Abstract
Background
Distant metastasis has been the main failure pattern for locoregionally advanced rectal cancer (LARC) patients, and intensified neoadjuvant chemotherapy has become a popular research topic. The present study aimed to compare the survival outcomes, acute toxicities and surgical complications in LARC patients who received preoperative chemoradiotherapy with triweekly oxaliplatin and capecitabine (triweekly XELOX) or capecitabine.
Methods: Between 2007 and 2017, patients with clinically staged II-III rectal cancer who were treated with preoperative chemoradiotherapy using either triweekly XELOX (oxaliplatin 130 mg/m2 plus capecitabine 825 mg/m2) or capecitabine were included. Variables potentially influencing chemotherapy treatment selection were used to generate propensity scores (PS). The association between chemotherapy regimens and survival endpoints, including distant metastasis-free survival (DMFS), overall survival (OS) and disease-free survival (DFS), were evaluated and adjusted with PS. The acute toxicities and surgical complications were also compared.
Results
A total of 810 patients were included in the analysis; 277 (34.2%) patients received triweekly XELOX, and 533 (65.8%) received capecitabine. The pathological complete response (pCR) rates were 20.2 and 19.9% (P = 0.912) for the groups treated with triweekly XELOX and capecitabine, respectively. The 5-year DMFS, OS and DFS with triweekly XELOX versus capecitabine were 75.6% vs. 77.6% (P = 0.555), 79.2% vs. 83.3% (P = 0.101), and 69.9% vs. 73.7% (P = 0.283), respectively. Triweekly XELOX was not associated with an increased risk of severe toxicity during chemoradiotherapy, but it increased the risk of postoperative complications compared to capecitabine. After PS adjustment, the differences between the two groups remained insignificant in pCR rate, survival outcomes, and acute toxicities, and the difference in surgical complications disappeared.
Conclusions
Triweekly XELOX or capecitabine concurrent with neoadjuvant radiotherapy leads to similar long-term survival outcomes, acute toxicities and surgical complications in LARC patients.
Similar content being viewed by others
Background
Fluorouracil-based preoperative chemoradiotherapy (CRT) followed by total mesorectal excision (TME) is the standard option for the initial treatment of locally advanced rectal cancer (LARC) in terms of downstaging to pathological complete response, increasing rates of sphincter-saving surgery and decreasing local recurrence [1,2,3]. However, this improvement in local control has not been paralleled by an increase in long-term overall survival (OS), largely owing to the persistently high rate of distant metastasis (29–39%) [2, 4]. Thus, it makes sense to achieve better control of systemic disease for longer survival.
Borrowing from the survival benefits of adding oxaliplatin to fluorouracil-based adjuvant chemotherapy in colon cancer [5,6,7], several large randomized trials have tested the combination in LARC, concomitantly with neoadjuvant radiotherapy. Apart from Jiao’s study [8] and the German CAO/ARO/AIO-04 trial [9], which reported improvements in distant metastasis-free survival (DMFS) and disease-free survival (DFS), respectively, most studies have failed to show survival benefits with the addition of oxaliplatin [10,11,12,13]. The unaltered survival implies that low-dose weekly oxaliplatin was unable to further eliminate tumour micrometastasis, providing a rationale for intensified systemic treatment. Our previous study and Gao et al. reported that oxaliplatin given triweekly at a dose of 130 mg/m2 added to capecitabine (triweekly XELOX) concomitant with preoperative radiotherapy was tolerable and associated with excellent compliance, as well as promising long-term outcomes [14, 15]. However, the reports were both based on small sample sizes, and no clear evidence exists for the superiority of triweekly XELOX compared to capecitabine alone with preoperative chemoradiotherapy in patients with LARC.
Therefore, we focused on the comparison of long-term survival outcomes and the adverse effects of capecitabine plus neoadjuvant radiation with or without triweekly oxaliplatin in LARC patients. Our findings could help to guide clinical decision-making regarding neoadjuvant chemoradiotherapy treatment strategies in LARC.
Methods
Patient selection
In this unicentral, retrospective, noninterventional study, we included patients with histologically diagnosed rectal cancer of clinical stages II-III treated at our hospital between September 2007 and October 2017. All patients were treated with concurrent chemoradiotherapy followed by definitive surgery. Patients with metastatic or recurrent rectal cancer or patients who underwent endoscopic mucosal resection or transanal local excision were excluded. Patients with nonmetastatic disease who were found to have metastasis in the reassessment before surgery or during surgery were also excluded to avoid misdiagnosis in the first assessment.
The retrospective study was approved by the Clinical Research Ethics Committee of the Fujian Medical University Union Hospital, and due to the retrospective nature of the study, the requirement for informed consent was waived. The study was performed in accordance with the Declaration of Helsinki.
