Abstract
Background
Noninvasive ventilation (NIV) is commonly used in patients with acute respiratory distress syndrome (ARDS). However, the incidence and distribution of treatment failure are unclear.
Methods
A comprehensive online search was conducted to select potentially eligible studies with reports of the rate of NIV failure in patients with ARDS. A manual search was also performed to identify additional studies. Data were extracted to calculate the pooled incidences of NIV failure and mortality. Based on oxygenation, the severity of the disease was classified as mild, moderate, or severe ARDS. Based on etiologies, ARDS was defined as being of pulmonary origin or extrapulmonary origin.
Results
We enrolled 90 studies in this meta-analysis, involving 98 study arms. The pooled incidence of NIV failure was 48% (n = 5847, 95% confidence interval [CI]: 43–52%). The pooled incidence of ICU mortality was 29% (n = 2363, 95%CI: 22–36%), and that of hospital mortality was 33% (n = 2927, 95%CI: 27–40%). In patients with mild, moderate, and severe ARDS, the pooled incidence of NIV failure was 30% (n = 819, 95%CI: 21–39%), 51% (n = 1332, 95%CI: 43–60%), and 71% (n = 525, 95%CI: 62–79%), respectively. In patients with pulmonary ARDS, it was 45% (n = 2687, 95%CI: 39–51%). However, it was 30% (n = 802, 95%CI: 21–38%) in those with extrapulmonary ARDS. In patients with immunosuppression, the incidence of NIV failure was 62% (n = 806, 95%CI: 50–74%). However, it was 46% (n = 5041, 95%CI: 41–50%) in those without immunosuppression.
Conclusions
Nearly half of patients with ARDS experience NIV failure. The incidence of NIV failure increases with increasing ARDS severity. Pulmonary ARDS seems to have a higher rate of NIV failure than extrapulmonary ARDS. ARDS patients with immunosuppression have the highest rate of NIV failure.
Similar content being viewed by others
Introduction
Acute respiratory distress syndrome (ARDS) was first described by Ashbaugh et al. in 1967 [1]. Its features are acute onset of hypoxemia, bilateral opacities not fully explained by effusions, lobar/lung collapse or nodules, respiratory failure not fully explained by cardiac failure or fluid overload, and PaO2/FiO2 less than 300 mmHg [2]. The etiologies of ARDS include pneumonia, pancreatitis, abdominal infection, blood transfusion, and trauma [3, 4]. ARDS is classified as pulmonary or extrapulmonary according to its cause. Based on oxygenation, it can be classified as mild, moderate, or severe [5]. To relieve respiratory distress, respiratory support is commonly used in ARDS patients.
Physiological studies have shown that noninvasive ventilation (NIV) decreases the work of breathing and improves oxygenation in patients with ARDS [6]. In contrast with invasive mechanical ventilation, NIV preserves the ability to swallow, cough, and communicate verbally; avoids intubation-associated complications; and reduces the likelihood of nosocomial pneumonia. Therefore, NIV has been commonly used in patients with ARDS [7]. However, the incidence and distribution of NIV failure in ARDS population are unclear. Here, we report the incidence of NIV failure in ARDS patients and further clarify the distributions of NIV failure in different subgroups.
Methods
This article reports the results of a systematic review and meta-analysis of NIV failure and mortality, focusing on patients with ARDS. It was performed in conformity with Preferred Reporting Items for Systematic Reviews and Meta-analysis statement [8].
Search techniques and selection criteria
We searched PubMed, Web of Science, the Cochrane library, and some Chinese databases (CBM, Wanfang Data, and CNKI), without any language limitation, for pertinent research published before September 30, 2022. We also performed manual searches of the reference lists of the identified articles and pertinent reviews to identify additional relevant articles. The search used the following key words: (“noninvasive ventilation” OR “noninvasive mechanical ventilation” OR “noninvasive positive pressure ventilation” OR “NIV” OR “NPPV” OR “NIPPV” OR “continuous positive airway pressure” OR “CPAP” OR “noninvasive pressure support ventilation” OR “noninvasive oxygen” OR “noninvasive oxygenation” OR “mask ventilation” OR “nasal ventilation” OR “helmet ventilation”) and (“ARDS” OR “acute respiratory distress syndrome” OR “ALI” OR “acute lung injury” OR “acute respiratory failure” OR “acute hypoxemic respiratory failure” OR “hypoxemic respiratory failure”).
