Abstract
Background
Acute kidney injury (AKI) represents a frequent and grave complication associated with acute pancreatitis (AP), substantially elevating both mortality rates and the financial burden of hospitalization. The aim of our study is to construct a predictive model utilizing automated machine learning (AutoML) algorithms for the early prediction of AKI in patients with AP.
Methods
We retrospectively analyzed patients who were diagnosed with AP in our hospital from January 2017 to December 2021. These patients were randomly allocated into a training set and a validation set at a ratio of 7:3. To develop predictive models for each set, we employed the least absolute shrinkage and selection operator (LASSO) algorithm along with AutoML. A nomogram was developed based on multivariate logistic regression analysis outcomes. The model’s efficacy was assessed using receiver operating characteristic (ROC) curves, calibration curves, and decision curve analysis (DCA). Additionally, the performance of the model constructed via AutoML was evaluated using decision curve analysis (DCA), feature importance, SHapley Additive exPlanations (SHAP) plots, and locally interpretable model-agnostic explanations (LIME).
Results
This study incorporated a total of 437 patients who met the inclusion criteria. Out of these, 313 were assigned to the training cohort and 124 to the validation cohort. In the training and validation cohorts, AKI occurred in 68 (21.7%) and 29(23.4%) patients, respectively. Comparative analysis revealed that the AutoML models exhibited enhanced performance over traditional logistic regression (LR). Furthermore, the deep learning (DL) model demonstrated superior predictive accuracy, evidenced by an area under the ROC curve of 0.963 in the training set and 0.830 in the validation set, surpassing other comparative models. The key variables identified as significant in the DL model within the training dataset included creatinine (Cr), urea (Urea), international normalized ratio (INR), etiology, smoking, alanine aminotransferase (ALT), hypertension, prothrombin time (PT), lactate dehydrogenase (LDH), and diabetes.
Conclusion
The AutoML model, utilizing DL algorithm, offers considerable clinical significance in the early detection of AKI among patients with AP.
Similar content being viewed by others
Introduction
Acute pancreatitis (AP) is a common disease characterized by inflammation of the pancreas, affecting adjacent local and peripancreatic tissues. Globally, it has a morbidity rate of 34 per 100,000 individuals [1]. And, cases are distributed without significant differences across age groups and genders [2, 3]. While the majority of individuals with AP experience spontaneous resolution of symptoms, around 20% are at risk of developing severe complications, such as systemic inflammatory response syndrome (SIRS) and persistent organ failure [4]. Acute kidney injury (AKI) is acknowledged as a common complication in AP cases, with its incidence ranging between 10 and 42%, particularly among critically ill patients [5, 6]. Moreover, the prognosis for AP patients who develop AKI is considerably worse, with mortality rates varying from 25 to 75%, accompanied by substantial healthcare expenditures [7, 8]. A study in the United States disclosed that the annual hospitalization costs for AKI exceeded $5.4 billion, ranking it as the second most expensive condition in the U.S. healthcare system, only surpassed by sepsis, which costs about $7.7 billion [9]. A significant portion of these expenses is attributed to cases of less severe AKI [10]. Additionally, the mildest form of AKI approximately doubles the mortality risk, which further escalates to 3–10 times in stage 2 and 3 AKI [11]. The intrinsic risks associated with AP can exacerbate these outcomes. Therefore, early and precise identification, coupled with timely intervention for AKI in AP patients, is crucial.
Previous studies in this field have predominantly utilized traditional regression methods to construct prediction models [12,13,14]. In these studies, they incorporated biological markers such as interleukin-6, serum cystatin C (CysC), and other variables, making it unfavorable for the model’s generalization and clinical application. Furthermore, these studies typically lacked comprehensive evaluation metrics such as receiver operating characteristic (ROC) curves, calibration curves, and decision curve analysis (DCA) to assess both model performance and clinical applicability.