Staging
Before treatment, patients underwent a routine staging procedure consisting of physical examination, digital rectal examination, colonoscopy, chest computed tomography, abdominal and pelvic magnetic resonance imaging or positron emission tomography-computed tomography scan. Routine laboratory tests consisted of complete blood count and chemistry, including renal function. Clinical stage was assessed according to the 7th edition of the International Union against Cancer/American Joint Committee on Cancer (UICC/AJCC) staging system.
Chemotherapy
The concurrent chemotherapy consisted of capecitabine alone or double chemotherapy with oxaliplatin and capecitabine. In the capecitabine alone group, patients received oral capecitabine 825 mg/m2 twice daily during radiotherapy. In the double chemotherapy group (triweekly XELOX), patients were administered 130 mg/m2 intravenous oxaliplatin on Day 1 plus 825 mg/m2 capecitabine twice daily from Days 1–14 every 21 days. The selection of the concurrent chemotherapy regimen mainly depended on the experiences of the surgeon and oncologist and patient preference, with some consideration of the economic situation of the patients.
Postoperative adjuvant chemotherapy was determined by the treating surgeon and the patient. Most adjuvant chemotherapy was an oxaliplatin-based regimen, and only two patients received capecitabine alone.
Radiotherapy
All patients received preoperative radiotherapy with either 3-dimensional conformal radiotherapy (3D-CRT) or intensity-modulated radiotherapy (IMRT). Radiation treatment planning was designed in accordance with previous studies conducted at the Fujian Medical University Union Hospital [14, 16]. Briefly, radiotherapy was delivered at 1.8 to 2.0 Gy (Gy) daily Monday through Friday for a total of 25 to 28 fractions over 5 to 6 weeks and a total dose of 45 Gy to 50.4 Gy. Radiation was delivered with a minimum energy of 6-MV photons through a three-field or five-field technique to the primary tumour and to mesorectal, presacral, and internal iliac lymph node drainage regions.
Surgery
Radical surgery according to the TME principle was performed for all patients. Pathological stage was determined based on the surgical specimen. Primary tumour downstaging was determined by comparing the pathologic T stage with the baseline clinical T stage, and a pathologic T stage lower than the baseline clinical T stage was considered tumour downstaging. A pathological complete response (pCR, ypT0N0) was defined as the complete absence of tumour cells at the primary site and without lymph node involvement. The rectal cancer regression grade (RCRG) after preoperative treatment was evaluated according to Wheelers’ classification [17]. The RCRG classification groups were as follows: RCRG1, sterilization or only microscopic foci of adenocarcinoma remaining, with marked fibrosis; RCRG2, marked fibrosis but macroscopic disease present; RCRG3, little or no fibrosis, with abundant macroscopic disease.
Statistical analysis
OS was calculated as the time from the first day of treatment to death from any cause or the last follow-up. DFS was defined as the time from the date of commencement of treatment to the first occurrence of any of the following events: local and/or regional recurrence, distant metastasis, or death from any cause. DMFS was defined as the time from the commencement of treatment to the date of metastasis. During therapy, adverse events were evaluated according to National Cancer Institute Common Terminology Criteria, version 4.0.
Baseline and clinical characteristics were used to examine balance between groups using absolute standardized mean differences (SMD), which describe between-group differences in units of SMD and are not substantially influenced by sample size in large cohorts [18]. Differences greater than 0.1 were considered clinically meaningful [18]. Survival was assessed using the Kaplan-Meier method, and categorical variables were compared using the log-rank test. The independent prognostic factors for rectal cancer were identified by the Cox proportional hazard regression model. To control for the misdistribution of the treatment period, we calculated the survival outcomes by comparing the triweekly XELOX cohort to the capecitabine cohort treated during 2012–2017. To focus on patients (clinical non lymph node metastasis (cN-) and pathological ypStage I-II) who likely benefited from the addition of oxaliplatin reported in the CAO/ARO/AIO-04 trial [9], we performed sensitivity analyses in the subgroups mentioned above.
An unadjusted comparison between patients receiving capecitabine and triweekly XELOX would be prone to bias. To enable a comparison in equivalent groups, a 1:2 propensity score (PS) analysis was performed between the two patient cohorts [18,19,20,21]. The propensity score was calculated for each patient using logistic regression with the variable that had a potential confounding effect, such as age at diagnosis, sex, interval from end of radiotherapy (RT) to surgery, clinical T stage, clinical N stage, RT method, and adjuvant chemotherapy. A PS-matched cohort was then created, yielding a cohort of 510 patients. PS-adjusted Cox regression models were used to evaluate the associations of concurrent chemotherapy regimens with treatment outcomes.
Statistical analyses were performed using SPSS software, version 23.0 (2015 IBM Corporation, Armonk, NY, USA), and the R programming language (R version 4.1.0). All tests used to explore statistical significance were 2 sided, and P < 0.05 was considered statistically significant.
