Abstract
Background
We evaluated a facilitation strategy to help clinical sites likely to experience challenges implement evidence-based Primary Care-Mental Health Integration (PC-MHI) care models within the context of a Department of Veterans Affairs (VA) initiative. This article describes our assessment of whether implementation facilitation (IF) can foster development of high quality PC-MHI programs that adhere to evidence, are sustainable and likely to improve clinical practices and outcomes.
Methods
Utilizing a matched pair design, we conducted a qualitative descriptive evaluation of the IF strategy in sixteen VA primary care clinics. To assess program quality and adherence to evidence, we conducted one-hour structured telephone interviews, at two time points, with clinicians and leaders who knew the most about the clinics’ programs. We then created structured summaries of the interviews that VA national PC-MHI experts utilized to rate the programs on four dimensions (overall quality, adherence to evidence, sustainability and level of improvement).
Results
At first assessment, seven of eight IF sites and four of eight comparison sites had implemented a PC-MHI program. Our qualitative assessment suggested that experts rated IF sites’ programs higher than comparison sites’ programs with one exception. At final assessment, all eight IF but only five comparison sites had implemented a PC-MHI program. Again, experts rated IF sites’ programs higher than their matched comparison sites with one exception. Over time, all ratings improved in five of seven IF sites and two of three comparison sites.
Conclusions
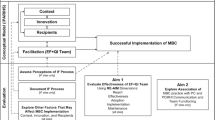
Implementing complex evidence-based programs, particularly in settings that lack infrastructure, resources and support for such efforts, is challenging. Findings suggest that a blend of external expert and internal regional facilitation strategies that implementation scientists have developed and tested can improve PC-MHI program uptake, quality and adherence to evidence in primary care clinics with these challenges. However, not all sites showed these improvements. To be successful, facilitators likely need at least a moderate level of leaders’ support, including provision of basic resources. Additionally, we found that IF and strength of leadership structure may have a synergistic effect on ability to implement higher quality and evidence-based programs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
In this article, we report our evaluation of a facilitation strategy to help clinical sites implement evidence-based practices in the context of the Veterans Health Administration (VHA) Primary Care-Mental Health Integration (PC-MHI) initiative. Implementation of evidence-based practices and programs (EBPPs) within healthcare systems can be widely variable. Numerous theories and substantial research evidence indicate that organizations and the individuals within them are often slow to adopt new practices (e.g., [1–6]). Such variability can result in lack of fidelity to evidence, poor program quality, and, ultimately poor outcomes. To spread EBPPs system-wide, many organizations simply mandate global implementation [7]. Such mandates may increase organizations’ motivation to change but not affect their capacity for change [8]. VHA, the largest integrated healthcare system in the United States, has been a forerunner in the development and promotion of EBPPs through clinical initiatives that include mandates, as well as other supports for change [9–11]. To transform mental health care for Veterans, VHA has provided mental health enhancement funds to increase staffing and has mandated provision of multiple specific EBPPs across VA healthcare facilities [12]. Sometimes mandates are poorly implemented because they receive little national implementation support [13, 14]. On the other hand, even when mandated EBPPs are highly supported, implementation can still be variable; this has been the case for several VHA programs including suicide prevention [11] and evidence-based psychotherapy treatments [9, 15]. VHA has also invested significant resources in its PC-MHI initiative [10]. Similar to the previous examples, all VA medical centers have implemented PC-MHI to some extent but not all have fully implemented evidence-based care models mandated by VA [10].
Variable implementation of EBPPs is likely inevitable given the dynamic and complex nature of healthcare systems and the vast number of factors that can foster or impede implementation success [16–18]. There is an extensive body of theory and evidence regarding contextual factors that affect organizations’ ability to change and implement new practices. For example, engaged and supportive leaders can sanction changes, reinforce priorities and authorize necessary resources for implementation efforts [19, 20]. An organizational climate that fosters stakeholder participation, innovation and collaboration can support EBPP adoption [8, 21]. Organizations need resources for staffing, education and training, and space; and they need the knowledge and capacity for planning change and monitoring and improving implementation processes; and stakeholders need protected time for implementation efforts [8, 22]. Stakeholder perceptions of the need for an innovation, the relative advantage of implementing it and the innovation’s fit with existing values and goals can also impact implementation efforts [16]. The presence of these contextual factors and many others are widely variable across healthcare organizations. Additionally, interactions between different contextual elements can vary as well [16, 23]. Although all healthcare settings experience implementation challenges, some may experience far more than others, lacking sufficient resources, infrastructure support and process knowledge needed to implement innovations [24].
Facilitation, a multi-faceted process of enabling and supporting individuals and groups, has been widely used in clinical settings, particularly primary care settings, as a strategy for overcoming barriers and leveraging strengths to foster implementation of EBPPs, prevention services and innovative care delivery models [24–26]. Facilitators can apply implementation science knowledge and interventions to help and enable others to understand what they need to change, plan and execute changes and address barriers to change efforts. Specifically, facilitators work with teams of key stakeholders to select innovations, adapt them to the local context, and select or develop interventions (e.g., formative evaluation, audit and feedback, clinical reminders) and tools (e.g., pocket cards, decision support) to support implementation [27–31]. Although facilitation has been widely used to address implementation challenges, we know little about whether this implementation strategy can help clinical settings with the most challenging contexts successfully implement PC-MHI programs. We applied and evaluated a facilitation strategy within the context of VHA’s PC-MHI initiative [10] to help sites with challenging clinical contexts implement PC-MHI and build capacity for future implementation efforts. This article describes one component of our evaluation of the implementation facilitation strategy, the assessment of the relative quality, fidelity to evidence, sustainability and likely level of improvement in PC-MHI programs.
Study context
VHA’s mandate to implement PC-MHI required that all VA primary care clinics serving more than 5000 Veterans implement a blend of two complex, evidence-based PC-MHI care models [12]. Both models require significant changes in the structure and processes of mental health care delivery. In one of the models, co-located collaborative care, mental health providers, embedded in primary care clinics, offer brief assessment, diagnosis and treatment for Veterans with mental health and substance abuse care needs [32, 33]. In the other highly evidence-based model, care management, care managers assist primary care clinicians in the care of this population by utilizing guideline-based protocols to provide clinical assessment, patient education and activation and follow-up care [34–36]. VA’s mandate to implement PC-MHI recommends, but does not require, that facilities implement either Translating Initiatives for Depression into Effective Solutions (TIDES) or Behavioral Health Laboratory (BHL), two specific care management models that have proven efficacious and effective within VA populations [37, 38]. VA delegated responsibility for PC-MHI implementation to regional network leaders and established a national program office to provide consultation, technical support, education and training, identification and dissemination of best practices, and development and dissemination of tools to support PC-MHI program implementation [39].
