Abstract
Background
The aim of the study was to determine the incidence and risk factors of retinopathy of prematurity (ROP) in premature, extremely low birth weight (BW, ELBW) and extremely low gestational age (GA, ELGA) infants.
Methods
The medical records of preterm infants who were screened for ROP between January 2012 and December 2020 were retrospectively reviewed. Only one eye of each infant with higher grade ROP was included in the study. BW; GA; medical characteristics; the presence, severity, and need for treatment of ROP were recorded. Infants were divided into groups according to BW (≤1000 g, 1001-1750 g, > 1750 g) and GA (≤25w, 26-28w, 29-31w, 32-34w, ≥35w) and data were analyzed.
Results
Data of 2186 infants were evaluated. The overall incidences of any stage ROP and ROP requiring treatment were 43.5 and 8.0%, respectively. These rates were 81.1 and 23.9% in ELBW (≤1000 g) infants and were 92.9 and 64.3% in ELGA (≤25w) infants, respectively. The rates of ROP, the median duration of oxygen therapy and systemic diseases increased significantly as BW and GA decreased. The median duration of oxygen therapy and the rates of sepsis, pulmonary dysplasia (BPD), and intraventricular hemorrhage (IVH) were statistically higher in infants with ROP compared to those without ROP (p < 0.001). Multivariate regression analysis demonstrated that low BW and GA; prolonged duration of oxygen therapy; presence of PDA and necrotizing enterocolitis (NEC) were important risk factors for ROP.
Conclusions
ELBW and ELGA infants develop higher rates of ROP and severe ROP. Prolonged duration of oxygen therapy, the presence of concomitant neonatal sepsis, BPD, IVH, PDA, and NEC further increases the risk of ROP.
Similar content being viewed by others
Introduction
The survival rates of very premature and low birth weight (BW) infants who are at risk of mortality and morbidity, including retinopathy of prematurity (ROP), have increased with advances in neonatal intensive care units [1, 2]. ROP is a leading cause of childhood blindness [3]. The incidence of ROP varies from country to country [4,5,6,7,8,9]. Several risk factors such as low BW [4,5,6, 8,9,10], low gestational age (GA) [4,5,6, 8,9,10], high oxygen saturation [11], mechanical ventilation [5, 6], phototherapy [8], intraventricular hemorrhage (IVH) [6, 7], anemia [6], blood transfusion [4, 6, 8, 9], patent ductus arteriosus (PDA) [6, 7, 9], respiratory distress syndrome (RDS) [6, 9], broncho pulmonary dysplasia (BPD) [6], necrotizing enterocolitis (NEC) [5, 6, 9], sepsis [4, 9], low Apgar scores [6] have been associated with the development of ROP.
Some studies have been published to determine the incidence and risk factors of ROP in extremely low BW (ELBW) infants [10, 12,13,14]. We planned this study to determine the incidence and risk factors of ROP in premature, ELBW and extremely low GA (ELGA) infants followed in our hospital, which is a tertiary referral and treatment center.
Materials and methods
Study design
This retrospective study was conducted at the Ondokuz Mayis University Hospital. The medical records of preterm infants who were screened for ROP between January 2012 and December 2020 were reviewed.
Subjects
Premature infants with a BW of ≤1700 g (g) and a GA of < 34 weeks (w), and with a BW of > 1700 g or a GA of ≥34 w who have received cardiopulmonary support therapy or were considered at risk for the development of ROP by the neonatologists were included in the study [15]. Premature infants who did not complete all screening sessions and had missing data were excluded from the study. This study was approved by the Ondokuz Mayis University Clinical Research Ethics Committee and was performed according to the principles of Declaration of Helsinki. Informed consent was obtained from the parents before the ophthalmological examination for ROP.