In recent years, the utilization of machine learning (ML) in the field of medicine, including both supervised and unsupervised approaches, is gaining popularity due to its ability to leverage large clinical datasets and efficient algorithms. In comparison to traditional logistic regression (LR), ML offers notable advantages in predicting complications and other relevant aspects [15, 16]. In the realm of ML models pertinent to this field, while some demonstrated commendable performance, there was a notable increase in complexity. This complexity arose from the incorporation of variables such as serum cystatin C (CysC), intra-abdominal pressure (IAP), and the Acute Physiology and Chronic Health Evaluation II (APACHE II) score. This complexity made them less practical for clinical application and posed challenges in achieving early AKI prediction [17,18,19]. Conventional machine learning encompasses algorithms such as Support Vector Machines (SVM), Random Forests, and similar approaches. However, a novel form of machine learning known as automated machine learning (AutoML) has emerged, which can intelligently choose from a range of algorithms and hyperparameters to tailor a model specifically for the target data. In comparison to traditional machine learning, the use of intelligent techniques like early stopping, cross-validation, regularization, and hyperparameter optimization significantly reduces the time required to develop more precise models. And, in our previous research on predicting severe acute pancreatitis (SAP), the algorithm performed well, with the best model achieving a test set area under the curve (AUC) of 0.945 [20].
In this study, we utilized routine serological indicators within 24 h of admission for patients with AP as variables. We employed the H2O AutoML platform to train and validate a series of ML models for early prediction of AKI in AP patients. Additionally, we compared their predictive performance with the traditional logistic regression (LR) method.
Materials & methods
Patients
From January 2017 to December 2021, a retrospective analysis was conducted at Changshu Hospital Affiliated to Soochow University. The patients were randomly allocated into a training group and a validation group at a ratio of 7:3. As a county hospital, Changshu Hospital has established five major centers, including the Chest Pain Center, Stroke Center, Atrial Fibrillation Center, and so on.
The diagnostic criteria for AP were established according to the revised 2012 Atlanta classification [21]. To confirm a diagnosis of AP, patients needed to satisfy at least two of the following three criteria: (1) experiencing typical abdominal pain; (2) having serum amylase levels surpassing three times the upper limit of normal; and (3) displaying imaging evidence demonstrating characteristic AP findings [21]. According to the the definition of kidney disease: Improving Global Outcomes (KDIGO) guidelines, AKI could be defined in one of the following situations: (1) an increase in serum creatinine (SCr) of 0.3 mg/dl (≥ 26.5µmol/L) within 48 h; (2) known or presumed kidney damage occurring within 7 days, with SCr rising to more than 1.5 times the baseline value; (3) urine output < 0.5 ml/(kg.h) for a continuous period of 6 h [22]. Adults aged over 18 years who met the above criteria would be enrolled in this study. Patients who met the diagnostic criteria for AKI during their entire hospitalization for AP were classified as belonging to the AKI group, whereas those who do not met the criteria were classified as non-AKI group. Patients with chronic liver disease, chronic kidney disease, hematological disorders, recurrent/chronic/traumatic/idiopathic pancreatitis, pancreatic cancer, history of pancreatic resection, those who underwent chemoradiotherapy, and pregnant patients were excluded from the study. All patients received treatment following the guidelines for managing AP. This study was approved by the ethics committee of Changshu Hospital Affiliated to Soochow University (L202324).
Data collection
Electronic medical records were used to extract demographic characteristics, clinical information, and information on concomitant diseases. These parameters mainly included the patient’s gender, age, etiology, history of hypertension, history of diabetes, systolic blood pressure, diastolic blood pressure, and mean arterial pressure (diastolic pressure + 1/3 of pulse pressure).And, the etiological diagnosis of AP could be primarily determined as follows: for biliary acute pancreatitis, the diagnosis primarily depended on comprehensive inpatient examinations such as abdominal ultrasound, magnetic resonance imaging (MRI), and computed tomography (CT) scans to determine the presence of gallstones and signs of infection in the biliary system, for hyperlipidemic acute pancreatitis, the key factor was whether the serum triglyceride(TG) levels at the time of onset exceed 11.3 mmol/L, for alcoholic acute pancreatitis, the primary consideration was whether there is a clear history of heavy alcohol consumption before the onset of the disease. If the condition did not fall into any of the above three categories, it was attributed to idiopathic acute pancreatitis.
