Abstract
Background
Folk medicine is crucial to healthcare delivery in the underdeveloped countries. It is frequently used as a primary treatment option or as a complementary therapy for malaria. Malaria is a deadly disease which greatly threatens global public health, claiming incredible number of lives yearly. The study was aimed at documenting the medicinal plants used for malaria treatment in folk medicine in Kwara State, Nigeria.
Methods
Ethnobotanical information was collected from selected consenting registered traditional medicine practitioners (TMPs) through oral face-to-face interviews using in-depth, semi-structured interview guide. The ethnobotanical data were analysed, and descriptive statistical methods were used to compile them.
Results
Sixty-two indigenous medicinal plants, including 13 new plants, used for malaria treatment were identified in this study. The TMPs preferred decoction in aqueous solvent (34%) and steeping in decaffeinated soft drink (19%) for herbal preparations. Oral administration (74%) was the main route of administration, while leaves (40%) and stem barks (32%) were the most dominant plant parts used in herbal preparations. The most cited families were Fabaceae (15%) and Rutaceae (6%), while Mangifera indica (77.14%), Enantia chlorantha (65.71%), Alstonia boonei (57.14%) followed by Cymbopogon citratus (54.29%) were the most used plants. Besides, the antimalarial activities of many of the plants recorded and their isolated phytocompounds have been demonstrated. Furthermore, the conservation status of 4 identified plants were Vulnerable.
Conclusion
The study showed strong ethnobotanical knowledge shared by the TMPs in the State and provides preliminary information that could be explored for the discovery of more potent antimalarial compounds.
Similar content being viewed by others
Background
Malaria is a deadly disease which has continued to plague global health for many centuries now, leading to an unimaginable loss of life annually. The malaria scourge is evolving, dynamic and diverse, and currently, it is concentrated in some of the poorest nations in the world [1]. In particular, the World Health Organization (WHO) African Region is affected the most where it causes huge economic setbacks, and mostly afflicts children below age 5; an outcome that is largely attributed primarily to Plasmodium falciparum, the most virulent of the five human malaria parasite species [2,3,4]. In 2021, an estimated 247 million malaria cases were reported in 84 malaria endemic countries worldwide and Africa, with 234 million cases, accounted for approximately 95% of all malaria cases globally. Death due to malaria in 2021 was estimated to be 619, 000 and the largest mortality was recorded in Nigeria [4]. The rapid spread of resistance of the malaria parasites to recommended drugs including artemisinin-based compounds [4,5,6,7,8], in addition to the high cost of antimalarial drugs and counterfeiting have made treatment and control of the disease very challenging and almost impossible [9]. Hence, this necessitates urgent need for more potent and safer alternative therapeutic agents with novel mode of action.
Since time immemorial, humans have relied upon medicinal plants for the prevention and cure of myriad of diseases and pathological disorders including malaria [10]. The application of the knowledge of medicinal plants for the treatment of various diseases has attracted the attention of researchers and formed the basis for modern pharmacology leading to the discovery and development of different therapeutic agents with plant origin [11, 12]. Today, the application of medicinal plants in folk medicine is still accepted as a preferred source of primary health care delivery in many nations including Nigeria and other parts of Africa [13,14,15,16] in spite of the paucity of pharmacological elucidation of their mechanisms of action and standard clinical trials. About 80% of the world’s population is reported to depend on the use of medicinal plants as essential sources of pharmaceutical and therapeutic needs largely because they are accessible and affordable [17,18,19].
Nigeria has a rich heritage in folk medicine in which herbal preparations comprising different medicinal plant parts are used as an alternative or to complement orthodox medicine in the prevention or treatment of many diseases and health disorders [18, 20, 21]. Till date, many Nigerians, especially those in rural and peri-urban areas, still depend on folk medicine as a major source of health care because it is readily available, conventional medicine is expensive, there is lack of adequate health care facilities, and also due to their limited access to allopathic medicine [13, 22]. Yet, others use herbal formulations most of the time simply because they trust them and their ancestors have been using them over the years.
A large number of medicinal plants that are used for malaria treatment in Nigeria, either singly or as polyherbal recipes, have been reported in previous studies [23,24,25,26]. Many of these plants have been demonstrated to possess antimalarial properties [27,28,29,30] and could serve as new leads for the discovery, design and development of more potent antimalarial agents [27]. This is predicated on the fact that the two most successful antimalarial drugs -quinine and artemisinin- were synthesised from plants [31, 32]. In the light of the above, exploration of the rich biodiversity of indigenous medicinal plant taxa through ethnobotanical and pharmacological studies becomes very important.
Ethnobotanical studies concentrate on the intricate relationship between indigenous people and local plants, including customs and cultural beliefs connected to various uses [33]. These studies help to collect vital ethnobotanical information from indigenous people including herbal practitioners in order to preserve the indigenous knowledge on the diagnosis of diseases, plant species used for treatment of diseases in folk medicine, their modes of preparation and administration, as well as the socio-cultural heritage of indigenous people for succeeding generations [34, 35]. In addition, ethnobotanical surveys are of great socio-economic importance to researchers as they are acknowledged as one of the most efficient ways of finding and documenting new medicinal plants with novel therapeutic properties and uses, and therefore enhance drug discovery and development approaches [36, 37]. This knowledge could be eroded and eventually lost to future generations without prompt and proper documentation since they are often passed through verbal communication and inappropriately documented [38, 39]. Besides, many of these plants are being destroyed due to industrialisation, urbanisation and expansion of housing programmes while some taxa are becoming endangered or threatened as a result of the loss of their natural habitats [40, 41].
The current study is premised on the observations of the historic application of medicinal plants in traditional medicine for the treatment of various human diseases among traditional medicine practitioners (TMPs) in the study area. We hypothesised that the TMPs that provide treatment for different human diseases and health disorders in the study area have valuable knowledge about the medicinal plants used for malaria treatment. Hence, this ethnobotanical survey was designed to identify and coherently document the indigenous medicinal plant taxa that are used in folk medicine for malaria treatment in Kwara State, Nigeria. Although, ethnobotanical studies of medicinal plants used in the treatment of malaria in north-eastern [42], south-western [25, 43,44,45,46], and south-eastern [23, 47, 48] regions of Nigeria have been reported, to the best of our knowledge, this is the first comprehensive account of the ethnobotanical resource of indigenous medicinal plants used for malaria treatment in the region, North Central Nigeria. The current survey was carried out as a preliminary to a larger study, the aim of which is to collect the five most used plants by the TMPs in the State for malaria treatment, evaluate them for antimalarial activity and isolate the bioactive principles in an effort to discovering new lead structures.
Methods
Description of the study area
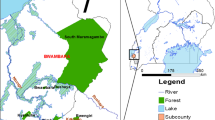
Kwara State is located in the North-Central region of the Federal Republic of Nigeria and lies between latitudes 7°45′ N and 9°30′ N and longitudes 2°30′ E and 6°25′ E. The state consists mostly of wooded savannah with forested regions in the south. It has a tropical wet and dry climate with a mean annual precipitation of about 1200 mm [49]. The distribution of rain is bimodal with long rains between April and September and short rains from October to November annually. The dry season is usually between November and March [50]. Its average annual temperature is about 26.2 ℃ and peaks at about 30 ℃ in March. Kwara State is divided into three Senatorial Districts: Kwara North, Kwara Central and Kwara South, and 16 Local Government Areas (LGAs) (Fig. 1).
The inhabitants of the State are predominantly of Yoruba-speaking ethnic group comprising Christians, Muslims and African Traditional worshippers. Others include Nupe, Bariba, Busa and Fulani minorities. They are dominated with farmers, traders, wage earners as well as those engaged in commercial services.
Selection of study participants and exclusion criteria
The respondents (Traditional medicine practitioners, TMPs) were purposively selected from the study area adopting the method of Vasileiou et al. [51]. In each location, the highest-ranking TMP was located with the help of the indigenous field assistants. We explained the aim of the study to the highest-ranking TMPs and got a verbal approval from them for the study. They subsequently gave us the list and contacts of other TMPs in the communities. All the TMPs willing to participate in the study were included in the survey. They were questioned individually on their knowledge of malaria and indigenous medicinal plant species used for malaria treatment.
Hawkers of medicinal plants used for malaria treatment were excluded from the survey. Also, indigenes and dwellers with some form of knowledge about medicinal plants used for malaria treatment were excluded from the study.
Ethnobotanical data collection and plant identification
The participants were questioned individually in their homes and workplaces on the knowledge of using medicinal plants for malaria treatment. Data on socio-demographic characteristic of the TMPs, diagnosis/symptoms of malaria and ethnobotanical uses of the indigenous medicinal plants were collected through oral face-to-face interviews by the use of in-depth, standardised semi-structured interview guide (Supplementary 1) [52]. The instrument was developed with the help of a statistician and sociologists in the Departments of Physical Sciences, and Sociology, respectively, Landmark University, Omu-Aran, Nigeria. However, the instrument was not pre-tested before its deployment since we used purposive sampling technique and did not anticipate any difficult problems during the survey. Also, the questions were easy to understand and not complex.
The survey was conducted between November and December, 2021. Ethnobotanical information including the vernacular names of commonly used plants for malaria treatment, frequently used plant parts, methods of herbal preparation, mode of administration, dosage, duration of malaria treatment and possible side effects were obtained from the participants and recorded using a recorder. They were later transcribed verbatim by the field assistants and properly documented. The bio-data of the participants including age, gender, town or village, religious affiliation, level of education and years of experience were also recorded. Three Yoruba-speaking field assistants, who are familiar with some of the medicinal plants used by the indigenous people of the State for malaria treatment, were engaged to administer and interpret the questions to the participants in their local languages, including Yoruba and Nupe, so as to facilitate efficient communication.
Photographs of the plants reported were taken to verify the taxonomic identification. Several plant identification guides were used to identify the family and species names of the medicinal plants obtained from the informants [53,54,55]. The scientific names of the plant species were checked with the “Plant List database” (www.theplantlist.org) [56] for accuracy. Thereafter, the voucher specimens were prepared and authenticated by experts, and deposited at the Forestry Research Institute of Nigeria (FRIN), Ibadan, National Institute of Pharmaceutical Research and Development (NIPRD), Abuja and University of Benin Herbarium (UBH), Benin City, Nigeria.
All methods were carried out in accordance with the relevant guidelines and regulations as stipulated in the International Union for Conservation of Nature (IUCN) Policy Statement on Research Involving Species at Risk of Extinction as well as the Convention on International Trade in Endangered Species (CITES) of Wild Fauna and Flora.
Data analysis
Data were computed and analysed using the Statistical Packages for the Social Sciences (IBM SPSS) Statistics software v25. Descriptive statistical method (percentage and/or frequency) was employed to summarise the ethnobotanical data. Results were reported as charts and tables.
Conservation status of the medicinal plants
The conservation status of the indigenous medicinal plants was collected and recorded for different conservation attributes, and classified according to the IUCN [57].
Results and discussion
Sociodemographic data of the informants
For the survey interview, a total of 35 indigenous TMPs comprising 10 (28.57%) males and 25 (71.43%) females (Supplementary 2) were randomly selected from 16 Towns/Villages across 9 Local Government Areas (LGAs) in the 3 Senatorial Districts of Kwara State. The educational background of the informants showed that 17 (48.6%), had no formal education. Seven of the informants had secondary (20%) and 6 had primary education (17.1%) respectively. Others attended Polytechnic (8.6%) and College of Education (5.7%).
The higher number of female respondents involved in herbalism could be attributed to the unique role women play both in the family and society. A similar trend was also observed in earlier reports [45, 58, 59]. The largest age group of the informants was 48–69 years old (51.43%); this was followed by those above or equal to 70 years old (25.71%) and 25–47 years old (20%) while younger TMPs less than 21 years old comprised the lowest percentage (2.86%) of informants. This corroborates earlier studies by Tchicaillat-Landou et al. [59] and Raimi et al. [60]. They reported that the majority of traditional healers who served as respondents in their ethnobotanical study was 40 and above. Also, our findings substantiate the report of Mudau et al. [61] who showed that most of the traditional healers in their survey were 41 years and above with adults between 21 and 40 years constituting the least.
Put together, the data support the concept that older generations are the main custodians of the knowledge of folk medicine and are more interested in traditional medicine practice compared with the younger generations. This development presents an enormous threat to the sustainable retention, transfer and enhancement of existing knowledge about the use of indigenous plants for folk medicine since it may eventually be lost following the demise of older generation. A possible reason for lower knowledge among young informants could be as a result of their limited interest in herbalism due to changes in lifestyle that are influenced by rapid sociocultural transformation, marked by industrialisation and modernisation. Also, due to the increasing literacy level, traditional medicine practice is becoming unpopular and unattractive to younger generations as such, they do not pay attention to the knowledge of using medicinal plants as sources of therapy.
Eleven (31.43%) of the informants had over 40 years’ experience as TMPs. This is followed by 13 (37.14%), 7 (11.25%) and 4 (11.43%), for 26–40, 11–25, and less than 10 years of experience, respectively. Regarding religious affiliation, most of the informants were Muslims (77%), followed by Christians (17%) and Traditionalist (6%). When we asked our informants how they gathered knowledge about plants and became TMPs, 48.6% stated that they were taught folk use of medicinal plants by their mother. Others received their education about herbal medicine from their father (25.7%), maternal grandparents (17.1%), maternal aunts (5.7%), and 2.9% was self-taught. This shows that transition of knowledge about the use of medicinal plants as sources of therapy is usually ancestral; passed down from one generation to another, supporting earlier findings [62, 63].
Documented medicinal plant species used for malaria treatment in Kwara State and their taxonomy
The ethnobotanical information inventoried in this survey is presented in Table 1. The vernacular/indigenous, common and scientific names of the plants, plant family, plant habit, plant parts used, use reports and citation frequency (%) are shown in the table. In this ethnobotanical investigation, a total of 62 indigenous medicinal plant species with their voucher numbers (Supplementary 3), belonging to 58 genera distributed across 36 families were revealed by the TMPs to be used in traditional health care system for malaria treatment (Table 1).