Data collection
ROP examinations were performed by two ophthalmologists (OEY and LN) experienced in ROP. BW, GA, multiple gestations, medical histories, the presence and severity of ROP, the need and type of treatment for ROP were recorded. ROP examination included assessment of pupillary dilation; vitreous clarity; presence of plus disease; presence, stage, location, and extent of ROP disease. ROP was classified according to the international classification of retinopathy [16]. ROP type 1 was defined as any stage ROP with plus disease (a degree of dilation and tortuosity of the posterior retinal blood vessels) at zone I, stage 3 ROP without plus disease at zone I, and stage 2 or 3 ROP with plus disease at zone II. ROP type 2 was defined as stage 1 or 2 ROP without plus disease at zone I and stage 3 ROP without plus disease at zone II [17]. ROP cases except types 1 and 2 were defined as mild ROP. Premature infants divided into groups according to BW (≤1000 g, 1001–1750 g, > 1750 g) and GA (≤25 w, 26–28 w, 29–31 w, 32–34 w, ≥35 w) and data were analyzed. Infants with a BW of ≤1000 g were considered as ELBW infants, and infants with a GA of ≤25 w were considered as ELGA infants [18, 19]. Data from only one eye of each infant with higher grade ROP were evaluated.
Statistical analysis
Statistical analysis was performed using V22 IBM SPSS (SPSS Inc., Chicago, IL, USA). The compliance of the data to normal distribution was evaluated with the Kolmogorov Smirnov test. Non-parametric tests were used because the data are not normally distributed. The Kruskal-Wallis and Mann-Whitney U tests were used for continuous independent variables and Chi-square test used for categorical variables. The results were given as median (minimum-maximum) and frequency (%). Binary logistic regression analysis was performed to determine the factors associated with ROP development. P values of < 0.05 were considered to be statistically significant.
Results
General characteristics of the patients
Two thousand one hundred and eighty-six premature infants with a median GA of 31 (22–38) w and a mean BW of 1500 (540–3450) g were included in the study. There were 347 ELBW infants with a median BW of 800 (540–1000) g and 28 ELGA infants with a median GA of 24 (22–25) w. Of the cases, 1075 (49.2%) were girls and 1111 (50.8%) were boys; 1571 (71.9%) were single, 529 (24.2%) were twins, and 86 (3.9%) were triplets. The distributions of BW and GA, and characteristics of infants in the groups are given in Table 1.
ROP rates
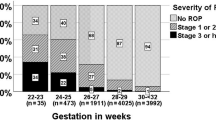
The number of infants with any stage of ROP was 952 (43.5%). The rates of mild ROP, type 2 ROP and type 1 ROP in infants was 751/2186 (34.4%), 51/2186 (2.3%) and 150/2186 (6.9%), respectively. The rate of ROP requiring treatment was 175/2186 (8.0%). Retinal laser diode photocoagulation was applied to 141 (80.6%) and intravitreal anti-VEGF (bevacizumab) injection was applied to 34 (19.4%) of 175 infants with ROP who needed treatment. ELBW infants had the highest rates of ROP (81%), severe (type 1) ROP (20.5%), and ROP requiring treatment (23.9%) compared to other BW groups (p < 0.001). In addition, ELGA infants had the highest rates of ROP (92.9%), severe (type 1) ROP (57.1%), and ROP requiring treatment (64.3%) compared to other GA groups (p < 0.001). ROP rates in BW and GA groups are presented in Table 2.
Risk factors
The incidence of ROP increased statistically as BW (p < 0.001) and GA (p < 0.001) decreased (Table 2). The incidence of ROP requiring treatment also increased statistically with decrease of BW (p = 0.002) and GA (p = 0.001) (Table 2). The median BW (1300 vs 1600 g; p < 0.001) and GA (30 vs 32 w; p < 0.001) were statistically lower in infants with ROP compared to those without ROP (Table 3).
The median duration of oxygen therapy increased statistically as BW (p < 0.001) and GA (p < 0.001) decreased (Table 1). The median duration of oxygen therapy (39 vs 20 days; p < 0.001) was longer in infants with ROP compared to those without ROP (Table 3).
The incidence of sepsis, BPD, and IVH (≥Grade II) increased statistically as BW (p < 0.001) and GA (p < 0.001) decreased (Table 1). The rates of sepsis, BPD, and IVH were statistically higher in infants with ROP compared to those without ROP (p < 0.001) (Table 3). While the NEC rate was higher in ELBW infants (p = 0.028), it did not differ according to GA and presence of ROP (Tables 1 and 3). While the rate of PDA was higher in both ELBW (p = 0.027) and ELGA (p = 0.016) infants, no difference was observed according to the presence of ROP (Tables 1 and 3).
