp J, Fischer L, et al. Escherichia coli O157:H7 strain EDL933 harbors multiple functional prophage-associated genes necessary for the utilization of 5-N-acetyl-9-O-acetyl neuraminic acid as a growth substrate. Appl Environ Microbiol. 2016;82(19):5940–50.
Johnson TJ, Wannemuehler Y, Johnson SJ, Stell AL, Doetkott C, Johnson JR, et al. Comparison of extraintestinal pathogenic Escherichia coli strains from human and avian sources reveals a mixed subset representing potential zoonotic pathogens. Appl Environ Microbiol. 2008;74(22):7043–50.
Desloges I, Taylor JA, Leclerc JM, Brannon JR, Portt A, Spencer JD, et al. Identification and characterization of OmpT-like proteases in uropathogenic Escherichia coli clinical isolates. Microbiologyopen. 2019;8(11): e915.
Hwang BY, Varadarajan N, Li H, Rodriguez S, Iverson BL, Georgiou G. Substrate specificity of the Escherichia coli outer membrane protease OmpP. J Bacteriol. 2007;189(2):522–30.
Ratledge C, Dover LG. Iron metabolism in pathogenic bacteria. Annu Rev Microbiol. 2000;54:881–941.
Caza M, Lépine F, Milot S, Dozois CM. Specific roles of the iroBCDEN genes in virulence of an avian pathogenic Escherichia coli O78 strain and in production of salmochelins. Infect Immun. 2008;76(8):3539–49.
Sorsa LJ, Dufke S, Heesemann J, Schubert S. Characterization of an iroBCDEN gene cluster on a transmissible plasmid of uropathogenic Escherichia coli: Evidence for horizontal transfer of a chromosomal virulence factor. Infect Immun. 2003;71(6):3285–93.
Dobrindt U, Blum-Oehler G, Nagy G, Schneider G, Johann A, Gottschalk G, et al. Genetic structure and distribution of four pathogenicity islands (PAI I536 to PAI IV536) of uropathogenic Escherichia coli strain 536. Infect Immun. 2002;70(11):6365–72.
Rendón MA, Saldaña Z, Erdem AL, Monteiro-Neto V, Vázquez A, Kaper JB, et al. Commensal and pathogenic Escherichia coli use a common pilus adherence factor for epithelial cell colonization. Proc Natl Acad Sci U S A. 2007;104(25):10637–42.
Low AS, Dziva F, Torres AG, Martinez JL, Rosser T, Naylor S, et al. Cloning, expression, and characterization of fimbrial operon F9 from enterohemorrhagic Escherichia coli O157:H7. Infect Immun. 2006;74(4):2233–44.
Mahajan A, Currie CG, Mackie S, Tree J, Mcateer S, Mckendrick I, et al. An investigation of the expression and adhesin function of H7 flagella in the interaction of Escherichia coli O157: H7 with bovine intestinal epithelium. Cell Microbiol. 2009;11(1):121–37.
Lymberopoulos MH, Houle S, Daigle F, Léveillé S, Brée A, Moulin-Schouleur M, et al. Characterization of Stg fimbriae from an avian pathogenic Escherichia coli O78:K80 Strain and assessment of their contribution to colonization of the chicken respiratory tract. J Bacteriol. 2006;188(18):6449–59.
Tenaillon O, Skurnik D, Picard B, Denamur E. The population genetics of commensal Escherichia coli. Nat Rev Microbiol. 2010;8(3):207–17.
Barth SA, Weber M, Schaufler K, Berens C, Geue L, Menge C. Metabolic traits of bovine shiga toxin-producing Escherichia coli (STEC) strains with different colonization properties. Toxins (Basel). 2020;12(6):414.
Stecher B. Establishing causality in Salmonella-microbiota-host interaction: the use of gnotobiotic mouse models and synthetic microbial communities. Int J Med Microbiol. 2021;311(3): 151484.
Ma KC, Mortimer TD, Hicks AL, Wheeler NE, Sánchez-Busó L, Golparian D, et al. Adaptation to the cervical environment is associated with increased antibiotic susceptibility in Neisseria gonorrhoeae. Nat Commun. 2020;11(1):4126.