Many of the medicinal plants identified in this study are also utilised for malaria treatment in other parts of Nigeria [24, 43, 64]; and several other nations in Africa including Benin Republic [65]; Cameroon, Kenya and Namibia [66]; Ethiopia [67, 68]; Togo [69] and Zimbabwe [70]. Additionally, they have also been reported to be used for the treatment and management of a wide range of ailments and health disorders such as chronic diarrhoea, jaundice, insomnia and rheumatism [71, 72]; asthma, warts, cancer, chicken pox and gonorrhoea [23, 73]; fever, hepatitis, tuberculosis and urinary tract infections [74, 75]; cough, bronchitis, laryngitis and hoarseness of voice [76,77,78]; nasopharyngeal, heamorrhoids, skin infections and infertility [79]; bronchitis, stomach ache, monorrhagia, high blood pressure and diabetes [80, 81].
When compared to similar ethnobotanical surveys carried out in north-eastern [42], south-western [9, 43,44,45, 64] and south-eastern Nigeria [23], this present study recorded a higher number of plant taxa used for malaria treatment. Nevertheless, it recorded fewer number of plants in comparison to Oyeyemi et al. [24] in south-western Nigeria. Noteworthily, 13 new plants which have not been reported in previous ethnobotanical surveys of plants used for malaria treatment in Nigeria were identified in this study (Acanthus montanus, Calliandra haematocephala, Pseudocedrela kotschyi, Vitellaria paradoxa, Ancistrophyllum secundiflorum, Waltheria indica, Byrsocarpus coccineus, Piliostigma thonningii, Cussonia barteri, Senna occidentalis, Tetracera potatoria, Ficus platyphylla and Entada africana). Altogether, the data obtained in this study contribute to the concerted effort globally to record local plants and their accompanying indigenous knowledge for the benefit of the present and succeeding generations [26, 82]. Additionally, the present study adds to the existing collection of medicinal plants in Nigeria [26, 43, 45, 73, 78, 83, 84].
Our results showed that a total of 36 medicinal plant families were used as traditional antimalarial medicine (Table 1). Regarding the number of identified species, the family Fabaceae was the most dominant plant family, represented with 9 species (15%). This was followed by Rutaceae 4 species (6%), Poaceae, Meliaceae, Malvaceae and Moraceae with 3 species (5%) each. Twenty-three (63.89%) other families were represented with a single species (2%) each. The dominance of Fabaceae as the most represented family has been reported in previous ethnobotanical surveys in Nigeria [45, 73, 78, 85, 86], as well as in other African countries including Ghana [87], South Africa [88], the Democratic Republic of Congo [89], and Uganda [90]. These data suggest a richness of their local flora species and affirm the popularity of Fabaceae for their curative effectiveness.
Habit of plants utilised for malaria treatment among the informants
The inventory of 62 indigenous medicinal plants was represented by different plant habits which were dominated by woody species (81%) comprising trees (57%) and shrubs (24%) (Fig. 2). The remaining 20% of the plants were distributed among herb (6%), climber (6%), grass (5%) and weed (2%). The strong connection between the predominant local plants corresponds to the dominance of plant habit used for medicine among the TMPs [87, 91]. In comparison with herbaceous plant species, the preponderance of trees and shrubs in folk medicine is linked to their comparatively longer accessibility and persistence of the various plant parts used [92].
Parts of the medicinal plants used for preparation of herbal medicine for malaria treatment
Figure 3 showed that leaves (40%) were the most utilised medicinal plant parts for preparation of herbal medicine for malaria treatment, solely or in combination with other plant parts. This was followed by stem bark (32%), root (18%) and fruit (6%) while the least commonly used parts were rhizome (2%), husk (1%).
Plant parts are capable of accumulating different important natural phytocompounds, which offer significant pharmaceutical potentials [93]. The dominance of leaves over other plant parts in preparing herbal remedies could be linked to their being the primary site of photosynthetic and other biosynthetic activities in plants, leading to the production and accumulation of photosynthates which contain higher concentrations of bioactive molecules including alkaloids and tannins with rich medicinal properties [94,95,96]. Another reason could be because of the ease of handling them [97]. Again, leaves are more readily available hence, they are easily accessible and harvested in large quantities for use when compared to other parts of the plant. Harvesting leaves does not exert much strain on plant regeneration and also does not extensively harm the plants compared to the use of stem barks, roots, and/or the whole plant [98, 99]. Furthermore, from a conservation and sustainability point of view, leaves are preferred over stem barks and roots since they are not as closely linked to the survival rate of plants. So, collecting the leaves biomass within acceptable limits does not cause serious interference with the plant life.
Traditional malaria medicine recipes, modes of preparation, routes of administration and duration of treatment
The TMPs revealed different recipes for preparing herbal medicine for malaria treatment, modes of preparation and administration, as well as duration of administration with possible side effects. Overall, 35 different polyherbal recipes were obtained from the informants. The recipes contain various plant parts including leaves, stem bark and roots, and ranged from 3 to 13 different plants used together. Although some of the plants can be used singly, earlier reports have claimed that the use of plant mixture in herbal preparation may enhance the synergism of phytochemicals of the different plants so as to elicit maximum therapeutic efficacy, and also cure several malaria-associated dysfunctions in the body [43, 90, 100].
Of the 11 different traditional methods reported to be used for preparation of malaria therapy by the TMPs, decoction in water (34%) was the most preferred method of extracting the bioactive ingredients from the plants (Fig. 4). The different components of the recipes are uniquely arranged with the stem bark and/or root usually cut into smaller pieces and placed at the bottom of the pots followed by the leaves, fruits and other plant parts. Other modes of preparation included steeping (infusion) in non-caffeinated soft drink (7up) (19%), steeping in alcohol “ogogoro” (11%) and steeping in water from fermented maize “omi ogi” or “omi idun” (10%). The least preferred methods were squeezing of leaves in lime juice (1%), steeping in coconut water (1%) and pulverization into powder (1%). The choice of decoction and steeping as the most preferred traditional methods of preparing malaria herbal remedies is consistent with earlier reports [101,102,103,104]. The main reason why these modes of preparation are widely utilised by the TMPs could be because they are simple, easy to handle and cheap [78]. In addition, decoction enhances extraction of bioactive ingredients from the plant parts much more in comparison to cold extraction. However, several factors including boiling duration, amount of solvent and plant material used may differ which could possibly affect the potency of the herbal preparations [61]. Also, both decoction and steeping do not provide long shelf life for the herbal preparations and as a result, the medicinal plants would need to be continuously harvested thereby putting them under considerable pressure that may lead to overexploitation.
Regarding administration of herbal preparation, oral administration (74%), bathing with water from decoction (15%), steam inhalation (9%) and pulverized form of plant parts taken with cornmeal (2%) were reported to be the routes of administration (Fig. 5). In this report, oral administration was considered to be the main route of administering herbal preparations, consistent with previous investigations [63, 105,106,107].
Pharmacological relevance of the extracts and phytocompounds isolated from the identified plants
As a result of the growing interest in phytoremediation as an alternative therapeutic strategy for combating malaria, many bioactive compounds have been extracted, isolated and characterised from medicinal plants using different methods including chromatographic and spectrophotometric techniques [108,109,110,111]. Interestingly, most of the medicinal plants recorded in this study have been demonstrated through in vitro and/or in vivo approaches to possess antimalarial activity (Table 2), supporting the traditional use of these plants for malaria treatment in the region. Over 50 compounds (Table 3), with varying antimalarial activities, have been isolated from some of the plants documented in this study including E. chlorantha, V. amygdalina, M. lucida, A. occidentale, H. madagascariensis, K. africana, G. kola, A. indica, X. aethiopica, C. papaya and P. americana. The following is a brief description of some of the medicinal plants and their constituents previously investigated for their antimalarial property.
Alstonia boonei De Wild is an evergreen tree, that is widely distributed in the tropical and rain forests zones of West and Central Africa. The plant is listed in African pharmacopoeia and is commonly applied in Nigeria for the treatment of a variety of ailments including malaria, chronic diarrhoea, insomnia, jaundice and rheumatic pains [72, 189]. Herbal tincture and decoction of the stem bark of the plant is also used as an effective antidote against scorpion or snake poison, as well as for inducing lactation and expelling retained products of afterbirth when administered to women [190]. Infected mice with the chloroquine-sensitive Plasmodium berghei NK65 parasite were used to study the in vivo anti-plasmodial effects (suppressive, curative and prophylactic models) of the ethanolic stem bark extract of the plant. The extract demonstrated significant (p < 0.05) suppressive (46.43–75%), curative (61.02–81.36%) and prophylactic (34.83–60.67%) antiplasmodial effects against the parasite in a dose-dependent (100, 200 and 400 mg/kg) manner [131]. Also, Agbedahunsi et al. [130] investigated the in vivo chemosuppressive effect of the ethanolic leaf extract of the plant in P. berghei–infected mice and their results revealed a chemosuppression of 0.2–74.8%, dose-dependently (12.5–400 mg/kg).
Anacardium occidentale L. is a nut tree crop widely cultivated in tropical regions of the world including Nigeria. Decoction of the leaves and stem bark have been reported to be used in ethnomedicine for malaria treatment and management of severe diarrhoea in Nigeria [64, 71]. The in vitro antiplasmodial investigation of ethanol extract of cashew apple demonstrated significant activity with an IC50 of 0.577 g/ml [126]. Besides, the phytocompounds cardol triene 1 (IC50 = 5.69 M) and 2-methylcardol triene 4 (IC50 = 5.39 M) isolated from the plant showed significant antimalarial activity in vitro when evaluated against P. falciparum D6 strain [126]. Additionally, anacardic acid, another phytocompound from A. occidentale, was demonstrated to possess antiplasmodial effect [127]. The compound interrupted the parasite’s transcription process by inhibiting P. falciparum histone acetyltransferase (PfGCN5) activity.
Azadirachta indica A. Juss. is a tropical and subtropical plant which is utilised for various medicinal purposes. In the tropics, the plant is used as traditional remedies for the treatment of malaria [43, 46]. Several studies have shown the seed kernel, leaf and stem bark extracts to possess antimalarial properties [191,192,193]. In an in vitro study, Hout et al. [194] reported a good activity (4.7 µg/ml IC50) of dichloromethane stem bark extract of A. indica against chloroquine resistant P. falciparum W2 strain. Tepongning et al. [114] reported that P. berghei-infected BALB/c mice treated with hydroethanolic leaf extract of the plant showed a significant (p ≤ 0.001) reduction of parasitaemia ranging from 49.75 ± 3.64 to 69.28 ± 1.36% dose-dependently. The antimalarial efficacy of tablet suspension of the leaf and bark of the plant were assessed on P. yoelli nigeriensis-infected mice. The tablet suspensions demonstrated high preventive to moderate suppressive and weak curative schizonticidal effects, respectively [195]. Furthermore, the in vitro antimalarial activities of various phytocompounds such as nimbolide, gedunin and azadirachtin isolated from A. indica have been reported [116,117,118].
Carica papaya L. is commonly administered in traditional settings for the treatment of malaria and various other maladies including cancer, asthma, warts, jaundice and malaria [23]. Using bioassay-guided fractions and dichloromethane extract, Teng et al. [135] evaluated the antimalarial activity of C. papaya leaf extracts in vitro against P. falciparum 3D7 and Dd2 strains. The hexane extract was the most potent of the extracts obtained from C. papaya leaves; it had an IC50 of 3.43 ± 0.41 and 1.52 ± 0.003 g/ml against the 3D7 and Dd2 strains, respectively. This was followed by dichloromethane leaf extract which had an IC50 of 7.67 ± 1.9 and 4.50 ± 0.17 g/ml against the 3D7 and Dd2 strains, respectively. The ethyl acetate leaf extract was moderately potent against the P. falciparum 3D7 strain, with an IC50 of 20.32 ± 3.5 g/mL, while the leaf juice at a dilution of 1/64 inhibited its development by 50%. In another study, Melariri et al. [136] demonstrated the antiplasmodial activity in vitro of the leaf extract of the plant. Their result showed that ethyl acetate crude extract had a very good activity with an IC50 of 2.96 µg/ml against P. falciparum D10 strain. The phytocompound carpaine isolated from C. papaya exhibited very good activity against P. falciparum 3D7 and Dd2 strains with IC50 of 2.01 ± 0.18 μg/ml (4.21 µM) and 2.19 ± 0.60 μg/ml (4.57 µM) against 3D7 and Dd2 strains, respectively [135]. Furthermore, both linoleic and linolenic acids isolated from the ethyl acetate leaf fraction of the plant showed good activity with IC50 of 6.88 μg/ml and 3.58 μg/ml against P. falciparum chloroquine-sensitive D10 and chloroquine-resistant DD2 strains, respectively [136].
Daniella oliveri (Rolfe) Hutch. & Dalziel grows predominantly in some parts of South America and Africa. In Nigeria, the plant is used in traditional medicine for the treatment of breast tumours, abscesses, and vestibule vagina fistula [196]. Also, herbal preparations with the leaves and stem bark are used for the treatment of gastrointestinal disorders, diabetes and diarrhoea, as well as malaria [197, 198]. The antiplasmodial effect of the methanol, aqueous, butanol and ethyl acetate stem bark extract revealed weak activity (IC50 of 23.14–32.97 µg/ml) against P. falciparum 3D7 and K1 strains [168]. But a suppression effect of 87% against P. berghei was recorded, though at a higher dose of 700 mg/kg [183].
Enantia chlorantha Oliv. is widely distributed along the coastal regions of West and Central Africa including Nigeria where it is utilised in traditional medicine for the treatment and management of several health disorders including urinary tract infections, jaundice, malaria, fever, tuberculosis and hepatitis [74, 75]. A decoction of 500 g of stem bark of the plant in 3 L of water for 20 min, taken orally, has been reported to treat malaria symptoms, aches, wounds, fever, and chills [199]. Boyom et al. [112] investigated the in vitro antiplasmodial activity of solvent fractions and ethanolic crude extract of E. chlorantha stem bark against P. falciparum W2 strain, and they reported a good activity at IC50 of 0.68 to 14.72 µg/ml. Also, the antimalarial activities of the protoberberine alkaloids compounds – jatrorrhizine, berberine and palmatine isolated from E. chlorantha have been tested both in vitro against P. falciparum and in vivo against P. berghei. They showed potency similar to that of quinine in vitro however, none of the compounds was active in vivo [113].
