Abstract
Background
Diet plays an important role in Helicobacter pylori (HP) infection, and our objective was to investigate potential connections between dietary patterns, specific food groups, and HP infection status in U.S. adults.
Methods
The data for this study was obtained from the NHANES (National Health and Nutrition Survey) database for the year 1999–2000. This cross-sectional study involved the selection of adults aged 20 years and older who had undergone dietary surveys and HP testing. Factor analysis was employed to identify dietary patterns, and logistic regression models were utilized to assess the association between these dietary patterns and specific food groups with HP infection status.
Result
Based on the inclusion and exclusion criteria, our final analysis included 2,952 individuals. The median age of participants was 51.0 years, and 48.7% were male. In the study population, the overall prevalence of HP infection was 44.9%. Factor analysis revealed three distinct dietary patterns: High-fat and high-sugar pattern (including solid fats, refined grains, cheese, and added sugars); Vegetarian pattern (comprising fruits, juices, and whole grains); Healthy pattern (encompassing vegetables, nuts and seeds, and oils). Adjusted results showed that the high-fat and high-sugar pattern (OR = 0.689, 95% CI: 0.688–0.690), vegetarian pattern (OR = 0.802, 95% CI: 0.801–0.803), and healthy pattern (OR = 0.717, 95% CI: 0.716–0.718) were all linked to a lower likelihood of HP infection. Further analysis of the high-fat and high-sugar pattern revealed that solid fats (OR = 0.717, 95% CI: 0.716–0.718) and cheese (OR = 0.863, 95% CI: 0.862–0.864) were protective factors against HP infection, while refined grains (OR = 1.045, 95% CI: 1.044–1.046) and added sugars (OR = 1.014, 95% CI: 1.013–1.015) were identified as risk factors for HP infection.
Conclusion
Both the Vegetarian pattern and the Healthy pattern are associated with a reduced risk of HP infection. Interestingly, the High-fat and High-sugar pattern, which is initially considered a risk factor for HP infection when the score is low, becomes a protective factor as the intake increases. Within this pattern, animal foods like solid fats and cheese play a protective role, while the consumption of refined grains and added sugars increases the likelihood of HP infection.
Similar content being viewed by others
Introduction
Helicobacter pylori (HP) has been a subject of significant concern since its discovery by Australian scholars Marshall and Warren in 1983 [1]. It affects more than half of the world’s population. Epidemiological studies indicate that the global HP infection rate is approximately 50%, and in developing countries, the rate is even higher, exceeding 70% [2, 3].HP infection is defined as an infectious disease according to the International HP Kyoto Consensus (2015) [4] and Masstricht VI Consensus (2022) [5]. It is recognized by the World Health Organization (WHO) and the International Agency for Research on Cancer (IARC) and is classified as a Group 1 carcinogen [6]. It is associated with various gastric conditions, such as peptic ulcer disease, gastric adenocarcinoma, and gastric mucosa-associated lymphoid tissue lymphoma, as well as extra-gastric diseases like metabolic syndrome, diabetes, and non-alcoholic liver disease [7].
Since the 1990s, researchers have increasingly focused on the connection between diet and HP. As research has progressed, numerous foods and ingredients have been linked to HP infection, including milk [8], honey [9], tea [9], garlic [10], soft drinks [11], and more. However, people’s daily diets are highly intricate, and the foods and nutrients they consume inevitably interact with one another. Therefore, dietary indices and dietary patterns are more suitable for understanding the relationship between HP and diet than evaluating individual foods or ingredients. Two separate studies in Iran have shown that the dietary antioxidant index [12] and glycemic index [13] are associated with HP infection. Additionally, Shi et al. [14] demonstrated that increased levels of the dietary inflammatory index are linked to a higher risk of HP infection in an American adult population. Although dietary indices can be complex to grasp and may be influenced by prior knowledge, dietary patterns are relatively easier to interpret.
Two observational studies in China have found that certain dietary patterns, such as a grain-vegetable pattern rich in whole grains and vegetables or a protein/cholesterol pattern characterized by a high intake of animal offal, animal blood, fish, seafood, and poultry, are associated with a reduced prevalence of Helicobacter pylori infection [15, 16]. Currently, the only two studies on the association between dietary patterns and HP have been conducted in China, and there are substantial differences between Chinese and Western diets. Since dietary patterns are influenced by geography and culture, it is essential to investigate the relationship between dietary patterns and HP infection in American adults in order to provide more accurate dietary recommendations for HP prevention and treatment.
