Abstract
Background
Chronic rhinosinusitis (CRS) increases the risk of developing nasopharyngeal carcinoma (NPC) while nasopharyngeal reflux is known to be one of the major aetiological factors of CRS. Bile acid (BA), the component of gastric duodenal contents, has been recognised as a carcinogen. BA-induced apoptosis was suggested to be involved in human malignancies. Cells have the potential and tendency to survive apoptosis. However, cells that evade apoptosis upon erroneous DNA repair may carry chromosome rearrangements. Apoptotic nuclease, caspase-activated deoxyribonuclease (CAD) has been implicated in mediating translocation in leukaemia. We hypothesised that BA-induced apoptosis may cause chromosome breaks mediated by CAD leading to chromosome rearrangement in NPC. This study targeted the AF9 gene located at 9p22 because 9p22 is one of the most common deletion sites in NPC.
Methods
We tested the ability of BA at neutral and acidic pH in inducing phosphatidylserine (PS) externalisation, reactive oxygen species (ROS) production, mitochondrial membrane potential (MMP) disruption, and caspase 3/7 activity in normal nasopharyngeal epithelial (NP69) and NPC (TWO4) cells. Inverse-PCR (IPCR) was employed to detect AF9 gene cleavages. To investigate the role of CAD in mediating these cleavages, caspase inhibition was performed. IPCR bands representing AF9 cleaved fragments were sequenced.
Results
BA-treated cells showed higher levels of PS externalisation, ROS production, MMP loss and caspase 3/7 activity than untreated control cells. The effect of BA in the induction of these intracellular events was enhanced by acid. BA at neutral and acidic pH also induced significant cleavage of the AF9 gene. These BA-induced gene cleavages were inhibited by Z-DEVD-FMK, a caspase-3 inhibitor. Intriguingly, a few chromosome breaks were identified within the AF9 region that was previously reported to participate in reciprocal translocation between the mixed lineage leukaemia (MLL) and AF9 genes in an acute lymphoblastic leukaemia (ALL) patient.
Conclusions
These findings suggest a role for BA-induced apoptosis in mediating chromosome rearrangements in NPC. In addition, CAD may be a key player in chromosome cleavages mediated by BA-induced apoptosis. Persistent exposure of sinonasal tract to gastric duodenal refluxate may increase genomic instability in surviving cells.
Similar content being viewed by others
Background
Nasopharyngeal carcinoma (NPC) is a malignant tumour derived from the nasopharyngeal epithelium. The development of NPC is a multi-step process. NPC pathogenesis is related to Epstein-Barr virus (EBV) infection [1], consumption of preserved foods [2], exposures to wood dust and industrial heat [3], epigenetic changes [4] and genetic predisposition [5]. Moreover, chronic inflammation of sinonasal tract and/or ear (rhinitis, sinusitis, postnasal drip, otitis media) is increasingly being recognised as one of the risk factors for NPC [6,7,8,9,10,11,12]. More recently, a retrospective cohort study involving 231,490 Taiwan Chinese individuals revealed that individuals with chronic rhinosinusitis (CRS) have a 3.55-fold higher risk of developing NPC as compared with those without CRS [12]. Similar findings had been found in a case-control study which comprised 2242 NPC patients and 6726 controls; the odd ratio of prior CRS for subjects with NPC is 3.83 (95% confidence interval [CI], 3.23–4.53) [11]. The investigators attributed these findings to the chronic inflammation which may predispose the nasopharyngeal mucosa to transformation by carcinogens [7,8,9, 11, 12]. However, the causative factors and the underlying mechanisms which contribute to the chronic inflammation of nose or ear leading to NPC remain enigmatic.
As has been addressed by several reviews, chronic inflammation of sinonasal tract or ear is one of the extraoesophageal manifestations of gastro-oesophageal reflux disease (GORD) [13,14,15,16,17]. The flowing back of gastric duodenal contents into the oesophagus results in GORD. GORD is usually presented with symptoms such as heartburn and acid regurgitation. Apart from these classical symptoms, the gastric duodenal refluxate may flow beyond the oesophagus and affect the airways (tracheobronchopulmonary tree, larynx, pharynx, sinonasal tract and middle ear) [15]. These atypical manifestations of GORD are not only classified as extraoesophageal reflux (EOR) but also as laryngopharyngeal reflux, supra-oesophageal reflux, gastro-oesophago-pharyngeal reflux or gastro-oesophago-laryngeal reflux [15, 18]. It is sometimes called ‘silent reflux’ as more than half of the patients with EOR may not show ‘typical GORD’ symptoms (heartburn and acid regurgitation) [16].
GORD is an important factor in the pathogenesis of various inflammatory diseases. These diseases include gastritis [19, 20], oesophagitis [21,22,23], laryngitis [24,25,26], pharyngitis [27, 28], post-nasal drip [29], otitis media [30,31,32,33] and asthma [34,35,36]. Furthermore, CRS, which is the inflammation of the nose and paranasal sinuses, has also been included in this growing list [28, 37,38,39]. A high prevalence of GORD has been reported in children [40, 41] and adults [37, 42] with CRS. One possible underlying mechanism by which acid pharyngeal reflux may affect the sinonasal cavity is that, gastric duodenal contents reflux into the nasopharynx and the posterior nasal passages; direct contact of gastric duodenal contents on nasal mucosa causes inflammation and oedema. Subsequently, these phenomena lead to sinus ostial obstruction and sinusitis [43]. Reflux to the nasopharynx has been documented in both paediatric [40, 41, 44, 45] and adult groups [29, 37, 38, 46]. In addition, GORD treatment has improved or resolved the sinus symptoms in most patients with medically or surgically refractory CRS [46, 47].
Apart from being a risk factor for inflammatory disorders, GOR has also been implicated in several malignancies. Indeed, chronic inflammation has been well recognised as a strong factor in carcinogenesis [48]. GORD-related cancers include stomach cancer [49, 50], oesophageal adenocarcinoma [51, 52], laryngeal cancer [53], pharyngeal cancer [54] and lung cancer [55]. Previous studies have also shown that GOR is strongly linked to Barrett’s oesophagus [56, 57], a preneoplastic disorder of the oesophagus [58].
Acid refluxate consists of pancreatic fluids, hydrochloric acid, pepsin and bile acids (BA) [59]. BA has been recognised as a carcinogen in malignancies of the gastrointestinal tract. These include malignancies of the oesophagus, stomach, small intestine, liver, biliary tract, pancreas, colon and rectum (reviewed in [60]). BA has also been known to be a potent apoptotic inducer in human colon adenocarcinoma cells [61] and human oesophageal adenocarcinoma cells [62]. Furthermore, BA-induced apoptosis has been well implicated in the pathogenesis of Barrett’s oesophagus, oesophagus adenocarcinoma and colon cancer [63,64,65].
Apoptosis is a type of genetically controlled cell suicide programme [66]. In non-apoptotic cells, caspase-activated deoxyribonuclease (CAD) exists as a heterodimer with the inhibitor of CAD (ICAD). ICAD possess two caspase-3 cleavage sites. When caspase-3 is activated by apoptotic stimuli, it cleaves ICAD at the two caspase-3 cleavage sites and thus releases CAD [67, 68]. This allows CAD to oligomerise and form a large functional complex which cleaves DNA [69].
Although apoptosis is a programmed cell death process, cells may recover through DNA repair [70]. However, erroneous DNA repair may cause cells that survive apoptosis to harbour certain types of chromosome rearrangements, such as chromosome translocation and deletion [71]. The initial event of chromosome rearrangement is chromosome breakage. The apoptotic nuclease CAD has been found to be involved in these chromosome breakage events in both leukaemia [70, 72,73,74] and NPC [75,76,77]. Based on the literature, GORD is a contributing factor to CRS while CRS is a precursor of NPC. At the same time, BA from GORD can induce oxidative stress as well as apoptosis. Oxidative stress can also induce apoptosis resulting in chromosome breaks in cultured nasopharyngeal epithelial cells [77]. Since CAD has been implicated in mediating the chromosome breakage events in both leukaemia and NPC chromosome rearrangements, we thus hypothesise that BA-induced apoptosis may cause chromosome breaks by CAD leading to chromosome rearrangement in NPC, and this process may involve induction of oxidative stress. This study focuses on the AF9 gene which is located at 9p22 because 9p22 is one of the deletion hotspots in NPC [78].
In this study, we report that BA induced PS externalisation, an early event of apoptosis, in normal nasopharyngeal epithelial and NPC cells. We demonstrated that BA-induced apoptosis triggered mitochondrial membrane potential (MMP) disruption, increased oxidative stress and activated caspase. Our findings also showed that these intracellular events were enhanced by acid. We further demonstrated that BA-induced apoptosis resulted in chromosome breaks within the AF9 gene. These chromosome breaks were inhibited by caspase inhibitor (CI), suggesting that CAD may be the major player in mediating these chromosome breaks. Interestingly, a few breakpoints were the same as those reported in the mixed lineage leukaemia (MLL)-AF9 fusion gene in an acute lymphoblastic leukaemia (ALL) patient. Lastly, we propose a potential schema for BA-induced apoptosis in mediating the chromosome breakages leading to chromosome rearrangements in NPC.
Methods
Cell line and chemicals
NP69 normal nasopharyngeal epithelial cell line was a kind gift from Prof. Tsao Sai Wah (The University of Hong Kong, Hong Kong, China) and Prof. Lo Kwok Wai (The Chinese University of Hong Kong, Hong Kong, China). TWO4 NPC cell line was a generous gift from Prof. Sam Choon Kook (formerly from University of Malaya, Malaysia). NP69 is an immortalised nasopharyngeal epithelial cell line which was established by transfection with SV40 large T oncogene. It retains some characteristics of normal nasopharyngeal epithelial cells and is non-tumourigenic. This cell line may provide potential nasopharyngeal epithelial cell model for studying mechanisms involved in the tumourigenesis of NPC [79]. TWO4 was derived from an undifferentiated NPC (WHO Type II B) of a 36-year-old Chinese female patient living in Taiwan [80].