Results
Patient characteristics
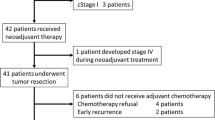
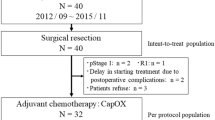
See Fig. 1 for a CONSORT diagram illustrating how we arrived at the analytic sample of 810 patients. From September 2007 to October 2017, 1477 patients were screened, and 810 patients met the eligibility criteria, of whom 277 (34.2%) were treated with triweekly XELOX and 533 (65.8%) with capecitabine. The distribution of baseline characteristics is reported in Table 1. In the triweekly XELOX group, 108 (39.0%) patients were treated in the early years (2007–2011), while only 2 patients (0.4%) in the capecitabine group were treated during the period (SMD = − 0.792). Regarding radiotherapy, 254 (91.7%) patients in the triweekly XELOX group and 525 (98.5%) patients in the capecitabine group received a dose of 50 or 50.4 Gy. The use of IMRT increased over time, so more patients in the capecitabine group were treated with IMRT (SMD = − 0.638). Patients receiving triweekly XELOX were younger (SMD = − 0.259) and tended to have a lower clinical T stage (SMD = − 0.145). After 1:2 matching based on PS, a total of 510 patients were included for subsequent analyses. Most variables were similarly distributed between the two groups after PS matching, except for the treatment period and the radiotherapy dose (Table 1).
Surgery and pathological findings
In the unadjusted cohort, the median time between surgery and chemoradiotherapy was 8 weeks (range, 4–20 weeks) for the triweekly XELOX group and 9 weeks (range, 4.3–19.9 weeks) for the capecitabine group (SMD = − 0.543) (Table 1). Anterior resection was the most common surgery type. The postoperative pathological findings are presented in Table 2. The pCR rate did not differ between the two groups (20.2% vs. 19.9% in the triweekly XELOX group and capecitabine group, respectively, SMD = 0.008). Regarding concerning primary tumour downstaging, it occurred less often in the triweekly XELOX group than in the capecitabine group (69.7% vs. 77.7%, SMD = 0.174). After PS adjustment, the difference in primary downstaging between the triweekly XELOX group and the capecitabine group disappeared (71.7% vs. 78.1%, SMD = − 0.101, Table 2). There was also no significant difference in the pCR rate (SMD = 0.037), ypT stage (SMD = 0.069), ypN stage (SMD = 0.073) or RCRG grade (SMD = − 0.096) in the PS-matched cohort. There were more lymph nodes retrieved in the capecitabine group than in the triweekly XELOX group (11 vs. 13, SMD = − 0.291). Survival analyses showed that dissection of ≥12 lymph nodes was not significantly associated with DMFS, DFS or OS in the unadjusted cohort (Additional Figure 1) or in the PS-matched cohort (Additional Figure 2).
Survival outcomes and prognostic factors
The median follow-up was 63 months (range, 6–152) for all patients and 89 months (range, 6–152) and 59 months (range, 6–109) for the triweekly XELOX group and capecitabine group, respectively. The 5-year DMFS, OS and DFS were 75.6% vs. 77.6% (P = 0.555), 79.2% vs. 83.3% (P = 0.101), and 69.9% vs. 73.7% (P = 0.283) for the patients treated with triweekly XELOX and capecitabine, respectively (Fig. 2). After PS adjustment, there remained no survival benefit to using triweekly XELOX in the PS-matched cohort (Fig. 3).
Considering that most patients in the capecitabine group were treated during 2012–2017, while the patients in the triweekly XELOX group were not, we repeated our survival analysis excluding patients treated during 2007–2011. We found similar survival outcomes in this analysis, including DMFS (P = 0.754), OS (P = 0.364) and DFS (P = 0.758) (Fig. 4). Triweekly XELOX was also not associated with improved DMFS, OS or DFS in the sensitivity analysis considering patients with different clinical cN stages and pathological ypStages or whether they received adjuvant chemotherapy (Fig. 4).
The results of multivariate analysis of the prognostic factors are shown in Table 3. In the whole cohort of patients, multivariate analysis showed that the regimen of concurrent chemotherapy had no effect on DMFS (P = 0.871), OS (P = 0.682) or DFS (0.820), while ypT stage and ypN stage were independent prognostic factors for DFS, OS and DFS (all P values < 0.001). In the PS-adjusted cohort, the regimen of concurrent chemotherapy was still not an independent prognostic factor for DMFS, OS, or DFS.
Compliance and acute toxicity
In an unadjusted analysis, 99.2% of patients in the triweekly XELOX group completed two cycles of chemotherapy, and 99.6% patients in the triweekly XELOX group and 100% patients in the capecitabine group received at least 45Gy; in the former group, only 1 patient received a dose of 41.4 Gy/23F due to poor health and treatment toxicity, while no patients in the latter group did.
