Abstract
Background
Undetected vision problems is an important cause of reduced academic achievement, performance in everyday life and self-esteem. This receives little attention in national health care services in Norway even though most of these vision problems are easily correctable. There are no published data on how many Norwegian schoolchildren are affected by correctable vision problems. This study aims to determine the vision status in primary and secondary schoolchildren referred from vision screening during the 10 year period of 2003–2013.
Methods
Of the 1126 children (15%) aged 7–15 years referred to the university eye clinic by the school screening program, all 782 who attended the eye clinic were included in the study. Patient records were retrospectively reviewed with regard to symptoms, refractive error, best corrected visual acuity (BCVA) of logMAR, binocular vision, ocular health and management outcomes.
Results
Previously undetected vision problems were confirmed in 650 (83%) of the children. The most frequent outcomes were glasses (346) or follow-up (209), but types of treatment modalities varied with age. Mean refractive errors were hyperopic for all age groups but reduced with age (ANOVA, p < 0.001). Overall, 51% were hyperopic, 32% emmetropic and 17% myopic. Refractive errors did not change across the decade (linear regression, all p > 0.05). Mean logMAR BCVAs were better than 0.0 and improved with age (ANOVA, p < 0.001). The most prevalent symptoms were headaches (171), near vision problems (149) and reduced distance vision (107).
Conclusions
The vision screening identified children with previously undetected visual problems. This study shows that the types of visual problems varied with age and that most problems could be solved with glasses. Our results stress the importance of regular eye examinations and that vision examinations should be included in primary health care services. Furthermore, there is a need for raised awareness among parents and teaching staff regarding vision problems in children.
Similar content being viewed by others
Background
We live in a visual world, and the ability to see effortlessly is something most people take for granted. The visual system changes in line with the growing child’s expanding visual and behavioural world, and the majority develop good vision, enabling the child to learn, master and achieve important goals in different phases of life and to contribute to society. In fact, good vision and eye health is a prerequisite for social, educational and economic independence and success [1,2,3].
Undetected vision problems are a major cause of reduced performance in everyday life, academic achievement and self-esteem [3,4,5,6,7,8]. Children are dependent on acquiring knowledge and skills through visual information, both in print and digital media, and by observing others. Children spend increasingly more time with their school- and homework as their academic requirements advance. At the same time, the font sizes in printed learning materials are gradually decreased. The fast development and use of digital technology further increase the visual demands in school [9, 10].
To keep up with learning and expected academic performance, children now need to spend more time reading and accessing digital information [11]. Furthermore, from a much earlier age, children spend more time using digital devices in their spare time [10,11,12,13,14]. This increases the workload on the visual system, especially the ability to sustain clear vision at near for longer periods at a time. Even problems like small refractive errors and accommodation or oculomotor control deficits can cause headaches, difficulty concentrating or poor coordination and may lead to unnecessary challenges in school [15,16,17,18]. If a child avoids activities such as reading and homework, there will be serious consequences for learning and for academic and social success [2, 4, 19, 20]. This means that good vision is more important than ever and that uncorrected vision problems should be identified.
In Norway, all children aged 6–16 years have free, compulsory education provided in comprehensive schools with one of the lowest pupil-teacher ratios (9:1) reported by the Organisation for Economic Co-operation and Development (OECD) [21, 22]. Both sexes have equal opportunity to education; the average number of years of education is 12.6, and most children (96%) are enrolled in pre-primary kindergartens [23]. The number of functional illiterates is low [24], and in 2018, Norway was ranked as number one for the global Human Development Index (HDI) [23]. Primary health care in Norway is provided free of charge by the national health authorities until the age of 16 years [25]. Vision and eye health are included in the public preventative health care programme up to 5 years of age [26] in order to prevent irreversible vision loss, but vision examination is not included in school health care services.
The prevalence of visual problems in primary and secondary schoolchildren (6–16 years) in Norway is unknown, but a recent Norwegian study in 16- to 19-year-olds found that more than half were hyperopes [27]. It is known that common vision problems, such as refractive errors, heterophorias and accommodative disorders, may have a profound effect on learning [4, 7]. Some of these conditions may be asymptomatic, and children are often not aware of their vision problems. Therefore, children’s vision problems may not be recognised by parents or teachers. In particular, hyperopia and near vision problems may only be detected through a thorough eye examination. Importantly, common vision problems are easy and cost-efficient to correct, and eye examinations should be considered included in school health services to promote learning, social interactions, future education, employment and socioeconomic benefits [2, 8, 20, 21].