Laboratory data, including blood routine examination, coagulation tests, and serum biochemical tests, were retrospectively collected within the initial 24 h after admission. These parameters included platelet count(PLT), white blood cell count(WBC), neutrophil count(N), lymphocyte count(L), hematocrit(HCT), red cell volume distribution width(RDW), lymphocyte percentage(Lr), creatinine(Cr), total bilirubin(TB), direct bilirubin(DB), urea(Urea), lactate dehydrogenase(LDH), serum calcium(Ca2+), triglycerides(TG), glucose(GLU), alanine aminotransferase(ALT), aspartate aminotransferase(AST), gamma-glutamyl transpeptidase(GGT), alkaline phosphatase(ALP), albumin(ALB), amylase(AMY), sodium(Na+), serum potassium(K+ ), prothrombin time(PT), international normalized ratio(INR), activated partial thromboplastin time(APTT), and C-reactive protein(CRP). In total, 36 variables were analyzed, as described in Supplementary Table S1. Any missing variables were recognized as missing data at random and were imputed using a random forest algorithm via the “mice” package in R software [23]. A flowchart depicting the study is presented in Fig. 1.
Statistical analysis
If continuous variables followed a normal distribution, they were presented as mean ± standard deviation (SD), whereas if they did not follow a normal distribution, they were presented as median (interquartile range). Categorical variables were presented as frequencies. For the comparison of two groups, categorical variables were analyzed using Pearson’s Chi-square test or Fisher’s exact test, while continuous variables were analyzed using Student’s t-test or nonparametric Mann-Whitney U test. A p-value less than 0.05 (two-sided) was considered statistically significant. All statistical analyses were conducted using R software (version 4.2.1) and the following packages: H2O (version 3.36.0.2), tableone (version 0.12.0), tidyverse (version 1.3.0), tidyquant (version 1.0.2), and lime (version 0.5.1).
Logistic regression algorithms and automated machine learning algorithms
The least absolute shrinkage and selection operator (LASSO) regression model with “λmin” as the criterion was employed for conducting univariate analysis to address the issue of multicollinearity among variables. Utilizing binary logistic backward stepwise regression analysis, we identified independent risk factors. And, these factors were employed to develop a nomogram. The predictive performance of the model was evaluated by ROC curve, calibration curve, and DCA.
The machine learning approach employed the H2O package (www.h2o.ai) for AutoML, which automatically selects appropriate algorithms and incorporates them into ensemble models. This includes the default Random Forest (DRF), a randomized grid of Gradient Boosting Machines (GBMs), an Extremely Randomized Forest (XRF), a randomized array of Deep Neural Nets (DLs), and a predefined array of Generalized Linear Models (GLMs). Hyperparameter optimization involved a 5-fold cross-validation grid search on the training set, assessing various hyperparameter combinations based on their performance measured by the area under the curve (AUCs). Visualizations such as feature importance, SHapley Additive exPlanation (SHAP) plots, and Local Interpretable Model Agnostic Explanation (LIME) were used for presenting the results. Feature importance quantified the impact of each feature on the machine learning model’s outcome. SHAP provided an explanation of the influence of each feature on individual predictions, offering more intuitive and interpretable model explanations. LIME analysis illustrated the contribution of each feature to the prediction outcome by randomly providing examples from the validation set.
Results
Baseline characteristics
We included a total of 499 patients in our study, of which 62(12.4%) were excluded due to incomplete data recording. The detailed clinical characteristics and baseline data of 437 patients are presented in Table 1. In the training dataset, males accounted for 56.2% (176/313), while females accounted for 43.8% (137/313). The median age in the non-AKI group was 52 years (interquartile range 40–65 years), whereas in the AKI group, the median age was 54 years (interquartile range 42–72 years). In the validation dataset, males accounted for 55.6% (69/124), while females accounted for 44.4% (55/124). The median age in the non-AKI group was 52 years (interquartile range 38-65.5 years), whereas in the AKI group, the median age was 58 years (interquartile range 44–71 years). Biliary obstruction or gallstones (42.3%) were the most common etiology of AP in our cohort, consistent with previous research. No statistically significant differences were observed in gender, age, smoking history, hypertension history, and diabetes history between the two groups in both datasets (p > 0.05).