Ficus platyphylla Del. Holl is a deciduous heavily branched tree that is distributed widely throughout the savannah region of West African coast. In folk medicine, decoction of the seeds, leaves and stem bark of F. platyphylla is taken as fertility enhancement in Nigeria [200]. It is also used for the management of epilepsy, psychosis [201], and tuberculosis [202]. In Burkina Faso, the stem bark of the plant is used traditionally for malaria treatment [203]. In vivo antiplasmodial activity of the ethanolic stem bark extract of the plant suppressed malaria dose-dependently in P. berghei-infected mice, by 43.50% at the highest dose of 300 mg/kg [185]. Additionally, treatment with the plant extract prevented severe reduction in packed-cell volume in the infected mice revealing its capacity to remedy anaemic conditions.
Garcinia kola Heckel is a flowering plant widely distributed in the tropical rain forest region of West and Central Africa, and it is largely valued for its nuts. The seeds, commonly known as “bitter kola”, are edible and are usually chewed as an adjuvant to the true kola (Cola nitida and C. accuminata). In traditional medicine, preparations with different parts of the plant including the seeds, leaves and stem bark are used extensively as purgatives, aphrodisiac, as well as for the treatment of diarrhoea, liver diseases, cough, hoarseness of voice, laryngitis and bronchitis [76, 77, 204]. In vitro and in vivo antimalarial studies have been carried out on the plant. The results from Tona et al. [157] revealed that the ethanolic extract of the stem bark and its petroleum ether fraction showed very good antiplasmodial activities with IC50 values of < 3 µg/ml in vitro. In another study by Ujomu et al. [158], it was observed that n-hexane, dichloromethane and methanol extracts of the seeds of G. kola were active in vitro against chloroquine-sensitive P. falciparum D10 strain (10.59–26 µg/mL IC50). They also reported that the n-hexane extract reduced parasitaemia in P. berghei-infected mice by 70% at 400 mg/kg, prolonging survival of the animals. Three biflavanones (I-4′,II-4′,I-5,II-5,I-7,II-7-hexahydroxy-I-3,II-8-biflavanone, II-3,I-4′,II-4’,I-5,II-5,I-7,II-7-heptahydroxy-I-3,II8-biflavanone and II-3, 3′,I-4′,II-4′,I-5,II-5,I-7, II-7-octahydroxy-I-3,II-8-biflavanone) isolated from G. kola demonstrated potent inhibitory activity in vitro against P. falciparum proliferation and against P. berghei in vivo [160]. I-4′,II-4′,I-5,II-5,I-7,II-7-hexahydroxy-I-3,II-8-biflavanone exhibited the strongest in vitro antimalarial potency on P. falciparum with an IC50 of 0.16 μM. In the in vivo antimalarial assay in P. berghei-infection in mice, I-4′,II-4′,I-5,II-5,I-7,II-7-hexahydroxy-I-3,II-8-biflavanone was found to exhibit antimalarial effect with an ED50 of about 100 mg/kg following oral treatment. I-4′,II-4′,I-5,II-5,I-7,II-7-hexahydroxy-I-3,II-8-biflavanone was also found to increase the average life span of the infected mice significantly when compared to that of the control (p < 0.01).
Harungana madagascariensis Lam. ex Poir. is found in tropical Africa with wide distribution in areas where annual rainfall is above 1300 mm. In traditional medicine, preparations from different parts of the plant including the stem bark and leaves are used in the treatment of urogenital infections, chest pain, river blindness, hepatitis, toothache, dysmenorrhea, asthma, and malaria [205]. Iwalewa et al. [145] evaluated the in vitro and in vivo antimalarial activity of the stem bark extract of H. madagascariensis. The IC50 of the ethanolic extract of the plant on P. falciparum was between 0.052 and 0.517 μg/ml as against the standard drugs artemether (0.021 g/ml) and chloroquine (0.0412 g/ml). The activities of the extract in an in vivo study on P. yoelii nigeriensis were between 28.6–44.8 and 30.2–78.2% at 20–80 mg/kg in both suppressive and prophylactic assays, respectively, in comparison to chloroquine (70.6%) and pyrimethamine (43.3%). However, in the curative test, only 80 mg/kg of the extract decreased the level of parasitaemia in comparison to the standard drug chloroquine. In a similar report, the methanolic leaf extract of the plant showed weak in vitro activity (39.07 ± 1.57 μg/ml) against P. falciparum D6 strain and moderate chemosuppression of parasitaemia (53.13%) [144]. The aqueous leaf extract was inactive in vitro but showed high chemosuppression of parasitaemia (88.04%). Lenta et al. [146] evaluated the antiplasmodial activity of compounds harunganin, harunganol A, harunganol B, feruginin and bazouanthrone isolated from H. madagascariensis against P. falciparum W2 strain. All the compounds showed varying activities against the malaria parasite with bazouanthrone being the most potent (IC50 = 1.80 μM).
Jatropha curcas Linn. is a semi-evergreen shrub found in abundance in Mexico, northeastern part of South America as well as in some tropical and sub-tropical regions in Asia and Africa. Herbal preparations of the stem, leaves and seeds are widely used for various medicinal purposes in traditional settings in Africa. Decoction of the leaves is utilised to treat cough while the seed oil is used for treating several skin diseases and soothing rheumatic pain. Decoction of the leaves is also applied as an antiseptic after child delivery [206]. Moreover, the seeds are used as purgative and laxative, and for the treatment of helminthic infections, paralysis, ascites, and gouts [207]. Furthermore, drops of diluted twig-sap are administered orally to babies affected by tetanus [208]. An in vitro study of the ethyl acetate, hexane and methanol leaf extract of the plant leaves demonstrated good to moderate antiplasmodial activity (2.39 ± 0.54- 31.09 ± 4.36 µg/ml IC50) against P. falciparum K1 and NF54 strains [209].
Kigelia africana (Lam.) Benth. is a monospecific genus under the family Bignoniaceae which grows along watercourses and in riverine areas in West, Central and South Africa [210]. Different parts of the plant are utilised in traditional medicine for the treatment of various ailments including rheumatism, heamorrhoids, nasopharyngeal, skin infections, malaria and for fertility enhancement [79, 211]. Imran et al. [9] evaluated the extracts of the leaf, stem bark, fruit and root of K. africana in a 4-day antiplasmodial test in P. berghei-infected mice, they all showed dose-dependent chemosuppressive activity at the three administered doses of 125, 250 and 500 mg/kg, respectively. The stem bark extract exhibited the highest chemosuppressive activity of 84.73%, followed by the root with 78.06% and then the leaf (72.94%) while the fruit had the least chemosuppressive activity of 70.33% at the maximum dose of 500 mg/kg compared to the standard drug chloroquine with 93.99% chemosuppression of parasitaemia. They also evaluated the antiplasmodial effect of different solvent fractions of the extract: the chemosuppressive activity of the ethyl acetate, n-butanol, and methanol fractions were 42.61, 56.05 and 69.94%, respectively in comparison with the positive control (chloroquine) which had chemosuppression of 86.17%. Zofou et al. [148] isolated the phytocompounds p-hydroxycinnamic acid, specicoside, 2β,3β,19α-trihydroxy-urs-12-en28-oic acid, and atranorin from the stem bark of the plant and evaluated them against the multidrug-resistant W2mef strain of P. falciparum. Three of the four compounds tested showed significant activity against W2mef: specicoside (IC50 = 1.02 ± 0.17 μM), 2β,3β,19α-trihydroxy-urs-12-en28-oic acid (IC50 = 1.86 ± 0.15 μM), and atranorin (IC50 = 1.78 ± 0.18 μM) while on the other hand, p-hydroxycinnamic acid showed a moderate activity (IC50 = 12.89 ± 0.87 μM).
Margaritaria discoidea (Baill.) G.L. Webster is a tree that can grow up to 30 m in height. The plant abounds in Senegal, Cameroun and other parts of tropical Africa. The stem bark of the plant is used in folk medicine to treat malaria and helminthic infections in Cote d’Ivoire [212]. It is also utilised for the treatment of onchocerciasis in Cameroon [213]. Additionally, the decoction of the stem bark is used in the Republic of the Congo to relieve stomach discomfort, and facilitate delivery during parturition [214]. The aqueous, ethanol and methanol leaf extracts of M. discoidea were assessed for their in vitro antiplasmodial activity against chloroquine sensitive (NF54) and multi-resistant (K1) strains of P. falciparum [115]. Moderate to weak activity (13.60 ± 1.80- 43.61 ± 0.92 µg/ml IC50) against the K1 and NF54 strains was reported.
Morinda lucida Benth is a small to medium-sized tree which grows in grassland, forests and occasionally in regularly flooded areas. Decoctions and infusions of the leaves, stem bark and root of the plant are utilised as remedies in traditional settings against trypanosomiasis, feverish condition during child delivery, yellow fever, and malaria [215]. The antimalarial effects of extracts of different parts of M. lucida have been demonstrated. Olasehinde et al. [121] reported the antiplasmodial effect in vitro of the methanolic extract of M. lucida leaf. An excellent activity against P. falciparum was recorded at IC50 of 25 nM. Treatment of P. berghei-infected mice with 200 and 400 mg/kg hydroethanolic (50:50) leaf extract of the plant significantly (p > 0.05) reduced the level of parasitaemia (96.24%), but was slightly lower than that recorded for the standard compound chloroquine (100%) [120]. In a similar manner, decoction of M. lucida root in distilled water demonstrated significant (p < 0.05) chemosuppressive (60.9 ± 0.01%), curative (85.1 ± 0.04%) and prophylactic (74.6 ± 0.03%) activities in P. berghei NK65-infected mice at 1200 mg/kg [119]. Additionally, the activity of three anthraquinone compounds – digitolutein, rubiadin 1-methyl ether, and damnacanthal isolated from the stem bark and the root of the plant against P. falciparum have been demonstrated in vitro. The number of parasites significantly decreased in a dose-dependent manner, and 100% inhibition was recorded with 30–40 μg of each compound [122]. In a similar manner, the two triterpenic acids – ursolic and oleanolic acids isolated from the plant showed very good to moderate antiplasmodial activity with IC50 values of 3.1 ± 1.3 and 15.2 ± 3.4 µg/ml, respectively [123].
Persea americana Mill. is a tropical plant that produces an edible fruit called avocado. The plant is used in ethnomedicine in Nigeria and other parts of Africa for the treatment of different health conditions including monorrhagia, rheumatism, stomach ache, high blood pressure, diarrhoea, bronchitis, diabetes and malaria [23, 80, 81]. The curative and suppressive antimalarial activities of extract and fractions of P. americana leaf have been demonstrated in vivo [176]. In the curative model, the extract produced inhibition (p < 0.05) of parasitaemia in a dose-dependent manner. The inhibition produced by 400 mg/kg of the extract (methanol in water) was 52.16 ± 2.77%, similar to that of the standard drug artemisinin-combination therapy (ACT) with 69.04 ± 3.02% inhibition. The extract produced significant (p < 0.05) chemosuppression (55.00 ± 0.06%) in parasitemia. Similarly, ethyl acetate, hexane, and aqueous fractions of the plant produced significant (p < 0.05) chemosuppressive effect by 40.00 ± 0.05, 56.03 ± 0.07 and 64.01 ± 0.08%, respectively at 200–400 mg/kg. However, the effects of the fractions were lower than the standard drug ACT (70.00 ± 0.06%) with only the aqueous fraction (64.01 ± 0.08%) producing a similar activity. The avocadenols, 1, 2, 4-trihydroxyheptadec-16-ene and 1, 2, 4, 15- tetrahydroxyheptadecane-6, 16-diene isolated from P. americana seeds showed promising antiplasmodial activity when investigated for their antiplasmodial effect in vitro [177].
Vernonia amygdalina Delile is a perennial rainforest herb which is commonly used as a vegetable in preparation of the popular bitter leaf soup as well as for other medicinal purposes in folk medicine in Nigeria [216]. Extracts of the herb have been utilised in ethnomedicine for the treatment of fevers, hiccups, stomach disorders, kidney problems, and malaria [217, 218]. The antimalarial effects of V. amygdalina have been reported. Omoregie et al. [137] demonstrated the in vitro and in vivo activities of ethanolic, aqueous and hydroethanolic (50:50) leaf extracts of the plant. In the in vitro study, the ethanolic extract produced the highest (p < 0.05) antiplasmodial activity (IC50 = 9.83 µg/ml) against P. falciparum 3D7 strain. Also, the ethanolic extract was significantly active in vivo against P. berghei, dose-dependently, with maximum activity observed at 1,000 mg/kg (82.3% % inhibition). In a 4-day chemosuppression test, a parasitaemia suppression of 67% in P. berghei-infected mice was demonstrated following oral administration of methanolic extract of the plant at a dose of 1,000 mg/kg [138]. Also, an aqueous extract of the plant administered orally to P. berghei-infected mice at 125 mg/kg reduced the parasitaemia by 63% [139]. Various compounds including steroidal saponins vernoniosides A1, A2, A3, A4 and B1, as well as sesquiterpenes vernodalin, vernodalol, vernolide and hydroxyvernolide isolated from the leaves of the plant have been shown to possess varying degrees of antimalarial effects in vitro. The compounds had antiplasmodial activities with IC50 between 4.0 and 46.1 µg/ml and vernodalin was observed to be the most potent (IC50 = 4.0 µg/ml) [140].
Xylopia aethiopica (Dunal) A. Rich is a sweet-smelling evergreen medicinal plant usually found in the forest-Savannah zone and the rainforest region of Africa [219]. Various parts of the plant including fruit, leaves, stem, stem bark and root are utilised in folk medicine for the treatment of different diseases and health disorders such as rheumatism, asthma, infertility, dysentery, epilepsy, candidiasis, fever, and malaria [220,221,222]. The in vitro antimalarial effect of the volatile oil of X. aethiopica seed was evaluated against P. falciparum 3D7 strain, a moderate activity against the malaria parasite was observed with an IC50 of 14 µg/ml [124]. In a similar vein, the antimalarial potential of the kaurene diterpene compound xylopic acid isolated from the fruit of the plant was assessed in P. berghei-infected mice and it showed promising activity (p < 0.05), comparable to that of the standard drug artemether/lumefantrine [125].
In addition to the plants above, the antiplasmodial activity of the underlisted plants have been evaluated in vitro and/or in vivo and are summarized in Table 2: M. indica, C. limon, C. citratus, L. inermis, N. latifolia, A. altilis, C. longa, T. cacao, L. alata, C. nucifera, S. acuta, Z. zanthoxyloides, C. aurantifolia, P. guajava, K. ivorensis, A. nilotica, B. coccineus, W. indica, A. leiocarpus, A. compressus, V. paradoxa, Z. officinale, P. kotschyi, A. comosus, O. gratissimum, P. africana, A. altilis, C. sinensis, F. exasperata, P. nigrescens, P. thonningii and P. guineense.