There are no published studies describing the visual status or extent of common vision problems in primary and secondary Norwegian schoolchildren. In Kongsberg, a school vision programme has existed since the 1970s as a collaboration between the municipality and the National Centre of Optics, Vision and Eye Care (NCOVE) at the University of South-Eastern Norway. Here, all schoolchildren are offered vision screening at the ages of 7–15 years. This study aims to determine the vision status in primary and secondary school children referred from vision screening during the 10 year period of 2003–2013.
Methods
Study population
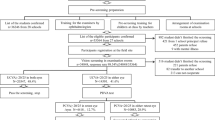
The annual school vision screening programme is a collaboration between the Kongsberg municipality and NCOVE and offers screening to all children in the 2nd, 5th and 10th grades (7-, 10- and 15-year-olds) in the 13 primary and secondary schools in Kongsberg. The majority of the school population has a Norwegian ethnic background (86%) similar to the rest of Norway [28]. During the decade of 2003–2013, 7658 children participated in the school vision screening programme (94% of all 8191 children eligible). Of these, 1126 (15%) children failed the vision screening and were referred to the NCOVE university eye clinic for a full eye examination.
Study sample
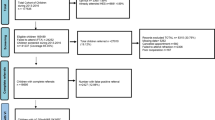
This study has retrospectively analysed data from the patient records of those 782 children who attended the NCOVE eye clinic. Several cohorts were screened two or three times, but only 40 (5%) children were referred more than once. See flow chart for details (Fig. 1). The study followed the tenets of the Declaration of Helsinki and was approved by the Ombudsman for Privacy in Research at the Norwegian Social Science Data Services.
Protocol
The patient records included a structured and age-appropriate history and symptoms interview. Reported refractive status was determined after retinoscopy and subjective refraction (at 6 m), generally obtained without cycloplegia, and best corrected visual acuity (BCVA) was recorded. Ocular alignment was assessed by cover test (at 6 m and 40 cm). Near point of convergence (NPC) and monocular and binocular accommodation amplitude (ACC) were assessed using an RAF ruler. In addition ocular adnexa, pupillary reactions, anterior and posterior segments were examined. The eye examination was performed by final-year optometry students under supervision, and the supervisor had the overall responsibility for choice of management and advice to the patient in line with the Norwegian Association of Optometry’s clinical guidelines.
For analysis, spherical equivalent refraction (SER) was calculated in dioptres (D). Refractive errors were defined as emmetropia (− 0.50 < SER < + 0.50 D), hyperopia (SER ≥ + 0.50 D), myopia (SER ≤ − 0.50 D), astigmatism (≤ − 0.75 DC) and anisometropia (≥ 1.00 D) [29, 30]. Orthophoria was defined as 2 prism dioptres (pd.) exophoria to 1 pd. esophoria for distance and 6 pd. exophoria to 0 pd. esophoria for near. Normal NPC was defined as ≤10 cm and normal ACC as ≤10 D, ≤ 9 D and ≤ 8 D for the 2nd-, 5th- and 10th-graders respectively. Thirty-four children with strabismus were excluded from the binocular vision analysis. Symptoms were grouped into six categories. For details, see Table 2.
Management outcomes
Reported management and advice given to the patient were defined by five main categories: 1) prescription, 2) vision training, 3) follow-up, 4) referral to ophthalmologist and 5) false referrals (referrals from screening where the eye examination concluded there were no symptoms and that vision was normal) (see Table 2). Prescriptions were glasses or contact lenses. Vision training was conventional home-based convergence, accommodation or facility training. Children with asthenopia or mild non-symptomatic hyperopia were scheduled for follow-up.
Statistical methods
One-way ANOVA and post hoc Tukey HSD tests were used to analyse differences between age groups. The threshold for statistical significance level was set at 5% (p < 0.05). Distributions and analyses using refractive errors include right eye only, as there were no significant differences between the right and left eyes (paired t-test, p > 0.05) for either age group, and SERs were normally distributed. Linear regression analyses were used to investigate refractive changes over time for each age group separately. All computations were performed using the statistical package SPSS Statistics 21 (International Business Machines, USA). Incomplete data sometimes occurred, explaining varying sample sizes for different parameters.