Multivariate logistic regression analysis
Compared to BW of > 1750 g, BW of ≤1000 g [OR (95% CI): 2.8 (1.8–4.4), p < 0.001] and BW of 1001–1750 g [1.6 (1.3–2.2), p < 0.001] were significant risk factors for ROP. Compared to GA of ≥35 w, GA of ≤25 w [66.5 (10.2–435.3), p < 0.001], GA of 26–28 w [34.7 (11.6–103.9), p < 0.001], GA of 29–31 w [10.5 (3.7–29.9), p < 0.001], and GA of 32–34 w [6.4 (2.3–18.4), p < 0.001] were significant risk factors for ROP. Additionally, increase in duration of oxygen therapy [1.03 (1.03–1.04), p < 0.001], presence of PDA [1.7 (1.2–2.4), p = 0.006], and presence of NEC [2.3 (1.2–4.1), p = 0.010] were determined as important risk factors for ROP.
Discussion
Premature, especially ELBW and ELGA infants are known to have increased systemic morbidities, including ROP [2, 20, 21]. This study revealed that the rate of any stage ROP was 81.1% and the rate of ROP requiring treatment was 23.9% in ELBW infants. In the Early Treatment for Retinopathy of Prematurity (ETROP) study, the incidences of ROP were reported as 92.7 and 75.8%, respectively, in infants with a BW < 750 g and 750–1000 g. In addition, the incidences of prethreshold or worse ROP were reported as 51.9 and 31.7% in infants with a BW < 750 g and 750–1000 g, respectively [20]. While the rate of any stage ROP in ELBW infants was higher than in our previous report (81% vs 70.7%), the rate of severe ROP requiring treatment decreased (23.9% vs 30.2%) [13]. In another previous report, in which we compared two consecutive 5-year periods, we found that although the incidence of any stage ROP increased, the incidence of ROP requiring treatment decreased [22]. This contrast situation may be explained by the improvement in neonatal care practices despite the fact that younger babies are now being kept alive. In the TR-ROP study, which is a multicenter study conducted in our country, the rate of any stage ROP (68%) in ELBW infants was lower than in the current study, while the rate of ROP requiring treatment (20.8%) was similar [4]. A recent meta-analysis reported that the rates of any stage ROP, severe ROP and ROP requiring treatment in infants with a BW < 1000 g were 49, 24 and 18%, respectively [21]. The rates reported by this meta-analysis, which included 192 studies, are lower than those reported in our country [21]. The incidence of ROP in ELBW infants from different countries is given in Table 4. In the current study, in infants with a GA of ≤25 w, the rates of any stage ROP and ROP requiring treatment were 100, and 66.7%, respectively. Although there are not many studies on the incidence of ROP in ELGA infants, high rates have been reported from different countries, generally similar to the results of this study (Table 4). We think that the number of the infants with a GA of ≤25 w was small in the current study and that further studies are needed with a larger number of ELGA infants.
Lower GA and BW are major risk factors for the development of ROP. In the current study, it was found that lower GA and BW were independent risk factors for the development of ROP, and any stage ROP and severe ROP requiring treatment mostly developed in ELBW and ELGA infants. Logistic regression analysis confirmed that reductions in both GA and BW, more pronounced in GA, are important risk factors for the development of ROP. Studies from different countries have been published reporting that both lower GA and BW are significant risk factors for ROP with multivariate logistic regression analysis [4, 6, 8]. In addition, Li et al. and Bortea et al. determined only low BW as a risk factor for ROP [9, 23]. Yau et al. and Fortes Filho et al. reported that small GA was identified as a risk factor for ROP in ELBW infants [10, 12]. Zarei et al. also reported that more advanced stages of ROP were observed in extreme prematurity and ELBW infants, and GA was a risk factor for ROP in ELBW infants on multivariate analysis [24].
