Abstract
Background
Preeclampsia is a life-threatening disorder during pregnancy and postpartum periods. Preeclampsia can affect the activity of many organs. It is very important because if this disorder is associated with changes in thyroid function, it can affect the results of maternal and fetal tests. Accordingly, the aim of this meta-analysis study was to assess the abnormalities in thyroid function tests in preeclampsia.
Methods
Studies were selected through a systematic search of the MEDLINE/PubMed, Scopus, Web of Science Core Collection, and Google Scholar databases in 31st August 2021. Also, reference lists of review articles and relevant studies were manual-searched to identify other potentially eligible studies. English studies that compared TSH, T4 and T3 of normal pregnant with preeclamptic women (Known to be normotensive before pregnancy; gestational age 20 week or more; singleton pregnancy; no previous history of thyroid dysfunction) were screened. Data sets were screened for eligibility by two independent reviewers. Articles were assessed by the Newcastle–Ottawa Scale. The Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach was used for quality assessment of evidence on outcome levels.
Results
After reviewing 886 published studies, 63 observational studies were selected and used for this meta-analysis. The study population included 21,528 pregnant women. The findings revealed that TSH (SMD = 1.70, 95%CI: 1.39 to 2.02; p < 0.001) was significantly higher in preeclamptic women. TT4 (SMD = -0.82, 95%CI: -1.16, -0.49; p < 0.001), TT3 (SMD = -0.88, 95%CI: -1.36 to -0.41; p < 0.001) and FT3 (SMD = -0.59, 95%CI: -0.91 to -0.27; p < 0.001) were less in preeclamptic women. There was no statistically significant difference in FT4 between two groups (SMD = 0.002, 95%CI: -0.27 to 0.27; p = .990). The results of publication bias and sensitivity analysis confirmed the reliability and stability of this meta-analysis. The quality of evidence was regarded as moderate, low, and very low for these risk factors according to the GRADE approach.
Conclusions
Findings of this meta-analysis indicated preeclamptic women were more at risk of changes in thyroid function tests. In order to prevent thyroid disorders, it is recommended that thyroid function tests be performed in women with pre-eclampsia.
Similar content being viewed by others
Background
Preeclampsia complicates 2–8% of pregnancies and is one main cause of maternal and neonatal mortality and morbidity worldwide [1, 2]. The American College of Obstetricians and Gynecologists (ACOG) defined preeclampsia in 2017 as having a blood pressure of more than 140/90 mmHg at intervals of four hours after the 20th week of pregnancy, as well as proteinuria greater than or equal to 300 mg in 24 h of urine collection [3]. Preeclampsia is a multi-systemic disorder [4].
During pregnancy, the physiological changes of the thyroid gland are completely normal and incompatibility with these changes leads to dysfunction of the thyroid gland [5]. Naturally, thyroid hormones increase by 40–100% to meet the needs of both mother and fetus [6]. In a normal pregnancy, Thyroid Stimulating Hormone (TSH) increases due to an increase in concentration of Human Chorionic Gonadotropin (HCG) [7]. Total Thyroxine(TT4) and Total Triiodothyronine(TT3) concentrations increase rapidly, while Free Triiodothyronine and Free Thyroxine (FT3 and FT4) increase at a slower rate [6]. Most serum thyroid hormones are protein bound and only 0.2% of T3 and 0.02% of T4 are free. About 45- 70% of thyroid hormones bind to thyroxine-binding globulin (TBG) and the rest to trans-thyrotin and albumin. [7].
Preeclampsia affects the function of many organs in the body, including the thyroid gland [8]. In general, the findings were not the same in all studies. For example, in one study, despite increased in TSH levels, T3 and T4 levels did not change notably in women with preeclampsia [9]. The findings of two studies revealed that TSH, TT4, and TT3 were not differ significantly between preeclamptic and normal pregnant women [3, 10]. The American Thyroid Association (ATA) guidelines report that there is no association between changes in thyroid function tests and preeclampsia [11]. Some findings resulted that preeclamptic women had higher incidence of the increase in TSH and low T4 in a comparison with normal pregnant women [10].
Some studies have shown that 29.3% of pregnant women with preeclampsia had hypothyroidism, 71.42% had subclinical hypothyroidism and the rest had overt hypothyroidism. [12]. Another studies showed that %16.7 and %43.7 of preeclamptic women had subclinical and overt hypothyroidism, respectively [13]. Subclinical hypothyroidism is defined by an increase in TSH concentration and normal FT4 concentration, and overt hypothyroidism is defined by an increase of TSH level and decrease free thyroxine [5].
The mechanism of hypothyroidism in pre-eclamptic women was not well understood. However, according to various theories, this mechanism in preeclampsia may be related to a decrease in plasma protein concentration and an increase in endothelin levels [14]. In addition, the high circulation of estrogens can change thyroid function. Also, decreased thyroid function may be due to anti-angiogenic factors in preeclampsia that reduce nitric oxide production [15, 16]. This in turn decreases capillary flow of thyroid which could lead to hypothyroidism [8]. According to various studies, changes in thyroid function tests can affect the outcome of pregnancy [17, 18]. The results of a survey showed that subclinical hypothyroidism (SCH) is almost twice as likely to cause severe preeclampsia [5]. Early subclinical hypothyroidism in pregnancy is a risk factor for the premature rupture of membranes PROM [19]. Another study found that mothers with SCH had increased risks for spontaneous abortions [20], intrauterine growth restriction (IUGR) and low birth weight(LBW) [21].