There was no incidence of treatment-related mortality. Grade 3–4 toxicities of any type during CRT were observed in 34 patients (12.3%) in the triweekly XELOX group and in 47 patients (8.8%) in the capecitabine group (P = 0.077). The most frequent toxicities were leukopenia, neutropenia and diarrhoea. In both the unadjusted analysis and PS-adjusted analysis, compared to capecitabine, triweekly XELOX did not increase the risk of severe acute toxicities during CRT (hazard ratio (HR) =1.447, 95% confidence interval (CI): 0.907–2.309, P = 0.121, and HR = 1.392, 95% CI: 0.760–2.550, P = 0.284, respectively) (Table 4).
The total rate of postoperative complications was 17.7% (49/277) with triweekly XELOX versus 12.2% (65/533) with capecitabine (P = 0.033). Pulmonary infection (5.4%) and bowel obstruction (4.3%) were more common with the triweekly XELOX regimen, whereas abdominal or pelvic infection (3.9%) was more frequent with the capecitabine regimen. Triweekly XELOX increased the risk of postoperative complications (HR = 1.574, 95% CI: 1.034–2.316, P = 0.034) in the unadjusted cohort (Table 4). After PS adjustment, the difference disappeared.
Discussion
Oxaliplatin is a common regimen added to capecitabine in the preoperative treatment of rectal cancer; however, the clinical value of oxaliplatin remains controversial. Several randomized clinical trials have attempted to compare capecitabine (fluorouracil) with or without oxaliplatin for preoperative chemoradiotherapy of LARC, and they have shown that double regimens did not improve tumour response or patient survival [10, 11, 13, 22]. A meta-analysis by Hoerndervangers reported that the addition of oxaliplatin might result in more pCR, but this benefit does not translate into less local recurrence or improved survival [23]. Notably, the oxaliplatin mentioned above was administered weekly at a low dose of 50 mg/m2, which was thought to act as a radiation sensitizer. Fluorouracil-based chemoradiotherapy and optimal TME surgery have already maximized local tumour control to more than 90% [2, 4, 9], and there is little or no room for further improvement with the incorporation of additional radiosensitizing agents. Distant metastasis becomes the main failure. CAO/ARO/AIO/04 showed that the gain of chemotherapy seen in the risk of distant metastasis occurred early in the course of treatment [9]. Hence, it could naturally be inferred that a full dose of oxaliplatin (130 mg/m2/triweekly), which has a different biologic mechanism compared to a low dose of oxaliplatin (50–60 mg/m2/weekly), combined with capecitabine during preoperative chemoradiotherapy could ameliorate long-term outcomes.
Our study was the first to compare the triweekly XELOX regimen with the capecitabine regimen during preoperative chemoradiotherapy for LARC. A previous study reported that an intensified full dose of XELOX concomitant with preoperative radiotherapy provided a promising long-term oncologic outcome for LARC patients, with 5-year DFS and OS of approximately 80% [14, 15]. In the present study, compared to the capecitabine regimen, intensified chemotherapy with triweekly XELOX offered no statistically significant survival advantage. The pathological responses to preoperative chemoradiotherapy were also equally distributed among patients treated with or without oxaliplatin, excluding even minor and/or qualitative effects. Sensitivity analysis confirmed the consistent results that no survival benefits were observed with the use of triweekly XELOX in patients treated with adjuvant chemotherapy or patients treated during 2012–2017. CAO/ARO/AIO/04 showed that the benefit of adding oxaliplatin was observed in patients with clinical cN- rather than cN1–2 and in patients with pathological stage I and II disease, rather than ypCR or stage III disease [9]. We performed sensitivity analysis in the subgroup patients mentioned above, and still not found survival improvement with the use of triweekly XELOX.