Increased duration of oxygen therapy was another risk factor for ROP. It has been reported in previous studies that supplementary oxygen administration and total days of oxygen are independent risk factors for ROP [4, 6, 8, 10]. Increased oxygen duration is a risk factor for ROP also confirmed by multivariate regression analysis in the current study. After birth, the encounter of the partially vascularized retina with hyperoxia leads to cessation of angiogenesis. Later, pathological neovascularization develops in the retina, which remains relatively hypoxic due to delayed vascularization relative to neuronal maturation [32]. Hyperoxia is a risk factor for eye, lung and cerebral injury in premature infants. In infants with GA of < 28 w, low saturation targets were associated with more deaths and more NEC, higher saturation targets (91–95%) were associated with more ROP. Despite this, it is recommended to target SpO2 at between 90 and 95% to reduce mortality in these infants [11, 33]. Stoll et al. reported that the rates of ROP decreased in infants with GA of 25–28 w, and this decrease might be related to oxygen saturation targets or to improved adherence to oxygen targets [1].
Some comorbidities may be risk factors for ROP. BPD is a lung disease that is defined as supplemental oxygen dependence beyond 28 days postpartum and results in prolonged oxygen exposure and is therefore associated with ROP. In our study BPD was found to be higher as BW and GA decreased, as well as in infants with ROP. Similar to some studies in the literature, higher rate of BPD was found in infants with ROP in our study [4, 6, 25, 34]. Yau et al. reported that BPD was identified as a risk factor for ROP in ELBW infants [10]. IVH was found to be higher in both ELBW and ELGA infants and infants with ROP. Although the association of IVH with ROP and severe ROP has been reported, the reason for the association is not known exactly [35]. In the current study, PDA was the most prevalent in ELBW and ELGA infants. While it could not be detected in the univariate analysis, the presence of PDA was found to be a risk factor for the development of ROP in the multivariate analysis. Several studies have reported that PDA or ligation of PDA is a risk factor for ROP [4, 6, 9, 25, 36]. Retinal hypoxia as a result of reduced perfusion in infants with PDA may lead to the development or progression of ROP. Weis et al. reported that while mortality was lower there was no difference in terms of ROP in babies with PDA who underwent ligation [37]. The frequency of NEC increased only with decreasing BW and was identified as a risk factor for the development of ROP by multivariate regression analysis. Li et al. and Kumar et al. reported an association between NEC and ROP by univariate regression analysis, but this association could not be demonstrated by multivariate regression analysis [9, 38]. Isaza et al. also reported that NEC is an independent risk factor for ROP [5]. Neonatal sepsis is one of the risk factors most commonly associated with the development of ROP [35]. Sepsis has also been identified as a risk factor for ROP in very low BW and ELBW infants [14, 25, 36]. Sepsis was found to be higher in both ELBW and ELGA infants and infants with ROP in the current study. Although its role in the pathogenesis is not fully elucidated, neonatal infection and perinatal inflammation are thought to play a role in the development of ROP [39].
Conclusions
Currently, ELBW and ELGA infants with increasing chances of survival have a high rate of developing ROP and severe ROP. In these infants, prolonged duration of supplemental oxygen therapy and increased systemic comorbidities increase the risk of ROP. Consequently, the lower the BW and GA, and the longer the exposure to supplemental oxygen, the greater the risk of developing any ROP and severe ROP. The presence of concomitant neonatal sepsis, BPD, IVH (≥Grade II), PDA, and NEC further increases the risk of ROP. Infants with these risk factors should be followed very closely for the development of ROP, and their families should be informed about risks.
Availability of data and materials
All data generated or analysed during this study are included in this published article.
Abbreviations
- ROP:
-
Retinopathy of prematurity
- BW:
-
Birth weight
- GA:
-
Gestational age
- IVH:
-
Intraventricular hemorrhage
- PDA:
-
Patent ductus arteriosus
- RDS:
-
Respiratory distress syndrome
- BPD:
-
Broncho pulmonary dysplasia
- NEC:
-
Necrotizing enterocolitis
- ELBW:
-
Extremely low birth weight
- ELGA:
-
Extremely low gestational age
- g:
-
Grams
- w:
-
Weeks
References
Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, Laptook AR, Sánchez PJ, Van Meurs KP, Wyckoff M, Das A, Hale EC, Ball MB, Newman NS, Schibler K, Poindexter BB, Kennedy KA, Cotten CM, Watterberg KL, D'Angio CT, DeMauro SB, Truog WE, Devaskar U, Higgins RD; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network (2015) Trends in Care Practices, Morbidity, and Mortality of Extremely Preterm Neonates, 1993-2012. JAMA. 314(10):1039–1051. https://doi.org/10.1001/jama.2015.10244.