Methods
Data collection and research population
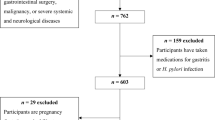
The data we analyzed was sourced from the National Health and Nutrition Examination Survey (NHANES) conducted in 1999–2000. NHANES is an ongoing survey program developed and administered by the National Center for Health Statistics (NCHS), which is part of the Centers for Disease Control and Prevention (CDC). In the 1999–2000 survey cycle, a total of 9,965 participants were excluded from our analysis. The reasons for exclusion included a lack of Helicobacter pylori antibody data (n = 2,472), absence of dietary data (n = 366), age less than 20 years old (n = 3,125), unreliable energy intake (less than 500 kcal or more than 5,000 kcal) (n = 95) [17], and missing values (n = 865) for any covariates in the model. Ultimately, 2,952 eligible subjects were included in our study. (Fig. 1)
Dietary pattern
NHANES employs a 24-hour dietary recall method to gather information about the types and quantities of food and beverages consumed by Americans. Subsequently, the Food Pattern Equivalent Database (FPED) is used to convert this dietary data into equivalent quantities for 32 food groups [18]. As FPED provides data in terms of food and beverage ingredients per 100 g, individual food intake quantities are adjusted by dividing them by 100 g and then multiplying them by the corresponding equivalent quantity in FPED. To streamline the analysis, we excluded five groups that represented the sum of various food subgroups (for instance, “Total Red and Orange Vegetables” included subgroups like “Tomatoes” and “Other Red and Orange Vegetables”). In the end, we worked with a total of 27 distinct food groups: Fruit, Fruit Juice, Dark Green Vegetables, Tomatoes, Other Red and Orange Vegetables, Potatoes, Other Starchy Vegetables, Other Vegetables, Legumes, Whole Grains, Refined Grains, Meat, Cured Meat, Organ Meat, Poultry, Seafood High in n-3 Fatty Acids, Seafood Low in n-3 Fatty Acids, Eggs, Soy Products, Nuts and Seeds, Milk, Yogurt, Cheese, Oils, Solid Fats, Added Sugars and Alcoholic Drinks. To identify dietary patterns, exploratory factor analysis was applied to these 27 food groups.
Review the scree plot and choose factors with eigenvalues exceeding 1. Apply Varimax rotation to facilitate the interpretation of identified patterns and reduce correlations between factors. Analyze the factor loadings to identify the primary contributors to each dietary pattern. Calculate factor scores for individuals based on factor loadings and their actual food intake. Finally, name the derived dietary pattern based on the food groups with the highest factor loadings (loadings>0.2) on that specific factor.
Evaluation of HP infection status
The Wampole Laboratory’s (Wampole) HP IgG enzyme-linked immunosorbent assay (ELISA) is designed to detect and quantitatively determine the concentration of HP IgG antibodies in human serum [14]. In comparison to other antibody serological tests, such as immunofluorescence, complement binding, hemagglutination, and radioimmunoassay, ELISA exhibits similar sensitivity, specificity, and repeatability. A serum IgG antibody value below 0.9 is considered negative, while an IgG value equal to or greater than 0.9 is considered positive, consistent with previous studies [19].
Other covariates
Our study examined various factors that could potentially impact HP infection, including family demographic information, lifestyle habits, and self-reported health status. Family demographic information included gender (male/female), age (in years), race (Mexican American, Other Hispanic, Non-Hispanic white, Non-Hispanic Black, Other Race), education level (Less Than 9th Grade, 9-11st Grade, High School Grade/Equivalent, Some College or Associate’s Degree, College Degree or above), and poverty income ratio (PIR). Drinking status was categorized as follows: Never (consumed fewer than 12 drinks in a lifetime), Former (consumed at least 12 drinks in one year but didn’t drink in the last year or consumed at least 12 drinks in a lifetime), Mild (consumed at least 1 drink per day for females or at least 2 drinks per day for males), Moderate (consumed at least 2 drinks per day for females or at least 3 drinks per day for males, or engaged in binge drinking on at least 2 days per month) and Heavy (consumed at least 3 drinks per day for females or at least 4 drinks per day for males) [20]. Smoking status was classified as: Never (smoked fewer than 100 cigarettes in a lifetime), Former (smoked more than 100 cigarettes in a lifetime but currently do not smoke at all), Current (smoked more than 100 cigarettes in a lifetime and currently smoke some days or every day) [20]. This information helped us assess potential associations between these factors and HP infection. We also considered Body Mass Index (BMI, Kg/m2) in our analysis. Current medical conditions taken into account included diabetes (yes/impaired fasting glucose (IFG)/no), hypertension (yes/no), heart disease (yes/no), and stroke (yes/no), determined based on self-reported information. These factors were incorporated to assess their potential influence on the study’s outcomes.