Keratinocyte-SFM medium, RPMI 1640 medium, fetal bovine serum, L-glutamine, penicillin/streptomycin and StemPro ACCUTASE Cell Dissociation Reagent were procured from GIBCO, Invitrogen, USA. Taurocholic acid sodium salt hydrate, sodium glycochenodeoxycholate, glycocholic acid sodium, sodium deoxycholate, sodium glycodeoxycholate, dibasic sodium phosphate and citric acid were bought from Sigma, USA. Caspase-3 inhibitor II (Z-DEVD-FMK) was obtained from Calbiochem, USA. Camptothecin (CPT) was purchased from Santa Cruz Biotechnology, California, USA. 2′,7′-Dichlorofluorescein diacetate (DCFH-DA) was bought from Sigma-Aldrich, Israel. Annexin V-Fluorescein isothiocyanate (FITC) Apoptosis Detection Kit I and Flow Cytometry Mitochondrial Membrane Potential Detection Kit were purchased from Becton Dickinson Biosciences, USA. Caspase-Glo 3/7 Assay Kit was bought from Calbiochem, USA. QIAquick Nucleotide Removal Kit and QIAquick Gel Extraction Kit were procured from QIAGEN, Germany. Ammonium acetate was obtained from Merck, Germany. Phenol and Sodium dodecyl sulfate (SDS) were procured from Amresco, USA. Chloroform was obtained from R&M Chemicals, UK. Isoamyl alcohol was purchased from Fluka, Switzerland. DNA Polymerase I, Large (Klenow) Fragment, T4 DNA Ligase and all the restriction enzymes were obtained from New England Biolabs (NEB), USA. Phusion High-Fidelity DNA Polymerase was purchased from Finnzymes, Finland. PCR primers were purchased from First Base Laboratories. dNTP mix was bought from Promega, USA.
Cell cultures
NP69 cells were grown in Keratinocyte-SFM medium supplemented with 100 U/ml penicillin, 100 μg/ml streptomycin, 2% (v/v) heat-inactivated fetal bovine serum, 4–5 ng/ml recombinant Epidermal Growth Factor (rEGF) and 40–50 μg/ml Bovine Pituitary Extract (BPE). TWO4 cells were cultured in RPMI 1640 medium supplemented with 100 U/ml penicillin, 100 μg/ml streptomycin, 10% (v/v) heat-inactivated fetal bovine serum and 2 mM L-glutamine. Cells were incubated at 37 °C with 5% CO2.
Preparations of BA cocktail and acidified media
The BA cocktail was prepared according to the studies of Dvorak and colleagues [62]. It consists of an equimolar mixture of sodium salts of glycocholic acid, taurocholic acid, glycodeoxycholic acid, glycochenodeoxycholic acid and deoxycholic acid. The BA cocktail used in this study reflects the mixture of BA to which the distal oesophagus is exposed to during GOR [81,82,83]. Total BA concentrations commonly observed in the refluxate of patients with Barrett’s oesophagus fall within the range from 0.03 to 0.82 mM [82]. BA concentrations beyond this range (as high as 7.6 mM) have also been reported in the refluxate of some patients with Barrett’s oesophagus [84]. Therefore, concentrations in the physiological range were used in our studies (from 0.5 to 1.0 mM).
Furthermore, nasopharyngeal pH variations (pH below 4, 5 or 6) were observed in patients with GORD-related nasopharyngitis, CRS and otitis media [38, 41, 44, 85]. In a 24-h pH monitoring study, acidic nasopharyngeal pH was observed in GOR patients with chronic respiratory disease (otitis, sinusitis, laryngitis, epiglottitis, recurrent stridor, asthma and recurrent pneumonia). Nasopharyngeal pH of 5.8 had been considered as the best cut-off point to indicate the presence of abnormal pH-metry in GOR patients with chronic respiratory diseases [85]. Thus, in our study, we tested the apoptosis-inducing effect of BA in NP69 and TWO4 cells at neutral pH (pH 7.4) and acidic pH (pH 5.8). The media used in BA treatment at acidic pH were acidified to pH 5.8 with citrate phosphate buffer.
Flow cytometric analysis of phosphatidylserine (PS) externalisation
NP69 cells (1 × 105) and TWO4 cells (1.5 × 105) were plated in 150 mm culture dishes and allowed to grow for 2 days. NP69 cells were either left untreated or treated with 0.5 mM of BA cocktail at pH 7.4 and pH 5.8 for 1 h. NP69 cells treated with 1.0 mM of CPT for 1 h were used to serve as a positive control. TWO4 cells were either left untreated or treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 3 h. TWO4 cells treated with 1.0 mM of CPT for 3 h were included as a positive control. CPT is a well-known apoptotic inducer. It has been shown that 2–10 μM of CPT with an exposure time of 24 h can induce apoptosis in NPC cells [86, 87]. After exposure, StemPro ACCUTASE Cell Dissociation Reagent was used to collect the cells. Using Annexin V-FITC Apoptosis Detection Kit I, the harvested cells were subjected to analysis of PS externalisation by a flow cytometer (FACSCalibur, Becton–Dickinson, USA) as previously described [77]. This analysis was performed in duplicate.
Measurement of reactive oxygen species (ROS)
The intracellular ROS was determined by employing 2′,7′-dichlorofluorescein diacetate (DCFH-DA), a nonpolar, cell-permeable fluorescent probe described by Bass et al. (1983). DCFH-DA is nonfluorescent until hydrolysed by intracellular esterase and oxidised by ROS. Upon cleavage of the acetate groups by intracellular esterase, DCFH-DA is hydrolysed to polar dichlorofluorescein (DCFH) which remains trapped in viable cells. The nonfluorescent DCFH is rapidly oxidised to highly fluorescent dichlorofluorescein (DCF) in the presence of ROS [88]. NP69 (2 × 103 cells per well) and TWO4 cells (3 × 103 cells per well) were seeded in a 96-well black plate and allowed to adhere overnight. NP69 cells were left untreated or treated with 0.5 and 1.0 mM of BA cocktail at pH 7.4 and pH 5.8 for 1 h. NP69 cells treated with 20 mM of H2O2 for 1 h were used as a positive control. TWO4 cells were left untreated or treated with 0.5 and 1.0 mM of BA cocktail at pH 7.4 and pH 5.8 for 3 h. TWO4 cells treated with 5 mM of H2O2 for 3 h were included as a positive control. After exposure, media was removed and the cells were incubated with media containing 10 μM DCFH-DA for 30 min at 37 °C. The cells were then washed once with culture media. Fluorescence intensity was measured using a microplate reader (Tecan Infinite 200 Pro, Austria), with excitation at 485 nm and emission at 538 nm. This assay was carried out in triplicate. The cells were then subjected to fluorescence microscopic analysis under an inverted fluorescence microscope (IX 71, Olympus, Japan). Morphology of cells was photographed using microscope digital camera (DP72, Olympus, Japan).
Flow cytometric analysis of mitochondrial membrane potential (MMP) disruption
NP69 cells (1 × 105) and TWO4 cells (1.5 × 105) were plated in 150 mm culture dishes and allowed to grow for 2 days. NP69 cells were either left untreated or treated with 0.5 mM of BA cocktail at pH 7.4 and pH 5.8 for 1 h. NP69 cells treated with 1.0 mM of CPT for 1 h were used to serve as a positive control. TWO4 cells were either left untreated or treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 3 h. TWO4 cells treated with 1.0 mM of CPT for 3 h were included as a positive control. Using Flow Cytometry Mitochondrial Membrane Potential Detection Kit, the collected cells were subjected to analysis of MMP disruption by a flow cytometer (FACSCalibur, Becton–Dickinson, USA) as previously described [77]. This assay was carried out in duplicate.
Measurement of Caspase-3/7 activity
NP69 (2 × 103 cells per well) and TWO4 cells (3 × 103 cells per well) were seeded in a white-walled 96-well plate and allowed to adhere overnight. The NP69 and TWO4 cells were either left untreated or pretreated with 50 μM of Z-DEVD-FMK (Caspase-3 Inhibitor II) for 1 h. Subsequently, the NP69 cells were either left untreated or cotreated for 1 h with 0.5 mM of BA cocktail, at pH 7.4 and pH 5.8, while the TWO4 cells were left untreated or cotreated for 3 h with 0.5 mM of BA cocktail, at pH 7.4 and pH 5.8. After exposure, the media was removed and the cells were washed once with 1 x PBS buffer. Using Caspase-Glo 3/7 Assay Kit (Promega), caspase-3/7 activity was determined by a microplate reader (Tecan Infinite 200 Pro, Austria) as previously described [77]. This assay was carried out in duplicate.
Identification of BA-induced chromosome breaks by nested inverse polymerase chain reaction (IPCR)
Apoptosis induction assay
NP69 cells (1.5 × 104) and TWO4 cells (2.5 × 104) were plated in 60 mm culture dishes and allowed to grow for 2 days. NP69 cells were left untreated or treated with 0.5 mM of BA cocktail at pH 7.4 and pH 5.8 for 1 h. TWO4 cells were left untreated or treated with 0.5 mM of BA cocktail at pH 7.4 and pH 5.8 for 3 h.
Genomic DNA extraction
Genomic DNA extraction was carried out using phenol/chloroform/isoamyl alcohol extraction method as previously described [77].
Manipulation of genomic DNA in preparation for nested IPCR
The extracted genomic DNA was manipulated for nested IPCR as previously described [77]. These manipulation steps were depicted in Additional file 1.
Nested IPCR
Using an ultraviolet-visible microvolume spectrophotometer (ND-1000, NanoDrop, USA), optical density (O.D.) of the purified DNA sample was determined. Nested IPCR was performed as previously described [77]. All the IPCR amplifications were carried out using a Veriti 96 Well Thermal Cycler (Applied Biosystems, USA).
Caspase inhibition assay
NP69 cells (1.5 × 104) were plated in 60 mm culture dishes and allowed to grow for 2 days. NP69 cells were either left untreated or pretreated with 50 μM of Z-DEVD-FMK for 1 h. Following that, the NP69 cells were either left untreated or cotreated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 1 h. Subsequently, genomic DNA extraction and IPCR detection of the chromosome breaks were performed as described above.