The follow-up time for our patients was equivalent to their length of hospital stay, ranging from 1 to 37 days (with an average of approximately 10.05 days). According to the KDIGO guidelines, 97 patients (22.2%) developed AKI during the follow-up period. Among the cases of AKI, 77cases (79.4%) were of the least severe manifestation, while stage 2 AKI was observed in 16 cases (16.5%), and stage 3 in 4 cases (4.1%). Additionally, 8 patients (8.3%) received Continuous Renal Replacement Therapy (CRRT). The onset of AKI in our patients occurred between 1 and 21 days (with an average of approximately 4.74 days). Among them, 17 cases (17.5%) were diagnosed on the first day of admission. To perform internal validation, all patients were randomly divided into a training set (n = 313, 70%) and a validation set (n = 124, 30%) using randomization software. In the training and validation sets, 68 patients (21.7%) and 29 patients (23.4%), respectively, developed AKI. Additionally, there were a total of 2 deaths (0.5%) among all patients, both of whom had AKI, and 1 patient (0.2%) had an unknow outcome.
Development of prediction model
Univariate analysis was performed using LASSO regression with the “λmin (0.020)” criterion, and 5-fold cross-validation was used to solve the problem of multicollinearity among the variables (Fig. 2). Multivariate analysis was performed using stepwise LR, and four variables were identified as independent risk factors from 36 variables, and a nomogram was plotted (Fig. 3). The calibration curves of the training and validation sets are shown in Fig. 4, with mean absolute errors of 0.025 and 0.048, respectively. This indicates a high degree of reliability of the LASSO model in estimating risk compared to the observed risk. The DCA of the validation set indicated that within the predicted AKI probability threshold range of 20–90% by the LASSO model, an intervention could potentially provide additional benefits in the range of 1–12%. If a clinician believed there was a 40% likelihood of a patient would develop AKI, the DCA analysis from the validation set suggested that the patient could potentially benefit by 10% from early intervention. This would be equivalent to identifying 10 AKI patients and advising against any unnecessary treatment for every 100 patients. This constitutes a straightforward comparison with the “treat none” strategy (represented by the horizontal line in Fig. 5), which inherently results in zero true positives and zero false positives [24]. The net benefit indicates that implementing the LASSO model would enhance patient outcomes, without regard to the preferences of either the patient or the doctor. The ROC curve for the validation set is displayed in Supplementary Figure S1, with an AUC value of 0.799, as depicted in Table 2.
We developed a total of 65 models using four machine learning algorithms (DL, GBM, GLM, and DRF) and excluded stacked ensemble models due to their poor interpretability. The DL model outperformed the other models, primarily because it achieved the highest AUC of 0.830 in the validation cohort, which was a comprehensive evaluation for imbalanced samples. As shown in Fig. 6, in the GBM model, creatinine (Cr), albumin (ALB) and lactate dehydrogenase (LDH) were the three most important features, followed by C-reactive protein(CRP)., blood urea nitrogen (Urea), sodium (Na+), prothrombin time (PT), serum potassium(K+ ), serum calcium(Ca2+)and white blood cell count(WBC). Additionally, Cr, LDH, and ALB were significant variables shared between the GBM model and the LASSO model. As shown in Fig. 7, in the SHapley Additive exPlanations (SHAP) plot of the GBM model, creatinine (Cr), albumin (ALB), lactate dehydrogenase (LDH), alanine aminotransferase(ALT), sodium (Na+), urea(Urea), C-reactive protein(CRP), gamma-glutamyl transpeptidase(GGT), activated partial thromboplastin time(APTT), and serum potassium(K+ ) were the top ten important variables. Variables with values approaching 1 exhibited a stronger correlation with an elevated risk of AKI progression in patients. For instance, the red segment of LDH, which was predominantly located to the right of the axis at 0, indicated that higher levels of LDH in AP patients were associated with an increased likelihood of developing AKI.
The locally interpretable model-agnostic explanations (LIME) plot of the DRF model on the validation set demonstrated the contributions of several important variables to the development of AKI. As shown in Fig. 8, the DRF model predicts that both case 1 and case 2 have a high probability of developing AKI, with probabilities exceeding 0.80. In both cases, albumin (ALB) was a supportive feature for the development of AKI, while urea (Urea), white blood cell count (WBC), creatinine (Cr), and C-reactive protein (CRP) did not support its occurrence. The DCA results for the validation set demonstrated that if the threshold probability of AKI predicted by the AutoML models fell within the range of 10–100%, an intervention could potentially yield an additional benefit in the range of approximately 1–15%. According to the DCA of the validation set, when a clinician considered the patient had a 10% chance of developing AKI, the patient might gain at least about 15% of the benefit from an early intervention. This is a direct comparison with treat none (the horizontal line in Supplementary Figure S2), which has zero true positives and zero false positives by default.