Statistical analyses
Data acquisition and calculations were carried out using R 4.2.1 software, while all analyses were performed using SPSS 26.0 software. To account for the complex survey design of NHANES data, we adhered to survey guidelines and applied appropriate sampling weights. Participants were categorized into two groups based on their HP antibody status.
In our study, we utilized “mean ± standard deviation” (mean ± SD) to represent quantitative data with a normal distribution. To test for differences between groups, we employed one-way analysis of variance (F). For quantitative data that did not follow a normal distribution, we expressed them as “median and quartiles” [M (Q1, Q3)] and compared groups using the Mann-Whitney U test. Categorical variables were described as “n (%)”, and group comparisons were conducted using the chi-square test (χ2). Statistical significance was considered at p-values less than 0.05.
Each dietary factor was classified into one of four categories based on quartiles of the score (P25, P50, P75), which were represented by numbers 1 to 4 (1 = < P25、2 = P25 ∼ P50、3 = P50 ∼ P75、4 = ≥ P75 percentile). Higher scores indicated higher intake of that dietary factor. Differences in the characteristics of each dietary pattern were assessed using one-way ANOVA for continuous variables and the chi-square test (χ2) for categorical variables. Multivariate logistic regression was employed to examine the relationship between dietary patterns and HP infection status. The independent variables were the quartiles of the factor scores for the identified dietary patterns, with the first quartile serving as the reference for each dietary pattern. Additionally, we investigated the association between specific food groups (Solid Fats, Refined Grains, Cheese, Added Sugars) and HP infection status.
Results
Characteristics of the study sample by HP infection status
A total of 2,952 individuals participated in our study, and Table 1 presents the baseline data for all participants categorized by their HP infection status. The median age of the participants was 51.0 years (with a range of 37.0 to 66.0), and the overall HP positivity rate was 44.9%. Individuals with HP infection tended to be female (P < 0.001), older (P < 0.001), have a higher BMI (P < 0.001), possess lower education levels (P < 0.001), have lower income (P < 0.001), smoke (P < 0.001), consume alcohol more frequently (P < 0.001), and had a higher prevalence of hypertension, diabetes, heart disease, and stroke (P < 0.001).
Description of dietary patterns
Following varimax rotation, factor analysis identified three dietary patterns, and the primary food factor loadings for each pattern are presented in Table 2. These three dietary patterns collectively explained 22.1% of the total variance. Based on their contributions to the total variance, the three dietary patterns were defined as follows: Factor 1 was labeled the “High fats and high sugars pattern,” characterized by dietary intake primarily consisting of solid fats, refined grains, cheese, and added sugars. Factor 2 was identified as the “Vegetarian pattern,” characterized by a dietary intake predominantly composed of fruits, fruit juices, and whole grains. Factor 3 was designated the “Healthy pattern,” marked by dietary intake primarily featuring dark vegetables, other vegetables, nuts, and vegetable oils.
Characteristics of the study sample by dietary patterns
Dietary pattern scores were categorized into quartiles to facilitate comparisons across different intake levels. The three dietary patterns exhibit their fundamental characteristics across these quartiles.
Individuals in the highest quartile of the “High fats and high sugars pattern” were generally younger, more likely to be male, more educated, had a higher BMI, higher income, were more frequent smokers and drinkers, and had a higher prevalence of high blood pressure, heart disease, and stroke, but a lower prevalence of diabetes (Supplementary Table 1).Participants with the highest scores in the “Vegetarian pattern” were older, more likely to be female, more educated, had a lower BMI, higher income, were less frequent smokers and drinkers, and had a higher prevalence of hypertension, diabetes, and heart disease, but a lower likelihood of stroke (Supplementary Table 2).Individuals with the highest scores in the “Healthy pattern” were younger, more likely to be male, more educated, had a lower BMI, higher income, smoked and drank less, and had a lower prevalence of hypertension, diabetes, heart disease, and stroke (Supplementary Table 3).