This study was part of larger VA-funded project (HSR&D SDP 08-316, J, Kirchner, PI), where we applied and evaluated an implementation facilitation (IF) strategy both to implement and foster sustainment of PC-MHI programs at sites that would have been unable to implement PC-MHI without assistance. When the project began in 2009, many VA facilities had not implemented PC-MHI. We believed that some sites, in time, would be able to implement PC-MHI but those that lacked sufficient resources, infrastructure support and process knowledge would need help. The IF strategy incorporated a blend of external and internal facilitation [40]. A national expert (JEK) in implementation science, PC-MHI models and evidence for them, and facilitation strategies, worked in tandem with network-level internal regional facilitators to support PC-MHI implementation. Facilitation activities included multiple evidence-based implementation strategies. The frequency and timing of facilitators’ application of these strategies varied by site based on the phase of the implementation process, site-level contextual factors and the perspectives of local site stakeholders regarding the evidence supporting PC-MHI [41, 42]. We describe the IF strategy, including facilitation activities, in the methods section.
The project was comprised of three primary studies. In one, we demonstrated that compared to non-IF clinics, primary care patients at IF clinics were significantly more likely to be seen in PC-MHI, primary care providers were more likely to refer patients to PC-MHI, and a greater proportion of those providers’ patients were referred to PC-MHI [43]. In a second study, we conducted a qualitative analysis of how the facilitation intervention unfolds over time and in response to local context. We also explored how it fit into a broad conceptual framework for planned organizational change [41, 42, 44, 45]. In the third study, described in this article, we explored two questions: 1) whether IF could help clinics with challenging contexts to implement PC-MHI programs and 2) whether IF could foster development of PC-MHI programs that are of high quality, adhere to evidence, are sustainable and are likely to lead to improvement of clinical practices and outcomes as assessed by experts. Because of our small sample size we did not have sufficient power to conduct statistical analyses. Thus we employed and present a qualitative assessment of our findings.
Methods
We applied qualitative descriptive methods to assess whether IF can foster implementation of PC-MHI programs, including facilitating the ability to create high quality programs that adhere to evidence. An independent evaluation team conducted all research activities and facilitators were blind to the evaluation findings until after the intervention ended. The VA Central Institutional Review Board approved the research, which was conducted between February 2009 and August 2013.
Study setting
We conducted the study in sixteen VA primary care clinics located in four VA networks. We matched clinics at both the network and clinic levels, resulting in two matched pairs of networks and eight matched pairs of clinics (four clinics within each network). Because our IF strategy and site recruitment were complex and the intervention occurred within a rapidly unfolding policy context, we could not wait to select all four networks before condition assignment. We therefore assigned the first two networks to the IF intervention. We then matched these two networks to two others for comparison purposes. It was not important when the comparison networks entered the study because we allowed the natural process to unfold in these networks without interference. Given these factors, however, randomization was not possible. Because the structure and operational authority of network-level leadership can influence efforts to facilitate implementation of PC-MHI, we selected one IF network (A) and one comparison network (B) with strong mental health service line structures [46]. Mental health leaders in these networks had dedicated budgets and input into the selection and evaluation of VA Medical Center (VAMC) mental health leaders and network policies and procedures. In the other networks, IF network C and comparison network D, mental health leaders did not have a dedicated budget and had only very limited input into selection and evaluation of VAMC mental health leaders and network policies and procedures. Although all network mental health leaders were extremely supportive of PC-MHI implementation, we wanted to control for the influence of network mental health service line structure on implementation. Additional information about the network selection and matching process is described in a previous publication [43].
To select clinics in IF networks, mental health leaders, based on their knowledge and experience with implementation of PC-MHI and other clinical initiatives at facilities in their networks, identified clinics that 1) had not yet implemented and would have difficulty implementing a policy-compliant PC-MHI program without assistance, 2) delivered services or had the potential to deliver services to 5000 or more primary care patients a year, and 3) planned to implement a PC-MHI program in the first year of the study. To select clinics in comparison networks, mental health leaders identified a list of clinical sites using the same clinic selection criteria. We then selected clinics from that list that best matched IF clinics on facility type, clinic size, number of primary care providers, clinic location (rural/urban), academic affiliation, perceived need for PC-MHI and innovativeness [43]. In each network, one of the clinics was located at a VAMC and the other three were community based outpatient clinics (CBOCs).
At baseline, none of the study clinics had a PC-MHI program that met VA policy requirements to implement 1) a blend of co-located collaborative care and care management and 2) evidence-based care models (see Table 1). To prepare for program implementation, we required intervention clinics to identify existing staff or hire new staff who could serve as PC-MHI providers. Facilitators conducted the first site visit once clinics identified or hired appropriate staff. Four of eight IF clinics had co-located mental health providers in primary care prior to the intervention, but those providers were delivering specialty mental health services rather than the brief models of care associated with PC-MHI. One IF clinic had identified PC-MHI staff members who were co-located but providing only mental health assessments. Two other IF clinics had identified or hired providers for PC-MHI but none were providing PC-MHI services and one IF clinic had no PC-MHI staff. In the comparison networks, only one clinic had identified and hired PC-MHI staff but those staff members were not yet providing PC-MHI services. None of the other comparison clinics had designated PC-MHI staff.
The implementation facilitation strategy
The multifaceted IF strategy that we evaluated incorporated a blend of external and internal facilitation [40]. The national expert facilitator (JEK) worked with and mentored two internal regional facilitators who were clinical personnel experienced in PC-MHI practices. Each of the regional facilitators worked on the project for 50% of their time. Below we describe the facilitation activities they performed.
In preparation for helping sites implement PC-MHI, the facilitators began engaging clinic leaders and provided information about PC-MHI programs as well as what to expect during the facilitation process. Facilitators used formative evaluation techniques [47] to assess clinics’ current practice patterns and anticipate barriers and facilitators to PC-MHI implementation. They also identified key stakeholders who should participate in designing an implementation plan (see below) to adapt the PC-MHI program to meet site needs, preferences and priorities for local implementation. Facilitators obtained additional site level information from administrative data (e.g., number of patients seen in primary care) and medical center webpages (e.g., organizational charts).
The facilitators conducted site visits in August and September 2009 to begin guiding local teams in designing their implementation plans. Prior research [40, 48–51] informed site visit agendas and activities designed to engage key stakeholders in implementation planning processes. During site visits, the expert facilitator conducted academic detailing [52] for leaders and other stakeholders and assessed clinics’ readiness to plan the implementation process. She then held a final leadership briefing during which she provided her assessment of the clinic’s ability to implement a PC-MHI program and discussed barriers and facilitators to implementation identified to date. This was an important activity in the implementation planning process because it provided facilitators an early opportunity to identify potential problems and work directly with leaders to develop solutions to address implementation barriers. In addition to the academic detailing provided during the initial site visit, the facilitators conducted education and marketing activities throughout the intervention, providing ongoing training, engaging stakeholders and helping sites identify and resolve problems.
