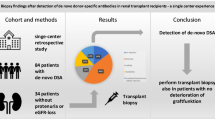
Abstract
Background
Donor-specific anti-human leukocyte antigen (HLA) antibodies (DSA) can be preformed or de novo (dn). Strategies to manage preformed DSA are well described, but data on the management and outcomes of dnDSA are lacking.
Methods
We performed a retrospective analysis of data from a single centre of the management and outcomes of 22 patients in whom a dnDSA was identified with contemporary and follow up biopsies.
Results
Evolution from baseline to follow up revealed a statistically significant loss of kidney function (estimated glomerular filtration rate: 45.9 ± 16.7 versus 37.4 ± 13.8 ml/min/1.73 m2; p = 0.005) and increase in the proportion of patients with transplant glomerulopathy (percentage with cg lesion ≥1: 27.2% vs. 45.4%; p = 0.04). Nine patients were not treated at the time of dnDSA identification, and 13 patients received various drug combinations (e.g., corticosteroids, plasmapheresis, thymoglobulins and/or rituximab). No significant pathological changes were observed for the various treatment combinations.
Conclusion
Our retrospective analysis of a small sample suggests that dnDSA should be considered a risk factor for the loss of kidney function independent of the baseline biopsy, and multidisciplinary evaluations of the transplant patient are a necessary requirement. Further confirmation in a multicentre prospective trial is required.
Similar content being viewed by others
Background
Kidney transplantation (KT) remains associated with suboptimal 5- and 10-year graft survival (77% and 56%, respectively [1]), despite an excellent 1-year allograft survival (91%). Graft failure is primarily associated with antibody mediated rejection (ABMR) [2,3,4].
Donor-specific anti-human leukocyte antigen (HLA) antibodies (DSA) drive ABMR. Development of the solid phase assay (SPA) to detect DSA was significant in the diagnosis of DSA [5]. SPA can detect DSA at the time of transplantation even with a negative flow cytometry crossmatch. DSA is divided into two categories: preformed DSA and de novo DSA (dnDSA). Preformed DSA may be associated with hyperacute rejection or ABMR in the weeks following transplantation. The presence of preformed DSA is associated with graft failure [6,7,8].
DSA development after KT is known as dnDSA. A previous review [9] noted that dnDSA primarily occurred during immunosuppression [10] in the context of poor adherence [4, 11, 12]. Early calcineurin inhibitor minimization may also be associated with dnDSA development [9]. Wiebe et al. [12] reported a trend towards an association with a history of acute rejection as an independent risk factor for dnDSA. DnDSA is independently associated with poor long-term allograft outcomes [13]. SPA technique allows to further detect complement-binding DSA, and these DSAs are associated with reduced graft survival compared to non-complement-binding DSA [14, 15].
Knowledge of the presence of DSA at the time of transplantation allows implementation of specific therapeutic strategies (e.g., desensitization) to reduce the levels of preformed DSA [16]. Several studies demonstrated acceptable long-term patient survival compared to wait-listed patients on dialysis [16, 17]. However, the therapeutic management of dnDSA remains controversial. Plasma exchange, intravenous immunoglobulin (IVIG), anti-thymoglobulin antibodies (ATG), rituximab, bortezomib and treatment abstention are used in different combinations for the treatment of ABMR [18]. Data on the long-term efficacy of these regimens and treatment strategies guided by the presence of dnDSA (independent histology) are lacking.
The present study retrospectively analysed the diagnosis and clinical and histological evolution in patients who developed dnDSA.