Agarose gel electrophoresis and DNA sequencing of the cleavage bands
In order to visualise cleaved chromosomes, the IPCR products were analysed on 1% agarose gel and stained with ethidium bromide. To map the breakpoints, IPCR bands representing the AF9 cleaved fragments were excised, cleaned up with QIAGEN QIAquick Gel Extraction Kit and sequenced. The sequencing results were then annotated by blasting the human genome database (Nucleotide BLAST, http://blast.ncbi.nlm.nih.gov/Blast.cgi). By aligning the sequencing data with the AF9 gene sequence retrieved from Ensembl database [EMBL:ENSG00000171843] using Seqman DNASTAR software (Lasergene, USA), the breakpoints of the AF9 cleaved fragments were determined. A genomic map illustrating the breakpoints was constructed.
Quantification of gene cleavage frequency
IPCR amplification was performed in two to five sets per experiment. Each set of IPCR consisted of three to six replicates per cell sample. The gene cleavage frequencies represent the average number of the AF9 cleaved fragments detected in at least three independent experiments.
Statistical analysis
Data were presented as means with standard deviation (SD). Student’s t-test was used to evaluate the significance of differences between the untreated control and the treated groups in flow cytometric analyses of PS externalisation and MMP loss, determination of ROS level, measurement of caspase activity and IPCR assays. All statistical tests were two-sided. Differences were considered statistically significant at p-value < 0.05.
Results
Bile acid induces apoptosis
Bile acid induces phosphatidylserine externalisation
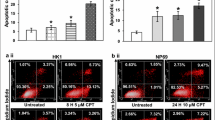
The potential role of BA in inducing apoptosis in NP69 and TWO4 cells was examined by flow cytometric analysis of PS externalisation. NP69 cells were treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 1 h. The percentages of apoptotic cells are 1.2-fold (p-value = 0.009) and 2.8-fold (p-value = 0.005) higher than that of the untreated control, for pH 7.4 and pH 5.8, respectively (Fig. 1a i). Similarly, TWO4 cells were treated with 0.5 mM of BA at pH 5.8 for 3 h. The percentage of apoptotic cells is 2.2-fold (p-value = 0.026) higher than that of the untreated control. However, only a minimal percentage of apoptotic cells were detected in TWO4 cells treated with 0.5 mM of BA at pH 7.4 for 3 h (p-value = 0.541) (Fig. 1b i). Camptothecin (CPT) was used as a positive control. Figures 1a ii and b ii show representative dot plot diagrams indicating the apoptotic populations of BA-treated NP69 and TWO4 cells. Taken together, these data indicate that the combination of BA and acid has a higher apoptotic effect in nasopharyngeal epithelial cells as compared with BA alone.
Bile acid induces phosphatidylserine externalisation in NP69 and TWO4 cells. NP69 cells were either left untreated or treated for 1 h with 0.5 mM of BA at pH 7.4 and pH 5.8. TWO4 cells were either left untreated or treated for 3 h with 0.5 mM of BA at pH 7.4 and pH 5.8. CPT was used as a positive control. After exposure, flow cytometric analysis of PS externalisation was performed in (a i) NP69 and (b i) TWO4 cells. Means and SD of three independent experiments performed in duplicate are shown. Data are expressed as fold increase normalised to the untreated control. Student’s t-test was used for statistical analysis to compare treated groups with the untreated control, * p < 0.05, ** p < 0.01. Representative dot plot diagrams showing the apoptotic populations of BA-treated (a ii) NP69 and (b ii) TWO4 cells detected by Annexin V-FITC and PI staining are shown. The lower left quadrants show viable cells; the lower right quadrants represent early apoptotic cells; the upper right quadrants show late apoptotic and necrotic cells
Bile acid induces mitochondrial membrane potential (MMP) disruption
Changes in MMP were assessed in BA-treated cells using flow cytometry. In NP69 treated for 1 h with 0.5 mM of BA at pH 7.4 and pH 5.8, the percentages of population with MMP disruption are 2.9-fold (p-value = 0.007) and 11.8-fold (p-value = 0.006) higher than that of the untreated control, respectively (Fig. 2a i). As shown in Fig. 2b i, the percentage of population with MMP loss in TWO4 cells treated with 0.5 mM of BA at pH 5.8 for 3 h is 2.1-fold (p-value = 0.009) higher than that in the untreated control. There was no significant change in MMP disruption when TWO4 cells were treated with 0.5 mM of BA at neutral pH (p-value = 0.737). Treatment of NP69 and TWO4 cells with CPT were used as positive controls. Figures 2a ii and b ii show representative contour plot diagrams indicating the populations with disruption of MMP in BA-treated NP69 and TWO4 cells. Collectively, these results show that the combination of BA and acid has a higher effect in triggering MMP loss in nasopharyngeal epithelial cells as compared with BA alone.
Bile acid induces mitochondrial membrane potential disruption in NP69 and TWO4 cells. NP69 cells were either left untreated or treated for 1 h with 0.5 mM of BA at pH 7.4 and pH 5.8. TWO4 cells were either left untreated or treated for 3 h with 0.5 mM of BA at pH 7.4 and pH 5.8. CPT was used as a positive control. After incubation, MMP disruption was assessed by flow cytometry in (a i) NP69 and (b i) TWO4 cells. Data presented are expressed as means and SD of two independent experiments performed in duplicate. The differences between untreated control and treated groups were compared by using Student’s t-test, * p < 0.05, ** p < 0.01. Representative contour plot diagrams showing the apoptotic populations of BA-treated NP69 (a ii) and TWO4 cells (b ii) determined by JC-1 staining are shown. The upper and lower quadrants represent the percentages of viable cells and apoptotic cells, respectively
Bile acid-induced apoptosis involves caspase activity that is reduced by a caspase inhibitor
To examine if BA-induced apoptosis is caspase-dependent, the activity of effector caspases, caspase-3 and caspase-7, was determined in BA-treated NP69 and TWO4 cells. NP69 cells treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 1 h showed 8% (p-value = 0.018) and 92% (p-value < 0.001) higher caspase 3/7 activities than that in the untreated control, respectively (Fig. 3a). The caspase 3/7 activities of TWO4 cells are 15% (p-value = 0.038) and 22% (p-value = 0.043) higher than that of the untreated control after 3 h treatment with 0.5 mM of BA at pH 7.4 and pH 5.8, respectively (Fig. 3b). Pretreatment with Caspase-3 Inhibitor II, Z-DEVD-FMK, significantly reduced caspase 3/7 activity in BA-cotreated NP69 and TWO4 cells. These findings suggest that caspase-3 and caspase-7 are involved in BA-induced apoptosis at both neutral and acidic pH.
Bile acid-induced apoptosis involves caspase activity that is reduced by a caspase inhibitor. NP69 and TWO4 cells were left untreated or pretreated with 50 μM of Z-DEVD-FMK (Caspase-3 Inhibitor II) for 1 h. Subsequently, NP69 cells were treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 1 h, whereas TWO4 were treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 3 h. The activity of caspase-3/7 was measured by using Caspase-Glo 3/7 Assay in (a) NP69 and (b) TWO4 cells. Means and SD of two independent experiments performed in duplicate are shown. Values were normalised to the percentage of untreated control. The differences between the untreated control and treated groups were compared by using Student’s t-test. An asterisk indicates a significant increase in BA-treated cells as compared with the untreated control, * p < 0.05, ** p < 0.01. An open diamond indicates a significant decrease in the sample with CI pretreatment as compared with its corresponding sample without CI pretreatment, ◊ p < 0.05, ◊◊ p < 0.01
Bile acid triggers production of intracellular reactive oxygen species (ROS)
In order to investigate if BA induces intracellular ROS generation, the BA-treated NP69 and TWO4 cells were subjected to ROS measurement and fluorescence microscopic analysis. NP69 cells were treated for 1 h with 0.5 mM of BA at pH 7.4, 1.0 mM of BA at pH 7.4, 0.5 mM of BA at pH 5.8 and 1.0 mM of BA at pH 5.8; the ROS generated are 10% (p-value < 0.001), 13% (p-value < 0.001), 57% (p-value < 0.001) and 28% (p-value = 0.002) higher than that of the untreated control, respectively (Fig. 4a). In TWO4 cells treated for 3 h with 0.5 mM of BA at pH 7.4, 1.0 mM of BA at pH 7.4, 0.5 mM of BA at pH 5.8 and 1.0 mM of BA at pH 5.8, the ROS generated are 2% (p-value < 0.001), 4% (p-value = 0.002), 9% (p-value < 0.001) and 11% (p-value < 0.001) higher than that of the untreated control, respectively (Fig. 5a). A well-known inducer of oxidative stress, hydrogen peroxide (H2O2), was used as a positive control. Representative fluorescence microscopic images showing the BA-treated NP69 and TWO4 cells were shown in Figs. 4b and 5b. These findings suggest that exposure of nasopharyngeal epithelial cells to BA resulted in intracellular ROS generation. Furthermore, the effect of BA on the induction of oxidative stress was enhanced by acid.
Bile acid triggers production of reactive oxygen species in NP69 cells. NP69 cells were treated with 0.5 and 1.0 mM of BA at pH 7.4 and pH 5.8 for 1 h. 20 mM of H2O2 was used as a positive control. After incubation with DCFH-DA, the cells were subjected to determination of ROS level and fluorescence microscopic analysis as described in “Methods” section. a Percentages of increased ROS level in NP69 cells after treatment with BA. Data are means and SD of five independent experiments performed in triplicate. Values are expressed in percentages with respect to values obtained from the untreated control. The differences between the untreated control and treated groups were compared by using Student’s t-test, * p < 0.01, ** p < 0.001. b Representative fluorescence microscopic images showing the BA-treated NP69 cells are shown. Magnification 100X, bar = 200 μm
Bile acid triggers production of reactive oxygen species in TWO4 cells. TWO4 cells were treated with 0.5 and 1.0 mM of BA at pH 7.4 and pH 5.8 for 3 h. 5 mM of H2O2 was included as a positive control. After exposure, ROS determination and fluorescence microscopy analysis were performed as described in “Methods” section. a Percentages of increased ROS generation in BA-treated TWO4 cells. Data are means and SD of five independent experiments carried out in triplicate. Values are presented in percentages with respect to values obtained from the untreated control. The differences between the untreated control and treated groups were compared by using Student’s t-test, * p < 0.01, ** p < 0.001. b Representative fluorescence microscopic images showing the BA-treated TWO4 cells are shown. Magnification 100X, bar = 200 μm
BA-induced apoptosis results in chromosome breaks in the AF9 gene
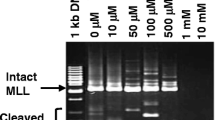
To test if BA-induced apoptosis leads to cleavage in the AF9 gene, BA-treated NP69 and TWO4 cells were subjected to genomic DNA extraction and subsequently IPCR. Two breakpoint cluster regions (BCR) have been reported in the AF9 gene. These two BCRs were denominated as BCR1 and BCR2 [89, 90]. In the present study, nested IPCR was used to detect chromosome breaks within the AF9 BCR1 which is located at the telomeric end of intron 4. Based on the primers position, the intact IPCR product is 944 bp (~ 950 bp). Consequently, IPCR bands smaller than 950 bp will be detected if there is any chromosome breakage within the region of study.