During a normal pregnancy, changes in thyroid function are well documented, but information on thyroid function in complicated pregnancies is scant [22, 23]. The association of preeclampsia with thyroid function has been studied but with different results. However, Due to the high prevalence of preeclampsia and the contradictory findings of thyroid function tests in pre-eclampsia, the aim of this study was to Meta-analyze the data examining changes in thyroid function tests in preeclampsia. The results of the data combination may provide useful information for patient counseling and clinical management of women with preeclampsia.
Objectives
The purpose of this systematic review and meta-analysis is to evaluate of thyroid function test abnormalities in preeclampsia.
Materials and methods
Protocol and registration
This study was reported based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist for systematic review and meta-analysis. The report was prepared in accordance with PRISMA 2020 guidelines [24], and the completed PRISMA 2020 checklist can be found in supplemental appendix 1.
The study protocol was registered in the PROSPERO (Registration number: CRD42020213560).
Search strategy for the identification of studies
A comprehensive electronic search in the databases PubMed/Medline (NLM), SCOPUS, and Web of Science Core Collection was carried out in 31 August 2021. Google Scholar was also used to search for online research-related articles that may not be in the search databases to increase the comprehensiveness of the search. Medical Subject Headings (Mesh) and Embase Subject Headings (Emtree) were used to finding keywords. Keywords that were obtained from Emtree or Mesh were included in our search strategy without any changes. The search terms used were: “pregnancy toxemia”, “toxemia of pregnancy”, “preeclampsia”, “pre-eclampsia”, “preeclamptic women”, “pre-eclamptic women”, “TSH”, “thyroid stimulating hormone”, “thyrotropin”, “T3”, “triiodothyronine”, “T4”, “thyroxine”, “tetraiodothyronine”, “hypothyroidism”, “hypothyroidism” and “thyroid”. These keywords were combined with “AND” or “OR” browsers during the advanced search (see Appendix 2 for electronic database search strings). A snowball search was also used to find relevant articles. In this method, reference lists of review articles and related studies were manually searched to identify other potentially eligible studies.
All searches were restricted to English language. References were managed with EndNote 8.0 software.
Study selection
Two independent reviewers (MF and SF) screened identified eligible studies based on their titles and abstracts. Then, all potentially relevant full texts were read to produce a final list of included studies for rejection recorded. The list of studies rejected at this stage and reasons for rejection can be found in supplemental appendix 3.
Any disagreements were resolved with discussion or through adjudication by a third review (SHT). The results of Cohen’s kappa indicated a perfect agreement between two authors (Kappa = 0.84).
Studies were incorporated if they were: original and peer-reviewed researches; one author conducted the initial screening analysis. After removing duplicates and scanning the titles and abstracts of articles, those meeting the inclusion criteria were reviewed.
Study eligibility criteria
Studies that met all of the following criteria in this review:
Included criteria
-
Compared TSH, T4 and T3 of normal pregnant with preeclamptic women (Known to be normotensive before pregnancy; gestational age 20 week or more; singleton pregnancy; no previous history of thyroid dysfunction);
-
English studies;
-
Published or in-press articles until August 31, 2021.
Excluded criteria
-
Articles without appropriate case or control groups
-
Animal studies
-
Case reports
-
Letter to the editor without quantitative data
-
Qualitative studies
-
Systematic reviews and meta-analyses
-
Abstract without sufficient data or full-text for quality assessment.
For prevention misleading conclusions about excluding abstracts or full-texts without sufficient data, the first author sent five emails (every three days) to correspondence of studies. unfortunately, she did not receive any replies.
Extracted information
-
- Research information (the first author, geographic location, year of publication, research design, sample size, finding);
-
- Characteristics of the participants (age, gestational age, number of participants, inclusion and exclusion criteria);
-
- Comparison of the details (number of groups, results of TSH, T4 and T3 tests);
Data extraction
First outcome measurement in this meta-analysis were TSH, TT3, TT4, FT3 and FT4. Additionally, age, gestational age, birth weight, BMI, SBP, DBP, and parity were considered secondary outcomes measurement. Data were carefully and independently extracted from all eligible studies by two reviewers (MF and SF according to the inclusion criteria mentioned above using a Microsoft Excel spreadsheet). The extracted data included study characteristics (e.g., first author name, year of publication, country, sample size, patient age and gestational age. Disagreement was resolved by discussion or consulting with a third reviewer (SHT).
Risk of bias (quality) assessment
In this study for evaluating the quality of studies, was used the Newcastle–Ottawa Scale (NOS) for case–control and clinical trial studies. The NOS is the most commonly tool for assessing the quality of observational cohort and cross-sectional studies [25], and each study is awarded scores for eight items: selection (maximum 4 scores for cohort and 5 scores for cross-sectional studies), comparability (maximum 2 scores), and Exposure (maximum 3 scores). Scores of the quality of articles was used as variable for subgroup analysis.