Results
Of the 1126 children referred, 782 (70%) attended the eye examination, with 241 (31%), 241 (31%) and 300 (38%) children in the three age groups (2nd, 5th and 10th grades, respectively).
Refractive error and visual acuity
Monocular and binocular spherical equivalent refractive errors (SERs) (mean [95% CI]) are shown in Table 1. For all three age groups the mean SER was skewed towards hyperopia. A one way ANOVA showed a significant difference in SER between age groups (ANOVA, F (2, 776) = 26.8, p < 0.001). Post hoc comparisons using the Tukey HSD test showed a significant reduction in hyperopia (SER) between all age groups with increased age (p < 0.05 for all comparisons).
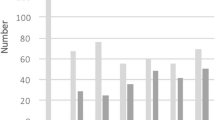
Table 2 shows that one third of the children were classified as emmetropic (33%) and slightly more than one third as low-grade hyperopic (≥ + 0.50D, < + 2.00D, 38%). Hyperopia ≥ + 2.00 D decreased with age and was present in 7% of the 2nd-grade, 6% of the 5th-grade and 1% of the 10th-grade children. Myopia increased with age, with 3% of 2nd-grade, 15% of 5th-grade and 27% of the 10th-grade children being myopic. Although almost one third of the 10th-grade children were classified as myopic, only 1% had myopia of − 3.00 D or higher. Clinically significant astigmatism (≥ 0.75 DC) was present in 5% of all children and clinically significant anisometropia (≥ 1.00 D) in 3%.
The distribution of refractive errors was similar for all three age groups over the 10-year period (Fig. 2). Linear regression shows no significant change in either myopia or hyperopia over the decade for the 2nd-grade (R2 = 0.01, F = 2.9, p > 0.05), 5th-grade (R2 = 0.01, F = 1.4, p > 0.05) or 10th-grade children (R2 = 0.002, F = 0.45, p > 0.05).
Most children obtained very good correctable vision (BCVA, Table 1). As expected, binocular BCVA was slightly better than monocular. Overall, 91% of the children had BCVA of logMAR 0.0 (decimal acuity 1.0) or better in one or both eyes (Table 2). Table 2 shows there was a slight but significant improvement with age in RE BCVA (ANOVA, F (2, 772) = 28.2, p < 0.001) and binocular BCVA (ANOVA, F (2, 766) = 44.8, p < 0.001). Tukey HSD post hoc analysis showed that 2nd-graders had lower RE and binocular visual acuity than both older age groups (p < 0.01 for all comparisons), but there was no significant difference between 5th- and 10th-graders (p > 0.1). Reduced vision was found in 2% of the children, including one 2nd-grade child with BCVA > logMAR 0.5 (decimal acuity < 0.3), who was referred to an ophthalmologist due to ocular pathology.
Binocular vision and accommodation
Table 2 shows that distance and near horizontal orthophoria were present in 78 and 77% of children, respectively. The mean [95% CI] heterophoria was 0.8 [0.6, 1.0] exophoria for distance, and 2.3 [2.0, 2.6] exophoria for near. Exophoria was present in 13 and 14% of the children for distance and near, respectively (see Table 2 for criteria). Esophoria was less common and present in 7 and 9% of children for distance and near, respectively. Mean [95% CI] binocular accommodation was 14.0 [13.4, 14.6] D, 12.9 [12.3, 13.5] D, and 11.8 [11.3, 12.2] D for 2nd-, 5th- and 10th-graders, respectively (Table 1), and reduced binocular accommodation was found in 15% of all children (Table 2). ANOVA and post hoc analyses showed a significant difference in accommodation between the three age groups for binocular measures (ANOVA, F (2, 774) = 18.9, p < 0.001; Tukey HSD, p < 0.007 for all comparisons). NPC was 8.2 [7.7, 8.7] cm across all children, and NPC ≥ 10 cm was found in 19%. There was no significant difference between the age groups for NPC (ANOVA, F (2, 774) = 0.241, p = 0.786).
Symptoms
Of the 782 children, 26% experienced symptoms of vision problems, and 25% had more than one symptom. The most prevalent symptoms were headaches (22%), near vision problems (19%) and reduced distance vision (14%).