Studies were enrolled based on the following inclusion criteria: ARDS or ALI was diagnosed, adult patients were involved, and NIV was used as a first-line intervention. The following works were excluded: reviews, case reports, editorials, letters, and conference abstracts; articles with no available data for NIV failure; studies that used NIV as preoxygenation before intubation; and studies that used NIV as a ventilator weaning strategy.
Data extraction and quality assessment
All studies were independently selected by two investigators (JW and JD). Any discrepancies were resolved by consensus. If the researchers failed to reach consensus, a third investigator (LZ) reviewed the data and made a determination. We extracted the data as follows: first author’s name, country, publication year, proportion of male patients, proportion of immunosuppressed patients, age, PaO2/FiO2, number of total patients, number of patients with ARDS, number of NIV failures in patients with ARDS, number of deaths in patients with ARDS, NIV mode, NIV interface, causes of ARDS, and the proportion of mild, moderate, and severe ARDS. NIV failure and mortality data were also collected.
We intended to report the incidence of treatment failure and mortality in ARDS patients who received NIV; thus, the quality of enrolled studies was assessed using Murad’s tool for non-comparative studies [9, 10]. This method includes five questions: (1) did the patients represent all of the cases seen by the medical center, (2) was the diagnosis correctly made, (3) were other important diagnosis excluded, (4) were all important data cited in the report, and (5) was the outcome correctly ascertained? Each question was assigned 1 (yes) or 0 (no) points. The quality of an enrolled study was classified as high, moderate, or low if the total scores were 5, 4, or ≤ 3, respectively.
Statistical analysis
The data were analyzed using R (version 4.2.2). The Shapiro-Wilk test of normality was used to analyze the distribution of the incidence of NIV failure and mortality among the included studies. Values of p more than 0.05 were taken to indicate that the rate of the relevant event was normally distributed. If it was abnormally distributed, the data were transferred to normally distributed data using logarithmic transformation, sine transformation, or arc sine transformation. I2 was used to describe heterogeneity, where I2 ≥ 50% represents significant heterogeneity. A random-effects model was used to pool the data, and a subgroup analysis was performed to explore potential heterogeneity. If no heterogeneity was observed, a common-effects model (also known as a fixed-effects model) was used.
The mean-pooled incidences of treatment failure and mortality were estimated. The corresponding confidence intervals (95% CIs) were also estimated. Egger’s test was used to assess the possibility of publication bias [11]. A funnel plot (plot of treatment effect against trial precision) was created to visualize publication bias.
Results
Characteristics of the included studies
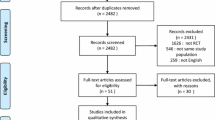
In all, 2219 studies were obtained using the search strategy, and 23 studies were identified from other sources (Fig. 1). After screening the titles and abstracts and reviewing full papers, we enrolled 90 studies involving 98 study arms. These studies were published between 1999 and 2022 (Table 1). Egger’s test (p = 0.69) and a funnel plot showed that there was no publication bias (Supplementary Fig. 1). The quality of most of the studies was high or moderate (Supplementary Fig. 2).
NIV failure and mortality in ARDS patients
In total, 5847 ARDS patients were enrolled in studies reporting NIV failure (Fig. 2). Significant heterogeneity was found between studies (I2 = 95%). Therefore, a random-effects model was selected. The pooled incidence of NIV failure was 48% (95%CI: 43–52%). In all, 2363 ARDS patients were enrolled in studies reporting ICU mortality (Fig. 3). The pooled incidence of ICU mortality was 29% (95%CI: 22–36%). In total, 2927 ARDS patients were enrolled in studies reporting hospital mortality (Fig. 4). The pooled incidence of hospital mortality was 33% (95%CI: 27–40%).
NIV failure in different subgroups
In all, 24, 20, and 17 study arms reported the rate of NIV failure in patients with mild, moderate, and severe ARDS, respectively (Fig. 5). The pooled incidence of NIV failure was 30% (95%CI: 21–39%), 51% (95%CI: 43–60%), and 71% (95%CI: 62–79%) in these ARDS groups, respectively.