Quality of evidence
The Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach was recommended by the Cochrane collaboration and can be used for assessing the quality of study outcome levels. There are four levels of certainty of evidence: high, moderate, low, and very low [26]. For the GRADE approach, we used the software GRADEpro,
Effect size
In this study, effect sizes were the difference between the mean of thyroid function indices (TSH, TT4, TT3, FT4 and FT3) divided by their standard errors related. The effect size was calculated for each study that had the inclusion criteria and was eventually used for combination in the meta-analysis.
Strategy for data synthesis
The standardized mean difference (SMD) and 95% confidence interval (CI) were applied to compare TSH, TT3, TT4, FT3 and FT4 in two groups. For each variable, to test SMD = 0, we used this formula:
Prior to the meta-analysis of the studies, a sensitivity test was performed and studies that were not within the confidence interval were excluded from the meta-analysis. To evaluate the heterogeneity between the studies, Cochran's Q test (p < 0.10 indicated significance) with I2 (0–25%: low heterogeneity; 25–50%: moderate heterogeneity; > 50 high heterogeneity). I2 measure the inconsistency across study and in general when \({\mathrm{I}}^{2}\) > 50%, a random-effects model and if I2 < 50%, a fixed effect model is suitable for pooling the effect size. Therefore, a random effects model (the DerSimonian and Laird method) was used to estimate the pooled effect size (EF) and each target variable. The results of meta-analysis were reported as a form of a forest plot which containing SMD and 95% CI.
To evaluate the degree of heterogeneity between the mean studies, Q-Cochran test (p < 0.1 as heterogeneity) and I2 index (I2 > 50% indicating heterogeneity between studies) were used. If significant heterogeneity was observed between studies, meta-analysis was performed in layers of variables such as age, gestational age, birth weight, BMI (Body Mass Index), SBP (Systolic Blood Pressure), DBP (Diastolic Blood Pressure), and parity. When heterogeneity was observed in the layers of each of the mentioned variables, we related the reasons for possible heterogeneity to the mentioned variables. We also performed meta-regression for variables whose results in the subgroup test are significant.
Egger’s regression test was used to evaluate the publication bias between simultaneous studies. Hence, when the potential publication bias observed, the fill and trim method was performed. Also, all meta-analyzes were performed in STATA software version 14.
We concluded that there is a potential bias among studies. Then we use the “trim and fill” method to consider the missing potential studies on both sides of the funnel piece and then recalculate the effect size considering the inclusion of the missing potential studies [27].
Results
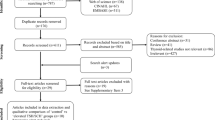
Our search resulted in 886 articles. The article selection process was such that articles that did not qualify and did not meet the inclusion criteria were removed from the initial screening (based on the title or abstract of the article). Finally, we selected 63 articles for further reviewing (Fig. 1).
All of the studies selected after the search process met the inclusion criteria and had high quality according to Newcastle checklist. Mean age was 26.59 years in preeclamptic group and 25.67 in normal pregnant women. Mean gestational age was 35.32 years in preeclampsia and 38.2 in normal pregnancy.
The final study population comprised 21,528 pregnant women participants. Of whom 4765 (22.13%) of pregnant women were preeclamptic and 16,763 (77.87%) normotensives. Included study characteristics are summarized in Table 1.
Sensitivity analysis
Initially, we performed sensitivity analyses by consecutive removal of a study at a time to evaluate the change in the pooled SMD and 95% CI of each thyroid hormone. The results of sensitivity analysis showed that the pooled effect size of thyroid hormones is 95% confidence interval. In other words, when each study was removed, there was no significant change in the pooled SMD.
Since there was a significant heterogeneity between studies, subgroup analysis was performed. The subgroup analysis was performed according to the age, gestational age, birth weight, BMI, SBP, DBP, parity (Table 2). In order to evaluate the impact of heterogeneous studies on the pooled estimates, we conducted a sensitivity analysis. For this purpose, we excluded studies serially and obtained pooled estimates from the remaining studies. This enabled us to evaluate whether single studies with highly heterogeneous results were affecting the overall pooled estimates. Results of this study, there is a potential bias among the studies. We then use the "draw and fill" method to consider the missing potential studies on either side of the funnel piece and recalculate the effect size by considering the inclusion of the lost potential studies For meta-analysis of compare gestational age of preeclampsia and normal pregnant women 37 studies were applied. The results showed in Table 3. Fifty-six studies comparing TSH were performed in preeclampsia and normal pregnant women. The results presented in Table 3 and Fig. 2. Thirty-eight studies have been performed to estimate the total SMD of TT4 and TT3. We found TT4 (Table 3 and Fig. 3) and TT3 (Table 3 and Fig. 4) for preeclampsia women significantly were less than normal women. Twenty-two studies were performed to compare FT4 in preeclampsia and normal pregnant women. The results showed that there was no statistically significant difference between the two groups (Table 3).
Based on the results of pooled SMD in 28 studies, FT3 was significantly lower in women with preeclampsia than in normal women (Table 3 and Fig. 5).