Gröschel MI, Meehan CJ, Barilar I, Diricks M, Gonzaga A, Steglich M, et al. The phylogenetic landscape and nosocomial spread of the multidrug-resistant opportunist Stenotrophomonas maltophilia. Nat Commun. 2020;11(1):2044.
Gao Q, Wang X, Xu H, Xu Y, Ling J, Zhang D, et al. Roles of iron acquisition systems in virulence of extraintestinal pathogenic Escherichia coli: Salmochelin and aerobactin contribute more to virulence than heme in a chicken infection model. BMC Microbiol. 2012;12:143.
McPhee JB, Small CL, Reid-Yu SA, Brannon JR, Le Moual H, Coombes BK. Host defense peptide resistance contributes to colonization and maximal intestinal pathology by Crohn’s disease-associated adherent-invasive Escherichia coli. Infect Immun. 2014;82(8):3383–93.
Fjell CD, Jenssen H, Fries P, Aich P, Griebel P, Hilpert K, et al. Identification of novel host defense peptides and the absence of α-defensins in the bovine genome. Proteins Struct Funct Genet. 2008;73(2):420–30.
Lynn DJ, Higgs R, Gaines S, Tierney J, James T, Lloyd AT, et al. Bioinformatic discovery and initial characterisation of nine novel antimicrobial peptide genes in the chicken. Immunogenetics. 2004;56(3):170–7.
Li Z, Hu Y, Yang Y, Lu Z, Wang Y. Antimicrobial resistance in livestock: antimicrobial peptides provide a new solution for a growing challenge. Anim Front. 2018;8(2):21–9.
Liu Q, Yao S, Chen Y, Gao S, Yang Y, Deng J, et al. Use of antimicrobial peptides as a feed additive for juvenile goats. Sci Rep. 2017;7(1):12254.
Xiao H, Shao F, Wu M, Ren W, Xiong X, Tan B, et al. The application of antimicrobial peptides as growth and health promoters for swine. J Anim Sci Biotechnol. 2015;6(1):19.
Barth S, Schwanitz A, Bauerfeind R. Polymerase chain reaction-based method for the typing of F18 fimbriae and distribution of F18 fimbrial subtypes among porcine Shiga toxin-encoding Escherichia coli in Germany. J Vet Diagnostic Investig. 2011;23(3):454–64.
Frydendahl K, Jensen TK, Andersen JS, Fredholm M, Evans G. Association between the porcine Escherichia coli F18 receptor genotype and phenotype and susceptibility to colonisation and postweaning diarrhoea caused by E. coli O138:F18. Vet Microbiol. 2003;93(1):39–51.
Barth S, Tscholshiew A, Menge C, Weiß R, Baljer G, Bauerfeind R. Virulence and fitness gene patterns of Shiga toxin-encoding Escherichia coli isolated from pigs with edema disease or diarrhea in Germany. Berl Munch Tierarztl Wochenschr. 2007;120(7–8):307–16.
Cummins ML, Reid CJ, Djordjevic SP. F plasmid lineages in Escherichia coli ST95: implications for host range, antibiotic resistance, and zoonoses. mSystems. 2022;7(1):e0121221.
Mora A, López C, Herrera A, Viso S, Mamani R, Dhabi G, et al. Emerging avian pathogenic Escherichia coli strains belonging to clonal groups O111: H4-D-ST2085 and O111: H4-D-ST117 with high virulence-gene content and zoonotic potential. Vet Microbiol. 2012;156(3–4):347–52.
Dodt M, Roehr JT, Ahmed R, Dieterich C. FLEXBAR-flexible barcode and adapter processing for next-generation sequencing platforms. Biology (Basel). 2012;1(3):895–905.
Roehr JT, Dieterich C, Reinert K. Flexbar 3.0 - SIMD and multicore parallelization. Bioinformatics. 2017;33(18):2941–2.
Andrews S, Krueger F, Seconds-Pichon A, Biggins F, Wingett S. FastQC. A quality control tool for high throughput sequence data. Babraham Bioinformatics. Babraham Inst. 2015. http://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
Ewels P, Magnusson M, Lundin S, Käller M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics. 2016;32(19):3047–8.
Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol. 2012;19(5):455–77.
Gurevich A, Saveliev V, Vyahhi N, Tesler G. QUAST: quality assessment tool for genome assemblies. Bioinformatics. 2013;29(8):1072–5.
Seemann T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics. 2014;30(14):2068–9.
Beghain J, Bridier-Nahmias A, Le Nagard H, Denamur E, Clermont O. ClermonTyping: an easy-to-use and accurate in silico method for Escherichia genus strain phylotyping. Microb Genomics. 2018;4(7):e000192.
Page AJ, Cummins CA, Hunt M, Wong VK, Reuter S, Holden MTG, et al. Roary: rapid large-scale prokaryote pan genome analysis. Bioinformatics. 2015;31(22):3691–3.
Löytynoja A. Phylogeny-aware alignment with PRANK. Methods Mol Biol. 2014;1079:155–70.
Stamatakis A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014;30(9):1312–3.
Lewis PO. A likelihood approach to estimating phylogeny from discrete morphological character data. Syst Biol. 2001;50(6):913–25.
Didelot X, Wilson DJ. ClonalFrameML: efficient inference of recombination in whole bacterial genomes. PLoS Comput Biol. 2015;11(2): e1004041.
Cheng L, Connor TR, Sirén J, Aanensen DM, Corander J. Hierarchical and spatially explicit clustering of DNA sequences with BAPS software. Mol Biol Evol. 2013;30(5):1224–8.
Tonkin-Hill G, Lees JA, Bentley SD, Frost SDW, Corander J. RhierBAPS: an R implementation of the population clustering algorithm hierBAPS. Wellcome Open Res. 2018;3:93.
Van Der Maaten LJP, Hinton GE. Visualizing high-dimensional data using t-sne. J Mach Learn Res. 2008;9:2579–605.
Van Der Maaten L. Accelerating t-SNE using tree-based algorithms. J Mach Learn Res. 2015;15(93):3221–45.
Letunic I, Bork P. Interactive Tree Of Life (iTOL): an online tool for phylogenetic tree display and annotation. Bioinformatics. 2007;23(1):127–8.
Argimón S, Abudahab K, Goater RJE, Fedosejev A, Bhai J, Glasner C, et al. Microreact: visualizing and sharing data for genomic epidemiology and phylogeography. Microb Genomics. 2016;2(11): e000093.
Venables WN, Ripley BD. Ripley BD. Modern applied statistics with S (Fourth Edition). 2002.
R Core Team (2020). R: a language and environment for statistical computing. R Found. Stat. Comput. Vienna, Austria. 2020.
Lees JA, Galardini M, Bentley SD, Weiser JN, Corander J. pyseer: A comprehensive tool for microbial pangenome-wide association studies. Bioinformatics. 2018;34(24):4310–2.
Epping L, Walther B, Piro RM, Knüver MT, Huber C, Thürmer A, et al. Genome-wide insights into population structure and host specificity of Campylobacter jejuni. Sci Rep. 2021;11(1):10358.
Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25(14):1754–60.
Datsenko KA, Wanner BL. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A. 2000;97(12):6640–5.
Tiwari SK, van der Putten BCL, Vinh TN, Bootsma M, Oldenkamp R, La Ragione R, et al. The impact of host restriction of Escherichia coli on transmission dynamics and spread of antimicrobial Resistance. NCBI BioProject. https://identifiers.org/bioproject:PRJNA739205 (2021).
Comparative genomics of Avian Pathogenic Escherichia coli (APEC). NCBI BioProject. https://identifiers.org/bioproject:PRJNA523197 (2019).
Funding
Open Access funding enabled and organized by Projekt DEAL. The HECTOR research project was supported under the framework of the JPIAMR—Joint Programming Initiative on Antimicrobial Resistance—through the 3rd joint call, thanks to the generous funding by the Netherlands Organisation for Health Research and Development (ZonMw, grant number 547001012), the Federal Ministry of Education and Research (BMBF/DLR grant numbers 01KI1703A, 01KI1703C and 01KI1703B), the State Research Agency (AEI) of the Ministry of Science, Innovation and Universities (MINECO, grant number PCIN-2016-096), and the Medical Research Council (MRC, grant number MR/R002762/1).