There were 46 study arms that involved 2687 patients with pulmonary ARDS that reported NIV failure (Supplementary Fig. 3), and 19 study arms that involved 802 patients with extrapulmonary ARDS that reported NIV failure (Supplementary Fig. 4). The pooled incidence of NIV failure was 45% (95%CI: 39–51%) and 30% (21–38%) in patients with pulmonary and extrapulmonary ARDS, respectively (Fig. 6A). For pulmonary ARDS in particular, the pooled incidence of NIV failure was 42% (95%CI: 31–52%) and 47% (95%CI: 41–54%) in patients with and without COVID-19, respectively (Fig. 6B, supplementary Figs. 5 and 6).
The pooled incidence of NIV failure was 62% (95%CI: 50–74%) and 46% (95%CI: 41–50%) in patients with and without immunosuppression, respectively (Fig. 6C, supplementary Figs. 7 and 8). NIV failure was 49% (95%CI: 44–53%) in observational studies and 34% (95%CI: 16–53%) in randomized control trials (Fig. 6D, supplementary Figs. 9 and 10). In patients who were ventilated using BiPAP, the NIV failure was 48%, the same as in those ventilated via CPAP (Fig. 6E, supplementary Figs. 11 and 12).
The pooled incidence of NIV failure was 44% (95%CI: 32–57%) before 2005 (Fig. 7). Between 2006 and 2010, it was 47% (33–62%). However, this did not change after 2010 when patients with COVID-19 were excluded (52% for 2011–2015, 49% for 2016–2020, and 52% for 2021 and 2022).
Discussion
This meta-analysis shows that the incidence of NIV failure was high in patients with ARDS. A gradual increase in NIV failure was found in ARDS patients with an increase in disease severity. Patients with pulmonary ARDS had a higher rate of NIV failure than those with extrapulmonary ARDS. ARDS patients with immunosuppression had the highest failure rate.
An international, multicenter, prospective cohort study reported that 15.5% of patients with ARDS had received NIV [6]. This indicates that the use of NIV in patients with ARDS is not rare. However, ARDS is a risk factor for NIV failure in patients who have acute respiratory failure [12,13,14]. In our analyses, nearly half of the ARDS patients experienced NIV failure. ARDS patients commonly developed excessive inspiratory effort and high transpulmonary pressures; this can lead the patient to self-inflicted lung injury (P-SILI) [15, 16]. Therefore, P-SILI greatly contributes to the high rate of NIV failure in ARDS patients. Esophageal pressure monitoring can detect the risk for P-SILI, which can help identify NIV failure early [17].
This meta-analysis shows that pulmonary ARDS leads to a higher rate of NIV failure than extrapulmonary ARDS, in line with a previous study [18]. Pulmonary ARDS is mainly caused by pneumonia, and extrapulmonary ARDS is mainly caused by sepsis [19, 20]. In patients with pulmonary ARDS, consolidation in chest CT scans is greater, and the response to the lung recruitment maneuver is worse than that in patients with extrapulmonary ARDS [21, 22]. This may be the reason for the lower NIV failure rate in patients with extrapulmonary ARDS. However, our study was a meta-analysis and the demographic information between two groups could not be comparable. Studies are required to further clarify this issue.
It is challenging to avoid intubation in immunocompromised patients with acute respiratory failure [23]. Patients with immunosuppression were more likely to receive NIV as a first-line therapy [24]. Relative to conventional oxygen therapy, use of NIV reduces the rate of intubation in patients with immunosuppression [25]. However, in our analyses, the pooled incidence of NIV failure in the immunocompromised group was 62%, the highest of all subgroups. Patients who experienced NIV failure had a higher likelihood of death in hospital than those who directly received intubation [26]. Therefore, the early identification of high-risk patients followed by the early application of intubation would be an alternative solution to reduce mortality.