For preeclamptic women, meta-regression results showed that with increasing age (β = -0.16, p = 0.022) and SBP (β = -0.09, p = 0.043) significantly FT3 was decreased. Also, FT4 increased significantly with increasing SBP (β = 0.46, p = 0.031). In the group of normal pregnant women, FT3 decreased significantly with age (β = -0.14, p = 0.037) (Table 2 and Fig. 6).
Publication bias
Publication bias in meta-analysis was evaluated by performing Egger rank correlation test for statistical analysis of plots symmetry. The results showed that except for TSH (t = -9.09, p < 0.001), TT4 (t = -3.32, p = 0.002) and FT3 (t = -2.09, p = 0.043) no observed publication bias among studies for other variables.
Quality of Evidence
We are moderately confident in the effect estimate TT4, FT4 and FT3: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different For TT3, our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect. Also, for TSH, we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect.
According to results of GRADE assessment the risk of bias, inconsistency, indirectness and imprecision for all effect sizes were not serious. The detailed table is presented in Table 4.
Discussion
In this meta-analysis study, for the first time, thyroid function tests in preeclampsia were compared with normal pregnant women. The results of this study showed that the mean TSH level is higher in preeclamptic women, but TT4, TT3 and FT3 are lower. Additionally, there was no statistically significant difference in FT4 between the two groups.
The results of this study showed that the higher TSH mean level in preeclamptic patients, which is consistent with other studies [35, 64, 82]. Even though placental function abnormalities can interfere with estrogen production, leading to decrease of TBG levels and it can be a reason for elevated TSH secretion by anterior pituitary [59, 61] but in the some studies, TSH level in preeclamptic women was not higher than normal pregnant women [3, 41].
Consistent with our findings, several studies have observed lower TT4 and TT3 levels in preeclampsia [7, 16, 35]. According to other studies, women with preeclampsia have increased antiangiogenic factors, which in turn reduce nitric oxide production. This in turn reduces thyroid capillary flow, which can lead to hypothyroidism [82]. In a normal pregnancy, estrogen increases serum TBG, but in preeclampsia, lack of estrogen production leads to a decrease in TBG. Also, placental dysfunction and decreased estrogen production in preeclampsia can reduce TT3 and TT4. [38, 73]. Also, in women with hypertension, the presence of protein-binding hormones in the urine and a decrease in serum albumin due to proteinuria can lead to a decrease in TT3 levels. Increased TSH levels in preeclampsia are associated with concomitant decreases in TT4, TT3 and endothelin levels. Accordingly, endothelial dysfunction is seen in extensive vasospasm and poor perfusion of many tissues such as the thyroid in preeclampsia. [73]. Other investigations have reported higher TT4 and TT3levels in preeclamptic women [9, 48].
Our finding observed FT3 in preeclamptic women was significantly different from normal pregnant. Low FT3 levels were observed in preeclamptic patients in a few studies, too [3, 33, 41, 58]. The hypothesis appropriate to these conditions is that the conversion of T4 to T3 is impaired in the liver, which could be the reason for the low level of FT3 in women with preeclampsia [30]. No difference in FT3 between the two groups was observed in other studies [30, 64, 82]. The findings of present study showed lower mean FT4 level in preeclamptic patients is similar to the results of other studies [30, 38, 41] while finding of some investigations showed a higher mean FT4 level in preeclampsia [14, 23].
In our study, results of meta-regression presented that in preeclamptic women with increasing SBP significantly FT3 was decreased. Also, FT4 increased significantly with increasing SBP. Maduka and et al. did not reported any correlation between FT3 or FT4 with systolic blood pressure in the preeclamptic women [29].
In another study, TSH is correlated positively with systolic blood pressure in preeclampsia [31] but this relationship did not find in this metanalysis. The differences observed in researches could be as a result of differences in the race, diets, and geographical locations of participants [29].In general, the results of current meta-analysis showed that an increase serum TSH with a meaningless difference in FT4 in preeclamptic women is called subclinical hypothyroidism [5]; this finding is supported by previous studies [42, 44, 64].
The relation between changes in thyroid function and preeclampsia may be reciprocal [15]. This means that the thyroid disorder is one of the predisposing causes for pre-eclampsia [57] and hypothyroidism is one of the pathophysiologic causes of pre-eclampsia.. Hypothyroidism can play an important role in smooth muscle contraction in the renal and systemic arteries, leading to increased peripheral vascular resistance, diastolic blood pressure, and decreased tissue perfusion [57, 60]. Therefore, the identification of thyroid abnormalities and their proper management can affect the incidence of preeclampsia [60]. It was suggested that evaluation of thyroid function could be useful in predicting preeclampsia [55]. In this regard, TSH plays a central role in screening and diagnosis of many thyroid disorders [5].
On the other hands, preeclampsia can be a key cause in the pathogenesis of hypothyroidism. Effects of preeclampsia in thyroid function is not yet elucidated, but increased levels of endothelin as a vasoconstrictor produced by vascular endothelium are involved in the pathogenesis of subclinical hypothyroidism in preeclampsia [44]. Out of 95 preeclamptic women, 44.2% were thyroid dysfunction with 38.9% patients had subclinical hypothyroidism and 4.2% overt hypothyroidism and 1% hyperthyroidism [83].