Management
Most children (650, 83%) referred from the school screening had vision problems requiring treatment or follow-up. Glasses (spectacle prescription) for distance or near work was the most common management strategy (55%), followed by vision training (7%) and glasses combined with vision training (4%). Glasses were recommended primarily for low hyperopia in 2nd- and 5th-grade children and for myopia in 10th-grade children. Binocular vision and near problems due to reduced accommodation or poor NPC were prescribed low plus lenses, vision training or both. Of the 16% of children with reduced accommodation, 42% were given glasses, 19% were prescribed vision training and 11% a combination. Of the 14% of children with reduced NPC, 40% were given glasses, 31% vision training and 20% both glasses and training. Vision training was more commonly recommended for 5th- and 10th-grade children, while the 2nd-grade children were more likely to receive follow-up. Follow-up (33%) was recommended when the child had very slight symptoms, refractive errors or binocular problems and when no immediate management was required. Overall 25 (4%) children were referred to an ophthalmologist; however, most were 2nd-grade children requiring glasses to be covered by the National Insurance Scheme.
Discussion
This study describes vision status based on a comprehensive eye examination in 782 of 1126 (70%) children referred from the Kongsberg vision screening programme, for which 7658 2nd-, 5th- and 10th-grade children attended during 2003–2013. Most children had normal functional vision and eye health. As expected, there was a slight but significant improvement in BCVA with age, and only a few children had reduced visual acuity. However, of those 782 attending the eye examination, 83% were confirmed to be true referrals, indicating that the vision screening programme identified vision problems previously not detected. This suggests that many children, parents and teachers are unaware of vision problems that may influence academic performance and quality of life, and supports previous studies [2, 4, 15,16,17, 20, 31, 32].
Importantly, our study confirms that most vision problems can easily be managed by glasses or vision training [31,32,33]. A substantial proportion of the children had low to moderate hyperopia or accommodative or binocular deficits. These deficits may interfere with the ability to do sustained near work [17, 18, 34, 35] but can be difficult to detect because of normal distance vision and absence of explicit symptoms. As children spend a considerable amount of time on near activities at school and in their spare time [10, 13], eye and vision examinations should be available through the school health care services to prevent unnecessary academic achievement gaps.
Even though one third of children in this study were emmetropic, all age groups showed a slight hyperopic mean refractive error which reduced with age, as expected [36,37,38,39,40]. Interestingly, we did not find any change in refractive errors during the 10-year period for either age group, nor did we find the high proportion of myopia reported in many studies [41,42,43,44,45]. Although we found that myopia increased with age, only four children (1%) had myopia > 3.0 D (SER). The relatively low proportion of myopia supports previous studies showing that hyperopia is frequent in Nordic children and youths [27, 46, 47] and implies that refractive status has been relatively stable over the past few decades. This is also similar to data reported in white American children [40, 48]. One limitation to the reported refractive errors in this study is that cycloplegic refraction was not routinely used, as this was not considered standard clinical practice for schoolchildren in Norway until 2015. However, a cycloplegic refraction would skew the refractive error for all age groups towards higher values of hyperopia, and as such, the values and numbers of hyperopia in our study would increase.
Most children had good accommodation and binocular vision, but a proportion were given glasses or vision training. Accommodation and binocular vision problems may cause symptoms like headache, eyestrain, blurred vision, intermittent diplopia, poor concentration and comprehension when performing near tasks [18, 35, 49,50,51]. However, children do not necessarily complain of symptoms if not asked specifically. It is likely that a substantial portion of the children found to have vision problems in this study would have remained undiagnosed in absence of the vison screening.
Our results show that children in different age groups have different visual challenges that need different treatments. Academic achievements and success depend on effective and efficient reading and learning. Vision changes along with increased visual demands in school with age, and eye examinations at regular intervals should be emphasised. This study suggests that children would benefit from a comprehensive vision examination during the 2nd, 5th and 10th grades at school, as this is likely to promote academic success and social inclusion. Furthermore, it would educate and raise awareness among children, parents and teaching staff of the importance of good vision for good health.