Conservation status of the medicinal plants
The conservation status of the 62 medicinal plant species reported in this study showed that 30 plants were found to be Least Concern (LC), data for 24 were Not Evaluated (NE), 4 each were both Data Deficient (DD) and Vulnerable (VU) i.e., they meet any of the criteria A-E for Vulnerable [57, 223]. Hence, this implies that the VU plants are more likely to go extinct in the wild [223]. Findings from this present study showed that none of the documented indigenous medicinal plant species was found to be Near Threatened (NT), Endangered (EN), Critically Endangered (CR), Extinct in the Wild (EW) and Extinct (EX) suggesting that they have not been overexploited. However, conservation strategies should be intensified to preserve and prevent the plants, especially the VU, from becoming Endangered or Extinct so as to ensure their sustainable availability and biodiversity. This can be achieved by planting these trees through afforestation and forestation programmes to guarantee the sustainable use of the exploited plant taxa [224, 225].
Conclusion
This is the first comprehensive ethnobotanical study carried out in the region revealing important medicinal plant taxa diversity and ethnomedicinal knowledge held by the TMPs. The current study highlighted the use of indigenous medicinal plants for malaria treatment and fills a gap in our ethnomedicinal knowledge about Kwara State and Nigeria in general. We showed that folk medicine is still being practiced in the State which harbours a wide variety of ethnoflora. A total of 62 medicinal plant species including 13 new plants used for malaria treatment in the State were identified. Although, these medicinal plants are usually combined as recipes for herbal preparations, many of them and their isolated phytocompounds have shown in vitro and/or in vivo antiplasmodial activities against P. falciparum and P. berghei while some are yet to be evaluated.
Due to their high percentage citation frequency, M. indica, E. chlorantha, A. boonei, C. citratus and N. latifolia were identified as the most widely used medicinal plants for malaria treatment in folk medicine among the TMPs thus, have great potential to be used in further ethnopharmacological research. This could help contribute to the provision of “Good Health and Well-being”, Goal 3 of the United Nations Sustainable Development Goals (UN SDGs), for the hundreds of millions of people infected with the human malaria parasites, and the achievement of the WHO Global Technical Strategy for Malaria goal – “reduction of global malaria burden by 90% in 2030”.
Altogether, the data in this current study contributes to both national and international efforts aimed at documenting the local use of indigenous medicinal plants with antimalarial potentials and provides preliminary information for future pharmacological, toxicological and conservation studies.
Availability of data and materials
The data generated or analysed during this study are included in this article.
Abbreviations
- CITES:
-
Convention on International Trade in Endangered Species
- CR:
-
Critically Endangered
- DD:
-
Data Deficient
- EN:
-
Endangered
- EW:
-
Extinct in the Wild
- EX:
-
Extinct
- FHI:
-
Forest Herbarium Ibadan
- FRIN:
-
Forestry Research Institute of Nigeria
- IC50 :
-
Half maximal inhibitory concentration
- IUCN:
-
International Union for Conservation of Nature
- LC:
-
Least Concern
- LGA:
-
Local Government Area
- LUCRID:
-
Landmark University Centre for Research, Innovation and Discoveries
- NE:
-
Not Evaluated
- NIPRD:
-
National Institute of Pharmaceutical Research and Development
- NT:
-
Near Threatened
- TMP:
-
Traditional medicine practitioner
- UBH:
-
University of Benin Herbarium
- VU:
-
Vulnerable
References
Alonso PL. Malaria: a problem to be solved and a time to be bold. Nat Med. 2021;27(9):1506–9. https://doi.org/10.1038/s41591-021-01492-6.
Oboh MA, Badiane AS, Ntadom G, Ndiaye YD, Diongue K, Ndiaye D. Molecular identification of Plasmodium species responsible for malaria reveals Plasmodium vivax isolates in Duffy negative individuals from southwestern Nigeria. Malar J. 2018;17(1):1–12. https://doi.org/10.1186/S12936-018-2588-7.
Oboh MA, Singh US, Ndiaye D, Badiane AS, Ali NA, Bharti PK, Das A. Presence of additional Plasmodium vivax malaria in Duffy negative individuals from Southwestern Nigeria. Malar J. 2020;19(1):1–8. https://doi.org/10.1186/S12936-020-03301-W.
World Health Organization. World Malaria Report, 2022. Geneva: WHO Press; 2022.
Ariey F, Witkowski B, Amaratunga C, Beghain J, Langlois AC, Khim N, Kim S, Duru V, Bouchier C, Ma L, Lim P. A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature. 2014;505(7481):50–5. https://doi.org/10.1038/nature12876.
Birnbaum J, Scharf S, Schmidt S, Jonscher E, Hoeijmakers WA, Flemming S, Toenhake CG, Schmitt M, Sabitzki R, Bergmann B, Fröhlke U. A Kelch13-defined endocytosis pathway mediates artemisinin resistance in malaria parasites. Science. 2020;367(6473):51–9. https://doi.org/10.1126/science.aax4735.
Delandre O, Daffe SM, Gendrot M, Diallo MN, Madamet M, Kounta MB, Diop MN, Bercion R, Sow A, Ngom PM, Lo G. Absence of association between polymorphisms in the pfcoronin and pfk13 genes and the presence of Plasmodium falciparum parasites after treatment with artemisinin derivatives in Senegal. Int J of Antimicrob Agents. 2020;56(6):106190. https://doi.org/10.1016/j.ijantimicag.2020.106190.
Uwimana A, Legrand E, Stokes BH, Ndikumana JL, Warsame M, Umulisa N, Ngamije D, Munyaneza T, Mazarati JB, Munguti K, Campagne P. Emergence and clonal expansion of in vitro artemisinin-resistant Plasmodium falciparum kelch13 R561H mutant parasites in Rwanda. Nat Med. 2020;26(10):1602–8. https://doi.org/10.1038/s41591-020-1005-2.
Imran IZ, Elusiyan CA, Agbedahunsi JM, Omisore NO. Bioactivity-directed evaluation of fruit of Kigelia africana (Lam.) benth. Used in treatment of malaria in Iwo, Nigeria. J Ethnopharmacol. 2021;268:113680. https://doi.org/10.1016/j.jep.2020.113680.
Yineger H, Yewhalaw D. Traditional medicinal plant knowledge and use by local healers in Sekoru District, Jimma Zone Southwestern Ethiopia. J Ethnobiol Ethnomed. 2007;3(1):1–7. https://doi.org/10.1186/1746-4269-3-24.
Chintamunnee V, Mahomoodally MF. Herbal medicine commonly used against non-communicable diseases in the tropical island of Mauritius. J Herb Med. 2012;2(4):113–25. https://doi.org/10.1016/j.hermed.2012.06.001.
James PB, Wardle J, Steel A, Adams J. Traditional, complementary and alternative medicine use in Sub-Saharan Africa: a systematic review. BMJ Glob Health. 2018;3(5):e000895. https://doi.org/10.1136/bmjgh-2018-000895.
Okaiyeto K, Oguntibeju OO. African herbal medicines: Adverse effects and cytotoxic potentials with different therapeutic applications. Int J Environ Res Public Health. 2021;18(11):5988. https://doi.org/10.3390/ijerph18115988.
Asfaw A, Lulekal E, Bekele T, Debella A, Abebe A, Degu S. Documentation of traditional medicinal plants use in Ensaro District, Ethiopia: Implications for plant biodiversity and indigenous knowledge conservation. J Herb Med. 2023;38:100641. https://doi.org/10.1016/j.hermed.2023.100641.
Matowa PR, Gundidza M, Gwanzura L, Nhachi CF. A survey of ethnomedicinal plants used to treat cancer by traditional medicine practitioners in Zimbabwe. BMC Complement Med Ther. 2020;20(1):1–13. https://doi.org/10.1186/s12906-020-03046-8.
Van Vuuren SF, Motlhatlego KE, Netshia V. Traditionally used polyherbals in a southern African therapeutic context. J Ethnopharmacol. 2022;288:114977. https://doi.org/10.1016/j.jep.2022.114977.
Jamshidi-Kia F, Lorigooini Z, Amini-Khoei H. Medicinal plants: Past history and future perspective. J Herbmed Pharmacol. 2018;7(1):1–7. https://doi.org/10.15171/jhp.2018.01.
Li S, Odedina S, Agwai I, Ojengbede O, Huo D, Olopade OI. Traditional medicine usage among adult women in Ibadan, Nigeria: a cross-sectional study. BMC Complement Med Ther. 2020;20(1):1–7. https://doi.org/10.1186/s12906-020-02881-z.
Mlilo S, Sibanda S. An ethnobotanical survey of the medicinal plants used in the treatment of cancer in some parts of Matebeleland. Zimbabwe S Afr J Bot. 2022;146:401–8. https://doi.org/10.1016/j.sajb.2021.11.022.
Mgbeahuruike EE, Holm Y, Vuorela H, Amandikwa C, Fyhrquist P. An ethnobotanical survey and antifungal activity of Piper guineense used for the treatment of fungal infections in West-African traditional medicine. J Ethnopharmacol. 2019;229:157–66. https://doi.org/10.1016/j.jep.2018.10.005.
Balogun JA. Emerging Developments in Traditional Medicine Practice in Nigeria. In: The Nigerian Healthcare System. Cham: Springer; 2021. p. 235–275.
Osuchukwu NC, Eko JE, Abia RP, Ochei KC. Use of herbal medicine among adult residents in Calabar Metropolis, Cross River State Nigeria. J Complement Altern Med Res. 2017;2(3):1–14.
Odoh UE, Uzor PF, Eze CL, Akunne TC, Onyegbulam CM, Osadebe PO. Medicinal plants used by the people of Nsukka Local Government Area, south-eastern Nigeria for the treatment of malaria: an ethnobotanical survey. J Ethnopharmacol. 2018;218:1–15. https://doi.org/10.1016/j.jep.2018.02.034.
Oyeyemi IT, Akinseye KM, Adebayo SS, Oyetunji MT, Oyeyemi OT. Ethnobotanical survey of the plants used for the management of malaria in Ondo State Nigeria. S Afr J Bot. 2019;124:391–401. https://doi.org/10.1016/j.sajb.2019.06.003.
Adewole KE. Nigerian antimalarial plants and their anticancer potential: a review. J Integr Med. 2020;18(2):92–113. https://doi.org/10.1016/j.joim.2020.01.001.
Ajao AAN, Mukaila YO, Sabiu S. Wandering through southwestern Nigeria: an inventory of Yoruba useful angiosperm plants. Heliyon. 2022;e08668. https://doi.org/10.1016/j.heliyon.2021.e08668
Evbuomwan IO, Alejolowo OO, Elebiyo TC, Nwonuma CO, Ojo OA, Edosomwan EU, Chikwendu JI, Elosiuba NV, Akulue JC, Dogunro FA, Rotimi DE, Osemwegie OO, Ojo AB, Ademowo OG, Adeyemi OS, Oluba OM. In silico modeling revealed phytomolecules derived from Cymbopogon citratus (DC.) leaf extract as promising candidates for malaria therapy. J Biomol Struct Dyn. 2023;1–18. https://doi.org/10.1080/07391102.2023.2192799.
Oluba OM. Ganoderma terpenoid extract exhibited anti-plasmodial activity by a mechanism involving reduction in erythrocyte and hepatic lipids in Plasmodium berghei-infected mice. Lipids Health Dis. 2019;18(1):1–9. https://doi.org/10.1186/s12944-018-0951-x.
Adepiti AO, Reuben TT, Odeyinka ID, Akinwunmi KF. Assessment of the activities of Chasmanthera dependens Hochst. combined with other plants on chloroquine-sensitive and chloroquine-resistant Plasmodium berghei. Sci Afr. 2022;15:e01103. https://doi.org/10.1016/j.sciaf.2022.e01103
Wu XR, Shen Y, Diao HM, Dong X, Xiao CJ, Jiang B. Sesquiterpenoids and their glycosides from Dobinea delavayi (Baill.) Baill. Phytochemistry. 2022;193:112999. https://doi.org/10.1016/j.phytochem.2021.112999.
Antoine T, Fisher N, Amewu R, O’Neill PM, Ward SA, Biagini GA. Rapid kill of malaria parasites by artemisinin and semi-synthetic endoperoxides involves ROS-dependent depolarization of the membrane potential. J Antimicrob Chemother. 2014;69(4):1005–16. https://doi.org/10.1093/jac/dkt486.
Gachelin G, Garner P, Ferroni E, Tröhler U, Chalmers I. Evaluating Cinchona bark and quinine for treating and preventing malaria. J R Soc Med. 2017;110(1):31–40. https://doi.org/10.1177/0141076816681421.
Arshad M, Ahmad M, Ahmed E, Saboor A, Abbas A, Sadiq S. An ethnobiological study in Kala Chitta hills of Pothwar region, Pakistan: multinomial logit specification. J Ethnobiol Ethnomed. 2014;10(1):1–7. https://doi.org/10.1186/1746-4269-10-13.
Gaikwad J, Wilson K, Kohen J, Vemulpad S, Jamie J, Ranganathan S. Combining ethnobotany and informatics to discover knowledge from data. Ethnomedicinal Plants: revitalization of traditional knowledge of herbs. New Hampshire: Science Publishers; 2010. p. 444–457.
Mahwasane ST, Middleton L, Boaduo N. An ethnobotanical survey of indigenous knowledge on medicinal plants used by the traditional healers of the Lwamondo area, Limpopo province South Africa. S Afr J Bot. 2013;88:69–75. https://doi.org/10.1016/j.sajb.2013.05.004.
Amjad MS, Zahoor U, Bussmann RW, Altaf M, Gardazi SMH, Abbasi AM. Ethnobotanical survey of the medicinal flora of Harighal, Azad Jammu & Kashmir Pakistan. J Ethnobiol Ethnomed. 2020;16(1):1–28. https://doi.org/10.1186/s13002-020-00417-w.
Pirintsos S, Panagiotopoulos A, Bariotakis M, Daskalakis V, Lionis C, Sourvinos G, et al. From Traditional ethnopharmacology to modern natural drug discovery: a methodology discussion and specific examples. Molecules. 2022;27(13):4060. https://doi.org/10.3390/molecules27134060.