Nowadays, some researchers have cited the relationship between maternal and fetal complications with thyroid dysfunction is a problem of concern [5, 23, 33]. Therefore, obstetricians and endocrinologists should be increasingly aware of the potential complications of hypothyroidism in pregnancy [49] and should recommend thyroid function screening in pregnancy for early diagnosis; follow-up in the third trimester of pregnancy, especially in preeclamptic women and treatment of thyroid dysfunction to prevent further complications [50, 57, 61]. Fortunately, the evaluation of thyroid function tests (serum T3, T4 and TSH) is reliable, simple, economical and sensitive [65].
The results of this study showed that except for TSH, there was no diffusion bias between studies for other variables. These results could mean that only research that has published positive results and published negative or insignificant statistical studies will not be published because they see it as a failed researcher that is not true. [84].
The quality of evidence for TSH was rated as very low by the GRADE that any effect of preeclampsia on TSH is very uncertain. The quality of evidence for TT4, FT4, and FT3 were rated as moderate. It shows in future studies, preeclampsia has likely an important effect on them and may change the estimate. For TT3, the quality of evidence was rated as low which present future studies, preeclampsia is very likely to have an important effect on TT3 and is likely to change the estimate.
Researches showed drug therapy may reduce the complications of pregnancy in women with thyroid dysfunction and recommended thyroid function be checked in preeclamptic women [85, 86]. Further studies are needed to investigate the odds of preeclampsia among hypothyroid women.
Present study has several strengths. This is the first systematic review and meta-analysis about the relationship between preeclampsia and the thyroid function tests. Accordingly, the finding of current study showed more reliable and evident results than the separate studies. Moreover, included studies were performed in four continents of Asia, Africa, America and Europe. Forth, the results of this meta-analysis have important implications for care from preeclamptic women and screening of thyroid hormone levels during pregnancy. We used the GRADE approach to appraise the quality of evidence and assessed the risk of bias of studies.
The limitation of the current study is that the great majority of studies comprising the meta-analysis reported findings for mixed groups of mild and severe preeclampsia. However, few studies reported results separately for mild and severe. Accordingly, we could not find the relation between changes in thyroid function tests with the severity of preeclampsia.
Conclusion
Findings of this meta-analysis indicated preeclamptic women were more at risk of changes in thyroid function tests. Therefore, thyroid function tests should be considered in preeclamptic women. Identification of changes in thyroid hormones in preeclampsia might be of help in preventing the thyroid disorders.
These results provide insights into the optimizing clinical decision-making strategies that should provide thyroid screening in women with preeclampsia.
Availability of data and materials
All data analyzed during this study are included in this published article.
Abbreviations
- ACOG:
-
American College of Obstetricians and Gynecologists
- ATA:
-
American Thyroid Association
- BMI:
-
Body Mass Index
- CI:
-
Confidence Interval
- DBP:
-
Diastolic Blood Pressure
- EF:
-
Effect size
- FT3:
-
Free Triiodothyronine
- FT4:
-
Free Thyroxine
- GRADE:
-
Grading of Recommendations Assessment, Development and Evaluation
- HCG:
-
Human Chorionic Gonadotropin
- IUGR:
-
Intrauterine Growth Restriction
- LBW:
-
Low birth weight
- LT4:
-
Levothyroxine
- NOS:
-
Newcastle–Ottawa Scale
- PRISMA:
-
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- PROM:
-
Premature Rupture Of Membranes
- SBP:
-
Systolic Blood Pressure
- SMD:
-
Standardized Mean Difference
- SCH:
-
Subclinical Hypothyroidism
- TBG:
-
Thyroxine Binding Globulin
- TSH:
-
Thyroid Stimulating Hormone
- TT3:
-
Total Triiodothyronine
- TT4:
-
Total Thyroxine
References
Sadek A, et al. Serum vitamin D3 levels in pregnant women with preeclampsia at third trimester of pregnancy. Baghdad J Biochem Appl Biol Sci. 2021;2(03):131–7.
Nasser N, et al. The association between urinary placental protein 13 and soluble fms-like tyrosine kinase-1 in preeclamptic women in the third trimester of pregnancy. Baghdad J Biochem Appl Biol Sci. 2020;1(01):49–55.
Sattar R, Ahmed E, Abbasi SQ. Thyroid Hormones Pattern in Preeclampsia. Ann King Edward Med Univ Lahore Pakistan. 2018;24:863–6.
Popovski N, Nikolov A. Practice Bulletin of the American College of Obstetrics and Gynaecologists 2019 on Magement of Hypertensive Disorders in Pregnancy-A Short Review of the Current Recommendations. Biomed J Sci Tech Res. 2019;23(2):17198–201.
Dashe JS, et al. Williams obstetrics. New York: McGraw-Hill Professional; 2018.
Saki F, et al. Thyroid function in pregnancy and its influences on maternal and fetal outcomes. Int J Endocrinol Metab. 2014;12(4):e19378.
Chowdhary S, et al. Thyroid Function Tests in Preeclampsia. Int J Med Sci Clin Invention. 2018;5(03):3606–9.
Sadiq AM, et al. Subclinical hypothyroidism with preeclampsia. Res J Pharm Biol Chem Sci. 2016;7(3):1536–44.