Methods
Patient selection and follow up
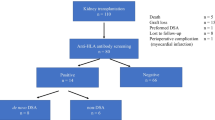
We retrospectively analysed the stored sera available from all consecutive patients who underwent kidney transplantation in Geneva University Hospitals from 1986 to 2012. Patients with combined transplantation (kidney – liver, kidney – heart or kidney – pancreas) were excluded. The cumulative incidence of dnDSA is 15% in our centre. We identified 22 patients with known dnDSA from the 315 patients transplanted from 1986 to 2012 for whom we had available sera. DnDSA was defined as a DSA that developed within six months after kidney transplantation with a mean fluorescence intensity (MFI) > 500. All 22 patients were transplanted with a negative CDC crossmatch (B and T) and/or FACS crossmatch. Clinical data were recorded during regular follow up at our institution. Graft function was assessed using the estimated glomerular filtration rate (eGFR). We used the CKD-EPI formula for each patient. The eGFR was calculated at the time of biopsy. A patient’s return to haemodialysis defined graft failure. We analysed graft biopsies at the time of dnDSA detection (± 3 months) in these patients and therapy administration according to the histological data. We identified a positive DSA in 2012 and retrospectively evaluated the sera from these patients to identify the first positive serum and matched the biopsy at the time of first identification of the dnDSA. The biopsies matched with the dnDSA were a protocol or indication biopsy because of the retrospective nature of this study.
The ethical committee approved the study (N° 6-208) which is in accordance with the regulations of the Geneva University Hospitals.
Immunosuppression dnDSA/histological-based treatment
Induction therapy evolved over time and consisted of no induction or basiliximab on post-operative days (PODs) 0 and 4. Antithymocyte globulin (ATG) or daclizumab reflected the use of a steroid-free protocol in selected patients. The immunosuppressive regimen consisted of tacrolimus or cyclosporine A, corticosteroids, and mycophenolate mofetil, mycophenolic acid or azathioprine. We reported the identified treatment combination at the time of dnDSA and biopsy as reported in medical files. The reported combinations included high doses of steroids, ATG, anti-CD20 and/or plasma exchange. These treatments were adjusted according to histopathological findings and patient’s age and comorbidities (e.g., cancer, previous infections).
Graft biopsies
Protocol biopsies are performed in our centre at one year post-transplantation, but the present analysis focused on kidney biopsies performed at approximately the time of development of dnDSA, regardless of kidney function. The clinician may not have known of the presence of dnDSA at the time of the biopsy. A control biopsy at the last follow up was also included to evaluate the histological changes following treatment of the dnDSA. The next available graft biopsy was used in patients who were not treated. Criteria from the Banff 2013 classification were used for the retrospective grading of biopsies [19].
Determination of DSA using Luminex solid-phase assay
Patient sera were analysed for the presence of anti-HLA class I and class II antibodies using solid phase assays on Luminex and the Labscreen Mix assay for HLA class I and HLA class II following the recommendations of the manufacturer (One Lambda, Canoga Park, CA), as previously described [20]. Sera collected before and after transplantation of all positive individuals were subsequently tested for anti-HLA class I- and class II-specific antibodies using the Luminex single antigen beads (one Lambda). Briefly, colour-coded microspheres coated with major HLA class I and II antigens were incubated with 10 μl serum for 30 min at room temperature in the dark. Samples were washed three times and incubated with 100 μL of 1:100 phycoerythrin-conjugated goat anti-human IgG (One Lambda Inc.). Samples were washed twice, and the fluorescent signal intensity for each microsphere was measured using a LABScan 100 flow analyser (One Lambda Inc.). The cut-off for positive samples was the normalized background (NBG) ratio recommended by the manufacturer, which was performed using HLA Fusion software (One Lambda). An MFI > 500 was considered positive, as recommended by the manufacturer, and clinical relevance was considered at MFI > 1000. Donor-specific antigens were classified as immunogenic HLA or non-immunogenic HLA based on the presence of specific antibodies in the recipient directed towards the donor-specific antigens.
Post-transplantation screening consisted of Luminex solid-phase assays performed at 1 month, 3 months, 6 months, 9 months, 12 months and annually. Each increase in serum creatinine > 25% required a new Luminex solid-phase assay.
Statistical analysis
Statistical analyses were performed using STATA v. 14.0 (College Station, TX). We used descriptive statistics to estimate the frequencies (%) and means (±SD) of study variables. Comparisons of two means were performed using t tests, and comparisons of the frequencies between groups were evaluated using Fisher’s exact test because of the small sample size. We did not perform further analyses because of the retrospective nature of the descriptive data.