Association between dietary patterns and HP infection status
The associations between dietary patterns and HP infection status are presented in Table 3. The “High fats and high sugars pattern” exhibited a negative association with the overall prevalence of HP infection (P < 0.001). After adjusting for confounding factors, it was observed that the prevalence of HP infection among participants in the second quartile of the “High fats and high sugars pattern” was 1.18 times higher than that of participants in the first percentile (95% CI, 1.177–1.179). However, as the score increased, the “High fats and high sugars pattern” gradually transitioned into a protective factor, with a multivariable-adjusted odds ratio (OR) for the prevalence of HP infection in participants in the highest quartile being 0.689 (95% CI, 0.688–0.690).
The “Vegetarian pattern” and the “Healthy pattern” were both linked to a reduced prevalence of HP infection (P < 0.001), and these associations remained consistent after adjusting for confounding variables, with ORs of 0.802 (95% CI, 0.801–0.803) and 0.717 (95% CI, 0.716–0.718), respectively.
Association between solid fats, refined grains, cheese, added sugars and HP infection status
As solid fats, refined grains, cheese, and added sugars are fundamental components of the “High fats and high sugars pattern,” we examined their intake in relation to HP infection status(Table 4). Adjusted refined grains and added sugars consistently demonstrated as risk factors for HP infection (P < 0.001). In contrast, solid fats and cheese from animal sources exhibited a protective effect with increasing intake. The ORs decreased from 1.189 (95% CI, 1.187–1.190) to 0.717 (95% CI, 0.716–0.718) for solid fats and from 1.116 (95% CI, 1.115–1.118) to 0.863 (95% CI, 0.862–0.864) for cheese, respectively.
Discussion
A total of 2,952 participants who met the criteria were included in this cross-sectional study. This study represents the first attempt to assess the association between dietary patterns and HP infection status in U.S. adults. Our findings indicated that the “Vegetarian pattern” and the “Healthy pattern” were negatively associated with HP infection rates. Conversely, the “High fats and high sugars pattern,” characterized by solid fats, refined grains, cheese, and added sugars, transitioned from a risk factor to a protective factor for HP infection as intake increased. Further analyses indicated that within the “High fats and high sugars pattern,” refined grains and added sugars were the primary factors contributing to an increased risk of HP infection. On the other hand, solid fats and cheese from animal sources exhibited potential protective effects.
The protective effect observed in the “High fats and high sugars pattern” may not be entirely consistent with previous research. However, this discrepancy can be explained by our further analysis of food groups. Our findings suggest that solid fats and cheeses from animal sources ultimately play a protective role. Foods of animal source are rich in vitamins and trace minerals such as vitamin A, D, selenium and zinc, which inhibit the colonization of HP and the inflammation induced by HP [21], and even influence HP eradication [22]. In a recent case-control study in Iran [23], there was no significant difference in fat intake between healthy participants and HP-infected individuals when dietary intake was used as a variable (P = 0.398). However, when the variable in this study was the food group, animal foods (including meat and processed meats) were considered risk factors for HP infection. We believe that broadly grouping meat with processed meats such as sausages may obscure the benefits of animal-based foods. As another Iranian study concluded by grouping meat samples more carefully [24], it is sausages, burgers, and fatty mayonnaise that are truly positively associated with HP infection, rather than meat and fat. The effects of refined carbohydrates and added sugars are similar to those of processed meats, as these ultra-processed foods are often overloaded with salt and sugar for taste, preservation, and cost, which tend to increase the colonization of HP as well as interfere with the protective effects of intragastric mucus and gastric mucosa [13, 25]. Animal foods have been shown to provide better nutritional status to combat HP infection [26, 27], and recent research in other areas has shifted the previous view of animal foods [28, 29]. However, it is undeniable that consuming too much animal food can bring about obesity and a variety of chronic metabolic diseases, so our results need to be taken with a grain of salt, besides more research is needed to elucidate its role.