The results were unexpected. Theoretically, intensified treatment would further downstage the tumour and nodal disease prior to surgery and/or target potential micrometastatic disease [23, 24]. STAR 01 reported a lower frequency of extrapelvic metastases found at surgery in patients treated with oxaliplatin [10]. Jiao et al. reported that adding oxaliplatin to capecitabine-based preoperative chemoradiotherapy could significantly reduce metastasis by 11.66% (P = 0.045) [8]. Meta-analyses provided further evidence for the application of oxaliplatin [25, 26]. One possible reason for the negative results of the current study was that the number of retrieved lymph nodes in the triweekly XELOX group was significantly smaller than that in the capecitabine group (11 vs. 13, SMD = − 0.291). A greater number of nodes increases the likelihood of proper staging [27, 28]. Some patients who might benefit from adjuvant therapy are misclassified as node-negative due to incomplete sampling of lymph nodes [29]. Kenjiro Kotake et al. reported that, with one increase in the number of lymph nodes retrieved, the mortality risk decreased by 2.1% for Stage II and by 0.8% for Stage III [30]. Previous studies have also reported that the number of retrieved lymph nodes was an important prognostic factor [31, 32], although in the present study, examination of at least 12 lymph nodes did not significantly improve survival. The detection of lymph nodes was associated with the experience of the surgeon and the pathologist, as well as the preoperative treatment. Neoadjuvant therapy can lead to a significantly decreased number of lymph nodes detected due to radiation-induced fibrosis, lymphocyte depletion, tissue contraction, adipocyte replacement and interstitial atrophy, making it more difficult to detect lymph nodes during surgery or pathological examination [33, 34]. Previous criteria for dissecting at least 12 lymph nodes might not be suitable for patients who receive neoadjuvant CRT. The minimal number of lymph nodes for patients who receive neoadjuvant CRT has still acquired a consensus. Further large trials are needed to validate the effects of retrieved lymph nodes. Another possible reason was that the treatment era was different between the two groups. A total of 27.2% of patients in the triweekly XELOX group received treatment in the earlier years (2007–2011), while only 0.6% of patients in the capecitabine group were treated at that time. This difference might somewhat offset the benefits of triweekly XELOX since the technique level of radiotherapy and surgery evolved over time. The FOWARC trial conducted in China investigated the addition of relatively high-dose oxaliplatin (85 mg/m2) to neoadjuvant fluorouracil-based chemoradiotherapy and reported similar results; no improvement in DFS or OS was shown among patients with stage II to III rectal cancer despite a higher pCR rate [35].
The cumulative dose of oxaliplatin has been reported to affect patient survival. Chang et al. reported that patients with a cumulative dose of oxaliplatin less than 460 mg/m2 had poorer OS, DMFS and DFS [36]. The CAO/ARO/AIO-04 trial added oxaliplatin both to preoperative chemoradiotherapy and adjuvant chemotherapy, had a high cumulative dose of oxaliplatin of 1000 mg/m2, and significantly increased both the pCR rate and 3-year DFS by a rate of 4% [9]. Similarly, a trial by Jiao et al. and the ADORE trial administered cumulative doses of oxaliplatin with 680 mg/m2 and 750–920 mg/m2, respectively, showing superiority of adding the oxaliplatin regimen in reducing distant metastasis and ameliorating DFS at 3 years [8, 37]. The trials that reported negative results had a relatively low cumulative dose of oxaliplatin (STAR-01360 mg/m2, ACCORD12 250 mg/m2 and NSABP R-04250 mg/m2). In our study, the cumulative dose of oxaliplatin in the triweekly XELOX group was similar to that in the ACCORD12 and NASABP-04 l trials (260 mg/m2), inducing consistently negative results with these trials.
To achieve better tumour control, oncologists have been exploring new neoadjuvant treatment strategies. An alternative strategy known as total neoadjuvant therapy was deemed to be a promising strategy in LARC [38]. Two recently reported phase III, randomized trials, PRODIGE 23 [39] and RAPIDO [40], reported that adding neoadjuvant chemotherapy to either standard short-course radiation or standard long-course chemoradiation in LARC patients reduced the risk of metastasis and significantly delayed disease-related treatment failure and increased disease-free survival. In an attempt to intensify neoadjuvant treatment, additional agents, such as bevacizumab, cetuximab, and veliparib, were also tested but failed to achieve their primary endpoints or led to higher rates of postoperative complications [41].
In both the full cohort and the PS-adjusted cohort, the addition of oxaliplatin did not significantly increase the risks of severe acute toxicities during CRT or postoperative complications. In our study, the incidence of grade 3–4 toxicities was very low (2.3%) in the triweekly XELOX group. In contrast, previous studies have shown that the addition of oxaliplatin to preoperative chemoradiotherapy significantly worsened toxicity, with grade 3–4 toxicity rates of up to 21.4–49.1% [8,9,10, 23]. The difference between our study and previous studies was that the oxaliplatin used in the previous studies was a weekly regimen for 5–6 cycles. This regimen suggested that oxaliplatin administered triweekly for 2 cycles was less toxic and more tolerable than the weekly strategy. Triweekly regimen administration compared to weekly regimen administration has been reported to reduce toxicities for several chemotherapeutic drugs, such as triweekly cisplatin in head and neck cancer and triweekly paclitaxel in breast cancer [42, 43]. A possible explanation was that, compared to the weekly dose-dense administration, there was a longer time free from chemotherapy in the triweekly regimen treatment strategy. During this period, patients could recover from the side effects.
The major deficiency of this study is its retrospective nature. Considerable heterogeneity between patients undergoing triweekly XELOX or standard capecitabine therapy could have influenced both primary outcomes. To control for bias, we performed PS analysis, and comparison of baseline characteristics showed that the patients in the two groups were comparable. Another limitation was that the time of follow-up was not sufficient in the capecitabine group. Considering the favourable prognosis of rectal cancer, further follow-up is needed to fully evaluate the long-term survival and late toxicities.