This study had several limitations. First, there were various definitions of NIV failure among the studies considered. Some studies defined it as intubation, whereas others defined it as intubation, death, or crossover to a high-flow nasal cannula. However, because it was commonly defined as intubation, the incidence of NIV failure may have been overestimated. Second, only four study arms, involving 133 patients, could be used to pool the incidence of helmet NIV failure. As helmet NIV shows much more favorable outcomes than oronasal NIV, the pooled incidence of helmet NIV failure may be overestimated [27]. Third, the incidence of NIV failure was high in the ARDS population. This does not imply that patients with ARDS cannot obtain benefits from NIV. In the future, randomized controlled trials should be performed to further investigate this issue.
Conclusions
Nearly half of ARDS patients experience NIV failure. With increasing ARDS severity, the pooled incidence of NIV failure increased. Patients with pulmonary ARDS seem to experience more NIV failure than those with extrapulmonary ARDS. ARDS patients with immunosuppression may be at highest risk for NIV failure.
Data availability
The datasets analyzed during current study are available from the corresponding author on reasonable request.
Abbreviations
- NA:
-
Not available
- NIV:
-
Noninvasive ventilation
- BiPAP:
-
Bi-level positive airway pressure
- CPAP:
-
Continuous positive airway pressure
- ARDS:
-
Acute respiratory distress syndrome
- CI:
-
Confidence interval
- P-SILI:
-
Patient self-inflicted lung injury
References
Ashbaugh DG, Bigelow DB, Petty TL, et al. Acute respiratory distress in adults. Lancet. 1967;2:319–23.
Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA. 2012;307:2526–33.
Rocco PR, Zin WA. Pulmonary and extrapulmonary acute respiratory distress syndrome: are they different? Curr Opin Crit Care. 2005;11:10–7.
Huang X, Zhang R, Fan G, et al. Incidence and outcomes of acute respiratory distress syndrome in intensive care units of mainland China: a multicentre prospective longitudinal study. Crit Care. 2020;24:515.
Fan E, Del Sorbo L, Goligher EC, et al. An official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: mechanical ventilation in adult patients with Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med. 2017;195:1253–63.
L’Her E, Deye N, Lellouche F, et al. Physiologic effects of noninvasive ventilation during acute lung injury. Am J Respir Crit Care Med. 2005;172:1112–8.
Bellani G, Laffey JG, Pham T, et al. Noninvasive ventilation of patients with Acute Respiratory Distress Syndrome. Insights from the LUNG SAFE study. Am J Respir Crit Care Med. 2017;195:67–77.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700.
Murad MH, Sultan S, Haffar S, et al. Methodological quality and synthesis of case series and case reports. BMJ Evid Based Med. 2018;23:60–3.
Haffar S, Bazerbachi F, Prokop L, et al. Frequency and prognosis of acute Pancreatitis associated with fulminant or non-fulminant acute hepatitis A: a systematic review. Pancreatology. 2017;17:166–75.
Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Ozyilmaz E, Ugurlu AO, Nava S. Timing of noninvasive ventilation failure: causes, risk factors, and potential remedies. BMC Pulm Med. 2014;14:19.
Duan J, Chen L, Liu X, et al. An updated HACOR score for predicting the failure of noninvasive ventilation: a multicenter prospective observational study. Crit Care. 2022;26:196.
Antonelli M, Conti G, Moro ML, et al. Predictors of failure of noninvasive positive pressure ventilation in patients with acute hypoxemic Respiratory Failure: a multi-center study. Intensive Care Med. 2001;27:1718–28.
Grieco DL, Menga LS, Eleuteri D, et al. Patient self-inflicted lung injury: implications for acute hypoxemic Respiratory Failure and ARDS patients on non-invasive support. Minerva Anestesiol. 2019;85:1014–23.
Sklienka P, Frelich M, Burša F. Patient self-inflicted Lung Injury-A Narrative Review of Pathophysiology, Early Recognition, and Management options. J Pers Med 2023;13.
Tonelli R, Fantini R, Tabbì L, et al. Early Inspiratory Effort Assessment by Esophageal Manometry predicts noninvasive ventilation outcome in De Novo Respiratory Failure. A pilot study. Am J Respir Crit Care Med. 2020;202:558–67.
Shu W, Guo S, Yang F, et al. Association between ARDS Etiology and risk of noninvasive ventilation failure. Ann Am Thorac Soc. 2022;19:255–63.