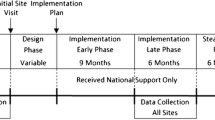
Once facilitators determined that a site was ready to begin in-depth planning, they used a checklist [53] to guide the decision-making process (e.g., identifying PC-MHI target population and exclusion criteria, PC-MHI staff members’ roles and responsibilities, and how to monitor the implementation process). Facilitators documented specific action items and, when appropriate, identified parties responsible for each action item. Although the facilitators initially intended to conduct this planning process during the first site visit, only two sites were ready at that time. At the other six sites, facilitators continued working with teams until they were ready to develop an implementation plan (see Fig. 1). It took five of the remaining six sites from a month to approximately a year to become ready to complete the implementation planning checklist. Site C4, a CBOC whose parent VAMC did not have a PC-MHI program, proved particularly challenging. After 6 months of working with this site, facilitators determined that without resources and support from the VAMC, this site would be unable to implement PC-MHI. Facilitators then provided support and consultation to the parent VAMC, which successfully implemented its own program and in November 2011, started implementing a PC-MHI program at site C4.
Length of time before sites were able to create implementation plans. Bars indicate the period of time during which facilitators helped intervention sites prepare to develop a PC-MHI program implementation plan. The beginning of the bar indicates facilitators’ first site visit. The end of the bar signals the completion of this plan. Two sites, A3 and C3, completed the plan during the first site visit
Once sites created their program plans, they could begin implementation of the plan. Facilitators monitored the implementation process, utilizing administrative data and assessing site progress in completing action items and achieving performance targets specified in the local implementation plan. The internal regional facilitator worked individually with each site to address site level needs such as additional provider education on the PC-MHI model and how to implement it, refinement of the site’s implementation plan, and marketing the potential outcomes and value of the PC-MHI program to primary care providers, Veterans and leadership. The external expert/internal regional facilitator pair in each network also established a learning collaborative [54] of IF site PC-MHI providers and champions to review implementation progress, share lessons learned, identify barriers and facilitators and create potential solutions to implementation problems.
Facilitators helped sites integrate the PC-MHI program into existing clinical programs and services, as well as continued problem identification and resolution and audit and feedback of the implementation process. Further, the internal regional facilitator continued to engage local leadership through regular briefings and email communication. Over the course of the implementation process, the expert facilitator mentored the internal regional facilitator in implementation strategies, transferring the knowledge and skills necessary to support ongoing program sustainment. Thus, these skills were retained within the IF networks once project funding ended. It is important to note that though we have described facilitation activities in a linear phasic framework, the IF process was actually dynamic and iterative, with activities overlapping and repeating to continually monitor and adjust local implementation processes to maximize potential for success [41, 42].
Measure development
Assessing program implementation typically involves assessing fidelity, or the degree to which a program is implemented as its developers intended [55–58]. Scholars suggest that fidelity should be assessed using measures of adherence to a program, dose or amount of the program that is delivered, quality of program delivery, participant responsiveness and program component differentiation [59]. A description of a program’s components (strategies, activities, behaviors, media products and technologies, intended recipients and delivery settings) is the ideal basis for measuring the extent of implementation [60]. However, we faced a number of challenges to conducting a traditional fidelity assessment of the extent of PC-MHI implementation in this study. VA policy mandated implementation of a “blend” of two different types of PC-MHI care, co-located collaborative care and care management, both of which required significant structural and procedural changes in the delivery of mental health care. Further, although facilities were expected to implement evidence-based care models, there are many models that fit this criterion [61, 62]. Specific model selection was left to the discretion of the VA facilities. Additionally, given the variability in resources, structures and processes across VA, facilities also needed to make adaptations to their local contexts. Because there was not a clearly defined list of required program components and VA administrative data did not include quantitative measures of program components, we developed a holistic qualitative assessment instrument to document the PC-MHI program components each site implemented.
We developed the PC-MHI Program Component Assessment instrument using several inter-related sources. The first source was a set of quality improvement (QI) tools that LEP created for facilities to self-assess their fidelity with three different PC-MHI models of care, Translating Initiatives in Depression into Effective Solutions (TIDES) [37], the Behavioral Health Laboratory (BHL) [63], and Co-located Collaborative Care (CCC) [32]. To develop these tools, she collaborated with VA’s PC-MHI model developers, conducting in-depth interviews with them and reviewing program materials (e.g., training manuals and assessment tools), journal articles and conference presentations describing the models’ goals and outcomes. She then iteratively revised these tools with the model developers. The second source was VA’s first national “VAMC Primary Care – Mental Health Integration Survey.” The PC-MHI National Evaluation Team, charged with assessing implementation progress across VA, needed an instrument to assess PC-MHI program implementation. Although they were able to capture some measures of implementation outcomes through VA administrative data, there was no data available that could measure if PC-MHI was being fully implemented. As described above, because of the complex multifaceted nature of PC-MHI programs and the large number of variations that would meet policy requirements for implementing evidence-based programs, it was not possible to develop quantitative implementation measures. Thus the VAMC survey was a holistic qualitative measure. To develop that instrument, LEP and MJR collaborated with VA’s PC-MHI National Evaluation Team to combine the QI fidelity tools into a single self-administered survey. To create the PC-MHI Program Component Assessment instrument for the current study, we modified the national PC-MHI survey for telephone administration, retaining the closed response set but including opportunities for informants to clarify their answers. Our resulting PC-MHI Program Component Assessment instrument (see Additional file 1) addresses the following domains:
-
Staffing issues including: PC-MHI occupations and availability of care managers, co-located prescribers and therapists.
-
Mental health conditions addressed.
-
Mental health services provided.
-
Program policies and procedures including those for referral and length and number of sessions available, suicide prevention, program monitoring, evaluation and quality improvement.
-
Types of assessment and communication tools utilized.
-
Extent of communication between primary care and mental health services providers and staff members.
-
Types of PC-MHI training available to providers and staff members.
Data collection: site programs
Internal regional facilitators at IF sites and VAMC and network mental health leaders for comparison sites identified study sites that had PC-MHI programs and the VA clinicians providing services or the leaders for those programs, i.e., those responsible for implementing or overseeing implementation of PC-MHI program components. Only sites with PC-MHI programs had PC-MHI service providers or leaders. To recruit participants, facility mental health leaders sent letters to site providers and/or leaders briefly describing the study and introducing them to the study team. We followed up with an email asking if they would be interested in learning more about the study and possibly participating in it. We sent a letter containing the required elements of informed consent, including the study’s purpose and procedures, privacy protections and the risks, benefits and voluntary nature of participation to those who were interested. We recruited a total of twenty-six unique individuals; two of those declined participation for a total study N = 24 (see Table 2).