Baydoun S, Chalak L, Dalleh H, Arnold N. Ethnopharmacological survey of medicinal plants used in traditional medicine by the communities of Mount Hermon Lebanon. J Ethnopharmacol. 2015;173:139–56. https://doi.org/10.1016/j.jep.2015.06.052.
Che CT, George V, Ijinu TP, Pushpangadan P, Andrae-Marobela K. Traditional medicine. Pharmacognosy 2017;15–30.https://doi.org/10.1016/b978-0-12-802104-0.00002-0.
Sen T, Samanta SK. Medicinal plants, human health and biodiversity: a broad review. Adv Biochem Eng Biotechnol. 2014;59–110. https://doi.org/10.1007/10_2014_273.
Arjona-García C, Blancas J, Beltrán-Rodríguez L, López Binnqüist C, Colín Bahena H, Moreno-Calles AI, Sierra-Huelsz JA, López-Medellín X. How does urbanization affect perceptions and traditional knowledge of medicinal plants? J Ethnobiol Ethnomed. 2021;17(1):1–26. https://doi.org/10.1186/s13002-021-00473-w.
Ene AC, Atawodi SE, Ameh DA, Kwanashie HO, Agomo PU. Locally used plants for malaria therapy amongst the Hausa, Yoruba and Igbo communities in Maiduguri, northeastern Nigeria. Indian J Tradit Knowl. 2010;9(3):486–90.
Olorunnisola OS, Adetutu A, Balogun EA, Afolayan AJ. Ethnobotanical survey of medicinal plants used in the treatment of malarial in Ogbomoso Southwest Nigeria. J Ethnopharmacol. 2013;150(1):71–8. https://doi.org/10.1016/j.jep.2013.07.038.
Ishola IO, Oreagba IA, Adeneye AA, Adirije C, Oshikoya KA, Ogunleye OO. Ethnopharmacological survey of herbal treatment of malaria in Lagos Southwest Nigeria. J Herb Med. 2014;4(4):224–34. https://doi.org/10.1016/j.hermed.2014.08.001.
Chukwuma DM, Chukwuma EC, Adekola OO. An ethnobotanical survey of Malaria-treating plants in Ado-Ekiti Local Government Area, Ekiti State, Nigeria. Ethnobot Res Appl. 2019;18:1–10. https://doi.org/10.32859/era.18.37.1-10.
Oladeji OS, Odelade KA, Oloke JK. Phytochemical screening and antimicrobial investigation of Moringa oleifera leaf extracts. Afr J Sci Technol Innov Dev. 2020;12(1):79–84.
Ukaga CN, Nwoke BE, Onyeka PI, Anosike JC, Udujih OS, Udijih OG, et al. The use of herbs in malaria treatment in parts of Imo State, Nigeria. Tanzan J Health Res. 2006;8(3):183–5.
Omosun G, Okoro IA, Ekundayo E, Ojimelukwe PC, Ibe O. Ethnobotanical study of medicinal plants useful for malaria therapy in eight Local Government Areas of Abia State Southeast Nigeria. Adv Med Plant Res. 2013;1(2):39–44.
Orosun MM, Oniku SA, Peter A, Orosun RO, Salawu NB, Hitler L. Magnetic susceptibility measurement and heavy metal pollution at an automobile station in Ilorin, North-Central Nigeria. Environ Res Commun. 2020;2(1):015001. https://doi.org/10.1088/2515-7620/ab636a.
Elijah OA, Adedapo A. 2006. The effect of climate on poultry productivity in Ilorin Kwara State, Nigeria. Int J Poult Sci. 2006;5(11):1061–1068.
Vasileiou K, Barnett J, Thorpe S, Young T. Characterizing and justifying sample size sufficiency in interview-based studies: systematic analysis of qualitative health research over a 15-year period. BMC Med Res Methodol. 2018;18(1):1–18. https://doi.org/10.1186/s12874-018-0594-7.
Harris LR, Brown GT. Mixing interview and questionnaire methods: Practical problems in aligning data. Pract Assess Res Eva. 2010;15(1):1. https://doi.org/10.7275/959j-ky83.
Steentoft M. Flowering plants in west Africa. Cambridge: Cambridge University Press; 1988. https://doi.org/10.1017/CBO9780511551505.
Aigbokhan EI, Berner DK, Musselman LJ, Mignouna HD. Evaluation of variability in Striga aspera, Striga hermonthica and their hybrids using morphological characters and random amplified polymorphic DNA markers. Weed Res. 2000;40(4):375–86.
Dressler S, Schmidt M, Zizka G. Introducing African Plants—a photo guide—an interactive photo database and rapid identification tool for continental Africa. Taxon. 2014;63(5):1159–64.
The Plant List database. http://www.theplantlist.org. Accessed 21 May 2022.
International Union for Conservation of Nature. The IUCN Red List of Threatened Species. v2021–3, https://www.iucnredlist.org. Accessed 9 May 2022.
Benarba B, Belabid L, Righi K, Amine Bekkar A, Elouissi M, Khaldi A, Hamimed A. Ethnobotanical study of medicinal plants used by traditional healers in Mascara (North West of Algeria). J Ethnopharmacol. 2015;175:626–37. https://doi.org/10.1016/j.jep.2015.09.030.
Tchicaillat-Landou M, Petit J, Gaiani C, Miabangana ES, Kimbonguila A, Nzikou JM, Scher J, Matos L. Ethnobotanical study of medicinal plants used by traditional healers for the treatment of oxidative stress-related diseases in the Congo Basin. J Herb Med. 2018;13:76–90. https://doi.org/10.1016/j.hermed.2018.05.002.
Raimi IO, Kopaopa BG, Mugivhisa LL, Lewu FB, Amoo SO, Olowoyo JO. An ethnobotanical survey of medicinal plants used by traditional healers for the treatment of cancer in Hammanskraal and Winterveld, Tshwane Metropolitan Municipality South Africa. Afr Health Sci. 2021;21(4):1746–53. https://doi.org/10.4314/ahs.v21i4.31.
Mudau TE, Olowoyo JO, Amoo SO. Ethnobotanical assessment of medicinal plants used traditionally for treating diabetes in Vhembe district, Limpopo Province South Africa. S Afr J Bot. 2022;146:304–24. https://doi.org/10.1016/j.sajb.2021.10.016.
Junsongduang A, Kasemwan W, Lumjoomjung S, Sabprachai W, Tanming W, Balslev H. Ethnomedicinal knowledge of traditional healers in Roi Et, Thailand. Plants. 2020;9(9):1177. https://doi.org/10.3390/plants9091177.
Bachir B, Belhouala K. Medicinal plants used by traditional healers in Algeria: a multi-regional ethnobotanical study. Front Pharmacol. 2021;12:760492. https://doi.org/10.3389/fphar.2021.760492.
Odugbemi TO, Akinsulire OR, Aibinu IE, Fabeku PO. Medicinal plants useful for malaria therapy in Okeigbo, Ondo State, Southwest Nigeria. Afr J Tradit Complement Altern Med. 2007;4(2):191–8. https://doi.org/10.4314/ajtcam.v4i2.31207.
Yetein MH, Houessou LG, Lougbégnon TO, Teka O, Tente B. Ethnobotanical study of medicinal plants used for the treatment of malaria in plateau of Allada, Benin (West Africa). J Ethnopharmacol. 2013;146(1):154–63. https://doi.org/10.1016/j.jep.2012.12.022.
Chinsembu KC. Plants as antimalarial agents in sub-Saharan Africa. Acta Trop. 2015;152:32–48. https://doi.org/10.1016/j.actatropica.2015.08.009.
Alebie G, Urga B, Worku A. Systematic review on traditional medicinal plants used for the treatment of malaria in Ethiopia: trends and perspectives. Malar J. 2017;16(1):1–13. https://doi.org/10.1186/s12936-017-1953-2.
Nigussie G, Wale M. Medicinal plants used in traditional treatment of malaria in Ethiopia: a review of ethnomedicine, anti-malarial and toxicity studies. Malar J. 2022;21(1):1–16. https://doi.org/10.1186/s12936-022-04264-w.
Agbodeka K, Gbekley HE, Karou SD, Anani K, Agbonon A, Tchacondo T, Batawila K, Simpore J, Gbeassor M. Ethnobotanical study of medicinal plants used for the treatment of malaria in the plateau region Togo. Pharmacognosy Res. 2016;8(Suppl 1):12–8. https://doi.org/10.4103/0974-8490.178646.
Ngarivhume T, van’t Klooster CI, de Jong JT, Van der Westhuizen JH. Medicinal plants used by traditional healers for the treatment of malaria in the Chipinge district in Zimbabwe. J Ethnopharmacol. 2015;159:224–37. https://doi.org/10.1016/j.jep.2014.11.011.
Etuk EU, Ugwah MO, Ajagbonna OP, Onyeyili PA. Ethnobotanical survey and preliminary evaluation of medicinal plants with antidiarrhoea properties in Sokoto State Nigeria. J Med Plant Res. 2009;3(10):763–6.
Idu M, Erhabor JO, Efijuemue HM. Documentation on medicinal plants sold in markets in Abeokuta, Nigeria. Trop J Pharma Res. 2010;9(2). https://doi.org/10.4314/tjpr.v9i2.53696.
Lawal IO, Rafiu BO, Ale JE, Majebi OE, Aremu AO. Ethnobotanical survey of local flora used for medicinal purposes among indigenous people in five areas in Lagos State, Nigeria. Plants. 2022;11(5):633. https://doi.org/10.3390/plants11050633.
Adebiyi OE, Abatan MO. Phytochemical and acute toxicity of ethanolic extract of Enantia chlorantha (Oliv) stem bark in albino rats. Interdiscip Toxicol. 2013;6(3):145. https://doi.org/10.2478/intox-2013-0023.
Tcheghebe OT, Tatong FN, Seukep AJ. Traditional uses, phytochemical and pharmacological profiles, and toxicity of Enantia chlorantha (Oliver): An overview. Edorium J Med. 2016;3:12–8. https://doi.org/10.5348/M05-2016-4-RA-2.
Udenze ECC, Braide VB, Okwesilieze CN, Akuodor GC. Pharmacological effects of Garcinia kola seed powder on blood sugar, lipid profile and atherogenic index of alloxan-induced diabetes in rats. Pharmacologia. 2012;3(12):693–9.
Adedara IA, Awogbindin IO, Anamelechi JP, Farombi EO. Garcinia kola seed ameliorates renal, hepatic, and testicular oxidative damage in streptozotocin-induced diabetic rats. Pharm Biol. 2015;53(5):695–704. https://doi.org/10.3109/13880209.2014.937504.
Lawal IO, Olufade II, Rafiu BO, Aremu AO. Ethnobotanical survey of plants used for treating cough associated with respiratory conditions in Ede South local government area of Osun State, Nigeria. Plants. 2020;9(5):647. https://doi.org/10.3390/plants9050647.
Saini S, Kaur H, Verma B, Singh SK. Kigelia africana (Lam.) Benth.—an overview. Nat Prod Radiance. 2009;8(2):190–197.
Dike IP, Obembe OO, Adebiyi FE. Ethnobotanical survey for potential anti-malarial plants in south-western Nigeria. J Ethnopharmacol. 2012;144(3):618–26. https://doi.org/10.1016/j.jep.2012.10.002.
Traore MS, Baldé MA, Diallo MS, Baldé ES, Diané S, Camara A, Diallo A, Balde A, Keïta A, Keita SM, Oularé K. Ethnobotanical survey on medicinal plants used by Guinean traditional healers in the treatment of malaria. J Ethnopharmacol. 2013;150(3):1145–53. https://doi.org/10.1016/j.jep.2013.10.048.
Erinoso SM, Aworinde DO. Current outlook and future promise of ethnobotany in Nigeria: A review and personal observation. Afr J Plant Sci. 2018;12(4):73–80. https://doi.org/10.5897/AJPS2017.1571.
Soladoye MO, Chukwuma EC, Sulaiman OM, Feyisola RT. Ethnobotanical survey of plants used in the traditional treatment of female infertility in Southwestern Nigeria. Ethnobot Res Appl. 2014;12:81–90.
Afolayan FI, Sulaiman KA, Okunade WT. Ethnobotanical survey of plants used in cancer therapy in Iwo and Ibadan, South-Western of Nigeria. J Pharm Pharmacogn Res. 2020;8(5):346–67.
Kankara SS, Ibrahim MH, Mustafa M, Go R. Ethnobotanical survey of medicinal plants used for traditional maternal healthcare in Katsina State Nigeria. S Afr J Bot. 2015;97:165–75. https://doi.org/10.1016/j.sajb.2015.01.007.
Abd El-Ghani MM. Traditional medicinal plants of Nigeria: an overview. Agr Biol J N Am. 2016;7(5):220–47.
Boadu AA, Asase A. Documentation of herbal medicines used for the treatment and management of human diseases by some communities in southern Ghana. Evid Based Complement Alternat Med. 2017;3043061. https://doi.org/10.1155/2017/3043061.
Magwede K, Van Wyk BE, Van Wyk AE. An inventory of Vhavenḓa useful plants. S Afr J Bot. 2019;122:57–89. https://doi.org/10.1016/j.sajb.2017.12.013.
Iragi GK, Rusaati BI, Nfizi IB, Masumbuko CN, Gendusa PA, Furaha AM, Kang JW. Ethnomedicinal study of plants used in the Uvira Territory (Democratic Republic of Congo). Forest Sci Technol. 2021;17(3):144–54. https://doi.org/10.1080/21580103.2021.1963327.
Asiimwe S, Namukobe J, Byamukama R, Imalingat B. Ethnobotanical survey of medicinal plant species used by communities around Mabira and Mpanga Central Forest Reserves Uganda. Trop Med Health. 2021;49(1):1–10. https://doi.org/10.1186/s41182-021-00341-z.
Semenya SS, Maroyi A. Plants used by Bapedi traditional healers to treat asthma and related symptoms in Limpopo Province, South Africa. Evid Based Complement Alternat Med. 2018;2183705. https://doi.org/10.1155/2018/2183705.
Muleba I, Yessoufou K, Rampedi IT. Testing the non-random hypothesis of medicinal plant selection using the woody flora of the Mpumalanga Province South Africa. Environ Dev Sustain. 2021;23(3):4162–73. https://doi.org/10.1007/s10668-020-00763-5.
Jiang X, Liu Y, Li W, Zhao L, Meng F, Wang Y, Tan H, Yang H, Wei C, Wan X, Gao L. Tissue-specific, development-dependent phenolic compounds accumulation profile and gene expression pattern in tea plant [Camellia sinensis]. PloS One. 2013;8(4):e62315. https://doi.org/10.1371/journal.pone.0062315.