The vegetarian pattern, characterized by a high intake of fruits and vegetables, typically acts as a protective factor against various diseases, thus diminishing the role of HP [30, 31]. This protective effect is attributed to the presence of antioxidants like carotenoids, vitamin C, and vitamin E in fruits and vegetables, which also help deter the progression of HP-related diseases such as atrophic gastritis [32]. Nevertheless, it’s important to acknowledge certain shortcomings in vegan diets. People following plant-based diets may experience insufficient intake of protein, vitamins B12 and D, calcium, iron, and zinc due to either their low levels in plant-based foods or limited absorption [33]. Deficiencies in these macronutrients, vitamins, and micronutrients not only increase the risk of HP infection but also elevate the risk of conditions like hemorrhagic stroke and fractures [34]. The efficiency of protein utilization tends to decrease with age, and abstaining from animal foods may not fulfill the nutritional needs of older adults [35]. More significantly, these nutrient deficiencies are often more pronounced in children and pregnant or lactating women, leading to serious consequences like megaloblastic anemia and irreversible neurological damage [36].
The quality of a vegan diet plays a crucial role in reducing the risk of various chronic diseases. In the absence of dietary and medical supervision, the consumption of highly processed plant-based products can compromise the effectiveness of a plant-based diet and even have adverse effects. Additionally, this dietary pattern often includes the consumption of raw vegetables and fruits. As a foodborne [37] and waterborne [38] pathogen, HP can be transmitted to humans through this pathway. HP has the capacity to form biofilms and thrive as colonies on the surfaces of vegetables, greatly extending its survival [39]. This is why frequent consumption of raw vegetables like cucumbers [40], tomatoes, and peppers [41] has been associated with an increased risk of HP infection in some studies. However, relatively speaking, groups with higher fruit and vegetable consumption are usually more affluent and better educated [42], especially since our study population came from a developed country like the United States, which typically boasts better sanitary conditions compared to many other countries. The protective effect ultimately results from the combined influence of dietary and socio-economic factors.
We also note that some researchers have questioned the relationship between HP and diet [43], suggesting that HP is usually acquired in childhood and that dietary factors may play a role later in life after HP infection. The prevalence of HP infection in children has been reported to about 33% (with variations in different regions) [44], and the difference in the prevalence with adults suggests that HP infection status in adulthood may arise from two sources, maintenance of infection in childhood and acquisition in adulthood. There is still no consensus on screening in childhood [45], and obtaining and following up this part of the data becomes difficult. Children’s dietary habits change with age, in addition to the influence of geographic factors, economic status, and level of education, so the current study could not clarify the difference between the role of diet in the maintenance and acquisition of HP infection. However, diet remains worthy of investigation as the cheapest and most easily achievable variable factor in HP infection prevention and treatment for adults with stabilized dietary habits and physical status.
In summary, our research is the first to investigate the relationship between dietary patterns and the risk of HP infection among U.S. adults. Previous studies conducted in the provincial capital of Zhejiang, China [16] were limited by differences in economic income and education levels compared to other provinces and cities, which may affect the generalizability of the results. However, NHANES provided a representative sample of Americans through its sampling methodology, allowing our findings to be more widely applicable. Notably, in another study on dietary patterns [15], no significant association was observed between any food group and Helicobacter pylori infection, whereas our study identified distinct roles of specific food groups in HP infection. Nonetheless, our study has some limitations. Firstly, it was cross-sectional, and as such, it cannot establish a causal relationship between dietary patterns and the risk of HP infection. Secondly, there may be recall bias in dietary assessment due to the use of the Food Frequency Questionnaire (FFQ), which could lead to inaccuracies in food categorization and intake estimation. Thirdly, HP infection status was determined solely by HP-specific IgG antibodies, and this method may introduce bias in the diagnosis of infection.
Conclusion
Our study revealed that dietary patterns play a significant role in the prevalence of HP infection among U.S. adults. Specifically, a Vegetarian dietary pattern characterized by high fruit, fruit juice, and whole grain consumption, as well as a healthy dietary pattern rich in vegetables, nuts, and vegetable oils, were associated with a reduced risk of HP infection status. Interestingly, a dietary pattern high in fats and sugars, featuring increased intake of solid fats, refined grains, cheese, and added sugars, was also found to be a protective factor against HP infection. However, further analysis suggested that reducing the consumption of added sugars and refined grains, while not overly restricting the intake of cheese and solid fats, may help lower the risk of HP infection status.