Results
Patients’ characteristics
Table 1 summarises the patients’ characteristics. The characteristics of these patients were similar to the transplanted patients who do not develop dnDSA (data not shown). The mean age was 45 years (standard deviation (SD): ± 14), and males predominated (14/22; 63.6%). These kidney transplantations were primarily first ABO compatible, with greater than 3 HLA mismatches. A delayed graft function was observed in 8/22 patients (36.3%), and 8/22 patients (36.3%) did not receive induction therapy. Most patients who developed dnDSA received a triple immunosuppression regimen that was prescribed at baseline (21/22 patients; 95.4%). Eight of the 22 patients (36.3%) were treated for a previous episode of T-cell-mediated rejection (TCMR).
Characteristics of the dnDSA
Table 2 describes the characteristics of the dnDSA. DSA was primarily directed against MHC class II antigens in 15/22 patients (68.1%) or in association with Class I dnDSA (6/22; 27%) with a mean MFI of 5896. No difference in the mean MFI for dnDSA was observed from baseline to follow up (Fig. 1). Notably, 18 patients (81.8%) received a triple immunosuppressant regimen one year post-kidney transplantation, but only 5 of the 22 patients (22.7%) received the triple therapy at the time of dnDSA detection. Less than 20% of the 22 patients with dnDSA exhibited concomitant acute kidney injury at the time of dnDSA, which was defined as an increase in creatinine ≥25% from baseline and/or appearance of proteinuria > 0.5 g/24 h at the time of dnDSA with a mean eGFR of 45.9 ml/min/1.73 m2 (SD: ± 16.7). Half of the patients exhibited albuminuria at the discovery of dnDSA.
Histological parameters before and after therapy
A median time of 55.5 days was observed between first dnDSA identification and the initial graft biopsy. The range was 102 days prior to the biopsy to 863 days after the biopsy. Figure 2 shows the diagnosis at the initial and the follow up biopsies. We observed that 12/22 (54.5%) patients had a diagnosis of acute ABMR, chronic ABMR or mixed rejection at the time of dnDSA.
Follow up biopsies occurred at a median of 812 days after the baseline biopsy (392; 1624). Most patients presented with chronic active ABMR and transplant glomerulopathy at the follow up biopsy (11/22; 50%).
We did not observe a significant change in microvascular inflammation (MVI) parameters (g, ptc separately, g + ptc) or C4d on two consecutive biopsies (Table 3). However, a significant increase in the proportion of patients with a chronic transplant glomerulopathy (cg) score ≥ 1 from 27.2% to 45.4% (p = 0.04) was observed between the two biopsies (Table 3).
The eGFR dropped from a mean of 45.9 (SD: ± 16.7) at the first biopsy to 37.4 ml/min/1.73 m2 (SD: ± 13.8) at the follow up biopsy (Table 3; p = 0.005).
Thirteen of the 22 patients (59.1%) received a therapeutic regimen that included a steroid pulse (N = 8), plasma exchange (N = 6), IVIG (N = 5) and/or rituximab (N = 6). Evaluation of the overall effect of any treatment versus no treatment at the time of dnDSA on the evolution of MVI and cg score in the follow-up biopsy (Table 4) revealed no statistically significant difference. Treatment did not significantly influence the evolution of eGFR (Table 4).
We analysed each specific drug and the evolution of MVI score (Table 5) or cg score (Table 6). Steroids were associated with a reduction in the MVI score (Trend only, p > 0.05), and IVIG therapy was associated with a progression of MVI score (p < 0.05). None of the other treatments were significantly associated with a modification of scores even considering a clinically relevant baseline score for MVI (Table 5) and cg (Table 6).
The mean graft survival was 13.8 years (± 7.8 years: range 6.0-21.6) from transplantation date to the last follow up, and no graft loss was observed.
Discussion
Our retrospective analysis reports clinical and histological data from a single centre cohort of kidney transplant recipients who developed dnDSA. No improvement in histopathological parameters was observed with any therapeutic intervention. We observed a loss of kidney function and increase in cg score ≥ 1 at follow up. Graft survival was similar in patients who received treatment at the time of dnDSA and patients who did not receive therapy.