Fortini P, Di Marzio P, Guarrera PM, Iorizzi M. Ethnobotanical study on the medicinal plants in the Mainarde Mountains (central-southern Apennine, Italy). J Ethnopharmacol. 2016;184:208–18. https://doi.org/10.1016/j.jep.2016.03.010.
Tariq A, Sadia S, Pan K, Ullah I, Mussarat S, Sun F, Abiodun OO, Batbaatar A, Li Z, Song D, Xiong Q. A systematic review on ethnomedicines of anti-cancer plants. Phytother Res. 2017;31(2):202–64. https://doi.org/10.1002/ptr.5751.
Zahoor M, Yousaf Z, Aqsa T, Haroon M, Saleh N, Aftab A, Javed S, Qadeer M, Ramazan H. An ethnopharmacological evaluation of Navapind and Shahpur Virkanin district Sheikupura, Pakistan for their herbal medicines. J Ethnobiol Ethnomed. 2017;13(1):1–26. https://doi.org/10.1186/s13002-017-0151-1.
Sauini T, Stern da Fonseca-Kruel V, Baptistela Yazbek P, Matta P, Cassas F, da Cruz C, Hortal Pereira Barretto E, Alice dos Santos M, Angelica Silva Gomes M, José Francischetti Garcia R, Honda S. Participatory methods on the recording of traditional knowledge about medicinal plants in Atlantic Forest, Ubatuba, São Paulo, Brazil. Plos One. 2020;15(5):e0232288. https://doi.org/10.1371/journal.pone.0232288.
Zheng XL, Xing FW. Ethnobotanical study on medicinal plants around Mt. Yinggeling, Hainan Island, China. J Ethnopharmacol. 2009;124(2):197–210. https://doi.org/10.1016/j.jep.2009.04.042.
Namukobe J, Kasenene JM, Kiremire BT, Byamukama R, Kamatenesi-Mugisha M, Krief S, Dumontet V, Kabasa JD. Traditional plants used for medicinal purposes by local communities around the Northern sector of Kibale National Park Uganda. J Ethnopharmacol. 2011;136(1):236–45. https://doi.org/10.1016/j.jep.2011.04.044.
Idowu OA, Soniran OT, Ajana O, Aworinde DO. Ethnobotanical survey of antimalarial plants used in Ogun State Southwest Nigeria. Afr J Pharm Pharmacol. 2010;4(2):55–60. https://doi.org/10.5897/AJPP.9000138.
Ahmed HM. Ethnopharmacobotanical study on the medicinal plants used by herbalists in Sulaymaniyah Province, Kurdistan Iraq. J Ethnobiol Ethnomed. 2016;12(1):1–17. https://doi.org/10.1186/s13002-016-0081-3.
Malik K, Ahmad M, Bussmann RW, Tariq A, Ullah R, Alqahtani AS, Shahat AA, Rashid N, Zafar M, Sultana S, Shah SN. Ethnobotany of anti-hypertensive plants used in northern Pakistan. Front Pharmacol. 2018;9:789. https://doi.org/10.3389/fphar.2018.00789.
Jadid N, Kurniawan E, Himayani CE, Prasetyowati I, Purwani KI, Muslihatin W, Hidayati D, Tjahjaningrum IT. An ethnobotanical study of medicinal plants used by the Tengger tribe in Ngadisari village, Indonesia. PloS One. 2020;15(7):e0235886. https://doi.org/10.1371/journal.pone.0235886.
Kachmar MR, Naceiri Mrabti H, Bellahmar M, Ouahbi A, Haloui Z, El Badaoui K, Bouyahya A, Chakir S. Traditional knowledge of medicinal plants used in the Northeastern part of Morocco. Evid Based Complement Alternat Med. 2021;6002949. https://doi.org/10.1155/2021/6002949.
Faruque MO, Uddin SB, Barlow JW, Hu S, Dong S, Cai Q, Li X, Hu X. Quantitative ethnobotany of medicinal plants used by indigenous communities in the Bandarban District of Bangladesh. Front Pharmacol. 2018;9:40. https://doi.org/10.3389/fphar.2018.00040.
Kassa Z, Asfaw Z, Demissew S. An ethnobotanical study of medicinal plants in Sheka Zone of Southern Nations Nationalities and peoples regional state Ethiopia. J Ethnobiol Ethnomed. 2020;16(1):1–15. https://doi.org/10.1186/s13002-020-0358-4.
Munir M, Sadia S, Khan A, Rahim BZ, Gagosh Nayyar B, Ahmad KS, Khan AM, Fatima I, Qureshi R. Ethnobotanical study of Mandi Ahmad Abad, District Okara, Pakistan. PloS One 2022;17(4):e0265125. https://doi.org/10.1371/journal.pone.0265125.
Cao J, Yang M, Cao F, Wang J, Su E. Well-designed hydrophobic deep eutectic solvents as green and efficient media for the extraction of artemisinin from Artemisia annua leaves. ACS Sustain Chem Eng. 2017;5(4):3270–8. https://doi.org/10.1021/acssuschemeng.6b03092.
Adeyemi OS, Eseola AO, Plass W, Atolani O, Sugi T, Han Y, Batiha GE, Kato K, Awakan OJ, Olaolu TD, Nwonuma CO. Imidazole derivatives as antiparasitic agents and use of molecular modeling to investigate the structure–activity relationship. Parasitol Res. 2020;119(6):1925–41. https://doi.org/10.1007/s00436-020-06668-6.
Dawurung CJ, Noitem R, Rattanajak R, Bunyong R, Richardson C, Willis AC, Kamchonwongpaisan S, Yimnual C, Muanprasat C, Pyne SG. Isolation of CFTR and TMEM16A inhibitors from Neorautanenia mitis (A. Rich) Verdcourt: potential lead compounds for treatment of secretory diarrhea. Phytochemistry. 2020;179:112464. https://doi.org/10.1016/j.phytochem.2020.112464.
Dawurung CJ, Nguyen MT, Pengon J, Dokladda K, Bunyong R, Rattanajak R, Kamchonwongpaisan S, Nguyen PT, Pyne SG. Isolation of bioactive compounds from medicinal plants used in traditional medicine: Rautandiol B, a potential lead compound against Plasmodium falciparum. BMC Complement Med Ther. 2021;21(1):1–12. https://doi.org/10.1186/s12906-021-03406-y.
Boyom FF, Kemgne EM, Tepongning R, Ngouana V, Mbacham WF, Tsamo E, Zollo PH, Gut J, Rosenthal PJ. Antiplasmodial activity of extracts from seven medicinal plants used in malaria treatment in Cameroon. J Ethnopharmacol. 2009;123(3):483–8. https://doi.org/10.1016/j.jep.2009.03.008.
Vennerstrom JL, Klayman DL. Protoberberine alkaloids as antimalarials. J Med Chem. 1988;31(6):1084–7. https://doi.org/10.1021/jm00401a006.
Tepongning RN, Mbah JN, Avoulou FL, Jerme MM, Ndanga EKK, Fekam FB. Hydroethanolic extracts of Erigeron floribundus and Azadirachta indica reduced Plasmodium berghei parasitemia in Balb/c mice. Evid Based Complement Alternat Med. 2018. https://doi.org/10.1155/2018/5156710.
Koffi JA, Silué KD, Tano DK, Dable TM, Yavo W. Evaluation of antiplasmodial activity of extracts from endemic medicinal plants used to treat malaria in Côte d'Ivoire. BioImpacts. 2020;10(3):151. https://doi.org/10.34172/bi.2020.19.
Rochanakij S, Thebtaranonth Y, Yenjai C, Yuthavong Y. Nimbolide, a constituent of Azadirachta indica, inhibits Plasmodium falciparum in culture. Southeast Asian J Trop Med Public Health. 1985;16(1):66–72.
Khalid SA, Farouk A, Geary TG, Jensen JB. Potential antimalarial candidates from African plants: an in vitro approach using Plasmodium falciparum. J Ethnopharmacol. 1986;15(2):201–9. https://doi.org/10.1016/0378-8741(86)90156-X.
Billker O, Shaw MK, Jones IW, Ley SV, Sinden RE. Azadirachtin disrupts formation of organized microtubule arrays during microgametogenesis of Plasmodium berghei. J Eukaryot Microbiol. 2002;49(6):489–97. https://doi.org/10.1111/j.1550-7408.2002.tb00234.x.
Alaribe SC, Oladipupo AR, Uche GC, Onumba MU, Ota D, Awodele O, Oyibo WA. Suppressive, curative, and prophylactic potentials of an antimalarial polyherbal mixture and its individual components in Plasmodium berghei-Infected mice. J Ethnopharmacol. 2021;277:114105. https://doi.org/10.1016/j.jep.2021.114105.
Oladeji OS, Oluyori AP, Dada AO. Antiplasmodial activity of Morinda lucida Benth. leaf and bark extracts against Plasmodium berghei infected mice. Saudi J Biol Sci. 2022;29(4):2475–82. https://doi.org/10.1016/j.sjbs.2021.12.017.
Olasehinde GI, Ojurongbe O, Adeyeba AO, Fagade OE, Valecha N, Ayanda IO, Ajayi AA, Egwari LO. In vitro studies on the sensitivity pattern of Plasmodium falciparum to anti-malarial drugs and local herbal extracts. Malar J. 2014;13(1):1–7. https://doi.org/10.1186/1475-2875-13-63.
Koumaglo K, Gbeassor M, Nikabu O, De Souza C, Werner W. Effects of three compounds extracted from Morinda lucida on Plasmodium falciparum. Planta Med. 1992;58(06):533–4. https://doi.org/10.1055/s-2006-961543.
Cimanga RK, Tona GL, Mesia GK, Kambu OK, Bakana DP, Kalenda PD, Penge AO, Muyembe JJ, Totté J, Pieters L, Vlietinck AJ. Bioassay-guided isolation of antimalarial triterpenoid acids from the leaves of Morinda lucida. Pharm Biol. 2006;44(9):677–81. https://doi.org/10.1080/13880200601009123.
Abisoye LR. Antimalarial, antitrypanosomal, antimicrobial activities and volatile oil profile of Xylopia aethiopica (Dunal) Rich (Annonaceae). Lett Appl NanoBioScience. 2021;11(3):3897–3908. https://doi.org/10.33263/LIANBS113.38973908.
Boampong JN, Ameyaw EO, Aboagye B, Asare K, Kyei S, Donfack JH, Woode E. The curative and prophylactic effects of xylopic acid on Plasmodium berghei infection in mice. J Parasitol Res. 2013;356107. https://doi.org/10.1155/2013/356107.
Gimenez VM, Alvarenga TA, Groppo M, Silva ML, Cunha WR, Januário AH, Smilkstein MJ, Riscoe MK, Pauletti PM. Antiplasmodial evaluation of Anacardium occidentale and alkyl-phenols. Rev Bras Farmacogn. 2019;29:36–9. https://doi.org/10.1016/j.bjp.2018.11.003.
Cui L, Miao J, Furuya T, Fan Q, Li X, Rathod PK, Su XZ, Cui L. Histone acetyltransferase inhibitor anacardic acid causes changes in global gene expression during in vitro Plasmodium falciparum development. Eukaryot Cell. 2008;7(7):1200–10.
Melariri P, Campbell W, Etusim P, Smith P. In vitro antiplasmodial activities of extracts from five plants used singly and in combination against Plasmodium falciparum parasites. J Med Plant Res. 2012;6(47):5770–9. https://doi.org/10.5897/JMPR11.1187.
Chukwuocha UM, Fernández-Rivera O, Legorreta-Herrera M. Exploring the antimalarial potential of whole Cymbopogon citratus plant therapy. J Ethnopharmacol. 2016;193:517–23. https://doi.org/10.1016/j.jep.2016.09.056.
Agbedahunsi JM, Adepiti AO, Adedini AA, Akinsomisoye O, Adepitan A. Antimalarial properties of Morinda lucida and Alstonia boonei on sulphadoxine-pyrimethamine and Curcuma longa on quinine in mice. J Herbs Spices Med Plants. 2016;22(1):1–10. https://doi.org/10.1080/10496475.2014.999151.
Iyiola OA, Tijani AY, Lateef KM. Antimalarial activity of ethanolic stem bark extract of Alstonia boonei. Asian J Biol Sci. 2011;4(3):235–43.
Edagha IA, Peter AI, Aquaisua AN. Histopathological effect of Nauclea latifolia ethanolic leaf extract and artemether/lumefantrine on the hippocampus of P berghei-infected mice. Int J Brain Cogn Sci. 2017;6:9–16. https://doi.org/10.5923/j.ijbcs.20170601.02.
Hafid AF, Septiani RP, Fabriana LH, Febrianty N, Ranggaditya D, Widyawaruyanti A. Antimalarial activity of crude extracts of Artocarpus heterophyllus, Artocarpus altilis, and Artocarpus camansi. Asian J Pharm Clin Res. 2016;9(1):261–3.
Hidayati AR, Widyawaruyanti A, Ilmi H, Tanjung M, Widiandani T, Syafruddin D, Hafid AF. Antimalarial activity of flavonoid compound isolated from leaves of Artocarpus altilis. Pharmacogn J. 2020;12(4). https://doi.org/10.5530/pj.2020.12.120.
Teng WC, Chan W, Suwanarusk R, Ong A, Ho HK, Russell B, Rénia L, Koh HL. In vitro antimalarial evaluations and cytotoxicity investigations of Carica papaya leaves and carpaine. Nat Prod Commun. 2019;14(1):1934578X1901400110.
Melariri P, Campbell W, Etusim P, Smith P. Antiplasmodial properties and bioassay-guided fractionation of ethyl acetate extracts from Carica papaya leaves. J Parasitol Res. 2011;104954. https://doi.org/10.1155/2011/104954.
Omoregie ES, Pal A, Darokar MP, Chanda D, Sisodia B. In vitro and in vivo antiplasmodial activity and cytotoxicity of extracts from Vernonia amygdalina Del Leaves. Malar J. 2010;9(2):1–2. https://doi.org/10.1186/1475-2875-9-S2-P30.
Challand S, Willcox M. A clinical trial of the traditional medicine Vernonia amygdalina in the treatment of uncomplicated malaria. J Altern Complement Med. 2009;15(11):1231–7. https://doi.org/10.1089/acm.2009.0098.