Comparisons models developed by LR and AutoML
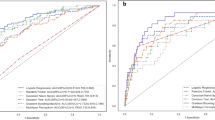
In the validation set, the AUC values of five models were as follows: DL had the highest AUC of 0.830, followed by GBM with 0.812, DRF with 0.800, LASSO with 0.799, and GLM with 0.734. Among these models, the DL model had the highest AUC and accuracy, both exceeding 0.80, and ranked first. And the AUC of machine learning models were consistently higher than that of the LR model, which is a comprehensive evaluation index for model performance. Details are listed in Table 2.
Discussion
AKI has been widely recognized as a major contributor to adverse outcomes in AP patients, with the ability to manifest at any point during the disease progression, thus exerting a significant impact on prognosis. Extensive research has revealed a substantial correlation between the occurrence of acute kidney injury and heightened risks of chronic kidney disease, myocardial infarction, stroke, and one-year mortality [25]. In this context, early and accurate prediction of which patients are more likely to develop AKI becomes crucial for proactive prevention and treatment.
In this research, we developed and validated a set of models using AutoML and LR, respectively, to enable early prediction of AKI. Compared to conventional univariate and sequential multivariate analyses, AutoML required less time and achieved higher accuracy, significantly enhancing work efficiency. Furthermore, the ensemble model integrated diverse ML algorithms and employed multiple-classifiers to predict the target outcome through a voting mechanism, thereby enhancing overall performance [26]. We employed four AutoML algorithms (GBM, DRF, GLM, and DL) to predict AKI early. All models outperformed traditional algorithm models, with the DL model ranking first in AUC on the validation set. AUC is a comprehensive indicator for evaluating model performance. As our goal is to early detect AP patients who may develop AKI, sensitivity is also important evaluation indicators. Both DL and GBM models have a sensitivity greater than 0.750. Therefore, in our study, the DL model performed the best.
In a research conducted in 2020, Wu et al. introduced a novel scoring system for forecasting organ failure in AP, which included LDH, creatinine, albumin, and serum calcium(Ca2+) as crucial variables [27]. LDH also played an important role in the classic RANSON score [28]. Several investigations have suggested the measurement of LDH activity (within 12 h of symptom onset) as a biomarker for early prognostic prediction in AP. These studies have disclosed that LDH activity can achieve a sensitivity of 63.6% and a specificity of 89.6% when discerning patients with distinct prognoses [29]. Yang et al. showed a significant correlation between elevated Cr and the high risk of AKI [19]. These findings are consistent with the results of this study, where LDH, creatinine and ALB were identified as important serum markers for early prediction of AKI in the GBM and LR models.
An increasing number of experimental and clinical studies have shown that the inflammatory response plays an indispensable role in the pathophysiology of AKI [29,30,31]. The inflammatory response has the potential to increase mucosal permeability, leading to the translocation of endotoxins. Endotoxins can promote the development of AKI by elevating endothelin levels, which in turn cause vasoconstriction, reduce renal blood flow, and result in tubular necrosis. Hence, systemic inflammatory markers like CRP and WBC may be useful in predicting AKI in AP patients, which is not surprising.
The levels of Urea can to a certain extent indicate volume depletion, kidney function, quality of resuscitation, and ischemic damage in patients with AP [32]. It has been reported that higher levels of Urea admission were associated with an increased risk of developing severe acute pancreatitis (SAP) [33]. Furthermore, Urea has been found to be a better predictor of persistent organ failure and pancreatic necrosis in acute pancreatitis compared to other laboratory indicators [34].
Multiple studies have indicated that imbalances in Na+ levels independently increase the risk of AKI [35, 36]. Given the kidneys’ pivotal role in maintaining the body’s fluid and electrolyte equilibrium, it’s not unexpected that renal dysfunction often correlates with disruptions in fluid balance and changes in serum electrolyte levels. Furthermore, research led by Lee et al. demonstrated that pre-existing hyponatremia elevates the probability of AKI occurrence in patients by approximately 30% [37].