The representative gel pictures indicating AF9 gene cleavages detected in NP69 and TWO4 cells after treatment with BA at pH 7.4 and pH 5.8 are shown in Figs. 6a and 7a, respectively. As assessed by the flow cytometric detection of apoptosis, a minimal amount of dying cells was detected in the untreated sample. These dying cells might undergo spontaneous chromosome breakages contributing to the background. In NP69 cells treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 1 h, the AF9 gene cleavage frequencies are 1.7-fold (p-value = 0.006) and 1.9-fold (p-value = 0.045) higher than that in the untreated cells, respectively (Fig. 6b). Similarly, in TWO4 cells treated with 0.5 mM of BA at pH 7.4 and pH 5.8 for 3 h, the gene cleavage frequencies are 1.8-fold (p-value = 0.004) and 1.6-fold (p-value = 0.036) higher than that in the untreated cells, respectively (Fig. 7b). Our data clearly indicate that BA-induced apoptosis in NP69 and TWO4 cells results in the AF9 gene cleavages at both neutral and acidic pH.
Bile acid induces AF9 gene cleavages in NP69 cells. IPCR was employed to identify the AF9 gene cleavages in NP69 cells after exposed to BA. a Representative gel picture showing the AF9 gene cleavages identified by IPCR. NP69 cells were left untreated (Lanes 1–6) or treated for 1 h with 0.5 mM of BA at pH 7.4 (Lanes 7–12) and pH 5.8 (Lanes 13–18). Genomic DNA extraction and nested IPCR were performed as described in “Methods” section. The side bracket represents the IPCR bands derived from the AF9 cleaved fragments. M: 100 bp DNA marker. N: negative control for IPCR. b Average number of the AF9 gene cleavages identified in BA-treated NP69 cells. Data are expressed as means and SDs of four independent experiments. Each experiment consisted of two to five sets of IPCR carried out in three to six replicates per set for each cell sample. Values are expressed as fold change normalised to the value of the untreated control. The differences between the untreated control and treated groups were compared by using Student’s t-test, * p < 0.05, ** p < 0.01
Bile acid induces AF9 gene cleavages in TWO4 cells. Genomic DNA was extracted from BA-treated TWO4 cells for nested IPCR as described in “Methods” section. a Representative gel picture showing the AF9 gene cleavages detected in BA-treated TWO4 cells. TWO4 cells were left untreated (Lanes 1–6) or treated for 3 h with 0.5 mM of BA at pH 7.4 (Lanes 7–12) and pH 5.8 (Lanes 13–18). The IPCR bands derived from the AF9 cleaved fragments were indicated by the side bracket. M: 100 bp DNA ladder. N: Negative control for IPCR. b Average number of the AF9 gene cleavages detected by IPCR. Data represents means and SDs of three independent experiments. Each experiment consisted of four sets of IPCR assays performed in three to six replicates per set for each cell sample. Values are expressed as fold change normalised to the value of the untreated control. The differences between the untreated control and treated groups were compared by using Student’s t-test, * p < 0.05, ** p < 0.01
Caspase inhibitor reduces AF9 gene cleavages in bile acid-cotreated NP69 cells
To investigate if CAD is responsible for mediating the chromosome cleavages during BA-induced apoptosis, caspase inhibition assay was conducted. In normal healthy cells, CAD exists naturally with its chaperone, ICAD, in the cytoplasm. When apoptosis is induced by its stimulus such as BA, caspase-3 will be triggered and cleaves the ICAD. CAD will be subsequently released from ICAD and enters the nucleus to cleave the chromosomal DNA [67, 68]. DEVD inhibitor was found to be the most effective caspase-3 inhibitor [91]. Thus, if CAD is responsible for mediating chromosome cleavages during BA-induced apoptosis, by inhibiting caspase-3 using DEVD inhibitor, chromosome breaks in BA-treated cells will be reduced, if not totally eliminated.
NP69 cells were either pretreated or left untreated with Z-DEVD-FMK. Subsequently, the cells were either left untreated or cotreated with BA at neutral and acidic pH. After BA treatment, IPCR was performed as described above. The representative gel pictures showing the IPCR results of BA-treated NP69 cells in the absence (Fig. 8a i) and presence (Fig. 8a ii) of Z-DEVD-FMK were shown in Fig. 8. Pretreatment with Z-DEVD-FMK has significantly reduced the gene cleavage frequency for approximately 1.7-fold in NP69 cells treated with BA at neutral pH (p-value = 0.006) and 3.0-fold in NP69 cells treated with BA at acidic pH (p-value = 0.010) (Fig. 8b). These findings together suggest that CAD is an essential player in mediating the chromosome breaks triggered by BA.
Caspase inhibitor reduces AF9 gene cleavages in bile acid-cotreated NP69 cells. a Representative gel pictures showing the IPCR detection of the AF9 gene cleavages in BA-treated NP69 cells: (i) without CI pretreatment (ii) with CI pretreatment. NP69 cells were left untreated or pretreated with 50 μM of Z-DEVD-FMK (Caspase-3 inhibitor II) for 1 h. The NP69 cells were subsequently left untreated (Lanes 1–6) or treated for 1 h with 0.5 mM of BA at pH 7.4 (Lanes 7–12) and pH 5.8 (Lanes 13–18). Genomic DNA was extracted and modified for nested IPCR as described in “Methods” section. The IPCR bands derived from the AF9 cleaved fragments were indicated by the side brackets. b Average number of AF9 gene cleavages detected in BA-treated NP69 cells. Data are presented as means and SDs of four independent experiments. Each experiment consisted of two to four sets of IPCR performed in three to six IPCR replicates per set for each cell sample. Values are expressed as fold change normalised to the value of the untreated control. The differences between the untreated control and treated groups were compared by using Student’s t-test. An asterisk indicates a significant increase in BA-treated cells as compared with the untreated control (* p < 0.05, ** p < 0.01). An open diamond represents a significant decrease in the sample with CI pretreatment as compared with its corresponding sample without CI pretreatment (p < 0.01)
Sequencing results
To ascertain that these fragments were derived from the cleaved AF9 gene, the IPCR bands were cleaned up and sequenced. The sequencing results show that these fragments are all derived from the cleaved AF9 gene. The positions of chromosome breaks detected within the AF9 gene in BA-treated NP69 and TWO4 cells are shown in Tables 1 and 2, respectively. Intriguingly, four breakpoints (at coordinates 245,509, 245,527, 245,594 and 245,596) were mapped within the AF9 region (at coordinates 245,252–245,612) which was previously reported to be involved in t(9;11)(p22;q23) in an ALL patient. This chromosome translocation resulted in the formation of the MLL-AF9 fusion gene in the ALL patient [GenBank:AM050804]. It is noteworthy that in BA-treated TWO4 cells, we identified a breakpoint (at coordinate 245,612) which is identical with that in the ALL patient [GenBank:AM050804]. A breakpoint (at coordinate 245,596) was simultaneously identified in both NP69 and TWO4 cells treated with BA at neutral pH. Three breakpoints (at coordinates 245,594, 245,596 and 246,116) are similar with those identified in CEM cells (at coordinate 246,114) and cultured normal blood cells (at coordinate 245,593) exposed to etoposide (VP16) [74]. A few breakpoints (at coordinates 245,596, 245,664, 245,708, 245,803 and 246,116) are similar with those identified in H2O2-treated NP69 cells (at coordinates 245,591, 245,659, 245,703, 245,796, 246,113) and HK1 cells (at coordinate 245,590) reported in our previous study [77]. A genomic map illustrating the breakpoints within the AF9 BCR in BA-treated NP69 and TWO4 cells is shown in Fig. 9.
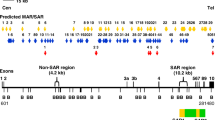
The positions of bile acid-induced chromosome breakages within the AF9 gene. aThe genomic map of AF9 gene from nucleotide positions 601–281,480 is illustrated [EMBL:ENSG00000171843]. Green boxes represent the two previously identified patient breakpoint cluster regions which are denoted as BCR1 and BCR2 [89]. Yellow boxes indicate the biochemically defined MAR/SARs reported in the previous study. These two MAR/SARs were denominated as SAR1 and SAR2 [89]. B: BamH I restriction sites. b Targeted region (10.2 kb) in the AF9 gene. Green and blue arrows indicate the primers used in the first (R1, AF9 236,451 R and F1, AF9 245,385 F) and second (R2, AF9 236,211 R and F2, AF9 245,507 F) rounds of nested IPCR, respectively. BamH I (B), Kpn I (K) and Nde I (N) restriction sites are shown. c Chromosome breakages detected in the present study by using IPCR. Red and green vertical lines indicate the presently identified breakpoints in BA-treated NP69 and TWO4 cells, respectively. All the chromosome breakages were mapped within BCR1 in close proximity with SAR1. Blue box represents the AF9 region (at coordinates 245,252–245,612) that was previously reported to translocate with the MLL gene resulting in the MLL-AF9 fusion gene in an ALL patient [GenBank:AM050804]
Discussion
In the present study, we intended to investigate the role of BA-induced apoptosis in mediating chromosome rearrangement in NPC. The externalisation of PS appears to be an early event of apoptosis [92]. Using flow cytometric analysis of PS externalisation, we observed significant percentages of apoptosis after BA treatment. This indicates that BA is a potential apoptotic inducer in nasopharyngeal epithelial cells. The results of this study reaffirm the findings of previous studies where BA could induce apoptosis in a number of cell types. These include normal oesophageal mucosal epithelial cells [93, 94], oesophageal adenocarcinoma cells [62, 95], Barrett epithelial cancer cells [96], normal colon epithelial cells [63], colon adenocarcinoma cells [61], hepatocellular carcinoma cells [97] and rat hepatocyte [98].