One year after IF clinics with PC-MHI programs completed the initial program design and implementation plan and approximately 1 year after that, we administered the Program Component Assessment (PCA) instrument to IF site participants (N = 17) during one-hour telephone interviews. At comparison sites with PC-MHI programs, we conducted the interviews with study participants (N = 7) at approximately the same time as their matched IF site. Two VA health services researchers with expertise in qualitative data collection and analysis conducted all interviews. Interviewers had no relationship with the participants before the study began. At the beginning of the interviews, the interviewers introduced themselves as VA researchers testing an innovative network-level facilitation method within the context of the VA’s requirements for integrating mental health services into primary care settings, reviewed the elements of consent in the letter participants had received, and obtained verbal informed consent. The primary interviewer (MJR), a female licensed clinical social worker and PhD candidate in Public Policy, administered the PCA, clarifying questions and responses. The second interviewer, a male who earned his DHSc in Health Sciences Leadership and Organizational Behavior during the project and who was also Project Coordinator, requested additional clarification as needed. Both interviewers documented informant responses, took notes on information that did not fit into the existing response set, and documented questions for which informants provided information that might need to be reviewed later. We audio-recorded all interviews. Interviewers met immediately after the interview to confirm responses and compare notes.
Because the PCA interviews created a large amount of raw data, we developed a structured summary template to facilitate comparison and rating of sites’ programs. The template used bulleted lists, tables and checkboxes to display program information so that it could be more easily reviewed (see Additional file 2). Utilizing this template, we then created structured summaries describing the components of the PC-MHI program for each site and time period. In addition to information that we obtained from the PCA interviews, the summaries also included information about clinic size and the availability of mental health specialty care services at the start of the study. We prepared a total of twenty-three program summaries for the sites that had PC-MHI programs (10 sites at the initial assessment and 13 at the final assessment). In preparation for the expert rating process described below, we removed study site identifiers and the time period of the PCA from the summaries.
Data collection and analysis: expert ratings of site programs
To assess how well sites implemented PC-MHI programs, VA PC-MHI experts rated the PCA summaries. Experts have an extensive body of domain knowledge organized abstractly in “mental models” of how things work [64, 65]. They also have refined reasoning processes that allow them to perceive and interpret complex patterns in a set of information, to detect problems or anomalies and to run mental simulations to predict how things will work in the future [65, 66]. Thus, we expected that VA PC-MHI experts would be able to review and assess each unique PC-MHI program based on the Program Component Summary we provided them. We recruited six national experts in PC-MHI care models and evidence-based interventions, as well as VA policy, organizational structures and processes, to rate the programs. Experts reviewed the program summaries and rated them on four dimensions: the extent to which the program made use of evidence-based strategies for improving mental health care within primary care, the overall program quality, the potential for long-term sustainability, and the likely level of improvement in quality of care the intervention would produce. Experts reviewed a mix of initial and final PCA summaries across IF and comparison sites and did not review the same site more than once. Following Parker and Rubenstein [48, 67], experts were blind to informant, site and time period and made their ratings independently using 7-point Likert scales. For each program summary, we collected ratings from a total of 3 reviewers. Because each expert had to review eleven or twelve program summaries and rate each one on four dimensions, a substantial time burden, we did not ask experts to explain their ratings. We calculated average ratings for each program summary on rated dimensions. Because of our small sample size, we did not have sufficient power to conduct statistical analyses. We therefore conduct a qualitative assessment.
Our intervention sites began receiving IF before we could finalize selection of comparison sites and Institutional Review Board approval in one of the networks. We were therefore unable to collect initial program component data at one comparison network (D). We were, however, able to determine whether sites in this network did or did not have programs. As only one out of four did during the initial period, we were in fact, missing initial expert ratings at one site. We were able to obtain data to rate for all networks for the second rating period.
Results
We found evidence supporting our hypothesis that facilitation could foster implementation of PC-MHI in VA primary care clinics. First, all sites receiving IF implemented PC-MHI programs but not all comparison sites were able to implement programs. At the time of the initial program assessment for each site, seven of eight IF sites and four of eight comparison sites had implemented a PC-MHI program (see Table 3). At the time of the final program assessment, 1 year later, all eight IF but only five comparison sites had implemented a PC-MHI program (see Table 4).
We were also interested in exploring whether IF could foster the development of high quality PC-MHI programs that adhere to evidence, are sustainable and are likely to lead to improvement of clinical practices and outcomes. At the time of the initial program assessment, experts rated IF programs higher than those at comparison sites with one exception. Program ratings for IF site C2 were very low. At the time of the final assessment, experts again rated IF site programs higher than their matched comparison sites with one exception. Site C1 was rated lower on all four dimensions than all other IF and comparison sites. At both time points, ratings for each site were generally similar across all four dimensions. In comparing the initial and final ratings, all ratings improved over the course of the study in five of the seven IF sites and two of the three comparison sites.
Discussion
Our qualitative analysis suggests that sites with challenging clinical contexts receiving the IF strategy were more likely to implement a PC-MHI program than were comparison sites. Indeed, whereas all the intervention sites implemented programs, some of comparison sites never did during the study period. Our analysis also suggests that IF is effective in fostering clinical practice quality as well as adherence to the evidence for PC-MHI at sites that lack knowledge, skills and resources for implementation. Further, during the second year of implementation, most sites receiving IF were not only able to sustain but actually able to continue improving program quality and adherence to evidence. These findings complement our findings from another project study that this strategy increases the likelihood that primary care providers will adopt the program, collaborating with and referring to PC-MHI providers [43]. Interestingly, in that other study, we did not find a difference in our quantitative fidelity measure (same day access). That measure only assessed one aspect of PC-MHI. PC-MHI, however, is extremely complex, with many interacting components and multiple evidenced-based methods with multiple acceptable implementation methods. As such, assessing its fidelity requires an in-depth program assessment beyond what one single quantitative measure can provide. Our study provides such an in-depth assessment. Additionally, our findings here are consistent with the growing body of evidence showing that facilitation improves the uptake of evidence-based practices and programs [25, 68, 69].
Our IF strategy appears to be effective in part because facilitators applied evidence-based implementation science strategies in their efforts to help sites implement PC-MHI. For example, at intervention sites, facilitators persistently sought to involve stakeholders across all organizational levels [49] and engage them in a QI dialogue [50]. It is likely that there was no one to do this at the three comparison sites that still had not implemented PC-MHI by the end of the study. Facilitators also assessed the organizational context and current practices and tailored implementation efforts to site context and needs, activities that evidence suggests will improve implementation [25, 70, 71]. We attribute the relatively high IF site program quality and adherence to evidence to facilitators’ efforts to 1) help site staff acquire the requisite knowledge and skills for delivering high quality PC-MHI care using evidence-based strategies, and 2) monitor programs and intervene to prevent program “drift,” e.g., by providing feedback to site stakeholders [58].