Thanna RC, Nigoskar S. Association of TSH (Thyroid Stimulating Hormone) with Preeclampsia as a Diagnostic Indicator. Int J Health Sci Res. 2015;5(6):107–10.
Khadem N, et al. Comparison of serum levels of Tri-iodothyronine (T3), Thyroxine (T4), and Thyroid-Stimulating Hormone (TSH) in preeclampsia and normal pregnancy. Iran J Reprod Med. 2012;10(1):47–52.
Alexander EK, et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid. 2017;27(3):315–89.
Prashansa S. Prevalence of hypothyroidism among women with pre-eclampsia: THYDOR study. Vellore: Christian Medical College; 2018.
Haldar R, et al. Correlation Between Maternal Serum Thyroid Profile And Preeclampsia At or Above 36 wks Gestation A Prospective Comparative Observational Study. IOSR J Dental Med Sci. 2017;16(3):41–5.
Harshvardhan L, et al. Study of Association of Thyroid Hormone in Pre-Eclampsia and Normal Pregnancy. J Assoc Physicians India. 2017;65(11):44–6.
Sogani S, Varma V, Sarkar PD. Estimation of thyroid hormones levels in preeclamptic pregnant women: an early predictor of the disease. Al Ameen J Med Sci. 2015;8(4):266–70.
Muraleedharan N, Janardhanan J. Thyroid hormone status in preeclampsia patients: A case-control study. Muller J Med Sci Res. 2017;8(2):68–68.
Mannisto T, et al. Thyroid dysfunction and autoantibodies during pregnancy as predictive factors of pregnancy complications and maternal morbidity in later life. J Clin Endocrinol Metab. 2010;95(3):1084–94.
Kharb S, Sardana D, Nanda S. Correlation of thyroid functions with severity and outcome of pregnancy. Ann Med Health Sci Res. 2013;3(1):43–6.
Wu M-Q, et al. The Impact of Subclinical Hypothyroidism on Adverse Perinatal Outcomes and the Role of Thyroid Screening in Pregnancy. Front Endocrinol. 2019;10(522):1–8.
Wang S, et al. Effects of maternal subclinical hypothyroidism on obstetrical outcomes during early pregnancy. J Endocrinol Invest. 2012;35(3):322–5.
Chen LM, et al. Effects of subclinical hypothyroidism on maternal and perinatal outcomes during pregnancy: a single-center cohort study of a Chinese population. PLoS ONE. 2014;9(10): e109364.
Jain P, Devi R. Thyroid hormonal status in pregnancy and pre-eclampsia and its correlation with maternal age and parity. Int J Basic Appl Med Sci. 2017;7(1):1–7.
Naykı Ü, et al. Maternal thyroid functions in pre-eclampsia. Med Bulletin Sisli Etfal Hospital. 2014;48(4):308–11.
Page MJ, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg. 2021;88:105906.
Ma L-L, et al. Methodological quality (risk of bias) assessment tools for primary and secondary medical studies: what are they and which is better? Mil Med Res. 2020;7(1):7.
Reeves BC, et al. Cochrane Handbook for Systematic Reviews of Interventions. Chapter 24: Including nonrandomized studies on intervention effects. Second Edition. London, England: Cochrane Collaboration; 2019.
Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56(2):455–63.
Muraleedharan N, Beegum MS. Association between Serum Albumin and Hypothyroidism in Preeclampsia: A Case-control Study. J Clin Diagn Res. 2021;15(6):BC26–30.
Maduka I, Obi N, Egwu M. Evaluation of Thyroid Hormones and Thyroid Antibodies in Nigerian Pregnant Women with Pre-eclampsia. J Med Lab. 2020;30(4):52–61.
Abdallatief A-S, et al. Comparison of thyroid hormones level in normal and preeclamptic pregnancy. Sohag Med J. 2019;24(1):26–32.
Patil N. Evaluation of thyroid function test in severity of preeclampsia. MedPulse Int J Biochem. 2020;16(3):29–35.
Kumar M, et al. Thyroid hormones: As biomarker of preeclampsia and an influencer in outcome of pregnancy. Int J Appl Res. 2020;6(3):01–3.
Misra M, et al. Comparative analysis of serum calcium level and thyroid profile in pregnant normotensive and preeclamptic women: a case-control study. J Clin Diagn Res. 2020;14(5):BC05–9.
Bozkurt ME, Yucel O, Saler T. An investigation into thyroid function tests in pregnant women and the relationship between thyroid autoantibodies and preeclampsia. Ann Med Res. 2020;27(7):1886–92.
Marwa AM, Haddad NI, Hussein EA. Correlations of Serum Vitamin D and Thyroid Hormones with Other Biochemical Parameters in Iraqi Pregnant Women with Preeclampsia Disease. J Glob Pharma Technol. 2019;11(2):441–50.
Prashanthi B, Himaja J, Himaja J. A Comparative Study of Thyroid Hormone Levels in Severe and Mild Pre-Eclmaptic Women and Normal Pregnant Women. Int J Pharm Biolog Sci. 2019;9(3):1097–103.
Tariq J, Aslam M, Zaidi YA. Frequency of altered thyroid hormone in cases of pre-eclampsia. Int J Adv Biotechnol Res. 2018;9(4):523–6.