Iwalokun B. Enhanced antimalarial effects of chloroquine by aqueous Vernonia amygdalina leaf extract in mice infected with chloroquine resistant and sensitive Plasmodium berghei strains. Afr Health Sci. 2008;8(1):25–35.
Ohigashi H, Huffman MA, Izutsu D, Koshimizu K, Kawanaka M, Sugiyama H, Kirby GC, Warhurst DC, Allen D, Wright CW, David PJ. Toward the chemical ecology of medicinal plant use in chimpanzees: The case of Vernonia amygdalina, a plant used by wild chimpanzees possibly for parasite-related diseases. J Chem Ecol. 1994;20(3):541–53. https://doi.org/10.1007/BF02059596.
Komlaga G, Forkuo AD, Suleman N, Nkrumah D, Nketia R, Bekoe SO. Antimalarial property and acute toxicity of the leaves of Theobroma cacao L. Evid Based Complement Alternat Med. 2021;2852442. https://doi.org/10.1155/2021/2852442.
Falade MO, Akinboye DO, Gbotosho GO, Ajaiyeoba EO, Happi TC, Abiodun OO, Oduola AM. In vitro and in vivo antimalarial activity of Ficus thonningii Blume (Moraceae) and Lophira alata Banks (Ochnaceae), identified from the ethnomedicine of the Nigerian Middle Belt. J Parasitol Res 2014;972853. https://doi.org/10.1155/2014/972853.
Adebayo JO, Balogun EA, Malomo SO, Soladoye AO, Olatunji LA, Kolawole OM, Oguntoye OS, Babatunde AS, Akinola OB, Aguiar AC, Andrade IM. Antimalarial activity of Cocos nucifera husk fibre: further studies. Evid Based Complement Alternat Med. 2013;742476. https://doi.org/10.1155/2013/742476.
Muthaura CN, Rukunga GM, Chhabra SC, Omar SA, Guantai AN, Gathirwa JW, Tolo FM, Mwitari PG, Keter LK, Kirira PG, Kimani CW. Antimalarial activity of some plants traditionally used in treatment of malaria in Kwale district of Kenya. J Ethnopharmacol. 2007;112(3):545–51. https://doi.org/10.1016/j.jep.2007.04.018.
Iwalewa EO, Omisore NO, Adewunmi CO, Gbolade AA, Ademowo OG, Nneji C, Agboola OI, Daniyan OM. Anti-protozoan activities of Harungana madagascariensis stem bark extract on trichomonads and malaria. J Ethnopharmacol. 2008;117(3):507–11. https://doi.org/10.1016/j.jep.2008.02.019.
Lenta BN, Ngouela S, Boyom FF, Tantangmo F, Tchouya GR, Tsamo E, Gut J, Rosenthal PJ, Connolly JD. Anti-plasmodial activity of some constituents of the root bark of Harungana madagascariensis L AM. (Hypericaceae). Chem Pharm Bull. 2007;55(3):464–467. https://doi.org/10.1248/cpb.55.464.
Zofou D, Kengne AB, Tene M, Ngemenya MN, Tane P, Titanji VP. In vitro antiplasmodial activity and cytotoxicity of crude extracts and compounds from the stem bark of Kigelia africana (Lam.) Benth (Bignoniaceae). Parasitol Res. 2011;108(6):1383–90. https://doi.org/10.1007/s00436-011-2363-y.
Zofou D, Tene M, Tane P, Titanji VP. Antimalarial drug interactions of compounds isolated from Kigelia africana (Bignoniaceae) and their synergism with artemether, against the multidrug-resistant W2mef Plasmodium falciparum strain. Parasitol Res. 2012;110(2):539–44. https://doi.org/10.1007/s00436-011-2519-9.
Adesina DA, Adefolalu SF, Jigam AA, Lawal B. Antiplasmodial effect and sub-acute toxicity of alkaloid, flavonoid and phenolic extracts of Sida acuta leaf on Plasmodium berghei-infected animals. J Taibah Univ Sci. 2020;14(1):943–53. https://doi.org/10.1080/16583655.2020.1790912.
Enechi OC, Amah CC, Okagu IU, Ononiwu PC, Nweke AC, Ugwuanyi TC, Ajibo EA, Nweze AC, Chukwurah BC. Sida acuta Burm. F. leaves ethanol extract ameliorates haematological and biochemical alterations induced by Plasmodium berghei ANKA-65 in mice. Clin Phytosci. 2021;7(1):1–12. https://doi.org/10.1186/s40816-021-00317-w.
Banzouzi JT, Prado R, Menan H, Valentin A, Roumestan C, Mallié M, Pelissier Y, Blache Y. Studies on medicinal plants of Ivory Coast: Investigation of Sida acuta for in vitro antiplasmodial activities and identification of an active constituent. Phytomedicine. 2004;11(4):338–41. https://doi.org/10.1078/0944711041495245.
Enechi OC, Amah CC, Okagu IU, Ononiwu CP, Azidiegwu VC, Ugwuoke EO, Onoh AP, Ndukwe EE. Methanol extracts of Fagara zanthoxyloides leaves possess antimalarial effects and normalizes haematological and biochemical status of Plasmodium berghei-passaged mice. Pharm Biol. 2019;57(1):577–85. https://doi.org/10.1080/13880209.2019.1656753.
Kassim OO, Loyevsky M, Elliott B, Geall A, Amonoo H, Gordeuk VR. Effects of root extracts of Fagara zanthoxyloides on the in vitro growth and stage distribution of Plasmodium falciparum. Antimicrob Agents Chemother. 2005;49(1):264–8. https://doi.org/10.1128/AAC.49.1.264-268.2005.
Rivaud M, Mendoza A, Sauvain M, Valentin A, Jullian V. Synthesis and antimalarial activity of fagaronine. Planta Med. 2012;78(11):39. https://doi.org/10.1055/s-0032-1320397.
Elufioye TO, Agbedahunsi JM. Antimalarial activities of Tithonia diversifolia (Asteraceae) and Crossopteryx febrifuga (Rubiaceae) on mice in vivo. J Ethnopharmacol. 2004;93(2–3):167–71. https://doi.org/10.1016/j.jep.2004.01.009.
Goffin E, Ziemons E, De Mol P, de Madureira MD, Martins AP, da Cunha AP, Philippe G, Tits M, Angenot L, Frederich M. In vitro antiplasmodial activity of Tithonia diversifolia and identification of its main active constituent: tagitinin C. Planta Med. 2002;68(06):543–5. https://doi.org/10.1055/s-2002-32552.
Tona L, Cimanga RK, Mesia K, Musuamba CT, De Bruyne T, Apers S, Hernans N, Van Miert S, Pieters L, Totté J, Vlietinck AJ. In vitro antiplasmodial activity of extracts and fractions from seven medicinal plants used in the Democratic Republic of Congo. J Ethnopharmacol. 2004;93(1):27–32. https://doi.org/10.1016/j.jep.2004.02.022.
Ujomu TS, Adegoke GO, Abiodun OO, Gbotosho GO. Antimalarial Activity and cytotoxicity profile of the seed extracts of Garcinia kola (Guttiferae). J Phytopharm. 2019;8(6):282–285. https://doi.org/10.31254/phyto.2019.8602.
Adaramoye O, Akinpelu T, Kosoko A, Okorie P, Kehinde A, Falade C, Ademowo O. Antimalarial potential of kolaviron, a biflavonoid from Garcinia kola seeds, against Plasmodium berghei infection in Swiss albino mice. Asian Pac J Trop Med. 2014;7(2):97–104. https://doi.org/10.1016/S1995-7645(14)60003-1.
Konziase B. Protective activity of biflavanones from Garcinia kola against Plasmodium infection. J Ethnopharmacol. 2015;172:214–8. https://doi.org/10.1016/j.jep.2015.06.038.
Ettebong E, Ubulom P, Etuk A. Antiplasmodial activity of methanol leaf extract of Citrus aurantifolia (Christm) Swingle. J Herbmed Pharmacol. 2019;8(4):274–280. https://doi.org/10.15171/jhp.2019.40.
Arrey Tarkang P, Okalebo FA, Ayong LS, Agbor GA, Guantai AN. Anti-malarial activity of a polyherbal product (Nefang) during early and established Plasmodium infection in rodent models. Malar J. 2014;13(1):1–11.
Muganza DM, Fruth BI, Lami JN, Mesia GK, Kambu OK, Tona GL, Kanyanga RC, Cos P, Maes L, Apers S, Pieters L. In vitro antiprotozoal and cytotoxic activity of 33 ethnopharmacologically selected medicinal plants from Democratic Republic of Congo. J Ethnopharmacol. 2012;141(1):301–8. https://doi.org/10.1016/j.jep.2012.02.035.
Tepongning RN, Lucantoni L, Nasuti CC, Dori GU, Yerbanga SR, Lupidi G, Marini C, Rossi G, Esposito F, Habluetzel A. Potential of a Khaya ivorensis–Alstonia boonei extract combination as antimalarial prophylactic remedy. J Ethnopharmacol. 2011;137(1):743–51. https://doi.org/10.1016/j.jep.2011.06.036.
Sadiq MB, Tharaphan P, Chotivanich K, Tarning J, Anal AK. In vitro antioxidant and antimalarial activities of leaves, pods and bark extracts of Acacia nilotica (L.) Del. BMC Complement Altern Med. 2017;17(1):1–8. https://doi.org/10.1186/s12906-017-1878-x.
Clarkson C, Maharaj VJ, Crouch NR, Grace OM, Pillay P, Matsabisa MG, Bhagwandin N, Smith PJ, Folb PI. In vitro antiplasmodial activity of medicinal plants native to or naturalized in South Africa. J Ethnopharmacol. 2004;92(2–3):177–91. https://doi.org/10.1016/j.jep.2004.02.011.
Akpan J, Akuodor G, Ezeokpo B, Essien A, Bassey A, Ezeonwumelu JOC. In vivo antiplasmodial activity of Byrsocarpus coccineus leaf extract in mice Infected with Plasmodium berghei. Ibnosina J Med Biomed Sci. 2012;4(3):78–83. https://doi.org/10.4103/1947-489X.210760.
Shuaibu MN, Wuyep PA, Yanagi T, Hirayama K, Tanaka T, Kouno I. The use of microfluorometric method for activity-guided isolation of antiplasmodial compound from plant extracts. Parasitol Res. 2008;102(6):1119–27. https://doi.org/10.1007/s00436-008-0879-6.
Salawu MO, Oyebode OT, Oloyede HOB, Adegoke MA, Muritala HF, Abubakar FA. Antimalarial potentials of aqueous root and leaf extracts of Axonopus compressus in Plasmodium berghei infected mice. Niger J Basic Appl Sci. 2016;29:2700–14.
Amlabu WE, Nock IH. Antimalarial efficacy of Vitellaria paradoxa Gaertn (Family: Sapotaceae) leaves and stem bark. FUW Trends Sci Technol J. 2018;3:605–9.
Jansen O, Angenot L, Tits M, Nicolas JP, De Mol P, Nikiéma JB, Frederich M. Evaluation of 13 selected medicinal plants from Burkina Faso for their antiplasmodial properties. J Ethnopharmacol. 2010;130(1):143–50. https://doi.org/10.1016/j.jep.2010.04.032.
Biruksew A, Zeynudin A, Alemu Y, Golassa L, Yohannes M, Debella A, Urge G, De Spiegeleer B, Suleman S. Zingiber officinale Roscoe and Echinops kebericho Mesfin showed antiplasmodial activities against Plasmodium berghei in a dose-dependent manner in Ethiopia. Ethiop J Health Sci. 2018;28(5). https://doi.org/10.4314/ejhs.v28i5.17.
Christian AG, Ahunna AG, Nwakaego EM, Chimsorom CK, Chile AE. Antimalarial potential of the ethanolic leaf extract of Pseudocedrala kotschyi. J Acute Dis. 2015;4(1):23–7. https://doi.org/10.1016/S2221-6189(14)60077-9.
Hay AE, Ioset JR, Ahua KM, Diallo D, Brun R, Hostettmann K. Limonoid orthoacetates and antiprotozoal compounds from the roots of Pseudocedrela kotschyi. J Nat Prod. 2007;70(1):9–13. https://doi.org/10.1021/np0680230.
Okokon JE, Okokon PJ, Sahal D. In vitro antiplasmodial activity of some medicinal plants from Nigeria. Int J Herb Med. 2017;5(5):102.
Uzor PF, Onyishi CK, Omaliko AP, Nworgu SA, Ugwu OH, Nwodo NJ. Study of the antimalarial activity of the leaf extracts and fractions of Persea americana and Dacryodes edulis and their HPLC analysis. Evid Based Complement Alternat Med. 2021;5218294. https://doi.org/10.1155/2021/5218294.
Falodun A, Erharuyi O, Imieje V, Ahomafor J, Akunyuli C, Jacobs M, Khan S, Hamann MT, Langer P. In vitro evaluation of aliphatic fatty alcohol metabolites of Persea americana seed as potential antimalarial and antimicrobial agents. Niger J Biotechnol. 2014;27:1–7.
Tchoumbougnang F, Zollo PA, Dagne E, Mekonnen Y. In vivo antimalarial activity of essential oils from Cymbopogon citratus and Ocimum gratissimum on mice infected with Plasmodium berghei. Planta Med. 2005;71(01):20–3. https://doi.org/10.1055/s-2005-837745.
Owuor BO, Ochanda JO, Kokwaro JO, Cheruiyot AC, Yeda RA, Okudo CA, Akala HM. In vitro antiplasmodial activity of selected Luo and Kuria medicinal plants. J Ethnopharmacol. 2012;144(3):779–81. https://doi.org/10.1016/j.jep.2012.09.045.
Muthaura CN, Keriko JM, Mutai C, Yenesew A, Gathirwa JW, Irungu BN, Nyangacha R, Mungai GM, Derese S. Antiplasmodial potential of traditional antimalarial phytotherapy remedies used by the Kwale community of the Kenyan Coast. J Ethnopharmacol. 2015;170:148–57. https://doi.org/10.1016/j.jep.2015.05.024.
Chinwuba P, Akah PA, Iiodigwe EE. In vivo antiplasmodial activity of the ethanol stem extract and fractions of Citrus sinensis in mice. Merit Res J Med Med Sci. 2015;3(4):140–6.