Despite the application of evidence-based implementation strategies, some of our study sites in IF Network C still had difficulty implementing PC-MHI. Site C1 was a VAMC primary care clinic and site C2 was the CBOC associated with that VAMC. Facilitators were unable to engage high-level leaders’ support at the parent VAMC, limiting resources available for needed structural change at both sites. However, with the facilitators’ assistance, site C1 was able to implement a program with quality and adherence to evidence ratings similar to other intervention sites by the end of the first year. Nevertheless, there was significant PC-MHI staff instability in the 6 months prior to our final assessment. At the time of our assessment, the new PC-MHI provider had just started; low ratings likely reflected this. Site C2 had low ratings at the time of the initial assessment. In addition to lack of high-level leadership support at the VAMC level, neither of the clinic leaders was engaged at the time of the first assessment and one of them was very resistant to change. PC-MHI provider time was diverted to conducting lengthy mental health assessments and referrals. The first set of ratings for this site reflects these circumstances. With intensive assistance, facilitators were able to help site C2 improve program quality and adherence to the evidence but only on a limited basis. Lack of VAMC support was also a challenge for helping implement PC-MHI at site C4. However, facilitators’ efforts were successful in engaging the site’s parent VAMC leadership to support implementation by the end of the study. By the time of our second assessment, this site was able to implement a program of moderate quality. Thus although IF clearly fosters implementation, facilitators will have difficulty being successful unless they obtain at least a moderate level of leaders’ support, including provisions of basic resources.
Implementation science scholars generally agree that the engagement and support of leadership at all levels is an important determinant of implementation success [8, 72–74]. In comparing all eight sites, it appears that the strength of network mental health service line structure influenced PC-MHI program implementation regardless of the initial engagement and support of leadership at lower levels. All sites in IF network A and comparison network B, which had strong network leadership structures, implemented programs and program quality and adherence to evidence improved over time. In IF clinics, facilitators engaged local leadership in network A and helped sites implement higher quality programs than comparison network B. In the two networks where the strength of mental health service line structures was relatively moderate, the results were variable. Only one site in IF network C implemented a high quality program and only one site in comparison network D implemented a PC-MHI program at all. Thus it appears that facilitation and strength of leadership structure may have a synergistic effect on ability to implement higher quality and evidence-based programs. Without either success is unlikely. With one or the other, success is possible but difficult and with both, success is most likely. See Fig. 2.
Combined effects of implementation facilitation and strength of leadership structure on implementation. Implementation facilitation and strength of leadership structure interact and affect how successful sites are in implementing evidence-based programs. Programs are most successful when there is a strong leadership structure and sites receive implementation facilitation
Although we cannot be sure why some comparison sites did better than others, one possible explanation concerns the level of commitment from the network mental health lead. As we described, our IF intervention required that clinics identify or hire PC-MHI staff. The mental health lead in the comparison network with a strong network structure had prior positive experience with PC-MHI. Other researchers have found that preexisting familiarity with the clinical intervention can foster adoption [14]. In our study, the network mental health lead in the comparison network with a strong network structure required clinics to assign staff for PC-MHI and clinics in this network were able to implement programs without IF assistance. Thus, IF might be most important for networks with weak leadership structures and support for programs. Future research should address this issue.
Future research should also explore whether there are other barriers to successful application of the IF strategy. Early identification of such barriers would allow researchers and/or clinical managers to delay facilitation efforts until barriers can be addressed, or to select sites that are more likely to benefit from IF. Finally, although the IF strategy we used was effective and built organizational capacity, it was also resource intensive. Given IF’s costs we recommend reserving it for only those sites most needing this assistance. Future research should develop tools for identifying those sites that are both able to avail themselves of IF assistance and that find implementation sufficiently difficult that it is worth the cost of intervening. Finding the balance will depend in part upon organizational priorities and resources.
In addition to improving PC-MHI program quality and adherence to evidence, the IF strategy we applied built capacity at the regional level for facilitating implementation of PC-MHI. At the beginning of the study, internal regional facilitators were IF novices. Over time, the expert facilitator mentored internal facilitators, helping them develop IF skills and handing off responsibility to them when they were ready. By the end of the study, internal facilitators were IF experts, applying evidence-based strategies and working to maintain and improve program quality and adherence to evidence. The IF skills they developed are readily transferrable to implementation of other evidence-based innovations. Implementation scientists generally seek to integrate evidence-based practices and programs into routine care by the end of a study. We recommend incorporating a process for handing off or transferring IF knowledge and skills to internal change agents. This could, as it did in this study, maximize potential for sustainment, continued improvement in quality and adherence to evidence, as well as build capacity for implementing other innovations. Although there is currently limited information regarding how to transfer IF knowledge and skills [29], another part of this project has begun to document this process [42].
Limitations
Although we believe that our study demonstrates that IF has great promise for helping challenged clinics to implement evidence based clinical programs, there were limitations. First, as we described earlier, we were unable to randomize to condition. Second, resources available for the study limited its scope. For example, we have less data on the comparison than on the intervention clinics. There may have been factors that we were unable to assess that could have contributed to or hindered adoption of PC-MHI at comparison clinics. Also, due to resource limitations, we were unable to involve more than eight intervention and eight comparison sites, thus limiting our ability to conduct statistical analyses on rating data. Additionally, we were only able to interview the people who knew the most about the PC-MHI program and services at each site, generally PC-MHI staff. It may have been beneficial to seek the perspectives of other stakeholders, including other clinicians and consumers. However, given the already broad scope of this study, we lacked resources to query additional stakeholders. A final study limitation is that we did not ask experts to give reasons for their ratings. We felt that the time burden for experts outweighed any potential benefits of additional explanation. Despite these limitations, we are confident that our evaluation and the tools we developed to assess implementation of these very complex programs should prove useful to others, both within and outside of VA, who are implementing and evaluating similarly complex, real-world quality improvement efforts.
Conclusions
Implementing complex evidence-based programs, particularly in settings that lack infrastructure, resources and support for such efforts, is challenging. We found that a blend of external expert and internal regional facilitation incorporating implementation science improved PC-MHI program uptake, quality and adherence to evidence in primary care clinics with these challenges. This study was conducted within the context of a VA policy initiative that required sites to implement a blend of two different, complex PC-MHI care models. The methods we developed for measuring program quality and adherence to evidence addressed this complexity.