A strength of this study is the large stable number of children attending the eye examination. The vision problems remained similar over the decade, which limits the effect of bias and contributes to the validity of this study. Furthermore, the Kongsberg school population has a Norwegian ethnic background similar to the rest of Norway, and the school vision screening has a very high attendance (94% of the population). This strengthens the potential generalisability of the study results to all Norwegian schoolchildren of the same age. A limitation of the study is the large amount of data collected over a long period by different optometry students, which may have influenced the quality of results and missing data, but in all cases, they were supervised and managed by experienced optometrists, and the results are comparable to other Nordic countries. The study does not include data after 2013 due to a time-limited approval of the study period. Furthermore, changes were made to the school vision screening protocol based on preliminary results from this study (e.g., include cycloplegic refraction). Based on the results from a recent study in Norwegian adolescents [27], we think our results would not be significantly different if more recent years were added. Despite not including recent years, our study contributes important knowledge of vision status and problems in schoolchildren that may guide further research, clinical practice and health care policies to include vision in school health care, as this still receives little attention in Norway and in many other countries [52, 53].
Conclusions
Even though most children attending the eye clinic had good vision, at least 11% of the population screened during 2003–2013 had previously undetected vision problems. This study shows that the types of visual problems vary with age and, importantly, that most problems were solved with glasses or vision training at a low cost. Our results stress the importance of regular eye examinations and that vision examinations should be included in primary health care services to reduce the number of schoolchildren with avoidable vision problems and potential socioeconomic burden. Furthermore, there is a need for raised awareness among parents and teaching staff on vision problems in children.
Availability of data and materials
The datasets analysed during the current study are available from HKF on reasonable request.
Abbreviations
- ACC:
-
Accommodation
- BCVA:
-
Best corrected visual acuity
- BIN:
-
Binocular
- cm:
-
Centimetre
- D:
-
Dioptres
- LE:
-
Left eye
- m:
-
Metre
- MAR:
-
Minimum angle of resolution
- NCOVE:
-
National Centre for Optics, Vision and Eye Care
- NPC:
-
Near point of convergence
- pd.:
-
Prism dioptres
- RE:
-
Right eye
- SER:
-
Spherical equivalent refraction
- VT:
-
Vision training
References
Marshall EC, Meetz RE, Harmon LL. Through our children's eyes--the public health impact of the vision screening requirements for Indiana school children. Optometry. 2010;81(2):71–82.
Lv B, Zhou H, Guo X, Liu C, Liu Z, Luo L. The relationship between academic achievement and the emotional well-being of elementary school children in China: the moderating role of parent-school communication. Front Psychol. 2016;7. https://doi.org/10.3389/fpsyg.2016.00948.
Basch CE. Vision and the achievement gap among urban minority youth. J Sch Health. 2011;81(10):599–605.
Dudovitz RN, Izadpanah N, Chung PJ, Slusser W. Parent, teacher, and student perspectives on how corrective lenses improve child wellbeing and school function. Matern Child Health J. 2016;20(5):974–83.
Grisham D, Powers M, Riles P. Visual skills of poor readers in high school. Optometry. 2007;78(10):542–9.
Ethan D, Basch CE. Promoting healthy vision in students: progress and challenges in policy, programs, and research. J Sch Health. 2008;78(8):411–6.
Evans JR, Morjaria P, Powell C. Vision screening for correctable visual acuity deficits in school-age children and adolescents. Cochrane Database Syst Rev. 2018;2:CD005023. https://doi.org/10.1002/14651858.CD005023.pub3.
Goldstand S, Koslowe KC, Parush S. Vision, visual-information processing, and academic performance among seventh-grade schoolchildren: a more significant relationship than we thought? Am J Occup Ther. 2005;59(4):377–89.
Narayanasamy S, Vincent SJ, Sampson GP, Wood JM. Visual demands in modern Australian primary school classrooms. Clin Exp Optom. 2016;99:233–240.
Norwegian Media Authority. Barn og medier 2018 [Children and media 2018]. Norwegian Media Authority; 2018. https://medietilsynet.no/barn-og-medier/barn-og-medier-undersokelsen/. Accessed 15 Nov 2018.
OECD. Students, Computers and Learning: Making the Connection. In: PISA. Paris: OECD Publishing; 2015.
Bakken A. Ungdata. Nasjonale resultater 2018 [Youth data. National results 2018]. NOVA-rapport 8/18. Oslo: Norwegian Social Research; 2018.
Przybylski AK, Weinstein N. Digital screen time limits and young Children's psychological well-being: evidence from a population-based study. Child Dev. 2019;90:e56-e65. https://doi.org/10.1111/cdev.13007.
Hushovd I-L, Falkenberg HK, Kristiansen B, Sundling V. Små barn med synssvekkelser i en digital Verden [Young children with visual impairments - included in the digital world]. Spesialpedagogikk. 2017;4:37–48.