Ruan SY, Huang CT, Chien YC, et al. Etiology-associated heterogeneity in acute respiratory distress syndrome: a retrospective cohort study. BMC Pulm Med. 2021;21:183.
Kallet RH, Zhuo H, Ho K, et al. Lung Injury etiology and other factors influencing the Relationship between Dead-Space Fraction and Mortality in ARDS. Respir Care. 2017;62:1241–8.
Goodman LR, Fumagalli R, Tagliabue P, et al. Adult respiratory distress syndrome due to pulmonary and extrapulmonary causes: CT, clinical, and functional correlations. Radiology. 1999;213:545–52.
Lim CM, Jung H, Koh Y, et al. Effect of alveolar recruitment maneuver in early acute respiratory distress syndrome according to antiderecruitment strategy, etiological category of diffuse lung injury, and body position of the patient. Crit Care Med. 2003;31:411–8.
De Jong A, Calvet L, Lemiale V, et al. The challenge of avoiding intubation in immunocompromised patients with acute Respiratory Failure. Expert Rev Respir Med. 2018;12:867–80.
Cortegiani A, Madotto F, Gregoretti C, et al. Immunocompromised patients with acute respiratory distress syndrome: secondary analysis of the LUNG SAFE database. Crit Care. 2018;22:157.
Zayed Y, Banifadel M, Barbarawi M, et al. Noninvasive oxygenation strategies in immunocompromised patients with Acute Hypoxemic Respiratory Failure: a pairwise and Network Meta-Analysis of Randomized controlled trials. J Intensive Care Med. 2020;35:1216–25.
Azoulay E, Pickkers P, Soares M, et al. Acute hypoxemic Respiratory Failure in immunocompromised patients: the Efraim multinational prospective cohort study. Intensive Care Med. 2017;43:1808–19.
Patel BK, Wolfe KS, Pohlman AS, et al. Effect of Noninvasive Ventilation delivered by Helmet vs Face Mask on the rate of endotracheal intubation in patients with Acute Respiratory Distress Syndrome: a Randomized Clinical Trial. JAMA. 2016;315:2435–41.
Funding
This study was supported by Chongqing medical scientific research project (Joint project of Chongqing Health Commission and Science and Technology Bureau) 2023MSXM067.
Author information
Authors and Affiliations
Contributions
JD and LZ contributed to the conception and design of the study. JW, JD and LZ contributed to literature research, article selection and data extraction. JD and LZ joined in data analysis and interpretation. All authors joined in manuscript preparation and revision, and approved the final version.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
: Supplementary material 1. Enrolled studies for meta-analysis. Supplementary Fig. 1. Funnel plot of the proportion of NIV failure. Supplementary Fig. 2. Quality assessment of enrolled studies. Supplementary Fig. 3. Pooled incidence of NIV failure in patients with pulmonary ARDS. Supplementary Fig. 4. Pooled incidence of NIV failure in patients with extra-pulmonary ARDS. Supplementary Fig. 5. Pooled incidence of NIV failure in pulmonary ARDS patients with COVID-19. Supplementary Fig. 6. Pooled incidence of NIV failure in pulmonary ARDS patients without COVID-19. Supplementary Fig. 7. Pooled incidence of NIV failure in patients with immunosuppression. Supplementary Fig. 8. Pooled incidence of NIV failure in patients without immunosuppression. Supplementary Fig. 9. Pooled incidence of NIV failure in observational studies. Supplementary Fig. 10. Pooled incidence of NIV failure in randomized control trials. Supplementary Fig. 11. Pooled incidence of NIV failure in patients who used BiPAP. Supplementary Fig. 12. Pooled incidence of NIV failure in patients who used CPAP. Supplementary Fig. 13. Pooled incidence of NIV failure in patients who used an oronasal mask. Supplementary Fig. 14. Pooled incidence of NIV failure in patients who used a helmet.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, J., Duan, J. & Zhou, L. Incidence of noninvasive ventilation failure and mortality in patients with acute respiratory distress syndrome: a systematic review and proportion meta-analysis. BMC Pulm Med 24, 48 (2024). https://doi.org/10.1186/s12890-024-02839-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12890-024-02839-8