Our findings have implications for research using implementation facilitation strategies, as well as for clinical initiatives implementing evidence-based interventions. In fact, VA leadership has already adopted this facilitation strategy to support implementation of PC-MHI, as well as other policy-mandated evidence-based innovations, at facilities across VA that struggle to implement new programs [9, 10, 15, 75]. Based on experiences of facilitators in this study, we developed IF training materials and the external expert facilitator (JEK) and one of the internal regional facilitators have been training clinical operations personnel in these methods [76, 77]. They have also trained other researchers in the application of implementation facilitation. Scholars have long advocated that implementing evidence-based practices and programs will improve healthcare quality and outcomes but clinical settings continue to struggle with implementation challenges. The IF strategy we describe enhances sites’ ability to successfully implement such innovations.
Abbreviations
- BHL:
-
Behavioral Health Laboratory
- CBOC:
-
Community based outpatient clinic
- CNE:
-
Carrie N Edlund
- IF:
-
Implementation facilitation
- i-PARIHS:
-
Integrated-Promoting Action on Research Implementation in Health Services
- JEK:
-
JoAnn E Kirchner
- LEP:
-
Louise E Parker
- MJR:
-
Mona J Ritchie
- MSW:
-
Master of social work
- PARIHS:
-
Promoting action on research implementation in health services
- PCA:
-
Program Component Assessment
- PC-MHI:
-
Primary Care-Mental Health Integration
- QI:
-
Quality improvement
- RN:
-
Registered nurse
- TIDES:
-
Translating initiatives for depression into effective solutions
- VA:
-
Department of Veterans Affairs
- VHA:
-
VA Medical Center
- VAMC:
-
Veterans Health Administration
References
Rogers EM. Diffusion of innovations. 5th ed. New York: Free Press; 2003.
Nutley S, Walter I, Davies HTO. From knowing to doing: a framework for understanding the evidence-into-practice agenda. Evaluation. 2003;9:125–48.
Burke WW. Organizational change: theory and practice. 4th ed. Thousand Oaks: Sage Publications; 2013.
Hovmand PG, Gillespie DF. Implementation of evidence-based practice and organizational performance. J Behav Health Serv Res. 2010;37:79–94.
Hannan MT, Freeman J. Structural inertia and organizational change. Am Sociol Rev. 1984;49:149–64.
Van de Ven AH, Sun K. Breakdowns in implementing models of organization change. Acad Manag Perspect. 2011;25:58–74.
Aarons GA, Farahnak LR, Ehrhart MG, Sklar M. Aligning leadership across systems and organizations to develop strategic climate to for evidence-based practice implementation. Annu Rev Publ Health. 2014;35:255–74.
Greenhalgh T, Robert G, MacFarlane F, Bate P, Kyriakidou O. Diffusion of innovations in service organizations: systematic review and recommendations. Milbank Q. 2004;82:581–629.
Karlin BE, Cross G. From the laboratory to the therapy room: national dissemination and implementation of evidence-based psychotherapies in the US Department of Veterans Affairs health care system. Am Psychol. 2014;69:19.
Pomerantz AS, Kearney LK, Wray LO, Post EP, McCarthy JF. Mental health services in the medical home in the Department of Veterans Affairs: factors for successful integration. Psychol Serv. 2014;11:243–53.
Katz IR, Kemp JE, Blow FC, McCarthy JF, Bossarte RM. Changes in suicide rates and in mental health staffing in the Veterans Health Administration, 2005 − 2009. Psychiatr Serv. 2013;64:620–5.
Veterans Health Administration. Uniform mental health services in VA medical centers and clinics, VHA handbook 1160.01. 2008.
McGuire AB, White DA, White LM, Salyers MP. Implementation of illness management and recovery in the Veterans Administration: an on-line survey. Psychiatr Rehabil J. 2013;36:264–71.
McGuire AB, Salyers MP, White DA, Gilbride DJ, White LM, Kean J, et al. Factors affecting implementation of an evidence-based practice in the Veterans Health Administration: Illness management and recovery. Psychiatr Rehabil J. 2015;38:300–5.
Rosen C, Matthieu MM, Wiltsey-Stirman S, Cook JM, Landes S, Bernardy NC, et al. A review of studies on the system-wide implementation of evidence-based psychotherapies for posttraumatic stress disorder in the Veterans Health Administration. Adm Policy Ment Health. 2016;43:957.
Lau R, Stevenson F, Ong BN, Dziedzic K, Treweek S, Eldridge S, et al. Achieving change in primary care − causes of the evidence to practice gap: systematic reviews of reviews. Implement Sci. 2015;11:40.
Flottorp SA, Oxman AD, Krause J, Musila NR, Wensing M, Godycki-Cwirko M, et al. A checklist for identifying determinants of practice: a systematic review and synthesis of frameworks and taxonomies of factors that prevent or enable improvements in healthcare professional practice. Implement Sci. 2013;8:35.
Krause J, Van Lieshout J, Klomp R, Huntink E, Aakhus E, Flottorp S, et al. Identifying determinants of care for tailoring implementation in chronic diseases: an evaluation of different methods. Implement Sci. 2014;9:102.
Aarons GA, Hurlburt M, Horwitz SM. Advancing a conceptual model of evidence-based practice implementation in public service sectors. Adm Policy Ment Health. 2011;38:4–23.
Ferlie EB, Shortell SM. Improving the quality of health care in the United Kingdom and the United States: a framework for change. Milbank Q. 2001;79:281–315.
Kirchner JE, Rule C, Bennett LA, Kramer TL. Blending education, research and service missions: The Arkansas Behavioral Health Model. Acad Med. 2007;82:107–12.
Fixsen DL, Naoom S, Blase KA, Friedman RM, Wallace F. Implementation research: a synthesis of the literature. Tampa: University of South Florida: Louis de la Parte Florida Mental Health Institute; 2005.
McCullough MB, Chou AF, Solomon JL, Petrakis BA, Kim B, Park AM, et al. The interplay of contextual elements in implementation: an ethnographic case study. BMC Health Serv Res. 2015;15:62.
Grumbach K, Bainbridge E, Bodenheimer T. Facilitating improvement in primary care: the promise of practice coaching. Issue Brief (Commonw Fund). 2012;15:1–14.
Baskerville NB, Liddy C, Hogg W. Systematic review and meta-analysis of practice facilitation within primary care settings. Ann Fam Med. 2012;10:63–74.
Nutting PA, Miller WL, Crabtree BF, Jaen CR, Stewart EE, Stange KC. Initial lessons from the first national demonstration project on practice transformation to a patient-centered medical home. Ann Fam Med. 2009;7:254–60.
Bidassie B, Williams LS, Woodward-Hagg H, Matthias MS, Damush TM. Key components of external facilitation in an acute stroke quality improvement collaborative in the Veterans Health Administration. Implement Sci. 2015;10:69.
Curran G, Mukherjee S, Allee E, Owen R. A process for developing an implementation intervention: QUERI Series. Implement Sci. 2008;3:17.