Murmu AK, et al. Correlation of Hypothyroidism with Pregnancy Outcome in Preeclampsia. OSR-JDMS. 2018;17(4):72–8.
Chaudhary RR, Muddeshwar M. A study of thyroid profile and serum albumin in preeclampsia women. Int J Cur Res Rev| Vol. 2016;8(23):11.
Ban-Amer M. Detection of Relationship between Maternal Thyroid Hormones and Severity of Preeclampsia. Int J Med Res Health Sci. 2018;7(9):127–31.
Amin K, Afzal I, Babur A. A Case Study on the Relation between the Preeclampsia Pregnancy and the Level of the Thyroid Harmone. Int J Adv Biotechnol Res. 2018;9(1):689–94.
Rani SU, Arumaikannu J, Shanthi S. Hypothyroidism as a bio marker of preeclampsia: Our experience. Int J Clin Obstetr Gynaecol. 2018;2(1):69–71.
Murmu AK, et al. Thyroid Stimulating Hormone As A Possible Predictor Of Preeclampsia. OSR-JDMS. 2018;17(4):01–9.
Grammatikakis I, et al. Prevalence of maternal hypothyroidism complicated with preeclampsia: a retrospective analysis of 60 cases. Med Surg J. 2017;121(2):253–7.
Dineshkumar R, Praneswari D, Zirsangliana-Chhangte M. Thyroid Status in Pre Eclampsia. J Dental Med Sci. 2017;16(6):141–3.
Businge C, et al. Iodine Deficiency in Pregnancy as a Predictor of Sub-clinical Hypothyroidism, Preeclampsia and Future Cardiovascular Disease. Asian J Clin Nutr. 2017;9(3):118–23.
Rajalaksmi V, Kumari A. Study on prevalence of hypothyroidism in women with preeclampsia. Med Sci. 2016;5(8):45–7.
Reddy AS. Study of Thyroid Hormones Free Tri-iodothyronine (T3), Free Thyroxine (T4) and Thyroid Stimulating Hormone (TSH) in Subjects with Preeclampsia. Int J Contemp Surg. 2016;4(1):182–6.
Tadas S, Tadas A. Thyroid hormone alteration in women with pre-eclampsia. Int J Res Med Sci. 2016;4(10):4520–3.
Chauhan M, Tomar S. A Clinical Study on Pre-Eclampsia and Thyroid Hormone Alteration in Women Presenting to Tertiary Care Institute of North India. JMSCR. 2016;4(10):12984–7.
Procopciuc LM, et al. D2-Thr92Ala, thyroid hormone levels and biochemical hypothyroidism in preeclampsia. Gynecol Endocrinol. 2017;33(2):136–40.
Umadevi N. Study on prevalence of hypothyroidism in women with preeclampsia, in Obstetrics and Gynaecology. India: Stanley Medical College, Chennai; 2015.
Rafeeinia A, Teymoori H, Marjani A. Serum thyroid hormone levels in preeclampsia women in Gorgan. J Med Sci (Faisalabad). 2015;15(1):38–43.
Deshpande S, et al. Maternal thyroid hormone status in pre-eclampsia: a tertiary care hospital based study. Int J Reprod Contracept Obstet Gynecol. 2015;4(6):1853–7.
Elhaj ET, et al. Thyroid Function/Antibodies in Sudanese Patients with Preeclampsia. Front Endocrinol (Lausanne). 2015;6:87.
Sheela S, Dayanand C, Sivaraj N. Evaluation of maternal thyroid hormone status and atherogenic potency in pre eclampsia in Kolar District-A South West of India. J Adv Med Med Res. 2015;10(4):1-8.
Satyanarayan AK, et al. Maternal thyroid profile in pre-eclampsia. Int J Med Sci Public Health. 2015;4(10):1401–3.
Camejo MI, Casart YC. Relation between human chorionic gonadotropin and thyroid hormones in preeclampsia. Open Access Library J. 2014;1:e652.
Kaveti SM, et al. Correlation of Thyroid and Lipid Profile in Preeclampsia Patients. MRIMS J Health Sci. 2014;2(2):84–8.
Bayejid M, et al. Association of maternal hypothyroidism with preeclampsia in Bangladeshi population. BioJ Sci Technol. 2014;1(1):1-7.
Manjunatha S, Basavaraja G, Ramesh S. Thyroid Dysfunction in Pregnancy and Preeclampsia. Sch J App Med Sci. 2014;2(6F):3297–9.
Kurlak LO, et al. Thyroid hormones and their placental deiodination in normal and pre-eclamptic pregnancy. Placenta. 2013;34(5):395–400.
Khanam M, Ilias M. Study of thyroid hormonal status in preeclamptic patients. Med Today. 2013;25(2):63–6.
Das S, et al. Evaluation of Thyroid Hormone Changes in Non-Pregnant, Normotensive Pregnant and Pregnancy with Preeclampsia. J Dent Med Sci. 2013;11(6):16–8.
Monika Y, Ranjana M, Prakash O. A Comparative Study of Thyroid Function Tests (serum T3, T4 and TSH) in Normal Pregnancy and Preeclampsia. IJSR. 2015;4(5):1277–8.