Data availability
All data of the study are presented in the text or supplementary materials. The datasets analyzed during the current study were publicly available from the NAHNES. Data from the NHANES can be found at https://www.cdc.gov/nchs/nhanes/index.htm. The FPED is available at https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/fped-overview/.
Abbreviations
- NHANES:
-
National Health and Nutrition Examination Survey
- HP:
-
Helicobacter pylori
- PIR:
-
Poverty Income Ratio
- MA:
-
Mexican American
- OH:
-
Other Hispanic
- NHW:
-
Non-Hispanic White
- NHB:
-
Non-Hispanic Black
- OR:
-
Other race
- DM:
-
Diabetes Mellitus
- IFG:
-
Impaired Fasting Glucose
- Q:
-
Quartiles
- CI:
-
Confidence interval
References
Marshall BJ, Warren JR. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet. 1984;1(8390):1311–5.
Mezmale L, Coelho LG, Bordin D, Leja M. Review: epidemiology of Helicobacter pylori. Helicobacter. 2020;25(Suppl 1):e12734.
Li M, Sun Y, Yang J, de Martel C, Charvat H, Clifford GM, Vaccarella S, Wang L. Time trends and other sources of variation in Helicobacter pylori infection in mainland China: a systematic review and meta-analysis. Helicobacter. 2020;25(5):e12729.
Sugano K, Tack J, Kuipers EJ, Graham DY, El-Omar EM, Miura S, Haruma K, Asaka M, Uemura N, Malfertheiner P, et al. Kyoto global consensus report on Helicobacter pylori gastritis. Gut. 2015;64(9):1353–67.
Malfertheiner P, Megraud F, Rokkas T, Gisbert JP, Liou JM, Schulz C, Gasbarrini A, Hunt RH, Leja M, O’Morain C et al. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut 2022.
Schistosomes. Liver flukes and Helicobacter pylori. IARC Monogr Eval Carcinog Risks Hum. 1994;61:1–241.
Tsay FW, Hsu PI. H. Pylori infection and extra-gastroduodenal diseases. J Biomed Sci. 2018;25(1):65.
Obaidat MM, Roess AA. First nationwide seroepidemiology and risk factors report of Helicobater Pylori in Jordan. Helicobacter. 2019;24(3):e12572.
Boyanova L, Ilieva J, Gergova G, Vladimirov B, Nikolov R, Mitov I. Honey and green/black tea consumption may reduce the risk of Helicobacter pylori infection. Diagn Microbiol Infect Dis. 2015;82(1):85–6.
Salih BA, Abasiyanik FM. Does regular garlic intake affect the prevalence of Helicobacter pylori in asymptomatic subjects? Saudi Med J. 2003;24(8):842–5.
Nseir W, Mograbi J, Di Castro N, Abu-Elheja O, Abu-Rahmeh Z, Khamaysi I, Samara M, Assy N. On the association between soft drink consumption and Helicobacter pylori infection. Dig Dis Sci. 2012;57(4):981–6.
Ebrahimi Z, Masoodi M, Aslani Z, Naghshi S, Khalighi Sikaroudi M, Shidfar F. Association between dietary antioxidant index and risk of Helicobacter pylori infection among adults: a case-control study. BMC Gastroenterol. 2022;22(1):413.
Sohouli MH, Haghshenas N, Pouladi F, Sayyari A, Olang B, Gaman MA, Kord-Varkaneh H, Fatahi S. Association between glycemic index and Helicobacter pylori infection risk among adults: a case-control study. Nutrition. 2021;83:111069.
Shi L, Zhang D. Association of Dietary Inflammation Index and Helicobacter pylori Immunoglobulin G Seropositivity in US Adults: A Population-Based Study. Mediators Inflamm 2023, 2023:8880428.
Xia Y, Meng G, Zhang Q, Liu L, Wu H, Shi H, Bao X, Su Q, Gu Y, Fang L, et al. Dietary patterns are Associated with Helicobacter Pylori infection in Chinese adults: a cross-sectional study. Sci Rep. 2016;6:32334.