PT representing the coagulation function of patients, was included in the list of the top 10 important features in the GBM model. This could be attributed to the fact that the hypercoagulable state of blood in patients with AP can contribute to tubular injury [38]. Moreover, hypocalcemia stands out as a key factor contributing to coagulation disturbances in individuals with AP [39]. On one hand, it is due to the release of pancreatic enzymes into the bloodstream, leading to the extensive breakdown of ALB and resulting in hypoalbuminemia, on the other hand, it is also due to the retention of calcium ions caused by the saponification of fatty acids released as a result of increased fat breakdown in abdominal adipose tissue [40]. Therefore, it is of great significance to strengthen the monitoring of Ca2+ and ALB in AP patients.
Lombardi G et al. demonstrated that deviations from normal potassium (K+) levels, as well as fluctuations within the normal range, were linked to the development of AKI. And, increased variability in K+ levels is independently associated with a higher risk of AKI, potentially due to its association with disturbances in acid-base balance [41].
The strength of this study lies in the use of AutoML to construct a series of models that can more accurately and sensitively predict AKI early compared to traditional algorithms. And the predictive factors in the models are routine detection indicators for AP, which have high clinical application value and are worth widespread application. However, this study also has some limitations. First, our analysis only used single-center data and included a relatively small number of patients. The performance of the predictive model may vary with data sets from other institutions with different patient characteristics. Second, we did not include novel biomarkers recently emphasized in research because they are not widely used in clinical practice. Third, we simply used serological indicators and did not incorporate variables such as body mass index (BMI), APACHE II, and IAP to comprehensively assess the risk of AKI in AP patients. This would affect the performance of our model. Fourth, our study is retrospective, and more prospective research is needed to further validate our findings.
Conclusions
The models developed based on the AutoML platform can assess the risk of AKI in patients with AP early and accurately. Their performance surpasses that of scoring systems constructed using traditional algorithms, holding significant clinical value. This may provide a direction for the application of AutoML in future medical research.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
References
Zhou Y, Ge YT, Shi XL, Wu KY, Chen WW, Ding YB, Xiao WM, Wang D, Lu GT, Hu LH. Machine learning predictive models for acute pancreatitis: a systematic review. Int J Med Inform. 2022;157:104641.
Greenberg JA, Hsu J, Bawazeer M, Marshall J, Friedrich JO, Nathens A, Coburn N, May GR, Pearsall E, McLeod RS. Clinical practice guideline: management of acute pancreatitis. Can J Surg J canadien de chirurgie. 2016;59(2):128–40.
Lankisch PG, Apte M, Banks PA. Acute pancreatitis. Lancet (London England). 2015;386(9988):85–96.
Xu F, Chen X, Li C, Liu J, Qiu Q, He M, Xiao J, Liu Z, Ji B, Chen D et al. Prediction of Multiple Organ Failure Complicated by Moderately Severe or Severe Acute Pancreatitis Based on Machine Learning: A Multicenter Cohort Study. Mediators Inflamm 2021, 2021:5525118.
Ljutić D, Piplović-Vuković T, Raos V, Andrews P. Acute renal failure as a complication of acute pancreatitis. Ren Fail. 1996;18(4):629–33.
Compañy L, Sáez J, Martínez J, Aparicio JR, Laveda R, Griñó P, Pérez-Mateo M. Factors predicting mortality in severe acute pancreatitis. Pancreatology. 2003;3(2):144–8.
Nasir K, Ahamd A. Clinical course of acute pancreatitis in chronic kidney disease patients in a single kidney center (PGTi) in Karachi. Arab J Nephrol Transplantation. 2012;5(2):87–90.
Petejova N, Martinek A. Acute kidney injury following acute pancreatitis: a review. Biomedical Papers of the Medical Faculty of the University Palacky Olomouc Czechoslovakia. 2013;157(2):105–13.
Hoste EAJ, Kellum JA, Selby NM, Zarbock A, Palevsky PM, Bagshaw SM, Goldstein SL, Cerdá J, Chawla LS. Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol. 2018;14(10):607–25.
Martinez DA, Levin SR, Klein EY, Parikh CR, Menez S, Taylor RA, Hinson JS. Early Prediction of Acute kidney Injury in the Emergency Department with Machine-Learning Methods Applied to Electronic Health Record Data. Ann Emerg Med. 2020;76(4):501–14.
Zeng X, McMahon GM, Brunelli SM, Bates DW, Waikar SS. Incidence, outcomes, and comparisons across definitions of AKI in hospitalized individuals. Clin J Am Soc Nephrology: CJASN. 2014;9(1):12–20.