Bruce A, Fairley L, Chambers B, Wright J, Sheldon TA. Impact of visual acuity on developing literacy at age 4-5 years: a cohort-nested cross-sectional study. BMJ Open. 2016;6(2):e010434.
Shankar S, Evans MA, Bobier WR. Hyperopia and emergent literacy of young children: pilot study. Optom Vis Sci. 2007;84(11):1031–8.
Williams W, Latif A, Hannington L, Watkins D. Hyperopia and educational attainment in a primary school cohort. Arch Dis Child. 2005;90(2):150–3.
The Vision In Preschoolers - Hyperopia In Preschoolers Study G, Kulp MT, Ciner E, Maguire M, Moore B, Pentimonti J, et al. Uncorrected hyperopia and preschool early literacy: results of the vision in preschoolers - hyperopia in preschoolers (VIP-HIP) study. Ophthalmology. 2016;123(4):681–9.
Flashman J. Academic achievement and its impact on friend dynamics. Sociol Educ. 2012;85(1):61–80.
Davidson S, Quinn GE. The impact of pediatric vision disorders in adulthood. Pediatrics. 2011;127(2):334–9.
OECD. Investing in Youth: Norway. 2018 Paris: OECD Publishing; 2018.
OECD. Education at a glance 2018: OECD indicators. Paris: OECD Publishing; 2018.
The United Nations Development Programme (UNDP). Human development indices and indicators: 2018 Statistical update. New York; 2018.
ELINET. Literacy in Norway. In: European literacy policy network; 2016.
Helse- og omsorgstjenesteloven. Lov om kommunale helse- og omsorgstjenester (LOV-2011-06-24-30), vol. 3–1; 2011.
Norwegian Directorate of Health. Retningslinjer for undersøkelse av syn, hørsel og språk hos barn [in Norwegian]. In: IS-1235, Nasjonale faglige retningslinjer; 2006.
Hagen LA, Gjelle JVB, Arnegard S, Pedersen HR, Gilson SJ, Baraas RC. Prevalence and possible factors of myopia in Norwegian adolescents. Sci Rep. 2018;8(1):13479.
Statistics Norway: Municipal population [Kongsberg]; 2018.
O'Donoghue L, Saunders KJ, McClelland JF, Logan NS, Rudnicka AR, Gilmartin B, Owen CG. Sampling and measurement methods for a study of childhood refractive error in a UK population. Br J Ophthalmol. 2010;94(9):1150–4.
Negrel AD, Maul E, Pokharel GP, Zhao J, Ellwein LB. Refractive error study in children: sampling and measurement methods for a multi-country survey. Am J Ophthalmol. 2000;129(4):421–6.
Kumaran SE, Balasubramaniam SM, Kumar DS, Ramani KK. Refractive error and vision-related quality of life in south Indian children. Optom Vis Sci. 2015;92(3):272–8.
Read SA, Vincent SJ, Collins MJ. The visual and functional impacts of astigmatism and its clinical management. Ophthalmic Physiol Opt. 2014;34(3):267–94.
Borsting E, Mitchell GL, Kulp MT, Scheiman M, Amster DM, Cotter S, et al. Improvement in academic behaviors after successful treatment of convergence insufficiency. Optom Vis Sci. 2012;89(1):12–8.
Hendricks TJ, DEB J, van Der Horst FG, Hendrikse F, Knottnerus JA. relationship between habitual refractive errors and headache complaints in schoolchildren. Optom Vis Sci. 2007;84(2):137–43.
Vilela MA, Pellanda LC, Fassa AG, Castagno VD. Prevalence of asthenopia in children: a systematic review with meta-analysis. J Pediatr. 2015;91(4):320–5.
Slataper FJ. Age norms of refraction and vision. Arch Ophthalmol. 1950;43(3):466–81.
Jones LA, Mitchell GL, Mutti DO, Hayes JR, Moeschberger ML, Zadnik K. Comparison of ocular component growth curves among refractive error groups in children. IOVS. 2005;46(7):2317–27.
Irving EL, Machan CM, Lam S, Hrynchak PK, Lillakas L. Refractive error magnitude and variability: relation to age. Aust J Optom. 2019;12:55-63.