Dogherty EJ, Harrison MB, Baker C, Graham ID. Following a natural experiment of guideline adaptation and early implementation: a mixed-methods study of facilitation. Implement Sci. 2012;7:9.
Rubenstein LV, Pugh J. Strategies for promoting organizational and practice change by advancing implementation research. J Gen Intern Med. 2006;21:S58–64.
Stetler CB, Legro MW, Rycroft-Malone J, Bowman C, Curran G, Guihan M, et al. Role of “external facilitation” in implementation of research findings: a qualitative evaluation of facilitation experiences in the Veterans Health Administration. Implement Sci. 2006;1:23.
Pomerantz AS, Sayers SL. Primary care-mental health integration in healthcare in the Department of Veterans Affairs. Fam Syst Health. 2010;28:78–82.
Watts BV, Shiner B, Pomerantz A, Stender P, Weeks WB. Outcomes of a quality improvement project integrating mental health into primary care. Qual Saf Health Care. 2007;16:378–81.
Williams Jr JW, Gerrity M, Holsinger T, Dobscha S, Gaynes B, Dietrich A. Systematic review of multifaceted interventions to improve depression care. Gen Hosp Psychiatry. 2007;29:91–116.
Katon W, Robinson P, Von Korff M, Lin E, Bush T, Ludman E, et al. A multifaceted intervention to improve treatment of depression in primary care. Arch Gen Psychiatry. 1996;53:924–32.
Gilbody S, Bower P, Fletcher J, Richards D, Sutton AJ. Collaborative care for depression: a cumulative meta-analysis and review of longer-term outcomes. Arch Intern Med. 2006;166:2314–21.
Rubenstein LV, Chaney EF, Ober S, Felker B, Sherman S, Lanto A, et al. Using evidence-based quality improvement methods for translating depression collaborative care research into practice. Fam Syst Health. 2010;28:91–113.
Oslin D, Ross J, Sayers S, Murphy J, Kane V, Katz I. Screening, assessment, and management of depression in VA primary care clinics. The behavioral health laboratory. J Gen Intern Med. 2006;21:46–50.
Post EP, Metzger M, Dumas P, Lehmann L. Integrating mental health into primary care within the Veterans Health Administration. Fam Syst Health. 2010;28:83–90.
Kirchner J, Edlund CN, Henderson K, Daily L, Parker LE, Fortney JC. Using a multi-level approach to implement a primary care mental health (PCMH) program. Fam Syst Health. 2010;28:161–74.
Parker LE, Ritchie MJ, Bonner LM, Kirchner JE. Examining inside the black box of implementation facilitation: Process and effects on program quality. Bethesda: Paper presented at the National Institutes of Health/AcademyHealth 7th Annual Conference on the Science of Dissemination and Implementation; 2014.
Ritchie MJ, Kirchner JE, Parker LE, Curran GM, Fortney JC, Pitcock JA, et al. Evaluation of an implementation facilitation strategy for settings that experience significant implementation barriers. Implement Sci. 2015;10 Suppl 1:A46.
Kirchner JE, Ritchie MJ, Pitcock JA, Parker LE, Curran GM, Fortney JC. Outcomes of a partnered facilitation strategy to implement primary care-mental health. J Gen Intern Med. 2014;29 Suppl 4:904–12.
Parker LE, Ritchie MJ, Bonner LM, Kirchner JE. Implementation facilitation and the evidence-based practice movement: a social movement analysis of practice transformation. Washington, D.C.: Poster presented at the National Institutes of Health/AcademyHealth 8th Annual Conference on the Science of Dissemination and Implementation; 2015.
Ritchie MJ, Parker LE, Kirchner JE. Evaluation of an implementation facilitation strategy for settings that experience significant implementation barriers. Bethesda: Paper presented at the National Institutes of Health/AcademyHealth 7th Annual Conference on the Science of Dissemination and Implementation; 2014.
Charns MP, Wray NP, Byrne MM, Meterko MM, Parker VA, Pucci LG, et al. Service Line Management Evaluation Project Final Report. Management Decision and Research Center; 2001. https://www.research.va.gov/resources/pubs/docs/service_line.pdf. Accessed 7 Apr 2017.
Stetler CB, Legro MW, Wallace CM, Bowman C, Guihan M, Hagedorn H, et al. The role of formative evaluation in implementation research and the QUERI experience. J Gen Intern Med. 2006;21:S1–8.
Parker LE, de Pillis E, Altschuler A, Rubenstein LV, Meredith LS. Balancing participation and expertise: a comparison of locally and centrally managed health care quality improvement within primary care practices. Qual Health Res. 2007;17:1268–79.
Kirchner JE, Parker LE, Bonner LM, Fickel JJ, Yano E, Ritchie MJ. Roles of managers, frontline staff and local champions, in implementing quality improvement: stakeholders’ perspectives. J Eval Clin Pract. 2012;18:63–9.
Parker LE, Kirchner JE, Bonner LM, Fickel JJ, Ritchie MJ, Simons CE, et al. Creating a quality improvement dialogue: utilizing knowledge from frontline staff, managers, and experts to foster health care quality improvement. Qual Health Res. 2009;19:229–42.
Parker LE, Ritchie MJ, Kirchner JE, Owen RR. Balancing health care evidence and art to meet clinical needs: policy makers’ perspectives. J Eval Clin Pract. 2009;15:970–5.
Grol R, Grimshaw J. Evidence-based implementation of evidence-based medicine. Jt Com J Qual Improv. 1999;25:503–13.
Fortney JC, Pyne JM, Smith JL, Curran GM, Otero JM, Enderle MA, et al. Steps for implementing collaborative care programs for depression. Popul Health Manag. 2009;12:69–79.
Nadeem E, Olin SS, Hill LC, Hoagwood KE, Horwitz SM. A literature review of learning collaboratives in mental health care: used but untested. Psychiatr Serv. 2014;65:1088–99.
Dusenbury L, Brannigan R, Falco M, Hansen WB. A review of research on fidelity of implementation: implications for drug abuse prevention in school settings. Health Educ Res. 2003;18:237–56.
Glasgow RE, McKay HG, Piette JD, Reynolds KD. The RE-AIM framework for evaluating interventions: what can it tell us about approaches to chronic illness management? Patient Educ Couns. 2001;44:119–27.
Glasgow RE. Translating research to practice. Lessons learned, areas for improvement, and future directions. Diabetes Care. 2003;26:2451–6.
Allen JD, Linnan LA, Emmons KM. Fidelity and its relationship to implementation effectiveness, adaptation, and dissemination. In: Brownson RC, Colditz GA, Proctor EK, editors. Dissemination and implementation research in health. New York: Oxford University Press; 2012. p. 281–304.
Dane AV, Schneider BH. Program integrity in primary and early secondary prevention: are implementation effects out of control? Clin Psychol Rev. 1998;18:23–45.