Mukhopadhyay K, Louis D, Mahajan R, Kumar P. Predictors of mortality and major morbidities in extremely low birth weight neonates. Indian Pediatr. 2013;50(12):1119–23. https://doi.org/10.1007/s13312-013-0305-8.
Blencowe H, Lawn JE, Vazquez T, Fielder A, Gibert C. Preterm-associated visual impairment and estimates of retinopathy of prematurity at regional and global levels for 2010. Pediatr Res. 2013;74:35–49. https://doi.org/10.1038/pr.2013.205.
Bas AY, Demirel N, Koc E, Ulubas Isik D, Hirfanoglu İM, Tunc T, et al. Incidence, risk factors and severity of retinopathy of prematurity in Turkey (TR-ROP study): a prospective, multicentre study in 69 neonatal intensive care units. Br J Ophthalmol. 2018;102(12):1711–6 https://doi.org/10.1136/bjophthalmol-2017-311789.
Isaza G, Arora S, Bal M, Chaudhary V. Incidence of retinopathy of prematurity and risk factors among premature infants at a neonatal intensive care unit in Canada. J Pediatr Ophthalmol Strabismus. 2013;50(1):27–32. https://doi.org/10.3928/01913913-20121127-02.
Yau GS, Lee JW, Tam VT, Liu CC, Yip S, Cheng E, et al. Incidence and risk factors of retinopathy of prematurity from 2 neonatal intensive care units in a Hong Kong Chinese population. Asia Pac J Ophthalmol (Phila). 2016;5(3):185–91. https://doi.org/10.1097/APO.0000000000000167.
Waheeb S, Alshehri K. Incidence of retinopathy of prematurity at two tertiary centers in Jeddah, Saudi Arabia. Saudi J Ophthalmol. 2016;30(2):109–12. https://doi.org/10.1016/j.sjopt.2016.02.006.
Alizadeh Y, Zarkesh M, Moghadam RS, Esfandiarpour B, Behboudi H, Karambin MM, et al. Incidence and Risk Factors for Retinopathy of Prematurity in North of Iran. J Ophthalmic Vis Res. 2015;10(4):424–8. https://doi.org/10.4103/2008-322X.176907.
Li ML, Hsu SM, Chang YS, Shih MH, Lin YC, Lin CH, et al. Retinopathy of prematurity in southern Taiwan: a 10-year tertiary medical center study. J Formos Med Assoc. 2013;112(8):445–53. https://doi.org/10.1016/j.jfma.2012.03.002.
Yau GS, Lee JW, Tam VT, Liu CC, Chu BC, Yuen CY. Incidence and risk factors for retinopathy of prematurity in extreme low birth weight Chinese infants. Int Ophthalmol. 2015;35:365–73. https://doi.org/10.1007/s10792-014-9956-2.
Saugstad OD, Aune D. Optimal oxygenation of extremely low birth weight infants: a meta-analysis and systematic review of the oxygen saturation target studies. Neonatology. 2014;105(1):55–63. https://doi.org/10.1159/000356561.
Fortes Filho JB, Borges Fortes BG, Tartarella MB, Renato Soibelmann Procianoy RS. Incidence and main risk factors for severe retinopathy of prematurity in infants weighing less than 1000 grams in Brazil. J Trop Pediatr. 2013;59(6):502–6. https://doi.org/10.1093/tropej/fmt036.
Demir S, Sayin O, Aygun C, Ortak H, Yucel OE, Kuçukoduk S, et al. Retinopathy of prematurity in extremely low birth weight infants in Turkey. J Pediatr Ophthalmol Strabismus. 2013;50:229–33. https://doi.org/10.3928/01913913-20130319-03.
Celebi AR, Petricli IS, Hekimoglu E, Demirel N, Bas AY. The incidence and risk factors of severe retinopathy of prematurity in extremely low birth weight infants in Turkey. Med Sci Monit. 2014;20:1647–53 https://doi.org/10.12659/MSM.892262.
Turkish Neonatology Association: Diagnosis and Treatment Guidelines. Turkish Retinopathy of Prematurity Guideline 2021 Update. http://wwwneonatologyorgtr (2021) Accessed Jan 2022.