It is important to note that, BA at acidic pH resulted in a higher percentage of apoptotic cells as compared with that at neutral pH. This is true for both NP69 and TWO4 cells. Our findings are consistent with previous work which demonstrated that the combination of acid and BA has a higher apoptosis-inducing effect as compared with either acid alone or BA alone [62]. Therefore, it is likely that there is a synergistic effect between BA and acidic pH in apoptosis induction. Indeed, there is abundant evidence that the combination of acid and BA has a higher injurious effect on the epithelial cells as compared with either acid alone or BA alone [82, 99,100,101].
Evidence has been given that BA triggers apoptosis via oxidative stress [61]. It is known that BA can induce the generation of intracellular ROS in human colon epithelial cells [61, 102], human oesophageal epithelial cells [62, 101] and rat hepatocyte [98]. In the present study, we tested the ability of BA in inducing ROS generation in NP69 and TWO4 cells. By using a dichlorofluorescein diacetate (DCFH-DA) fluorescence-based assay, we have demonstrated that BA at neutral and acidic pH induced the production of ROS in both cell lines. Our findings are consistent with previous work which demonstrated that BA at neutral and acidic pH (pH 6) induced the release of ROS within the cytoplasm of exposed oesophageal adenocarcinoma cells (OE33) and oesophageal squamous cell carcinoma cells (KYSE-3) [101]. However, it is noteworthy that in both NP69 and TWO4 cells, the effect of BA in triggering ROS generation was higher at acidic pH than at neutral pH. This supports an earlier study of Dvorak and colleagues which suggested that there is a synergism between acid and BA in oxidative stress induction. In their study, a significant increase in oxidative stress was found in oesophageal adenocarcinoma cells (Seg-1) exposed to pH 4 in combination with BA cocktail, but not in Seg-1 cells treated with acid alone or BA alone. Their study also showed that generation of 8-OH-dG, a marker of oxidative DNA damage, was significantly increased in biopsy specimens after incubation in medium acidified to pH 4 and BA cocktail, but not in specimens treated with acid only or BA only [62]. The authors have therefore proposed a few possible mechanisms of synergism between acid and BA in oxidative stress induction. One of the possible mechanisms is that acid and BA synergise to activate nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, through endosomal acidification by acid [103] and membrane perturbation by BA [102]. It has also been speculated that BA-induced ROS might be enhanced by acidic pH through iron-mediated Fenton reactions [104].
It is known that in BA-induced apoptosis, ROS is mainly generated through the activation of NADPH oxidases [61, 102]. However, it has also been observed that inhibition of NADPH oxidases activities did not completely protect BCS-TC2 cells from the cytotoxic effects of BA, indicating potential additional minor sources of cytotoxicity. Due to the hydrophobic nature of BA, BA may directly diffuse into the cytosol resulting in mitochondrial perturbations. This can lead to subsequent alteration in oxidative phosphorylation which causes excessive ROS formation [61]. This ROS generation is strongly associated with the onset of mitochondrial permeability transition (MPT) which is an important feature of BA-induced apoptosis [98]. One of the hallmarks of the induction of MPT is the loss of MMP [105].
In the current study, we have demonstrated that BA stimulated MMP disruption in nasopharyngeal epithelial cells. The capability of BA in stimulating MMP disruption, which represents MPT, has also been demonstrated in rat hepatocyte [98], human colon carcinoma tumourigenic cells (HCT-116) [102] and non-tumourigenic cells (BCS-TC2) [61]. In consistency with the ROS detection assay, the BA-triggered MMP loss was substantially enhanced by acidic pH. Since the collapse of MMP is an event greatly dependent on the signalling molecules, ROS, these findings therefore strengthen the suggestion that acid and BA synergise to stimulate oxidative stress (as discussed above).
Previous studies have shown that activation of MPT by oxidative stress is a crucial event for the downstream caspase activation and apoptosis. This BA-induced MPT can provoke the release of pro-apoptotic proteins (such as cytochrome c) to the cytosol leading to the activation of initiator caspase-9. Subsequently, effector caspase-3 is activated, followed by the activation of DNA degrading enzymes [61, 98]. Our current findings showed that BA triggered increased caspase-3 activity in NP69 and TWO4 cells. This suggests that this effector caspase is playing a role in the execution of BA-induced apoptosis. Indeed, BA-induced apoptosis has been shown to be caspase-3-dependent in human colon adenocarcinoma cells [61] and rat hepatocyte [98]. In this study, caspase-3 activity was significantly reduced by Z-DEVD-FMK in NP69 and TWO4 cells treated with BA at neutral and acidic pH. Z-DEVD-FMK is a synthetic caspase-3 inhibitor which has been developed based on the substrate cleavage site of caspase-3. It acts as a pseudosubstrate for caspase-3 and is therefore a competitive inhibitor [106]. By employing a positional scanning synthetic combinatorial library, the optimal peptide recognition motif for caspase-3 has previously been determined to be DEVD (Asp-Glu-Val-Asp) [107]. Hence, DEVD inhibitor is the most potent caspase-3 inhibitor with the lowest inhibitory constant (Ki) against caspase-3 (Ki = 0.23 nM) [91].
In addition, our results showed that NP69 cells (normal nasopharyngeal epithelial cells) were more sensitive to BA treatment as compared with TWO4 cells (NPC cells). Thus, shorter exposures were used to treat NP69 cells to avoid cell detachment. These observations were similar with those obtained by Dvorak and colleagues where normal squamous oesophageal HET-1A cells appeared to be more sensitive to BA treatment as compared with human oesophageal adenocarcinoma Seg-1 cells [62]. The fact that TWO4 cells were less sensitive than NP69 cells to BA driven cytotoxicity could be due to some unknown cellular defects which contribute to apoptotic resistance or deregulation of cell death.
As discussed above, our data indicated that BA could trigger apoptosis at both neutral and acidic pH. Knowing that chromosome breakages occur in both apoptosis and chromosome rearrangements, we intended to identify the chromosome breaks mediated by BA-induced apoptosis. This study targeted the AF9 gene located at 9p22 because 9p22 is a deletion hotspot in NPC [78]. Besides, the formation of the MLL-AF9 fusion gene has been associated with acute myelogenous leukaemia (AML), acute lymphoblastic leukaemia (ALL), myelodysplastic syndromes (MDS) and therapy-related AML (t-AML) [89, 108]. Previous studies have reported two BCRs in the AF9 gene, namely, BCR1 (within intron 4) and BCR2 (spans introns 7 and 8) [89, 90]. In the study of Strissel et al. (2000), two matrix association regions/scaffold attachment regions (MAR/SARs) have been isolated experimentally from the AF9 gene. These MAR/SARs were denominated as SAR1 (found in intron 4) and SAR2 (encompasses exons 5 to 7). The AF9 BCRs are bordered by these two MAR/SARs [89]. Using nested IPCR, we showed that the AF9 gene cleavage frequencies in BA-treated NP69 and TWO4 cells were significantly higher than those in the untreated control. Our findings demonstrated that BA-induced apoptosis caused cleavages within the AF9 BCR1. The AF9 BCR1 is bordered by SAR1 and SAR2 (Fig. 9). It was found that the AF9 BCRs share similar structural elements as the MLL BCR. These structural elements include MAR/SAR sequences, topo II cleavage sites and DNase I hypersensitive cleavage sites. The regions containing these similar structural elements have been found to serve as illegitimate recombination hot spots leading to the MLL/AF9 translocations in leukaemia [89]. These previous findings are in good agreement with our current results that a few chromosome breaks were mapped within the AF9 region that was previously reported being involved in t(9;11)(p22;q23) in an ALL patient. This chromosome translocation resulted in the formation of the MLL-AF9 fusion gene in the ALL patient [GenBank:AM050804]. In addition, we detected a breakpoint which is identical with that identified in the ALL patient [GenBank:AM050804].
Previous reports suggested that the apoptotic nuclease CAD mediates DNA cleavages which in turn leads to chromosome translocation in leukaemia [73, 109]. Our previous findings have also suggested that CAD participates in DNA cleavages in normal nasopharyngeal epithelial cells (NP69) and NPC cells (HK1) that are undergoing oxidative stress-induced apoptosis. These cleavages may subsequently lead to chromosome rearrangement in NPC [77]. Given the involvement of oxidative stress induction in BA-induced apoptosis, it is possible that the chromosome breaks mediated by BA-induced apoptosis are also CAD-dependent. Therefore, in the current study, the possible involvement of CAD in mediating BA-induced chromosome breaks was investigated.
In non-apoptotic condition, CAD exists naturally in the cytoplasm as a heterodimer with its inhibitor, ICAD. When there is an apoptotic inducer, caspase-3 will be activated and cleaves the ICAD which possesses two caspase-3 cleavage sites. Subsequently, CAD will be released from its chaperone ICAD and enters the nucleus to cleave DNA by generating double-stranded breaks [67, 68]. An in vitro CAD assay has been conducted by Wolf and colleagues (1999) to study the roles of caspases-3, − 6, − 7, − 8, and granzyme B in mediating ICAD inactivation and apoptotic DNA fragmentation. It was found that only the DEVD-cleaving caspases, namely caspase-3 and caspase-7, inactivated ICAD and induced DNA fragmentation. Their results suggested that caspase-6, − 8 and granzyme B caused ICAD inactivation and DNA fragmentation in an indirect manner, presumably via activation of caspase-3 and/or caspase-7. They further demonstrated that caspase-3 was more efficient than caspase-7 in promoting ICAD inactivation and DNA fragmentation. Furthermore, in caspase-3 null MCF7 cells (breast cancer cell line) and cytosolic extracts, caspase-7 was found to be unable to promote ICAD inactivation in its endogenous level. Their findings concluded that caspase-3 is the main player in ICAD inactivation and apoptotic DNA fragmentation [110].