Hirsch MJ. Predictability of refraction at age 14 on the basis of testing at age 6: interim report from the Ojai longitudinal study of refraction. Am J Optom Arch Am Acad Optom. 1964;41:567–73.
Mayro EL, Hark LA, Shiuey E, Pond M, Siam L, Hill-Bennett T, Tran J, Khanna N, Silverstein M, Donaghy J, et al. Prevalence of uncorrected refractive errors among school-age children in the School District of Philadelphia. J Am Assoc Pediatr Ophthalmol Strabismus. 2018;22(3):214–217.e212.
McCullough SJ, O'Donoghue L, Saunders KJ. Six year refractive change among white children and young adults: evidence for significant increase in myopia among white UK children. PLoS One. 2016;11(1):e0146332.
Tsai DC, Fang SY, Huang N, Hsu CC, Chen SY, Chiu AW, et al. Myopia development among young schoolchildren: the myopia investigation study in Taipei. IOVS. 2016;57(15):6852–60.
Bar Dayan Y, Levin A, Morad Y, Grotto I, Ben-David R, Goldberg A, et al. The changing prevalence of myopia in young adults: a 13-year series of population-based prevalence surveys. IOVS. 2005;46(8):2760–5.
Parssinen O. The increased prevalence of myopia in Finland. Acta Ophthalmol. 2012;90(6):497–502.
French AN, Morgan IG, Mitchell P, Rose KA. Risk factors for incident myopia in Australian schoolchildren: the Sydney adolescent vascular and eye study. Ophthalmology. 2013;120(10):2100–8.
Sandfeld L, Weihrauch H, Tubaek G, Mortzos P. Ophthalmological data on 4.5- to 7-year-old Danish children. Acta Ophthalmol. 2018;96(4):379–83.
Jacobsen N, Jensen H, Goldschmidt E. Prevalence of myopia in Danish conscripts. Acta Ophthalmol Scand. 2007;85(2):165–70.
Hashemi H, Fotouhi A, Yekta A, Pakzad R, Ostadimoghaddam H, Khabazkhoob M. Global and regional estimates of prevalence of refractive errors: systematic review and meta-analysis. J Currt Ophthalmol. 2018;30(1):3–22.
Rouse M, Borsting E, Mitchell GL, Cotter SA, Kulp M, Scheiman M, et al. Validity of the convergence insufficiency symptom survey: a confirmatory study. Optom Vis Sci. 2009;86(4):357–63.
Borsting E, Rouse MW, Deland PN, Hovett S, Kimura D, Park M, et al. Association of symptoms and convergence and accommodative insufficiency in school-age children. Optometry. 2003;74(1):25–34.
Hussaindeen JR, Shah P, Ramani KK, Ramanujan L. Efficacy of vision therapy in children with learning disability and associated binocular vision anomalies. Aust J Optom. 2018;11(1):40–8.
Nikander K, Kosola S, Kaila M, Hermanson E. Who benefit from school doctors’ health checks: a prospective study of a screening method. BMC Health Serv Res. 2018;18(1):501.
Langford R, Bonell C, Jones H, Pouliou T, Murphy S, Waters E, Magnus D, Campbell R, et al. The World Health Organization’s Health Promoting Schools framework: a Cochrane systematic review and meta-analysis. BMC Public Health. 2015;15(1):130.
Acknowledgements
The authors would like to thank all children, students and optometrists who participated in data collection. The authors would also like to thank statistician Tor Martin Kvikstad for his help.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
HKF designed the study, analysed and interpreted the data and wrote the first draft of the manuscript. ES made significant contributions to data analyses, interpretations and manuscript preparation. TL contributed to data interpretation and critically revising the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study followed the tenets of the Declaration of Helsinki. The study was approved by the Ombudsman for Privacy in Research at the Norwegian Social Science Data Services (reference number 35374), and all data were treated according to their regulations. Data collection was organised digitally while taking care of appropriate data protection. The confidentiality of participants was protected by using an encryption key prior to data analyses. The key was stored separately from study data. Because the project was defined as a Quality Assurance project, Ethics committee approval was deemed unnecessary according to national regulations (Act 2008-06-20 no. 44).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Falkenberg, H.K., Langaas, T. & Svarverud, E. Vision status of children aged 7–15 years referred from school vision screening in Norway during 2003–2013: a retrospective study. BMC Ophthalmol 19, 180 (2019). https://doi.org/10.1186/s12886-019-1178-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12886-019-1178-y