Conclusions
Although not conclusive, our study showed that neoadjuvant chemoradiotherapy with triweekly XELOX did not improve pathological response or long-term outcomes relative to capecitabine alone in locally advanced rectal cancer. Adding the full dose of oxaliplatin to capecitabine did not increase acute toxicities or postoperative complications. A large, randomized, phase III trial is warranted to confirm the safety and efficacy of the triweekly XELOX regimen.
Availability of data and materials
All data generated or analysed during this study are included in the supplementary information files.
Abbreviations
- LARC:
-
Locoregionally advanced rectal cancer
- XELOX:
-
Oxaliplatin and capecitabine
- PS:
-
Propensity scores
- DMFS:
-
Distant metastasis-free survival
- OS:
-
Overall survival
- DFS:
-
Disease-free survival
- pCR:
-
Pathological complete response
- CRT:
-
Chemoradiotherapy
- TME:
-
Total mesorectal excision
- UICC/AJCC:
-
Union against Cancer/American Joint Committee on Cancer
- 3D-CRT:
-
3-dimensional conformal radiotherapy
- IMRT:
-
Intensity modulated radiotherapy
- Gray:
-
Gy
- RCRG:
-
Rectal cancer regression grade
- RT:
-
Radiotherapy
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
- SMD:
-
Standardized mean difference
References
Bosset JF, Collette L, Calais G, Mineur L, Maingon P, Radosevic-Jelic L, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006;355(11):1114–23.
van Gijn W, Marijnen CA, Nagtegaal ID, Kranenbarg EM, Putter H, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 2011;12(6):575–82.
Zorcolo L, Rosman AS, Restivo A, Pisano M, Nigri GR, Fancellu A, et al. Complete pathologic response after combined modality treatment for rectal cancer and long-term survival: a meta-analysis. Ann Surg Oncol. 2012;19(9):2822–32.
Sauer R, Liersch T, Merkel S, Fietkau R, Hohenberger W, Hess C, et al. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol. 2012;30(16):1926–33.
Andre T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004;350(23):2343–51.
Andre T, Boni C, Navarro M, Tabernero J, Hickish T, Topham C, et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol. 2009;27(19):3109–16.
Yothers G, O'Connell MJ, Allegra CJ, Kuebler JP, Colangelo LH, Petrelli NJ, et al. Oxaliplatin as adjuvant therapy for colon cancer: updated results of NSABP C-07 trial, including survival and subset analyses. J Clin Oncol. 2011;29(28):3768–74.
Dexin JRZZ. Fluorouracil-based preoperative chemoradiotherapy with or without oxaliplatin for stage II/III rectal cancer:a 3-year follow-up study. Chin J Cancer Res. 2015;27(6):588–96.
Rödel C, Graeven U, Fietkau R, Hohenberger W, Hothorn T, Arnold D, et al. Oxaliplatin added to fluorouracil-based preoperative chemoradiotherapy and postoperative chemotherapy of locally advanced rectal cancer (the German CAO/ARO/AIO-04 study): final results of the multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2015;16(8):979–89.
Aschele C, Cionini L, Lonardi S, Pinto C, Cordio S, Rosati G, et al. Primary tumor response to preoperative Chemoradiation with or without Oxaliplatin in locally advanced rectal Cancer: pathologic results of the STAR-01 randomized phase III trial. J Clin Oncol. 2011;29(20):2773–80.
Gerard JP, Azria D, Gourgou-Bourgade S, Martel-Lafay I, Hennequin C, Etienne PL, et al. Clinical outcome of the ACCORD 12/0405 PRODIGE 2 randomized trial in rectal cancer. J Clin Oncol. 2012;30(36):4558–65.
Allegra CJ, Yothers G, O'Connell MJ, Beart RW, Wozniak TF, Pitot HC, et al. Neoadjuvant 5-FU or Capecitabine plus radiation with or without Oxaliplatin in rectal Cancer patients: a phase III randomized clinical trial. J Natl Cancer Inst. 2015;107(11):djv248.
Schmoll HJ, Stein A, Hofheinz RD, Price TJ, Nordlinger B, Daisne JF, et al. 467PD - preoperative chemoradiotherapy and postoperative chemotherapy with capecitabine and oxaliplatin vs. capecitabine alone in locally advanced rectal cancer: final analyses. Ann Oncol. 2016;27:i154.
Zheng R, Lian S, Huang X, Guan G, Li X, Chi P, et al. The survival benefit of intensified full-dose XELOX chemotherapy concomitant to radiotherapy and then resting-period consolidation chemotherapy in locally advanced rectal cancer. J Cancer. 2019;10(3):730–6.
Tang J, Wu X, Bai Y, Gao Y, Jiang W, Kong L, et al. Long-term outcome of Oxaliplatin and Capecitabine (XELOX) concomitant with Neoadjuvant radiotherapy and extended to the resting period in high risk locally advanced rectal Cancer. J Cancer. 2018;9(8):1365–70.