International Committee for the Classification of Retinopathy of Prematurity. The international classification of retinopathy of prematurity revisited. Arch Ophthalmol. 2005;123(7):991–9 https://doi.org/10.1001/archopht.123.7.991.
Good WV; Early Treatment for Retinopathy of Prematurity Cooperative Group. Final results of the early treatment for retinopathy of prematurity (ETROP) randomized trial. Trans Am Ophthalmol Soc. 2004;102:233–50.
Tyson JE, Parikh NA, Langer J, Green C, Higgins RD; National Institute of Child Health and Human Development Neonatal Research Network. Intensive care for extreme prematurity-moving beyond gestational age. N Engl J Med. 2008;358(16):1672–81. https://doi.org/10.1056/NEJMoa073059.
Yeo KT, Safi N, Wang YA, Marsney RL, Schindler T, Bolisetty S, et al. Prediction of outcomes of extremely low gestational age newborns in Australia and New Zealand. BMJ Paediatr Open. 2017;1(1):e000205. https://doi.org/10.1136/bmjpo-2017-000205.
Good WV, Hardy RJ, Dobson V, Palmer EA, Phelps DL, Quintos M, Tung B; Early Treatment for Retinopathy of Prematurity Cooperative Group. The incidence and course of retinopathy of prematurity: findings from the early treatment for retinopathy of prematurity study. Pediatrics. 2005;116:15–23. https://doi.org/10.1542/peds.2004-1413.
Ramaswamy VV, Abiramalatha T, Bandyopadhyay T, Shaik NB, Bandiya P, Nanda D, et al. ELBW and ELGAN outcomes in developing nations-systematic review and meta-analysis. PLoS One. 2021;16(8):e0255352. https://doi.org/10.1371/journal.pone.0255352.
Demir S, Yücel ÖE, Niyaz L, Karakuş G, Arıtürk N. Incidence of retinopathy of prematurity in the middle Black Sea region of Turkey over a 10-year period. J AAPOS. 2015;19(1):12–5. https://doi.org/10.1016/j.jaapos.2014.09.017.
Bortea CI, Stoica F, Boia M, Iacob ER, Dinu M, Iacob R, et al. Risk factors associated with retinopathy of prematurity in very and extremely preterm infants. Medicina (Kaunas). 2021;57(5):420 https://doi.org/10.3390/medicina57050420.
Zarei M, Bazvand F, Ebrahimiadib N, Roohipoor R, Karkhaneh R, Farahani Dastjani A, Imani Fouladi M, Riazi Esfahani M, Khodabande A, Davoudi S, Ghasemi H, S Modjtahedi B. Prevalence and risk factors of retinopathy of prematurity in Iran. J Ophthalmic Vis Res. 2019;14(3):291–298. https://doi.org/10.18502/jovr.v14i3.4785.
Wu T, Zhang L, Tong Y, Qu Y, Xia B, Mu D. Retinopathy of prematurity among very low-birth-weight infants in China: incidence and perinatal risk factors. Invest Ophthalmol Vis Sci. 2018;59(2):757–63. https://doi.org/10.1167/iovs.17-23158.
Fortes Filho JB, Eckert GU, Procianoy L, Barros CK, Procianoy RS. Incidence and risk factors for retinopathy of prematurity in very low and in extremely low birth weight infants in a unit-based approach in southern Brazil. Eye (Lond). 2009;23(1):25–30 https://doi.org/10.1038/sj.eye.6702924.
Li Q, Wang Z, Wang R, Tang H, Chen H, Feng Z. A prospective study of the incidence of retinopathy of prematurity in China: evaluation of different screening criteria. J Ophthalmol. 2016;2016:5918736. https://doi.org/10.1155/2016/5918736.
Isaza G, Arora S. Incidence and severity of retinopathy of prematurity in extremely premature infants. Can J Ophthalmol. 2012;47:296–300 https://doi.org/10.1016/j.jcjo.2012.03.027.
Kong M, Shin DH, Kim SJ, Ham DI, Kang SW, Chang YS, et al. Retinopathy of prematurity in infants born before 25 weeks gestation in a Korean single neonatal intensive care unit: incidence, natural history and risk factors. J Korean Med Sci. 2012;27(12):1556–62 https://doi.org/10.3346/jkms.2012.27.12.1556.