Wiebe et al. [12] analysed the clinical and histopathological correlations with dnDSA in 315 consecutive renal transplants without pre-transplant DSA. The mean follow-up was shorter (6.2 ± 2.9 years). Forty-seven of the 315 (15%) patients developed dnDSA 4.6 years post-transplant, which is earlier than our cases. They found that the two independent predictors of dnDSA were a mismatch for HLA-DRβ1 > 0 (OR 5.66, p < 0.006) and non-adherence (OR 8.75, p < 0.001). The authors demonstrated that the median 10-year graft survival for patients with dnDSA was lower than the no dnDSA group (57% vs. 96%, p < 0.0001). Their study found a non-significant trend with an odds ratio of 1.57 per rejection episode prior to an occurrence of dnDSA (p = 0.061). A high prevalence of TCMR episodes preceded the development of dnDSA (36.3%) in our study. Less than 20% of the 22 patients who developed dnDSA exhibited graft dysfunction at the time of diagnosis, which is consistent with Yamamoto et al., who reported a prevalence of 40% of patients with dnDSA and biopsy-proven subclinical ABMR [21]. Histological features at the time of dnDSA identification were not broadly consistent with ABMR in our study, and less than 50% of the biopsies exhibited an MVI score ≥ 2. C4d was negative in 59.1% of our baseline biopsies, which is consistent with previous reports [22, 23].
Further analyses of the incidence and impact of dnDSA in renal transplant recipients were performed. Forty-seven of the 189 consecutive non-sensitized, non-HLA-identical patients who received a kidney transplant between March 1999 and March 2006 (25%) developed dnDSA within 10 years [24]. The cumulative incidence 5 years post-transplantation was 20%, and more than half of these patients developed dnDSA in the first post-transplantation year. Patients of a younger age (< 35 years old), deceased donor, the presence of HLA antibodies (non-DSA) and DQ mismatch (as previously described [25]) were most likely to develop dnDSA in our study.
One strength of our analysis is the focus on the evolution of histological parameters in patients with dnDSA. Focusing on cases with negative crossmatch and DSA identified 6 months post-KT avoided the analysis of patients with preformed DSA. Our data evaluated the effect of each component of treatment on the evolution of the primary features of the Banff score associated with subtypes of ABMR [26]. Halloran et al. performed a recent principal component analysis of 164 indication biopsies [26] and found 3 main ABMR phenotypes: early ABMR with MVI lesions only (called pgABMR), late ABMR with cg lesions (cgABMR) and mixed feature (pgcgABMR).
The present study has several limitations. First, it used a retrospective analysis or different therapeutic regimens. However, we provide data on a broad heterogeneous kidney transplant recipient population that reflects routine clinical practice. Second, the small number of patients, without a control non-dnDSA group, prevents us from drawing any conclusions about the best protocol to treat dnDSA. We did not evaluate patient adherence, which is a known cause of dnDSA development and eventual graft failure [4]. Finally, an obvious pitfall was the non-standardisation of our treatment following dnDSA and biopsy results. The current evidence is insufficient to create an internationally approved protocol for the treatment of dnDSA. Our treatment protocol for ABMR evolved over time and reflects changes in practice based on the literature and a tailoring of therapy based on patient age, co-morbidities and cancer risk. Notably, the absence of IVIG as a component of the treatment given at the time of dnDSA was associated with MVI reduction. Current evidence is conflicting on the utility of IVIG in the treatment of antibody mediated injury. The benefit has been reported for active ABMR but its benefit on late/chronic ABMR is less straight forward [27].
Evidence supporting the use of rituximab for MVI or cg lesion is also scant [27, 28]. A recent randomized double-blind, placebo-controlled trial did not demonstrate a significant advantage of rituximab in biopsy-proven ABMR in creatinine level or proteinuria at 12 months [28]. The decision to treat dnDSA independently of histological features has not been addressed. Use of an intragraft transcript set measurement, as proposed in the 2013 Banff update [19], may further help the reclassification of these biopsies, but this methodology is not widely implemented yet [29].