Shu L, Zheng PF, Zhang XY, Feng YL. Dietary patterns and Helicobacter pylori infection in a group of Chinese adults ages between 45 and 59 years old: an observational study. Med (Baltim). 2019;98(2):e14113.
Kim WK, Shin D, Song WO. Are dietary patterns Associated with Depression in U.S. adults? J Med Food. 2016;19(11):1074–84.
Food Surveys Research Group. USDA ARS. [Food Surveys Research Group: USDA ARS.].
Huang JW, Xie C, Niu Z, He LJ, Li JJ. The relation between Helicobacter pylori immunoglobulin G seropositivity and leukocyte telomere length in US adults from NHANES 1999–2000. Helicobacter. 2020;25(6):e12760.
Chen C, Zhang H, Lan Y, Yan W, Liu S, Chen Y, Xie T, Ning J, Yan X, Shang L, et al. Statins as a risk factor for diabetic retinopathy: a mendelian randomization and cross-sectional observational study. J Transl Med. 2024;22(1):298.
Nabavi-Rad A, Azizi M, Jamshidizadeh S, Sadeghi A, Aghdaei HA, Yadegar A, Zali MR. The Effects of Vitamins and Micronutrients on Helicobacter pylori Pathogenicity, Survival, and Eradication: A Crosstalk between Micronutrients and Immune System. J Immunol Res 2022, 2022:4713684.
Han C, Ni Z, Yuan T, Zhang J, Wang C, Wang X, Ning HB, Liu J, Sun N, Liu CF, et al. Influence of serum vitamin D level on Helicobacter pylori eradication: a multi-center, observational, prospective and cohort study. J Dig Dis. 2019;20(8):421–6.
Nabavizadeh R, Sohouli MH, Santos HO, Roustaei M, Fatahi S, Ghodoosi N, Saeidi R. Higher dietary total antioxidant capacity is inversely associated with Helicobacter pylori infection among adults: a case-control study. Indian J Gastroenterol. 2022;41(3):258–65.
Mard SA, Khadem Haghighian H, Sebghatulahi V, Ahmadi B. Dietary factors in relation to Helicobacter pylori Infection. Gastroenterol Res Pract. 2014;2014:826910.
Fahey JW, Stephenson KK, Wallace AJ. Dietary amelioration of Helicobacter infection. Nutr Res. 2015;35(6):461–73.
Hall AG, Ngu T, Nga HT, Quyen PN, Hong Anh PT, King JC. An animal-source food supplement increases Micronutrient Intakes and Iron Status among Reproductive-Age women in Rural Vietnam. J Nutr. 2017;147(6):1200–7.
Oztekin M, Yilmaz B, Agagunduz D, Capasso R. Overview of Helicobacter pylori infection: clinical features, treatment, and nutritional aspects. Diseases 2021, 9(4).
Orlich MJ, Sabate J, Mashchak A, Fresan U, Jaceldo-Siegl K, Miles F, Fraser GE. Ultra-processed food intake and animal-based food intake and mortality in the Adventist Health Study-2. Am J Clin Nutr. 2022;115(6):1589–601.
Sun Z, Liu L, Wang PP, Roebothan B, Zhao J, Dicks E, Cotterchio M, Buehler S, Campbell PT, McLaughlin JR, et al. Association of total energy intake and macronutrient consumption with colorectal cancer risk: results from a large population-based case-control study in Newfoundland and Labrador and Ontario, Canada. Nutr J. 2012;11:18.
Wang XQ, Yan H, Terry PD, Wang JS, Cheng L, Wu WA, Hu SK. Interaction between dietary factors and Helicobacter pylori infection in noncardia gastric cancer: a population-based case-control study in China. J Am Coll Nutr. 2012;31(5):375–84.
Phuoc LH, Sengngam K, Ogawa T, Ngatu NR, Ikeda S, Hoc TH, Phu PV, Minh DT, Ngoan LT. Fruit and vegetable intake and stomach Cancer among male adults: a case-control study in Northern Viet Nam. Asian Pac J Cancer Prev. 2020;21(7):2109–15.
Izzotti A, Durando P, Ansaldi F, Gianiorio F, Pulliero A. Interaction between Helicobacter pylori, diet, and genetic polymorphisms as related to non-cancer diseases. Mutat Res. 2009;667(1–2):142–57.
Neufingerl N, Eilander A. Nutrient intake and status in adults consuming plant-based diets compared to Meat-Eaters: a systematic review. Nutrients 2021, 14(1).