Chai X, Huang HB, Feng G, Cao YH, Cheng QS, Li SH, He CY, Lu WH, Qin MM. Baseline serum cystatin C is a potential predictor for acute kidney Injury in patients with Acute Pancreatitis. Dis Markers. 2018;2018:8431219.
Wu C, Ke L, Tong Z, Li B, Zou L, Li W, Li N, Li J. Hypertriglyceridemia is a risk factor for acute kidney injury in the early phase of acute pancreatitis. Pancreas. 2014;43(8):1312–6.
Li H, Qian Z, Liu Z, Liu X, Han X, Kang H. Risk factors and outcome of acute renal failure in patients with severe acute pancreatitis. J Crit Care. 2010;25(2):225–9.
Arefan D, Mohamed AA, Berg WA, Zuley ML, Sumkin JH, Wu S. Deep learning modeling using normal mammograms for predicting breast cancer risk. Med Phys. 2020;47(1):110–8.
Le S, Hoffman J, Barton C, Fitzgerald JC, Allen A, Pellegrini E, Calvert J, Das R. Pediatric severe Sepsis prediction using machine learning. Front Pead. 2019;7:413.
Qu C, Gao L, Yu XQ, Wei M, Fang GQ, He J, Cao LX, Ke L, Tong ZH, Li WQ. Machine learning models of Acute kidney Injury Prediction in Acute Pancreatitis patients. Gastroenterol Res Pract. 2020;2020:3431290.
Yang D, Zhao L, Kang J, Wen C, Li Y, Ren Y, Wang H, Zhang S, Yang S, Song J, et al. Development and validation of a predictive model for acute kidney injury in patients with moderately severe and severe acute pancreatitis. Clin Exp Nephrol. 2022;26(8):770–87.
Yang Y, Xiao W, Liu X, Zhang Y, Jin X, Li X. Machine learning-assisted ensemble analysis for the prediction of Acute Pancreatitis with Acute kidney Injury. Int J Gen Med. 2022;15:5061–72.
Yin M, Zhang R, Zhou Z, Liu L, Gao J, Xu W, Yu C, Lin J, Liu X, Xu C, et al. Automated machine learning for the early prediction of the severity of Acute Pancreatitis in hospitals. Front Cell Infect Microbiol. 2022;12:886935.
Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS. Classification of acute pancreatitis–2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62(1):102–11.
Eckardt KU, Kasiske BL. Kidney disease: improving global outcomes. Nat Rev Nephrol. 2009;5(11):650–7.
Blazek K, van Zwieten A, Saglimbene V, Teixeira-Pinto A. A practical guide to multiple imputation of missing data in nephrology. Kidney Int. 2021;99(1):68–74.
Van Calster B, Wynants L, Verbeek JFM, Verbakel JY, Christodoulou E, Vickers AJ, Roobol MJ, Steyerberg EW. Reporting and interpreting decision curve analysis: a guide for investigators. Eur Urol. 2018;74(6):796–804.
Mitchell AM, Kline JA, Jones AE, Tumlin JA. Major adverse events one year after acute kidney Injury after contrast-enhanced computed Tomography. Ann Emerg Med. 2015;66(3):267–274e264.
Goh KH, Wang L, Yeow AYK, Poh H, Li K, Yeow JJL, Tan GYH. Artificial intelligence in sepsis early prediction and diagnosis using unstructured data in healthcare. Nat Commun. 2021;12(1):711.
Wu H, Li J, Zhao J, Li S. A new scoring system can be applied to predict the organ failure related events in acute pancreatitis accurately and rapidly. Pancreatology. 2020;20(4):622–8.
Mounzer R, Langmead CJ, Wu BU, Evans AC, Bishehsari F, Muddana V, Singh VK, Slivka A, Whitcomb DC, Yadav D, et al. Comparison of existing clinical scoring systems to predict persistent organ failure in patients with acute pancreatitis. Gastroenterology. 2012;142(7):1476–82. quiz e1415-1476.
Kolodecik TR, Reed AM, Date K, Shugrue CA, Patel V, Chung SL, Desir GV, Gorelick FS. The serum protein renalase reduces injury in experimental pancreatitis. J Biol Chem. 2017;292(51):21047–59.