Author information
Authors and Affiliations
Contributions
Conceptualization: S.K.T., B.C.L.v.d.P., T.N.V., S.M., N.T.H., T.S., and C.S. Data curation: S.K.T. and B.C.L.v.d.P. Formal analysis: S.K.T., B.C.L.v.d.P., and T.M.F. Funding acquisition: T.N.V., M.B., R.L.R., S.M., N.T.H., C.B., J.A., J.M.R., S.S., L.D., M.U.-R., A.F.-H., C.M., T.S., and C.S. Investigation: S.K.T., B.C.L.v.d.P., T.M.F., M.B., A.F., and H.S. Methodology: S.K.T., B.C.L.v.d.P., and T.M.F. Project administration: S.K.T., B.C.L.v.d.P., T.S., and C.S. Resources: S.K.T., B.C.L.v.d.P., T.M.F., T.N.V., M.B., R.O., R.L.R., S.M., N.T.H., C.B., J.L., J.A., M.F.-V., J.M.R., A.F., S.S., L.D., M.U.-R., A.B., C.H., V.J., I.S., L.H.W., C.E., A.F.-H., H.S., C.M., T.S., and C.S. Software: S.K.T., B.C.L.v.d.P., and T.S. Supervision: C.M., T.S., and C.S. Visualization: S.K.T., B.C.L.v.d.P., and T.M.F. Writing—original draft: S.K.T., B.C.L.v.d.P., and T.M.F. Writing—review and editing: S.K.T., B.C.L.v.d.P., T.M.F., T.N.V., M.B., R.O., R.L.R., S.M., N.T.H., C.B., J.L., J.A., M.F.-V., J.M.R., A.F., S.S., L.D., M.U.-R., A.B., C.H., V.J., I.S., L.H.W., C.E., A.F.-H., H.S., C.M., T.S., and C.S. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Fig. S1. Distribution of 1,198 isolates and enrichment analysis: A) The plot represents the proportion of E. coli isolates isolated from hosts in four countries. The number above each plot indicates the total number of isolates per host. B) Phylogroups enriched with different hosts (Pearson residual > 0 represents positive correlation indicating the enrichment of certain host-species in distinct clusters at p-value < 2.2e−16). Fig. S2. Core genome phylogeny of E. coli isolates from our collection (n=1,198) and reference strains (n=146) from the ECOR collection, RefSeq and cryptic clades annotated with their phylogroups (phylogroups were determined by ClermonTyper v. 1.3). Fig S3. Core-genome phylogeny and Accessory-genome clustering of 1,198 E. coli isolates showing different BAPS clusters. Each BAPS on accessory genome clustering consist of the same strains as on the core-genome phylogeny. Fig. S4. Minimum-spanning tree of MLST profiles of 1,198 E. coli isolates. Left: the number of isolates constituting an ST; Right: the proportion of hosts in each ST. Fig. S5. Genetic surroundings of the human-associated nan gene cluster in the genomes of all isolates in which it was identified.
Additional file 2:
Table S1. Metadata of 1,198 E. coli isolates. Table S2. Genes and genetic variants associated with different hosts that came as significant by both k-mer based as well as pan-genome based GWAS. Table S3. Prevalence of ompP, arlC and ompT in different STs in the RefSeq collection (n=17,994) and in our collection of E. coli isolates (n=1198). Table S4. Prevalence of the iroBCDEN gene cluster in sequence types and associated hosts. Details are shown for sequence types (STs) with at least ten isolates harboring iroBCDEN. Table S5. Genomes from our dataset used for annotating host-associated k-mers.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Tiwari, S.K., van der Putten, B.C.L., Fuchs, T.M. et al. Genome-wide association reveals host-specific genomic traits in Escherichia coli. BMC Biol 21, 76 (2023). https://doi.org/10.1186/s12915-023-01562-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12915-023-01562-w