Männistö T, et al. Preeclampsia, gestational hypertension and subsequent hypothyroidism. Pregnancy Hypertens. 2013;3(1):21–7.
Procopciuc LM, et al. The effect of the D1–C785T polymorphism in the type 1 iodothyronine deiodinase gene on the circulating thyroid hormone levels in Romanian women with preeclampsia Association with the degree of severity and pregnancy outcome of preeclampsia. Gynecol Endocrinol. 2012;28(5):386–90.
Alavi A, et al. Thyroid dysfunction and autoantibodies association with hypertensive disorders during pregnancy. J Pregnancy. 2012;2012:742695.
Riah HA, Ahmed F, Moneim HA. Thyroid Function and Preeclampsia. Population Sci. 2012;37:97–118.
Dhananjaya B, et al. Thyroid stimulating hormone (TSH) level as a possible indicator of pre-eclampsia. J Clin Diagn Res. 2011;5(8):1542–3.
Al-Naqeeb A. Correlation between Thyroid-related Hormones and Preeclampsia. Nurs Natl Iraqi Specility. 2010;3(Special):1–10.
Obiero RO. Maternal thyroid hormonal status in preelampsia and Eclampsia at Kenyatta National Hospital-a case control study. In: Study Obstetrics and Gynecology. Kenya: University of Nairobi; 2010.
Sardana D, Nanda S, Kharb S. Thyroid hormones in pregnancy and preeclampsia. J Turkish German Gynecol Assoc. 2009;10(3):168–71.
Pasupathi P, Deepa M. Evaluation of serum lipids and thyroid hormone changes in non-pregnant, pregnant, and preeclampsia women. Thyroid Sci. 2009;4(10):1–7.
Kumar A, Ghosh B, Murthy N. Maternal thyroid hormonal status in preeclampsia. Indian J Med Sci. 2005;59(2):57–63.
Larijani B, et al. Thyroid hormone alteration in pre-eclamptic women. Gynecol Endocrinol. 2004;18(2):97–100.
Başbuğ M, et al. Correlation between maternal thyroid function tests and endothelin in preeclampsia-eclampsia. Obstet Gynecol. 1999;94(4):551–5.
Khaliq F, et al. Thyroid functions in pre-eclampsia and its correlation with maternal age, parity, severity of blood pressure and serum albumin. Indian J Physiol Pharmacol. 1999;43(2):193–8.
Kaya E, et al. Relation between birth weight and thyroid function in preeclampsia-eclampsia. Gynecol Obstet Invest. 1994;37(1):30–3.
Lao T, et al. Maternal thyroid hormones and outcome of pre-eclamptic pregnancies. BJOG. 1990;97(1):71–4.
Lao T, Chin R, Swaminathan R. Thyroid function in pre-eclampsia. BJOG. 1988;95(9):880–3.
Muraleedharan N, Beegum M. Association between Serum Albumin and Hypothyroidism in Pre-eclampsia: A Case-control Study. J Clin Diagn Res. 2021;15(6):BC26–30.
Banik P, et al. Thyroid dysfunction in preeclampsia and related fetomaternal outcomes. Int J Reprod Contracept Obstet Gynecol. 2019;8(5):1928–33.
Nair AS. Publication bias-Importance of studies with negative results. Indian J Anaesth. 2019;63(6):505–7.
Maraka S, et al. Thyroid hormone treatment among pregnant women with subclinical hypothyroidism: US national assessment. BMJ. 2017;356:i6865.
Bein M, et al. Levothyroxine and the risk of adverse pregnancy outcomes in women with subclinical hypothyroidism: a systematic review and meta-analysis. BMC Endocr Disord. 2021;21(1):34.
Acknowledgements
The authors acknowledged the vice chancellor of research, Shiraz University of Medical Science, Iran for funding assistance with this project (Grant number: 21260).
Funding
This study was funded and supported by Shiraz University of Medical Sciences.
Author information
Authors and Affiliations
Contributions
MH and MD designed the search strategy, and conducted and re-ran the search. MH and SF conducted the title and full-text screening and performed data extraction on the initial group of studies. SH extracted data on studies identified upon re-running the search, performed the meta-analysis, and risk-of-bias assessment, MH wrote the initial manuscript draft. MD conceptualized the study, supervised SF, MH and SH through all steps of the systematic review, provided content and methodologic support, and revised all versions of the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Shiraz University of Medical Sciences (Ethical code: IR.SUMS.REC. 1399. 867). Ethical issues (Including plagiarism, data fabrication and/or falsification, double publication and/or submission, redundancy, etc.) have been completely observed by the authors. The consent of the participants in this article does not apply.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Supplemental appendix 1.
PRISMA 2020 Checklist.
Additional file 2: Supplemental appendix 2.
Electronic database search strings.
Additional file 3: Supplemental appendix 3
. Studies rejected at full-text review stage.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Hajifoghaha, M., Teshnizi, S.H., Forouhari, S. et al. Association of thyroid function test abnormalities with preeclampsia: a systematic review and meta-analysis. BMC Endocr Disord 22, 240 (2022). https://doi.org/10.1186/s12902-022-01154-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12902-022-01154-9