Zhao K, Chen C, Shi Q, Deng W, Zuo T, He X, Liu T, Zhao L, Wang W. Inhibition of glycogen synthase kinase-3β attenuates acute kidney injury in sodium taurocholate–induced severe acute pancreatitis in rats. Mol Med Rep. 2014;10(6):3185–92.
Nagao S, Taguchi K, Sakai H, Yamasaki K, Watanabe H, Otagiri M, Maruyama T. Carbon monoxide-bound hemoglobin vesicles ameliorate multiorgan injuries induced by severe acute pancreatitis in mice by their anti-inflammatory and antioxidant properties. Int J Nanomed. 2016;11:5611–20.
Lin S, Hong W, Basharat Z, Wang Q, Pan J, Zhou M. Blood Urea Nitrogen as a predictor of severe Acute Pancreatitis based on the revised Atlanta Criteria: timing of measurement and cutoff points. Can J Gastroenterol Hepatol. 2017;2017:9592831.
Li YL, Zhang DD, Xiong YY, Wang RF, Gao XM, Gong H, Zheng SC, Wu D. Development and external validation of models to predict acute respiratory distress syndrome related to severe acute pancreatitis. World J Gastroenterol. 2022;28(19):2123–36.
Koutroumpakis E, Wu BU, Bakker OJ, Dudekula A, Singh VK, Besselink MG, Yadav D, Mounzer R, van Santvoort HC, Whitcomb DC, et al. Admission hematocrit and rise in blood Urea Nitrogen at 24 h outperform other laboratory markers in Predicting Persistent Organ failure and pancreatic necrosis in Acute Pancreatitis: a Post Hoc Analysis of three large prospective databases. Am J Gastroenterol. 2015;110(12):1707–16.
Aronson D, Darawsha W, Promyslovsky M, Kaplan M, Abassi Z, Makhoul BF, Goldberg A, Azzam ZS. Hyponatraemia predicts the acute (type 1) cardio-renal syndrome. Eur J Heart Fail. 2014;16(1):49–55.
Adams D, de Jonge R, van der Cammen T, Zietse R, Hoorn EJ. Acute kidney injury in patients presenting with hyponatremia. J Nephrol. 2011;24(6):749–55.
Lee SW, Baek SH, Ahn SY, Na KY, Chae DW, Chin HJ, Kim S. The effects of Pre-existing Hyponatremia and subsequent-developing acute kidney Injury on In-Hospital mortality: a retrospective cohort study. PLoS ONE. 2016;11(9):e0162990.
Papachristou GI. Prediction of severe acute pancreatitis: current knowledge and novel insights. World J Gastroenterol. 2008;14(41):6273–5.
Singh S, Dodt J, Volkers P, Hethershaw E, Philippou H, Ivaskevicius V, Imhof D, Oldenburg J, Biswas A. Structure functional insights into calcium binding during the activation of coagulation factor XIII A. Sci Rep. 2019;9(1):11324.
Condon JR, Ives D, Knight MJ, Day J. The aetiology of hypocalcaemia in acute pancreatitis. Br J Surg. 1975;62(2):115–8.
Lombardi G, Gambaro G, Ferraro PM. Serum Potassium disorders Predict subsequent kidney Injury: a retrospective observational cohort study of hospitalized patients. Kidney Blood Press Res. 2022;47(4):270–6.
Funding
Gusu Health Personnel Training Project (GSWS2020109), and Suzhou City Clinical Key Disease Diagnosis and Treatment Technology Special Project (LCZX202334) sponsored by XX.
Author information
Authors and Affiliations
Contributions
R Z and M Y are responsible for data analysis and article writing. R Z was responsible for data collection and organization. A J assisted in data collection and statistical analysis. A J and S Z contributed to data cleaning and creating charts. X X and L L managed this project and provided the funding. All authors listed have made a substantial, and direct contribution to the work and reviewed the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study has obtained approval from the Ethics Committee of Changshu Hospital Affiliated to Soochow University. Informed consent was waived by the Ethics Committee of Changshu Hospital Affiliated to Soochow University. This study was conducted in accordance to the relevant guidelines and regulations.
Consent for publication
Not applicable.
Competing interests
No conflict of interest exits in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, R., Yin, M., Jiang, A. et al. Automated machine learning for early prediction of acute kidney injury in acute pancreatitis. BMC Med Inform Decis Mak 24, 16 (2024). https://doi.org/10.1186/s12911-024-02414-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12911-024-02414-5