Scheirer MA. Designing and using process evaluation. In: Wholey JS, Hatry HP, Newcomer KE, editors. Handbook of practical program evaluation. San Francisco: Josey-Bass; 1994. p. 40–68.
Butler M, Kane RL, McAlpine D, Kathol RG, Fu SS, Hagedom H, et al. Integration of Mental Health/Substance Abuse and Primary Care. Evidence Report/Technology Assessment No. 173 (Prepared by the Minnesota Evidence-based Practice Center under Contract No. 290-02-0009). Rockville: Agency for Healthcare Research and Quality; 2008. Report No. AHRQ Publication No. 09-E003.
Collins C, Hewson DL, Munger R, Wade T. Evolving models of behavioral health integration in primary care. New York: Milbank Memorial Fund; 2010.
Tew J, Klaus J, Oslin DW. The behavioral health laboratory: building a stronger foundation for the patient-centered medical home. Fam Syst Health. 2010;28:130–45.
Rouse WB, Morris NM. On looking into the black box: prospects and limits in the search for mental models. Psychol Bull. 1986;100:349–63.
Hoffman RR. How can expertise be defined? implications of research from cognitive psychology. In: Williams R, Faulkner W, Fleck J, editors. Exploring expertise. New York: Macmillan; 1998. p. 81–100.
Phillips JK, Klein G, Sieck WR. Expertise in judgment and decision making: a case for training intuitive decision skills. In: Koehler DJ, Harvey N, editors. Blackwell handbook of judgment and decision making. Malden: Blackwell Publishing; 2004. p. 297–315.
Rubenstein LV, Parker LE, Meredith LS, Altschuler A, de Pillis E, Hernandez J, et al. Understanding team-based quality improvement for depression in primary care. Health Serv Res. 2002;37:1009–29.
Parchman ML, Noel PH, Culler SD, Lanham HJ, Leykum LK, Romero RL, et al. A randomized trial of practice facilitation to improve the delivery of chronic illness care in primary care: initial and sustained effects. Implement Sci. 2013;8:93.
Kilbourne AM, Goodrich DE, Lai Z, Almirall D, Nord KM, Bowersox NW, et al. Reengaging veterans with serious mental illness into care: preliminary results from a national randomized trial. Psychiatr Serv. 2015;66:90–3.
Baker R, Camosso-Stefinovic Janette, Gillies C, Shaw EJ, Cheater F, Flottorp S, et al. Tailored interventions to address determinants of practice. Cochrane Database Syst Rev. 2015. doi:10.1002/14651858.CDO05470.pub3.
Kochevar LK, Yano EM. Understanding health care organization needs and context. Beyond performance gaps. J Gen Intern Med. 2006;21:S25–9.
Kaplan HC, Brady PW, Dritz MC, Hooper DK, Linam WM, Froehle CM, et al. The influence of context on quality improvement success in health care: a systematic review of the literature. Milbank Q. 2010;88:500–59.
VanDeusen LC, Engle RL, Holmes SK, Parker VA, Petzel RA, Nealon SM, et al. Strengthening organizations to implement evidence-based clinical practices. Health Care Manage Rev. 2010;35:235–45.
Bradley EH, Holmboe ES, Mattera JA, Roumanis SA, Radford MJ, Krumholz HM. The roles of senior management in quality improvement efforts: what are the key components? J Healthc Manag. 2003;48:15–29.
Kirchner JE, Kearney LK, Ritchie MJ, Dollar KM, Swensen AB, Schohn M. Research & services partnerships: lessons learned through a national partnership between clinical leaders and researchers. Psychiatr Serv. 2014;65:577–9.
Kirchner JE, Ritchie MJ, Dollar KM, Gundlach P, Smith JL. Implementation Facilitation Training Manual: Using External and Internal Facilitation to Improve Care in the Veterans Health Administration. 2013. http://www.queri.research.va.gov/tools/implementation/Facilitation-Manual.pdf. Accessed 7 Apr 2017.
Ritchie MJ, Dollar KM, Kearney LK, Kirchner JE. Responding to needs of clinical operations partners: transferring implementation facilitation knowledge and skills. Psychiatr Serv. 2014;65:141–3.
Acknowledgements
We would like to especially acknowledge the contributions of James Townsend, DHSc, MBA, MIS, who was the Project Coordinator and who assisted with data collection and creation of program summaries. The authors are grateful to the following (listed alphabetically) for their input and source materials concerning the content and fidelity assessment of PC-MHI programs: Edmund Chaney, PhD, Brady H. Cole, MA, MPH, Johanna Klaus, PhD, John F. McCarthy, PhD, MPH, David Oslin, MD, Andrew Pomerantz, MD and Lisa V. Rubenstein, MD, MSPH.
Funding
This study, “Blended Facilitation to Enhance PCMH Program Implementation,” (Service Directed Project #08-316) was funded by the United States (U.S.) Department of Veterans Affairs’ Health Services Research & Development Service (HSR&D) and Quality Enhancement Research Initiative (QUERI). The funding body had no involvement in the design of the study or in the collection, analysis and interpretation of data and in writing the manuscript. The views expressed in this article are those of the authors and do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
Availability of data and materials
The data generated by participant interviews during this study are not publicly available because data contain potentially identifying information and participants were assured during the consent process that information they provided would be publicly available only in aggregate.
Authors’ contributions
JEK, MJR and LEP conceptualized the study. LEP and MJR developed the program component assessment instrument and the expert rating instrument and process. MJR conducted all interviews and the expert rating process. MJR and LEP analyzed the data. MJR, LEP and CNE interpreted the data and drafted the manuscript. JEK reviewed the draft and contributed to refinement of the manuscript. All authors read and approved the final manuscript.
Authors’ information
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
The VA Central Institutional Review Board (#09-05) approved the conduct of the study and provided regulatory oversight. Prior to conducting program component assessment interviews, we sent potential participants a letter describing the study and containing the elements of informed consent. Prior to conducting the interviews, we reviewed this information and obtained verbal informed consent from each participant.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Authors and Affiliations
Corresponding author
Additional files
Additional file 1:
PC-MHI Program Component Assessment interview guide. Assessment instrument/interview guide for obtaining and documenting information about the components of each facility’s program for integrating mental health services into primary care. (DOCX 102 kb)
Additional file 2:
PC-MHI program components summary template. Template for creating a summary of the components of each facility’s program for integrating mental health services into primary care. Designed for use with the “PC-MHI Program Component Assessment Interview Guide.” (DOCX 52 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Ritchie, M.J., Parker, L.E., Edlund, C.N. et al. Using implementation facilitation to foster clinical practice quality and adherence to evidence in challenged settings: a qualitative study. BMC Health Serv Res 17, 294 (2017). https://doi.org/10.1186/s12913-017-2217-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12913-017-2217-0