Xu B, Guo Y, Chen Y, Lu H, Tang T, Yue Z, et al. Is the irradiated small bowel volume still a predictor for acute lower gastrointestinal toxicity during preoperative concurrent chemo-radiotherapy for rectal cancer when using intensity-modulated radiation therapy? Radiat Oncol. 2015;10:257.
Wheeler JM, Warren BF, Mortensen NJ, Ekanyaka N, Kulacoglu H, Jones AC, et al. Quantification of histologic regression of rectal cancer after irradiation: a proposal for a modified staging system. Dis Colon Rectum. 2002;45(8):1051–6.
Webster-Clark M, Sturmer T, Wang T, Man K, Marinac-Dabic D, Rothman KJ, et al. Using propensity scores to estimate effects of treatment initiation decisions: state of the science. Stat Med. 2021;40(7):1718–35.
Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399–424.
Austin PC. The use of propensity score methods with survival or time-to-event outcomes: reporting measures of effect similar to those used in randomized experiments. Stat Med. 2014;33(7):1242–58.
Haukoos JS, Lewis RJ. The propensity score. JAMA. 2015;314(15):1637–8.
O'Connell MJ, Colangelo LH, Beart RW, Petrelli NJ, Allegra CJ, Sharif S, et al. Capecitabine and oxaliplatin in the preoperative multimodality treatment of rectal cancer: surgical end points from National Surgical Adjuvant Breast and bowel project trial R-04. J Clin Oncol. 2014;32(18):1927–34.
Hoendervangers S, Burbach J, Lacle MM, Koopman M, van Grevenstein W, Intven M, et al. Pathological complete response following different Neoadjuvant treatment strategies for locally advanced rectal Cancer: a systematic review and Meta-analysis. Ann Surg Oncol. 2020;27(11):4319–36.
Nussbaum N, Altomare I. The neoadjuvant treatment of rectal cancer: a review. Curr Oncol Rep. 2015;17(3):434.
De Felice F, Benevento I, Magnante AL, Musio D, Bulzonetti N, Caiazzo R, et al. Clinical benefit of adding oxaliplatin to standard neoadjuvant chemoradiotherapy in locally advanced rectal cancer: a meta-analysis : Oxaliplatin in neoadjuvant treatment for rectal cancer. BMC Cancer. 2017;17(1):325.
Zheng J, Feng X, Hu W, Wang J, Li Y. Systematic review and meta-analysis of preoperative chemoradiotherapy with or without oxaliplatin in locally advanced rectal cancer. Medicine (Baltimore). 2017;96(13):e6487.
Jin M, Frankel WL. Lymph node metastasis in colorectal Cancer. Surg Oncol Clin N Am. 2018;27(2):401–12.
Awwad GE, Tou SI, Rieger NA. Prognostic significance of lymph node yield after long-course preoperative radiotherapy in patients with rectal cancer: a systematic review. Color Dis. 2013;15(4):394–403.
Tepper JE, O'Connell MJ, Niedzwiecki D, Hollis D, Compton C, Benson AR, et al. Impact of number of nodes retrieved on outcome in patients with rectal cancer. J Clin Oncol. 2001;19(1):157–63.
Kotake K, Honjo S, Sugihara K, Hashiguchi Y, Kato T, Kodaira S, et al. Number of lymph nodes retrieved is an important determinant of survival of patients with stage II and stage III colorectal cancer. Jpn J Clin Oncol. 2012;42(1):29–35.
Park IJ, Yu CS, Lim SB, Yoon YS, Kim CW, Kim TW, et al. Prognostic implications of the number of retrieved lymph nodes of patients with rectal cancer treated with preoperative chemoradiotherapy. J Gastrointest Surg. 2014;18(10):1845–51.
Vergara-Fernandez O, Navarro-Navarro A, Rangel-Rios HA, Salgado-Nesme N, Reyes-Monroy JA, Velazquez-Fernandez D. Oncological implications of lymph nodes retrieval and Perineural invasion in colorectal Cancer: outcomes from a referral center. Rev Investig Clin. 2018;70(6):291–300.
Damin DC, Rosito MA, Contu PC, Tarta C, Ferreira PR, Kliemann LM, et al. Lymph node retrieval after preoperative chemoradiotherapy for rectal cancer. J Gastrointest Surg. 2012;16(8):1573–80.
Fajardo LF. Effects of ionizing radiation on lymph nodes. A review. Front Radiat Ther Oncol. 1994;28:37–45.
Deng Y, Chi P, Lan P, Wang L, Chen W, Cui L, et al. Neoadjuvant modified FOLFOX6 with or without radiation versus fluorouracil plus radiation for locally advanced rectal Cancer: final results of the Chinese FOWARC trial. J Clin Oncol. 2019;37(34):3223–33.