Gunn DJ, Cartwright DW, Yuen SA, Gole GA. Treatment of retinopathy of prematurity in extremely premature infants over an 18-year period. Clin Exp Ophthalmol. 2013;41:159–66 https://doi.org/10.1111/j.1442-9071.2012.02839.x.
Aikawa H, Noro M. Low incidence of sight-threatening retinopathy of prematurity in infants born before 28 weeks gestation at a neonatal intensive care unit in Japan. Tohoku J Exp Med. 2013;230(3):185–90. https://doi.org/10.1620/tjem.230.185.
Chan-Ling T, Gole GA, Quinn GE, Adamson SJ, Darlow BA. Pathophysiology, screening and treatment of ROP: a multi-disciplinary perspective. Prog Retin Eye Res. 2018;62:77–119. https://doi.org/10.1016/j.preteyeres.2017.09.002.
Carlo WA, Finer NN, Walsh MC, Rich W, Gantz MG, Laptook AR, Yoder BA, Faix RG, Das A, Poole WK, Schibler K, Newman NS, Ambalavanan N, Frantz ID 3rd, Piazza AJ, Sánchez PJ, Morris BH, Laroia N, Phelps DL, Poindexter BB, Cotten CM, Van Meurs KP, Duara S, Narendran V, Sood BG, O'Shea TM, Bell EF, Ehrenkranz RA, Watterberg KL, Higgins RD.; SUPPORT Study Group of the Eunice Kennedy Shriver NICHD Neonatal Research Network. Target ranges of oxygen saturation in extremely preterm infants. N Engl J Med. 2010;362(21):1959–1969. https://doi.org/10.1056/NEJMoa0911781.
Gebeşçe A, Uslu H, Keleş E, Yildirim A, Gürler B, Yazgan H, et al. Retinopathy of prematurity: incidence, risk factors, and evaluation of screening criteria. Turk J Med Sci. 2016;46(2):315–20. https://doi.org/10.3906/sag-1407-127.
Kim SJ, Port AD, Swan R, Campbell JP, Chan RVP, Chiang MF. Retinopathy of prematurity: a review of risk factors and their clinical significance. Surv Ophthalmol. 2018;63(5):618–37. https://doi.org/10.1016/j.survophthal.2018.04.002.
Hwang JH, Lee EH, Kim EA. Retinopathy of prematurity among very-low-birth-weight infants in Korea: incidence, treatment, and risk factors. J Korean Med Sci. 2015;30(Suppl 1):S88–94. https://doi.org/10.3346/jkms.2015.30.S1.S88.
Weisz DE, Mirea L, Rosenberg E, Jang M, Ly L, Church PT, et al. Association of Patent Ductus Arteriosus Ligation with Death or neurodevelopmental impairment among extremely preterm infants. JAMA Pediatr. 2017;171(5):443–9. https://doi.org/10.1001/jamapediatrics.2016.5143.
Kumar P, Sankar MJ, Deorari A, Azad R, Chandra P, Agarwal R, et al. Risk factors for severe retinopathy of prematurity in preterm low birth weight neonates. Indian J Pediatr. 2011;78(7):812–6. https://doi.org/10.1007/s12098-011-0363-7.
Dammann O. Inflammation and retinopathy of prematurity. Acta Paediatr. 2010;99(7):975–7 https://doi.org/10.1111/j.1651-2227.2010.01836.x.
Acknowledgments
Not applicable.
Funding
No funding was received for this publication.
Author information
Authors and Affiliations
Contributions
All authors meet the criteria for authorship: Yucel, Eraydin and Niyaz designed the study; Yucel and Eraydin acquised of the data; Yucel and Terzi analyzed and interpreted the data; Yucel and Niyaz drafted the manuscript; Yucel and Terzi prepared the tables; Yucel, Eraydin, Niyaz and Terzi reviewed and finalized the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ondokuz Mayis University Clinical Research Ethics Committee (OMU KAEK 2021/469). Informed consent was obtained from the parents before the ophthalmological examination for ROP.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yucel, O.E., Eraydin, B., Niyaz, L. et al. Incidence and risk factors for retinopathy of prematurity in premature, extremely low birth weight and extremely low gestational age infants. BMC Ophthalmol 22, 367 (2022). https://doi.org/10.1186/s12886-022-02591-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12886-022-02591-9