Key TJ, Papier K, Tong TYN. Plant-based diets and long-term health: findings from the EPIC-Oxford study. Proc Nutr Soc. 2022;81(2):190–8.
Melina V, Craig W, Levin S. Position of the Academy of Nutrition and Dietetics: vegetarian diets. J Acad Nutr Diet. 2016;116(12):1970–80.
Wang T, Masedunskas A, Willett WC, Fontana L. Vegetarian and vegan diets: benefits and drawbacks. Eur Heart J. 2023;44(36):3423–39.
Quaglia NC, Dambrosio A. Helicobacter pylori: a foodborne pathogen? World J Gastroenterol. 2018;24(31):3472–87.
Aziz RK, Khalifa MM, Sharaf RR. Contaminated water as a source of Helicobacter pylori infection: a review. J Adv Res. 2015;6(4):539–47.
Ng CG, Loke MF, Goh KL, Vadivelu J, Ho B. Biofilm formation enhances Helicobacter pylori survivability in vegetables. Food Microbiol. 2017;62:68–76.
Goto Y, Syam AF, Darnindro N, Hapsari FC. Prevalence and risk factors for Helicobacter Pylori Infection among healthy inhabitants in Northern Jakarta, Indonesia. Asian Pac J Cancer Prev. 2016;17(10):4747–53.
Monno R, De Laurentiis V, Trerotoli P, Roselli AM, Ierardi E, Portincasa P. Helicobacter pylori infection: association with dietary habits and socioeconomic conditions. Clin Res Hepatol Gastroenterol. 2019;43(5):603–7.
Wang T, Cai H, Sasazuki S, Tsugane S, Zheng W, Cho ER, Jee SH, Michel A, Pawlita M, Xiang YB, et al. Fruit and vegetable consumption, Helicobacter pylori antibodies, and gastric cancer risk: a pooled analysis of prospective studies in China, Japan, and Korea. Int J Cancer. 2017;140(3):591–9.
Pellicano R. Diet, glycemic index, and Helicobacter pylori infection: which comes first? Nutrition. 2021;90:111416.
Zabala Torrres B, Lucero Y, Lagomarcino AJ, Orellana-Manzano A, George S, Torres JP, O’Ryan M. Review: prevalence and dynamics of Helicobacter pylori infection during childhood. Helicobacter 2017, 22(5).
Borka Balas R, Melit LE, Marginean CO. Worldwide Prevalence and Risk factors of Helicobacter pylori infection in children. Child (Basel) 2022, 9(9).
Acknowledgements
We sincerely thank Zhang Jing (Second Department of Infectious Disease, Shanghai Fifth People’s Hospital, Fudan University) for his work on the NHANES database. His outstanding work, nhanesR package and webpage, makes it easier for us to explore NHANES database.
Funding
This work was supported by Shaanxi Key Research & Development Projects (No. 2023-ZDLSF-35).
Author information
Authors and Affiliations
Contributions
Author Contributions: Conceptualization: HZY, SL, SYQ, WCC and ZH; Acquisition of data: ZH, ZJ, YJM, PQM and SL; Analysis of data: ZH, XC, YN, LZH and SL; Writing original draft: ZH, XC, and SL; Writing review and editing: HZY, SYQ and SL. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
As the data used in this study were obtained from publicly available NHANES data, all data-related research had previously received approval from their respective ethical review committees and had obtained written informed consent from the participants. Consequently, this study does not require additional ethical approval.
Consent for publication
We used anonymous de-identified data that are publicly available from the NHANES. The NHANES obtained informed consent from all participants. Information related to this process can be found at We used anonymous de-identified data that are publicly available from the NHANES. The NHANES obtained informed consent from all participants. Information related to this process can be found at https://wwwn.cdc.gov/nchs/nhanes/continuousnhanes/documents.aspx?BeginYear=2017 for NHANES 2017–2018 (accessed on 21 September 2023).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, H., Xu, C., Zhang, J. et al. The intake of solid fat and cheese may be associated with a reduced risk of Helicobacter pylori infection status: a cross-sectional study based on NHANES 1999–2000. BMC Infect Dis 24, 493 (2024). https://doi.org/10.1186/s12879-024-09392-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12879-024-09392-z