In the present study, we studied the role of CAD in mediating DNA cleavages in BA-induced apoptosis by inhibiting caspase-3 and caspase-7. As discussed above, we have clearly demonstrated that Z-DEVD-FMK could effectively inhibit the activity of caspase-3 and caspase-7. Therefore, if CAD is the main player in mediating chromosome breaks in BA-induced apoptosis, Z-DEVD-FMK treatment should reduce the number of chromosome breaks. Our data demonstrated that inhibition of caspase-3, which indirectly inhibits CAD, significantly reduced gene cleavage frequency in BA-treated NP69 cells. These findings suggest that BA induced chromosome breaks in a caspase-3-dependent manner. Given that caspase-3 is the main executioner caspase which activates CAD to cause apoptotic DNA fragmentation, thus CAD is most likely responsible for BA-induced chromosome breakages. Combining the findings from the current study and earlier work, we propose a model for BA-induced apoptosis in mediating chromosome breakages leading to chromosome rearrangements in NPC (Fig. 10). This model might also be applied to other types of cancers which have been linked to GORD or GORD-associated inflammatory diseases. These cancers include stomach, oesophageal, laryngeal, pharyngeal and lung cancers.
A proposed model for BA-induced apoptosis in mediating chromosome rearrangements in NPC. Exposure of nasopharyngeal epithelial cells to BA triggers intracellular ROS generation. The effect of BA in triggering ROS production is enhanced by acid through synergistic mechanisms. Increased ROS level induces PS externalisation and MMP loss. The former is acting as the apoptotic signalling which activates the downstream cascade, whereas the latter leads to MPT which in turn provokes the release of proapoptotic proteins such as cytochrome c. Subsequently, the main effector, caspase-3 is activated and cleaves the ICAD which possesses two caspase-3 cleavage sites. Once CAD is released from its chaperone ICAD, it enters the nucleus to cleave the DNA and causes DNA fragmentation. Cells may evade apoptosis and recover upon DNA repair. However, imprecise DNA repair may cause chromosome rearrangements in surviving cells. Repeated exposure to BA and acid (i.e. gastric duodenal refluxate) may increase the rate of mutations. The genomic instability may be exacerbated by chronic inflammation of sinonasal tissue which is repeatedly exposed to gastric duodenal refluxate. This is due to the fact that ROS production, apoptotic signalling and DNA damage may also be provoked by the inflammatory response. The cytotoxicity and genotoxicity of BA, especially in combination of acid, may therefore contribute to chromosome rearrangements in NPC
We note that direct association between GORD and NPC requires further investigation. Nevertheless, there is clear evidence that CRS has a positive association with an increased risk of developing NPC, and that GOR is one of the major aetiological factors of CRS. Early diagnosis of NPC is important for successful therapeutic intervention. Moreover, identification of NPC risk factors is crucial for risk prediction as well as prevention. Since the typical reflux symptoms (heartburn and regurgitation) are usually absent in patients with EOR, most of these patients are unaware of the acid refluxate exposure. Hence, patients with CRS and/or otitis media should be subjected to diagnosis for EOR such as nasopharyngeal pH monitoring. This is especially so for those who are unresponsive to conventional therapies. As a preventive measure, acid suppression therapy with antioxidant supplementation may be recommended for patients with EOR-related chronic inflammation of sinonasal tract. This may help in preventing the cytotoxicity and genotoxicity driven by both BA and acid.
Conclusions
In summary, our study is the first to demonstrate that BA could induce apoptosis, ROS generation, MMP loss and caspase activation in nasopharyngeal epithelial cells. The effect of BA was amplified by acidic pH, suggesting that there is a synergistic effect between acid and BA in the induction of these events. We further demonstrated that BA-induced apoptosis could lead to chromosome cleavages within the AF9 BCR. Besides, the apoptotic nuclease CAD could be a crucial player in mediating chromosome breakages in BA-induced apoptosis. Although apoptosis can act as a defence against cancer by removing cells harbouring DNA damage, cells may recover from apoptosis via DNA repair systems. However, erroneous DNA repair may cause chromosome rearrangements in surviving cells. Therefore, we speculate that repeated exposure of nasopharyngeal epithelium to BA and acid (i.e. a mixture of gastric duodenal refluxate) causes not only chronic inflammation but also elevates the risk of chromosomal alterations leading to NPC development. The increased genomic instability may be exacerbated by chronic inflammatory conditions that also induce ROS generation and DNA damage. It is also reasoned that the inflammatory conditions predispose the nasopharyngeal mucosa to transformation on repeated exposure to BA and acid. The identification of high-risk group makes early recognition and prevention of NPC a possible task. Nevertheless, further epidemiological studies of nasopharyngeal reflux and NPC are warranted.
Abbreviations
- ALL:
-
Acute lymphoblastic leukaemia
- AML:
-
Acute myelogenous leukaemia
- BA:
-
Bile acid
- BCR:
-
Breakpoint cluster region
- CAD:
-
Caspase-activated deoxyribonuclease
- CI:
-
Caspase inhibitor
- CPT:
-
Camptothecin
- CRS:
-
Chronic rhinosinusitis
- DCFH-DA:
-
2′,7′-dichlorofluorescein diacetate
- EBV:
-
Epstein-Barr virus
- ENT:
-
Ear, nose and throat
- EOR:
-
Extraoesophageal reflux
- FITC:
-
Annexin V-fluorescein isothiocyanate
- GORD:
-
Gastro-oesophageal reflux disease
- H2O2 :
-
Hydrogen peroxide
- ICAD:
-
Inhibitor of caspase-activated deoxyribonuclease
- IPCR:
-
Inverse polymerasechain reaction
- Ki :
-
Inhibitory constant
- MAR/SAR:
-
Matrix association region/scaffold attachment region
- MDS:
-
Myelodysplastic syndromes
- MLL :
-
Mixed lineage leukaemia
- MMP:
-
Mitochondria membrane potential
- MPT:
-
Mitochondrial permeability transition
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NPC:
-
Nasopharyngeal carcinoma
- PI:
-
Propidium iodide
- PS:
-
Phosphatidylserine
- ROS:
-
Reactive oxygen species
- SDS:
-
Sodium dodecyl sulfate
- t-AML:
-
Therapy-related acute myelogenous leukaemia
- VP16:
-
Etoposide
References
Dickens P, Srivastava G, Loke SL, Chan CW, Liu YT. Epstein-Barr virus DNA in nasopharyngeal carcinomas from Chinese patients in Hong Kong. J Clin Pathol. 1992;45:396–7.
Armstrong RW, Armstrong MJ, Yu MC, Henderson BE. Salted fish and inhalants as risk factors for nasopharyngeal carcinoma in Malaysian Chinese. Cancer Res. 1983;43:2967–70.
Armstrong RW, Imrey PB, Lye MS, Armstrong MJ, Yu MC, Sani S. Nasopharyngeal carcinoma in Malaysian Chinese: occupational exposures to particles, formaldehyde and heat. Int J Epidemiol. 2000;29:991–8.
Hutajulu SH, Indrasari SR, Indrawati LP, Harijadi A, Duin S, Haryana SM, et al. Epigenetic markers for early detection of nasopharyngeal carcinoma in a high risk population. Mol Cancer. 2011;10:48.
Lo KW, Huang DP. Genetic and epigenetic changes in nasopharyngeal carcinoma. Semin Cancer Biol. 2002;12:451–62.
Henderson BE, Louie E, SooHoo JJ, Buell P, Gardner MB. Risk factors associated with nasopharyngeal carcinoma. N Engl J Med. 1976;295:1101–6.
Yu MC, Garabrant DH, Huang TB, Henderson BE. Occupational and other non-dietary risk factors for nasopharyngeal carcinoma in Guangzhou, China. Int J Cancer. 1990;45:1033–9.
Yuan JM, Wang XL, Xiang YB, Gao YT, Ross RK, Yu MC. Non-dietary risk factors for nasopharyngeal carcinoma in shanghai, China. Int J Cancer. 2000;85:364–9.
Ekburanawat W, Ekpanyaskul C, Brennan P, Kanka C, Tepsuwan K, Temiyastith S, et al. Evaluation of non-viral risk factors for nasopharyngeal carcinoma in Thailand: results from a case-control study. Asian Pac J Cancer Prev. 2010;11:929–32.
Huang WY, Lin CC, Jen YM, Lin KT, Yang MH, Chen CM, et al. Association between adult otitis media and nasopharyngeal cancer: a nationwide population-based cohort study. Radiother Oncol. 2012;104:338–42.
Hung SH, Chen PY, Lin HC, Ting J, Chung SD. Association of rhinosinusitis with nasopharyngeal carcinoma: a population-based study. Laryngoscope. 2014;124:1515–20.
Tsou YA, Lin CC, Tai CJ, Tsai MH, Tsai TC, Chen CM. Chronic rhinosinusitis and the risk of nasopharyngeal cancer in a Taiwanese health study. Am J Rhinol Allergy. 2014;28:168–72.
Issing WJ, Karkos PD. Atypical manifestations of gastro-oesophageal reflux. J R Soc Med. 2003;96:477–80.
Fass R, Achem SR, Harding S, Mittal RK, Quigley E. Review article: supra-oesophageal manifestations of gastro-oesophageal reflux disease and the role of night-time gastro-oesophageal reflux. Aliment Pharmacol Ther. 2004;20(Suppl 9):26–38.
Poelmans J, Tack J. Extraoesophageal manifestations of gastro-oesophageal reflux. Gut. 2005;54:1492–9.
Farrokhi F, Vaezi MF. Extra-esophageal manifestations of gastroesophageal reflux. Oral Dis. 2007;13:349–59.
Madanick RD. Extraesophageal presentations of GERD: where is the science? Gastroenterol Clin N Am. 2014;43:105–20.
Mosca F, Rossillo V, Leone CA. Manifestations of gastro-pharyngo-laryngeal reflux disease. Acta Otorhinolaryngol Ital. 2006;26:247–51.
Fink SM, Barwick KW, DeLuca V, Sanders FJ, Kandathil M, McCallum RW. The association of histologic gastritis with gastroesophageal reflux and delayed gastric emptying. J Clin Gastroenterol. 1984;6:301–9.
Bowrey DJ, Clark GW, Williams GT. Patterns of gastritis in patients with gastro-oesophageal reflux disease. Gut. 1999;45:798–803.
Little AG, DeMeester TR, Kirchner PT, O'Sullivan GC, Skinner DB. Pathogenesis of esophagitis in patients with gastroesophageal reflux. Surgery. 1980;88:101–7.
Dodds WJ, Dent J, Hogan WJ, Helm JF, Hauser R, Patel GK, et al. Mechanisms of gastroesophageal reflux in patients with reflux esophagitis. N Engl J Med. 1982;307:1547–52.