Conclusions
We describe retrospective results from a single centre of the clinical and histological features at the time of dnDSA collection with a follow up biopsy after therapeutic management. We observed no difference in histological evolution with or without treatment. Evolution was marked by a decrease in kidney function and increase in proportion of patients with transplant glomerulopathy at the follow up biopsy. No statistically significant changes in the MVI or cg parameters were observed in the follow up biopsy of patients who received different treatments. Further intervention studies are needed to prospectively evaluate the treatment stratification and integration of the dnDSA status, biopsy histological and molecular features and patient’s functional status and comorbidities. Novel drugs are necessary to control dnDSA and its long-term effect on graft function.
Abbreviations
- ABMR:
-
Antibody mediated rejection
- ATG:
-
Antithymocyte globulin
- CDC:
-
Complement-dependent cytotoxicity
- dnDSA:
-
de novo donor specific antibody
- eGFR:
-
estimated glomerular filtration rate
- HLA:
-
Human leukocyte antigen
- IVIG:
-
Intravenous immunoglobulins
- MVI:
-
microvascular inflammation
- POD:
-
post-operative day
- SPA:
-
solid phase assay
- TCMR:
-
T-cell mediated rejection
References
Gondos A, Dohler B, Brenner H, Opelz G. Kidney graft survival in Europe and the United States: strikingly different long-term outcomes. Transplantation. 2013;95(2):267–74.
Nair R, Fraer M, Agrawal N, Suneja M. Acute transplant glomerulopathy is associated with antibody-mediated rejection and poor graft outcome. Transplant Proc. 2010;42(9):3507–12.
Einecke G, Sis B, Reeve J, et al. Antibody-mediated microcirculation injury is the major cause of late kidney transplant failure. Am J Transplant. 2009;9(11):2520–31.
Sellares J, de Freitas DG, Mengel M, et al. Understanding the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am J Transplant. 2012;12(2):388–99.
Riethmuller S, Ferrari-Lacraz S, Muller MK, et al. Donor-specific antibody levels and three generations of crossmatches to predict antibody-mediated rejection in kidney transplantation. Transplantation. 2010;90(2):160–7.
Mohan S, Palanisamy A, Tsapepas D, et al. Donor-specific antibodies adversely affect kidney allograft outcomes. J Am Soc Nephrol. 2012;23(12):2061–71.
Tsapepas DS, Vasilescu R, Tanriover B, et al. Preformed donor-specific antibodies and risk of antibody-mediated rejection in repeat renal transplantation. Transplantation. 2014;97(6):642–7.
Aubert O, Kamar N, Vernerey D, et al. Long term outcomes of transplantation using kidneys from expanded criteria donors: prospective, population based cohort study. BMJ. 2015;351:h3557.
O'Leary JG, Samaniego M, Barrio MC, et al. The influence of immunosuppressive agents on the risk of De novo donor-specific HLA antibody production in solid organ transplant recipients. Transplantation. 2016;100(1):39–53.
Ferrari-Lacraz S, Tiercy JM, Villard J. Detection of anti-HLA antibodies by solid-phase assay in kidney transplantation: friend or foe? Tissue Antigens. 2012;79(5):315–25.
Almeshari K, Pall A, Chaballout A, et al. Targeted monitoring of donor-specific HLA antibodies following renal transplantation. Clin Transpl. 2011:395–400.
Wiebe C, Gibson IW, Blydt-Hansen TD, et al. Evolution and clinical pathologic correlations of de novo donor-specific HLA antibody post kidney transplant. Am J Transplant. 2012;12(5):1157–67.
Mao Q, Terasaki PI, Cai J, et al. Extremely high association between appearance of HLA antibodies and failure of kidney grafts in a five-year longitudinal study. Am J Transplant. 2007;7(4):864–71.
Loupy A, Lefaucheur C, Vernerey D, et al. Complement-binding anti-HLA antibodies and kidney-allograft survival. N Engl J Med. 2013;369(13):1215–26.