Chang H, Tao YL, Jiang W, Chen C, Liu SL, Ye WJ, et al. Optimize the dose of oxaliplatin for locally advanced rectal cancer treated with neoadjuvant chemoradiotherapy followed by radical surgery and adjuvant chemotherapy. BMC Cancer. 2020;20(1):498.
Hong YS, Nam BH, Kim KP, Kim JE, Park SJ, Park YS, et al. Oxaliplatin, fluorouracil, and leucovorin versus fluorouracil and leucovorin as adjuvant chemotherapy for locally advanced rectal cancer after preoperative chemoradiotherapy (ADORE): an open-label, multicentre, phase 2, randomised controlled trial. Lancet Oncol. 2014;15(11):1245–53.
Kasi A, Abbasi S, Handa S, Al-Rajabi R, Saeed A, Baranda J, et al. Total Neoadjuvant therapy vs standard therapy in locally advanced rectal Cancer: a systematic review and Meta-analysis. JAMA Netw Open. 2020;3(12):e2030097.
Conroy T, Bosset JF, Etienne PL, Rio E, Francois E, Mesgouez-Nebout N, et al. Neoadjuvant chemotherapy with FOLFIRINOX and preoperative chemoradiotherapy for patients with locally advanced rectal cancer (UNICANCER-PRODIGE 23): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(5):702–15.
van der Valk M, Marijnen C, van Etten B, Dijkstra EA, Hilling DE, Kranenbarg EM, et al. Compliance and tolerability of short-course radiotherapy followed by preoperative chemotherapy and surgery for high-risk rectal cancer - results of the international randomized RAPIDO-trial. Radiother Oncol. 2020;147:75–83.
Ominelli J, Valadao M, Araujo R, Cristina DMA, Araujo LH. The evolving field of Neoadjuvant therapy in locally-advanced rectal Cancer: evidence and prospects. Clin Colorectal Cancer. 2021;20:288–98.
Geeta SN, Padmanabhan TK, Samuel J, Pavithran K, Iyer S, Kuriakose MA. Comparison of acute toxicities of two chemotherapy schedules for head and neck cancers. J Cancer Res Ther. 2006;2(3):100–4.
Katsumata N, Yasuda M, Takahashi F, Isonishi S, Jobo T, Aoki D, et al. Dose-dense paclitaxel once a week in combination with carboplatin every 3 weeks for advanced ovarian cancer: a phase 3, open-label, randomised controlled trial. Lancet. 2009;374(9698):1331–8.
Acknowledgments
We thank Shunan Qi, Doctor of Cancer Hospital Chinese Academy of Medical Sciences, for her guidance in statistical analysis and paper writing and thank Professor Jinhua Chen, a statistician working in the Department of Medical Administration in Fujian Medical University Union Hospital, for his reviewing for our data and statistical analysis.
Funding
This work was supported by the Fujian Provincial Science and Technology Innovation Joint Foundation Leading Program (Grant number 2017Y9026), the National Natural Science Foundation, China (Grant number 82173452), and the National Natural Science Foundation of Fujian Province, China (Grant number 2020 J011014).
Author information
Authors and Affiliations
Contributions
Anchuan Li and Rong Zheng collected the data, did statistical analysis and wrote the manuscript. Chi Pan collected the data and helped to write the manuscript. Zhihua Li did statistical analysis and prepared Fig. 4. Benhua Xu contributed to the study conception and design, helped write the manuscript. Tingxuan Huang contributed in data analysis and helped to response to the reviewer comments. Xiaozhong Wang contributed in statistical validation and guided to response to the reviewer comments and revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Consent to publication
Not applicable.
Ethics approval and consent to participate
The retrospective study was approved by the Clinical Research Ethics Committee of the Fujian Medical University Union Hospital. Due to the retrospective nature of the study, the Clinical Research Ethics Committee of the Fujian Medical University Union Hospital waived the informed consent.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure 1.
Unadjusted survival curves of all patients when the cutoff value of the total number of lymph nodes retrieved was 12. a) Unadjusted analysis of distant metastasis-free survival; b) Unadjusted analysis of disease-free survival; c) Unadjusted analysis of overall survival. Figure 2. Propensity score (PS)-matched survival curves of patients when the cutoff value of the total number of lymph nodes retrieved was 12. a) Unadjusted analysis of distant metastasis-free survival; b) Unadjusted analysis of disease-free survival; c) Unadjusted analysis of overall survival.
Additional file 2.
Original data.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Li, A., Huang, T., Zheng, R. et al. Preoperative chemoradiotherapy with capecitabine and triweekly oxaliplatin versus capecitabine monotherapy for locally advanced rectal cancer: a propensity-score matched study. BMC Cancer 22, 789 (2022). https://doi.org/10.1186/s12885-022-09855-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12885-022-09855-z