Shub MD, Ulshen MH, Hargrove CB, Siegal GP, Groben PA, Askin FB. Esophagitis: a frequent consequence of gastroesophageal reflux in infancy. J Pediatr. 1985;107:881–4.
Delahunty JE. Acid laryngitis. J Laryngol Otol. 1972;86:335–42.
Ward PH, Berci G. Observations on the pathogenesis of chronic non-specific pharyngitis and laryngitis. Laryngoscope. 1982;92:1377–82.
Ulualp SO, Toohill RJ, Hoffmann R, Shaker R. Pharyngeal pH monitoring in patients with posterior laryngitis. Otolaryngol Head Neck Surg. 1999;120:672–7.
Cherry J, Siegel CI, Margulies SI, Donner M. Pharyngeal localization of symptoms of gastroesophageal reflux. Ann Otol Rhinol Laryngol. 1970;79:912–4.
Barbero GJ. Gastroesophageal reflux and upper airway disease. Otolaryngol Clin N Am. 1996;29:27–38.
Wise SK, Wise JC, DelGaudio JM. Association of nasopharyngeal and laryngopharyngeal reflux with postnasal drip symptomatology in patients with and without rhinosinusitis. Am J Rhinol. 2006;20:283–9.
Poelmans J, Tack J, Feenstra L. Chronic middle ear disease and gastroesophageal reflux disease: a causal relation? Otol Neurotol. 2001;22:447–50.
Tasker A, Dettmar PW, Panetti M, Koufman JA, Birchall P, Pearson JP. Is gastric reflux a cause of otitis media with effusion in children? Laryngoscope. 2002;112:1930–4.
Serra A, Cocuzza S, Poli G, La M, Messina I, Pavone AP. Otologic findings in children with gastroesophageal reflux. Int J Pediatr Otorhinolaryngol. 2007;71:1693–1697.
Al Saab F, Manoukian JJ, Al Sabah B, Almot S, Nguyen LH, Tewfik TL, et al. Linking laryngopharyngeal reflux to otitis media with effusion: pepsinogen study of adenoid tissue and middle ear fluid. J Otolaryngol Head Neck Surg. 2008;37:565–71.
Sontag SJ, O'Connell S, Khandelwal S, Miller T, Nemchausky B, Schnell TG, et al. Most asthmatics have gastroesophageal reflux with or without bronchodilator therapy. Gastroenterology. 1990;99:613–20.
Schnatz PF, Castell JA, Castell DO. Pulmonary symptoms associated with gastroesophageal reflux: use of ambulatory pH monitoring to diagnose and to direct therapy. Am J Gastroenterol. 1996;91:1715–8.
Mujica VR, Rao SS. Recognizing atypical manifestations of GERD. Asthma, chest pain, and otolaryngologic disorders may be due to reflux. Postgrad Med. 1999;105:53–6.
Ulualp SO, Toohill RJ, Hoffmann R, Shaker R. Possible relationship of gastroesophagopharyngeal acid reflux with pathogenesis of chronic sinusitis. Am J Rhinol. 1999;13:197–202.
DelGaudio JM. Direct nasopharyngeal reflux of gastric acid is a contributing factor in refractory chronic rhinosinusitis. Laryngoscope. 2005;115:946–57.
Lin YH, Chang TS, Yao YC, Li YC. Increased risk of chronic sinusitis in adults with Gastroesophgeal reflux disease: a Nationwide population-based cohort study. Medicine (Baltimore). 2015;94:e1642.
Phipps CD, Wood WE, Gibson WS, Cochran WJ. Gastroesophageal reflux contributing to chronic sinus disease in children: a prospective analysis. Arch Otolaryngol Head Neck Surg. 2000;126:831–6.
Contencin P, Narcy P. Nasopharyngeal pH monitoring in infants and children with chronic rhinopharyngitis. Int J Pediatr Otorhinolaryngol. 1991;22:249–56.
DiBaise JK, Huerter JV, Quigley EM. Sinusitis and gastroesophageal reflux disease. Ann Intern Med. 1998;129:1078.
Loehrl TA. Sinonasal problems and reflux. Facial Plast Surg Clin North Am. 2012;20:83–6.
Contencin P, Viala P, Narcy P. Variations of nasopharyngeal pH in nasopharyngitis in children. Presse Med. 1991;20:1595–8.
Aydin E, Tastan E, Aydogan F, Arslan N, Karaca G. Role of nasopharyngeal reflux in the etiology of otitis media with effusion. J Otolaryngol Head Neck Surg. 2011;40:499–503.
Pincus RL, Kim HH, Silvers S, Gold S. A study of the link between gastric reflux and chronic sinusitis in adults. Ear Nose Throat J. 2006;85:174–8.
Dibaise JK, Olusola BF, Huerter JV, Quigley EMM. Role of GERD in chronic resistant sinusitis: a prospective, open label, pilot trial. Am J Gastroenterol. 2002;97:843–50.
Schottenfeld D, Beebe-Dimmer J. Chronic inflammation: a common and important factor in the pathogenesis of neoplasia. CA Cancer J Clin. 2006;56:69–83.
Hashimoto K. The effect of reflux of bile juice on the development of residual stomach cancer. Nihon Geka Gakkai Zasshi. 1987;88:26–34.
Buchmann F, Baumer F, Eilles C. Significance of duodenogastric reflux in the pathogenesis of primary stomach cancer. Zentralbl Chir. 1988;113:170–3.
Lagergren J, Bergstrom R, Lindgren A, Nyren O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N Engl J Med. 1999;340:825–31.
Shaheen N, Ransohoff DF. Gastroesophageal reflux, Barrett esophagus, and esophageal cancer: scientific review. JAMA. 2002;287:1972–81.
Qadeer MA, Colabianchi N, Vaezi MF. Is GERD a risk factor for laryngeal cancer? Laryngoscope. 2005;115:486–91.
Ward PH, Hanson DG. Reflux as an etiological factor of carcinoma of the laryngopharynx. Laryngoscope. 1988;98:1195–9.
Vereczkei A, Horvath OP, Varga G, Molnar TF. Gastroesophageal reflux disease and non-small cell lung cancer. Results of a pilot study. Dis Esophagus. 2008;21:457–60.
Winters C Jr, Spurling TJ, Chobanian SJ, Curtis DJ, Esposito RL, Hacker JF III, et al. Barrett’s esophagus. A prevalent, occult complication of gastroesophageal reflux disease. Gastroenterology. 1987;92:118–24.
Csendes A, Smok G, Burdiles P, Quesada F, Huertas C, Rojas J, et al. Prevalence of Barrett's esophagus by endoscopy and histologic studies: a prospective evaluation of 306 control subjects and 376 patients with symptoms of gastroesophageal reflux. Dis Esophagus. 2000;13:5–11.
Chevfec G, Schnell T, Sontag S. Barrett’s esophagus. A preneoplastic disorder. Am J Clin Pathol. 1992;98:5–7.
Blumin JH, Merati AL, Toohill RJ. Duodenogastroesophageal reflux and its effect on extraesophageal tissues: a review. Ear Nose Throat J. 2008;87:234–7.
Bernstein H, Bernstein C, Payne CM, Dvorakova K, Garewal H. Bile acids as carcinogens in human gastrointestinal cancers. Mutat Res. 2005;589:47–65.
Ignacio BJ, Olmo N, Perez-Ramos P, Santiago-Gomez A, Lecona E, Turnay J, et al. Deoxycholic and chenodeoxycholic bile acids induce apoptosis via oxidative stress in human colon adenocarcinoma cells. Apoptosis. 2011;16:1054–67.
Dvorak K, Payne CM, Chavarria M, Ramsey L, Dvorakova B, Bernstein H, et al. Bile acids in combination with low pH induce oxidative stress and oxidative DNA damage: relevance to the pathogenesis of Barrett's oesophagus. Gut. 2007;56:763–71.
Bernstein C, Bernstein H, Garewal H, Dinning P, Jabi R, Sampliner RE, et al. A bile acid-induced apoptosis assay for colon cancer risk and associated quality control studies. Cancer Res. 1999;59:2353–7.
Bernstein C, Payne CM, Bernstein H. Bile acids: promoters or carcinogens in Colon Cancer? J Carcinogene Mutagene. 2011;2:101e.
Fang Y, Chen X, Bajpai M, Verma A, Das KM, Souza RF, et al. Cellular origins and molecular mechanisms of Barrett's esophagus and esophageal adenocarcinoma. Ann N Y Acad Sci. 2013;1300:187–99.
Kerr JF, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer. 1972;26:239–57.
Enari M, Sakahira H, Yokoyama H, Okawa K, Iwamatsu A, Nagata S. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature. 1998;391:43–50.
Sakahira H, Enari M, Nagata S. Cleavage of CAD inhibitor in CAD activation and DNA degradation during apoptosis. Nature. 1998;391:96–9.
Liu X, Zou H, Widlak P, Garrard W, Wang X. Activation of the apoptotic endonuclease DFF40 (caspase-activated DNase or nuclease). Oligomerization and direct interaction with histone H1. J Biol Chem. 1999;274:13836–40.
Vaughan AT, Betti CJ, Villalobos MJ. Surviving apoptosis. Apoptosis. 2002;7:173–7.
Weinstock DM, Richardson CA, Elliott B, Jasin M. Modeling oncogenic translocations: distinct roles for double-strand break repair pathways in translocation formation in mammalian cells. DNA Repair (Amst). 2006;5:1065–74.
Betti CJ, Villalobos MJ, Diaz MO, Vaughan AT. Apoptotic triggers initiate translocations within the MLL gene involving the nonhomologous end joining repair system. Cancer Res. 2001;61:4550–5.
Sim SP, Liu LF. Nucleolytic cleavage of the mixed lineage leukemia breakpoint cluster region during apoptosis. J Biol Chem. 2001;276:31590–5.
Cynthia PN, Sim SP. Etoposide-induced apoptosis results in chromosome breaks within the AF9 gene: its implication in chromosome rearrangement in leukaemia. Adv Biosci Biotechnol. 2012;3:686–94.
Yee PH, Sim SP. High cell density and latent membrane protein 1 expression induce cleavage of the mixed lineage leukemia gene at 11q23 in nasopharyngeal carcinoma cell line. J Biomed Sci. 2010;17:77.