Bhat GP, Surolia N. In vitro antimalarial activity of extracts of three plants used in the traditional medicine of India. Am J Trop Med Hyg. 2001;65(4):304–8. https://doi.org/10.4269/ajtmh.2001.65.304.
Musa A, Namadi S, Falaki A, Nafiu L. In vivo antimalarial activity of the aqueous methanol leaf extract of Daniellia oliveri (Rolfe) Hutch. and Dalz. (Caesalpinioideae). Niger J Sci Res. 2018;17(2):205–110.
Okon OE, Gboeloh LB, Udoh S. Antiplasmodial effects of Ficus exasperata on albino mice experimentally infected with Plasmodium berghei. Univers J Pharm. 2013;2(5):29–35.
Shittu II, Emmanuel A, Nok AJ. Antimalarial effect of the ethanolic stem bark extracts of Ficus platyphylla del. J Parasitol Res. 2011. https://doi.org/10.1155/2011/618209.
Nafiu MO, Akanji MA, Raji ZA, Abdulsalam TA. Phytochemical analysis and in vivo antimalarial activities of aqueous extracts of Tithonia diversifolia and Parquetina nigrescens leaves in mice. Biokemistri. 2014;26(2):63–8.
Madara AA, Ajayi JA, Salawu OA, Tijani AY. Anti-malarial activity of ethanolic leaf extract of Piliostigma thonningii Schum. (Caesalpiniacea) in mice infected with Plasmodium berghei berghei. Afr J Biotechnol. 2010;9(23):3475–3480.
Kabiru AY, Ibikunle GF, Innalegwu DA, Bola BM, Madaki FM. In vivo antiplasmodial and analgesic effect of crude ethanol extract of Piper guineense leaf extract in Albino Mice. Scientifica. 2016;2016:1–6.
Majekodunmi SO, Adegoke OA, Odeku OA. Formulation of the extract of the stem bark of Alstonia boonei as tablet dosage form. Trop J Pharm Res. 2008;7(2):987–94. https://doi.org/10.4314/tjpr.v7i2.14683.
Adotey JPK, Adukpo GE, Opoku Boahen Y, Armah FA. A review of the ethnobotany and pharmacological importance of Alstonia boonei De Wild (Apocynaceae). Int Sch Res Notices. 2012. https://doi.org/10.5402/2012/587160.
Udeinya JI, Shu EN, Quakyi I, Ajayi FO. An antimalarial neem leaf extract has both schizonticidal and gametocytocidal activities. Ame J Ther. 2008;15(2):108–10. https://doi.org/10.1097/MJT.0b013e31804c6d1d.
Yerbanga RS, Lucantoni L, Ouédraogo RK, Da DF, Yao FA, Yaméogo KB, Churcher TS, Lupidi G, Taglialatela-Scafati O, Gouagna LC, Cohuet A. Transmission blocking activity of Azadirachta indica and Guiera senegalensis extracts on the sporogonic development of Plasmodium falciparum field isolates in Anopheles coluzzii mosquitoes. Parasit Vectors. 2014;7(1):1–10. https://doi.org/10.1186/1756-3305-7-185.
Dahiya N, Chianese G, Abay SM, Taglialatela-Scafati O, Esposito F, Lupidi G, Bramucci M, Quassinti L, Christophides G, Habluetzel A, Lucantoni L. In vitro and ex vivo activity of an Azadirachta indica A. Juss. seed kernel extract on early sporogonic development of Plasmodium in comparison with azadirachtin A, its most abundant constituent. Phytomedicine. 2016;23(14):1743–1752. https://doi.org/10.1016/j.phymed.2016.10.019.
Hout S, Chea A, Bun SS, Elias R, Gasquet M, Timon-David P, Balansard G, Azas N. Screening of selected indigenous plants of Cambodia for antiplasmodial activity. J Ethnopharmacol. 2006;107(1):12–8. https://doi.org/10.1016/j.jep.2006.01.028.
Isah AB, Ibrahim YKE, Iwalewa EO. Evaluation of the antimalarial properties and standardization of tablets of Azadirachta indica (Meliaceae) in mice. Phytother Res. 2003;17(7):807–10. https://doi.org/10.1002/ptr.1231.
Jegede IA, Nwinyi FC, Muazzam I, Akumka DD, Njan AA, Shok M. Micromorphological, anti-nociceptive and anti-inflammatory investigations of stem bark of Daniellia oliveri. Afr J Biotechnol. 2006;5(10):930–5.
Onwukaeme DN, Udoh F. Anti-ulcer activity of stem bark of Daniellia oliveri. Niger J Nat Prod Med. 1999;3:39–41.
Muhammad S, Amusa NA. The important food crops and medicinal plants of north-western Nigeria. Res J Agric Biol Sci. 2005;1(3):254–60.
Tsabang N, Fokou PV, Tchokouaha LR, Noguem B, Bakarnga-Via I, Nguepi MS, Nkongmeneck BA, Boyom FF. Ethnopharmacological survey of Annonaceae medicinal plants used to treat malaria in four areas of Cameroon. J Ethnopharmacol. 2012;139(1):171–80. https://doi.org/10.1016/j.jep.2011.10.035.
Ugwah-Oguejiofor CJ, Bello SO, Okolo RU, Etuk EU, Ugwah MO, Igbokwe VU. Ficus platyphylla promotes fertility in female Rattus norvegicus Wistar strain: a preliminary study. Reprod Biol Endocrinol. 2011;9(1):1–6.
Chindo BA, Anuka JA, McNeil L, Yaro AH, Adamu SS, Amos S, Connelly WK, Lees G, Gamaniel KS. Anticonvulsant properties of saponins from Ficus platyphylla stem bark. Brain Res Bull. 2009;78(6):276–82. https://doi.org/10.1016/j.brainresbull.2008.12.005.
Kubmarawa D, Ajoku GA, Enwerem NM, Okorie DA. Preliminary phytochemical and antimicrobial screening of 50 medicinal plants from Nigeria. Afr J Biotechnol. 2007;6(14):1690–6.
Nadembega P, Boussim JI, Nikiema JB, Poli F, Antognoni F. Medicinal plants in Baskoure, Kourittenga province, Burkina Faso: an ethnobotanical study. J Ethnopharmacol. 2011;133(2):378–95. https://doi.org/10.1016/j.jep.2010.10.010.
Adaramoye OA, Adeyemi EO. Hypoglycaemic and hypolipidaemic effects of fractions from kolaviron, a biflavonoid complex from Garcinia kola in streptozotocin-induced diabetes mellitus rats. J Pharm Pharmacol. 2006;58(1):121–8. https://doi.org/10.1211/jpp.58.1.0015.
Antia BS, Ita BN, Udo UE. Nutrient composition and in vitro antioxidant properties of Harungana madagascariensis stembark extracts. J Med Food. 2015;18(5):609–14. https://doi.org/10.1089/jmf.2014.0084.
Patil D, Roy S, Dahake R, Rajopadhye S, Kothari S, Deshmukh R, Chowdhary A. Evaluation of Jatropha curcas Linn. leaf extracts for its cytotoxicity and potential to inhibit hemagglutinin protein of influenza virus. Indian J Virol. 2013;24(2):220–226. https://doi.org/10.1007/s13337-013-0154-z.
Olatokun WM, Ayanbode OF. Use of indigenous knowledge by women in a Nigerian rural community. Indian J Tradit Knowl. 2009;8(2):287–95.
Abdelgadir HA, Van Staden J. Ethnobotany, ethnopharmacology and toxicity of Jatropha curcas L. (Euphorbiaceae): A review. S Afr J Bot. 2013;88:204–218. https://doi.org/10.1016/j.sajb.2013.07.021.
Abiodun O, Gbotosho G, Ajaiyeoba E, Happi T, Falade M, Wittlin S, Sowunmi A, Brun R, Oduola A. In vitro antiplasmodial activity and toxicity assessment of some plants from Nigerian ethnomedicine. Pharm Biol. 2011;49(1):9–14. https://doi.org/10.3109/13880209.2010.490224.
Grace OM, Light ME, Lindsey KL, Mulholland DA, Van Staden J, Jager AK. Antibacterial activity and isolation of active compounds from fruit of the traditional African medicinal tree Kigelia africana. S Afr J Bot. 2002;68(2):220–2. https://doi.org/10.1016/S0254-6299(15)30424-5.
Oyelami OA, Yusuf KO, Oyelami AO. The use of Kigelia africana in the management of polycystic ovary syndrome (PCOS). Chin Med. 2012;3(1). https://doi.org/10.4236/cm.2012.31001.
Bla KB, Trebissou JN, Bidie AP, Assi YJ, Zihiri-Guede N, Djaman AJ. Ethnopharmacological study of antimalarial plants used among the Baoulé-N’Gban of Toumodi in central Côte d’Ivoire. J Appl Biosci. 2015;85:7775–83.
Cho-Ngwa F, Abongwa M, Ngemenya MN, Nyongbela KD. Selective activity of extracts of Margaritaria discoidea and Homalium africanum on Onchocerca ochengi. BMC Complement Altern Med. 2010;10(1):1–7. https://doi.org/10.1186/1472-6882-10-62.
Adedapo AA, Sofidiya MO, Afolayan AJ. Anti-inflammatory and analgesic activities of the aqueous extracts of Margaritaria discoidea (Euphorbiaceae) stem bark in experimental animal models. Rev Biol Trop. 2009;57(4):1193–200.
Agyare C, Obiri DD, Boakye YD, Osafo N. Anti-inflammatory and analgesic activities of African medicinal plants. Med Plant Res Afr. 2013;725–752. https://doi.org/10.1016/B978-0-12-405927-6.00019-9.
Atangwho IJ, Ebong PE, Eyong EU, Williams IO, Eten MU, Egbung GE. Comparative chemical composition of leaves of some antidiabetic medicinal plants: Azadirachta indica, Vernonia amygdalina and Gongronema latifolium. Afr J Biotechnol. 2009;8(18):4685–9.
Oguwike FN, Offor CC, Onubeze DPM, Nwadioha AN. Evaluation of activities of bitterleaf (Vernonia Amygdalina) extract on haemostatic and biochemical profile of induced male diabetic albino rats. IOSR J Dent Med Sci. 2013;11(2):60–4.
Toyang NJ, Verpoorte R. A review of the medicinal potentials of plants of the genus Vernonia (Asteraceae). J Ethnopharmacol. 2013;146(3):681–723. https://doi.org/10.1016/j.jep.2013.01.040.
Macedo T, Ribeiro V, Oliveira AP, Pereira DM, Fernandes F, Gomes NG, Araújo L, Valentão P, Andrade PB. Anti-inflammatory properties of Xylopia aethiopica leaves: Interference with pro-inflammatory cytokines in THP-1-derived macrophages and flavonoid profiling. J Ethnopharmacol. 2020;248:112312. https://doi.org/10.1016/j.jep.2019.112312.
Erhirhie EO, Moke GE. Xylopia aethiopica: A review of its ethnomedicinal, chemical and pharmacological properties. Am J PharmTech Res. 2014;4:21–37.
Ehigiator BE, Adikwu E. Toxicity study of ethanolic stem bark extract of Xylopia aethiopica on fertility indices of male rats: an experimental study. Int J Reprod BioMed. 2020;18(4):265. https://doi.org/10.18502/ijrm.v13i4.6889.
Melo C, Perdomo R, Yerima F, Mahoney O, Cornejal N, Alsaidi S, Coron S, Bonnaire T, Zhang S, Zydowsky TM, Priano C. Antioxidant, antibacterial, and anti-SARS-CoV Activity of commercial products of Xylopia (Xylopia aethiopica). J Med Act Plants. 2021;10(1):11–23. https://doi.org/10.7275/9baf-e988.
International Union for Conservation of Nature Standards and Petitions Committee. Guidelines for Using the IUCN Red List Categories and Criteria. Version 15.1, https://www.iucnredlist.org/documents/RedListGuidelines.pdf. Accessed 3 June 2023.
Cunningham AB. Applied ethnobotany: people, wild plant use and conservation. London: Earthscan; 2001.
Hamilton AC. Medicinal plants, conservation and livelihoods. Biodivers Conserv. 2004;13(8):1477–517. https://doi.org/10.1023/B:BIOC.0000021333.23413.42.
Acknowledgements
The authors would like to express profound thanks to the Royal Society of Tropical Medicine and Hygiene (RSTMH, Grant No. 0021462) for supporting this research. Dr. Bamidele Rasak and Dr. Oluwakemi Iwelumor, Department of Sociology, Landmark University, are appreciated for designing the interview guide used for this study. We recognise the efforts of our research/field assistants: Mr. Omokolade Alejolowo, Ms Tobiloba Elebiyo and Mr. Oluwayomi Adesina who helped with the administration of the questions, collation of information from the TMPs and data analysis. We also acknowledge the heads of the TMPs and their members in the towns/villages visited for participating in this research and sharing necessary information with us.
Funding
This research was supported by RSTMH (RSTMH, Grant No. 0021462).
Author information
Authors and Affiliations
Contributions
IOE, OSA, OMO: Methodology, Conceptualization, Project administration, Investigation, Writing–original draft, Writing—review & editing. IOE, OSA: Funding acquisition. OSA, OMO: Supervision. All the authors listed in this paper have read and approved the final version of the submitted manuscript, and agreed to be accountable for the content of the work.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study protocol involving human participants were performed according to relevant guidelines in the Declaration of Helsinki. In additional, all methods, including the collection of medicinal plants complied with relevant guidelines and regulations outlined in the International Union for Conservation of Nature (IUCN) Policy Statement on Research Involving Species at Risk of Extinction, the Convention on International Trade in Endangered Species (CITES) of Wild Fauna and Flora. Ethical approval to carry out the research was granted by the Landmark University Centre for Research, Innovation and Discoveries (LUCRID) and the Research Ethical Committee for Landmark University (LUAC/2021/0067C). Informed consent to participate in the study was obtained from the participants.
Consent for publication
Not applicable.
Competing interests
None declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 2: Table S1.
Sociodemographic details of informants (n = 35).
Additional file 3: Table S2.
Ethnobotanical data including plant names and voucher numbers of the identified indigenous medicinal plants.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Evbuomwan, I.O., Stephen Adeyemi, O. & Oluba, O.M. Indigenous medicinal plants used in folk medicine for malaria treatment in Kwara State, Nigeria: an ethnobotanical study. BMC Complement Med Ther 23, 324 (2023). https://doi.org/10.1186/s12906-023-04131-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12906-023-04131-4