Sicard A, Ducreux S, Rabeyrin M, et al. Detection of C3d-binding donor-specific anti-HLA antibodies at diagnosis of humoral rejection predicts renal graft loss. J Am Soc Nephrol. 2015;26(2):457–67.
Lefaucheur C, Suberbielle-Boissel C, Hill GS, et al. Clinical relevance of preformed HLA donor-specific antibodies in kidney transplantation. Am J Transplant. 2008;8(2):324–31.
Montgomery RA, Lonze BE, King KE, et al. Desensitization in HLA-incompatible kidney recipients and survival. N Engl J Med. 2011;365(4):318–26.
Djamali A, Kaufman DB, Ellis TM, Zhong W, Matas A, Samaniego M. Diagnosis and management of antibody-mediated rejection: current status and novel approaches. Am J Transplant. 2014;14(2):255–71.
Haas M, Sis B, Racusen LC, et al. Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant. 2014;14(2):272–83.
Colombo MB, Haworth SE, Poli F, et al. Luminex technology for anti-HLA antibody screening: evaluation of performance and of impact on laboratory routine. Cytometry B Clin Cytom. 2007;72(6):465–71.
Yamamoto T, Watarai Y, Takeda A, et al. De novo anti-HLA DSA characteristics and subclinical antibody-mediated kidney allograft injury. Transplantation. 2016;100(10):2194–202.
Halloran PF, Pereira AB, Chang J, et al. Microarray diagnosis of antibody-mediated rejection in kidney transplant biopsies: an international prospective study (INTERCOM). Am J Transplant. 2013;13(11):2865–74.
Gupta A, Broin PO, Bao Y, et al. Clinical and molecular significance of microvascular inflammation in transplant kidney biopsies. Kidney Int. 2016;89(1):217–25.
Everly MJ, Rebellato LM, Haisch CE, et al. Incidence and impact of de novo donor-specific alloantibody in primary renal allografts. Transplantation. 2013;95(3):410–7.
Rebellato LM, Everly MJ, Haisch CE, et al. A report of the epidemiology of de novo donor-specific anti-HLA antibodies (DSA) in "low-risk" renal transplant recipients. Clin Transpl. 2011:337–40.
Halloran PF, Merino Lopez M, Barreto PA. Identifying subphenotypes of antibody-mediated rejection in kidney transplants. Am J Transplant. 2016;16(3):908–20.
Tedla FM, Roche-Recinos A, Brar A. Intravenous immunoglobulin in kidney transplantation. Curr Opin Organ Transplant. 2015;20(6):630–7.
Sautenet B, Blancho G, Buchler M, et al. One-year results of the effects of rituximab on acute antibody-mediated rejection in renal transplantation: RITUX ERAH, a multicenter double-blind randomized placebo-controlled trial. Transplantation. 2016;100(2):391–9.
Halloran PF, Famulski KS, Reeve J. Molecular assessment of disease states in kidney transplant biopsy samples. Nat Rev Nephrol. 2016;12(9):534–48.
Acknowledgments
None.
Funding
No funding was received to prepare this manuscript. The authors of this manuscript have no conflicts of interest to disclose, as described by BMC Nephrology.
Availability of data and materials
The dataset generated and analysed during the current study is not publicly available because it contains information that could compromise research participant privacy and consent, but data are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Contributions
YB and SFL drafted the manuscript. SFL and JV performed the anti-HLA antibodies analyses. SM and OS reviewed the kidney biopsies. PYM participated in the writing of the manuscript, and KH managed the cases and thoughtfully revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The ethics committee approved the study, which was performed in accordance with the regulations of Geneva University Hospitals. The Commission cantonale d’éthique de la recherche granted approval for this retrospective study (CCER, cantonal ethical commission for research of Geneva; reference N° 6-208). Written informed consent for participation was waived because of the retrospective nature of the study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Bouatou, Y., Seyde, O., Moll, S. et al. Clinical and histological evolution after de novo donor-specific anti-human leukocyte antigen antibodies: a single centre retrospective study. BMC Nephrol 19, 86 (2018). https://doi.org/10.1186/s12882-018-0886-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12882-018-0886-5