Boon SS, Sim SP. Inhibitor of caspase-activated DNase expression enhances caspase-activated DNase expression and inhibits oxidative stress-induced chromosome breaks at the mixed lineage leukaemia gene in nasopharyngeal carcinoma cells. Cancer Cell Int. 2015;15:54.
Tan SN, Sim SP, Khoo AS. Potential role of oxidative stress-induced apoptosis in mediating chromosomal rearrangements in nasopharyngeal carcinoma. Cell Biosci. 2016;6:35.
Shao JY, Wang HY, Huang XM, Feng QS, Huang P, Feng BJ, et al. Genome-wide allelotype analysis of sporadic primary nasopharyngeal carcinoma from southern China. Int J Oncol. 2000;17:1267–75.
Tsao SW, Wang X, Liu Y, Cheung YC, Feng H, Zheng Z, et al. Establishment of two immortalized nasopharyngeal epithelial cell lines using SV40 large T and HPV16E6/E7 viral oncogenes. Biochim Biophys Acta. 2002;1590:150–8.
Lin CT, Chan WY, Chen W, Huang HM, Wu HC, Hsu MM, et al. Characterization of seven newly established nasopharyngeal carcinoma cell lines. Lab Investig. 1993;68:716–27.
Kauer WK, Peters JH, DeMeester TR, Feussner H, Ireland AP, Stein HJ, et al. Composition and concentration of bile acid reflux into the esophagus of patients with gastroesophageal reflux disease. Surgery. 1997;122:874–81.
Nehra D, Howell P, Williams CP, Pye JK, Beynon J. Toxic bile acids in gastro-oesophageal reflux disease: influence of gastric acidity. Gut. 1999;44:598–602.
Theisen J, Nehra D, Citron D, Johansson J, Hagen JA, Crookes PF, et al. Suppression of gastric acid secretion in patients with gastroesophageal reflux disease results in gastric bacterial overgrowth and deconjugation of bile acids. J Gastrointest Surg. 2000;4:50–4.
Iftikhar SY, Ledingham S, Steele RJ, Evans DF, Lendrum K, Atkinson M, et al. Bile reflux in columnar-lined Barrett's oesophagus. Ann R Coll Surg Engl. 1993;75:411–6.
Junqueira JC, Penna FJ. Nasopharyngeal pH and gastroesophageal reflux in children with chronic respiratory disease. J Pediatr. 2007;83:225–32.
Zhang Q, Zhou K, Ling G. Apoptosis of nasopharyngeal carcinoma cells induced by topoisomerase I inhibitor. Zhonghua Er Bi Yan Hou Ke Za Zhi. 1997;32:332–5.
Zhang Y, Chen Y, Zhou K, Cai K. Mitochondrial mechanisms of CPT-induced apoptosis in nasopharyngeal carcinoma cells. Lin Chuang Er Bi Yan Hou Ke Za Zhi. 2001;15:309–10.
Bass DA, Parce JW, Dechatelet LR, Szejda P, Seeds MC, Thomas M. Flow cytometric studies of oxidative product formation by neutrophils: a graded response to membrane stimulation. J Immunol. 1983;130:1910–7.
Strissel PL, Strick R, Tomek RJ, Roe BA, Rowley JD, Zeleznik L. DNA structural properties of AF9 are similar to MLL and could act as recombination hot spots resulting in MLL/AF9 translocations and leukemogenesis. Hum Mol Genet. 2000;9:1671–9.
Strick R, Zhang Y, Emmanuel N, Strissel PL. Common chromatin structures at breakpoint cluster regions may lead to chromosomal translocations found in chronic and acute leukemias. Hum Genet. 2006;119:479–95.
Garcia-Calvo M, Peterson EP, Leiting B, Ruel R, Nicholson DW, Thornberry NA. Inhibition of human caspases by peptide-based and macromolecular inhibitors. J Biol Chem. 1998;273:32608–13.
Fadok VA, Voelker DR, Campbell PA, Cohen JJ, Bratton DL, Henson PM. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J Immunol. 1992;148:2207–16.
Zhang R, Gong J, Wang H, Wang L. Bile salts inhibit growth and induce apoptosis of culture human normal esophageal mucosal epithelial cells. World J Gastroenterol. 2005;11:6466–71.
Li Y, Reuter NP, Li X, Liu Q, Zhang J, Martin RC. Colocalization of MnSOD expression in response to oxidative stress. Mol Carcinog. 2010;49:44–53.
Looby E, Abdel-Latif MM, Athie-Morales V, Duggan S, Long A, Kelleher D. Deoxycholate induces COX-2 expression via Erk1/2-, p38-MAPK and AP-1-dependent mechanisms in esophageal cancer cells. BMC Cancer. 2009;9:190.
Burnat G, Majka J, Konturek PC. Bile acids are multifunctional modulators of the Barrett's carcinogenesis. J Physiol Pharmacol. 2010;61:185–92.
Higuchi H, Bronk SF, Takikawa Y, Werneburg N, Takimoto R, El Deiry W, et al. The bile acid glycochenodeoxycholate induces trail-receptor 2/DR5 expression and apoptosis. J Biol Chem. 2001;276:38610–8.
Yerushalmi B, Dahl R, Devereaux MW, Gumpricht E, Sokol RJ. Bile acid-induced rat hepatocyte apoptosis is inhibited by antioxidants and blockers of the mitochondrial permeability transition. Hepatology. 2001;33:616–26.
Kauer WK, Peters JH, DeMeester TR, Ireland AP, Bremner CG, Hagen JA. Mixed reflux of gastric and duodenal juices is more harmful to the esophagus than gastric juice alone. The need for surgical therapy re-emphasized. Ann Surg. 1995;222:525–31.
Lang J, Blikslager A, Regina D, Eisemann J, Argenzio R. Synergistic effect of hydrochloric acid and bile acids on the pars esophageal mucosa of the porcine stomach. Am J Vet Res. 1998;59:1170–6.
Jenkins GJ, D'Souza FR, Suzen SH, Eltahir ZS, James SA, Parry JM, et al. Deoxycholic acid at neutral and acid pH, is genotoxic to oesophageal cells through the induction of ROS: the potential role of anti-oxidants in Barrett's oesophagus. Carcinogenesis. 2007;28:136–42.
Payne CM, Weber C, Crowley-Skillicorn C, Dvorak K, Bernstein H, Bernstein C, et al. Deoxycholate induces mitochondrial oxidative stress and activates NF-kappaB through multiple mechanisms in HCT-116 colon epithelial cells. Carcinogenesis. 2007;28:215–22.
Reinehr R, Becker S, Braun J, Eberle A, Grether-Beck S, Haussinger D. Endosomal acidification and activation of NADPH oxidase isoforms are upstream events in hyperosmolarity-induced hepatocyte apoptosis. J Biol Chem. 2006;281:23150–66.
Schafer FQ, Buettner GR. Acidic pH amplifies iron-mediated lipid peroxidation in cells. Free Radic Biol Med. 2000;28:1175–81.
Lemasters JJ, Nieminen AL, Qian T, Trost LC, Elmore SP, Nishimura Y, et al. The mitochondrial permeability transition in cell death: a common mechanism in necrosis, apoptosis and autophagy. Biochim Biophys Acta. 1998;1366:177–96.
Ekert PG, Silke J, Vaux DL. Caspase inhibitors. Cell Death Differ. 1999;6:1081–6.
Thornberry NA, Rano TA, Peterson EP, Rasper DM, Timkey T, Garcia-Calvo M, et al. A combinatorial approach defines specificities of members of the caspase family and granzyme B. Functional relationships established for key mediators of apoptosis. J Biol Chem. 1997;272:17907–11.
Swansbury GJ, Slater R, Bain BJ, Moorman AV, Secker-Walker LM. Hematological malignancies with t(9;11)(p21-22;q23)--a laboratory and clinical study of 125 cases. European 11q23 workshop participants. Leukemia. 1998;12:792–800.
Stanulla M, Wang J, Chervinsky DS, Thandla S, Aplan PD. DNA cleavage within the MLL breakpoint cluster region is a specific event which occurs as part of higher-order chromatin fragmentation during the initial stages of apoptosis. Mol Cell Biol. 1997;17:4070–9.
Wolf BB, Schuler M, Echeverri F, Green DR. Caspase-3 is the primary activator of apoptotic DNA fragmentation via DNA fragmentation factor-45/inhibitor of caspase-activated DNase inactivation. J Biol Chem. 1999;274:30651–6.
Acknowledgements
We are deeply grateful to Prof. Tsao Sai Wah (The University of Hong Kong, Hong Kong, China) and Prof. Lo Kwok Wai (The Chinese University of Hong Kong, Hong Kong, China) for providing the normal nasopharyngeal epithelial cell line (NP69). We gratefully acknowledge Prof. Sam Choon Kook (formerly from University of Malaya, Malaysia) for providing the NPC cell line (TWO4). We express our gratitude to Ms. Tay Siow Phing (Universiti Malaysia Sarawak) and Ms. See Hui Shien (Biomarketing Services Sdn. Bhd.) for their technical support and advice with the flow cytometry assays. We acknowledge Universiti Malaysia Sarawak for all infrastructure support. We thank Ministry of Higher Education Malaysia for the MyBrain15 (MyPhd) Postgraduate Scholarship and the Fundamental Research Grant [FRGS/SKK01(01)/977/2013 (18)] that supported this work.
Funding
The project was funded by Ministry of Higher Education Malaysia [MOHE Fundamental Research Grant, grant number: FRGS/SKK01(01)/977/2013 (18)]. The funding body only reviewed the research proposal and approved with a budget adjustment. They did not involve in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Availability of data and materials
All data generated or analysed during this study are included in this published article.
Author information
Authors and Affiliations
Contributions
SNT and SPS together participated in the conception and design of the study. SNT conducted the experimental work, performed data analyses and wrote the manuscript. SPS interpreted the data, edited the manuscript and supervised the whole study. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional file
Additional file 1:
Flow chart depicting the simplified DNA manipulation steps in preparation for nested IPCR. (PDF 64 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Tan, SN., Sim, SP. Bile acids at neutral and acidic pH induce apoptosis and gene cleavages in nasopharyngeal epithelial cells: implications in chromosome rearrangement. BMC Cancer 18, 409 (2018). https://doi.org/10.1186/s12885-018-4327-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12885-018-4327-4