Abstract
Background
Toxoplasma gondii is an opportunistic protozoan parasite that can infect all warm-blooded animals including humans and cause serious clinical manifestations. Toxoplasmosis can be diagnosed using histological, serological, and molecular methods. In this study, we aimed to detect T. gondii RE gene in various human samples by in house and commercial real time polymerase chain reactions.
Methods
A total of 38 suspected cases of toxoplasmosis [peripheral blood (n:12), amnion fluid (n:11), tissue (n:9), cerebrospinal fluid (n:5), and intraocular fluid (n:1)] were included to the study. An in house and a commercial RT-PCR were applied to investigate the T. gondii RE gene in these samples.
Results
The compatibility rate of the two tests was 94.7% (37/38). When the commercial RT-PCR kit was taken as reference, the sensitivity and specificity of in house RT-PCR test was 87.5 and 100%. When the in house RT-PCR test was taken as reference, the commercial RT-PCR kit has 100% sensitivity and 96.8% specificity. Incompatibility was detected in only in a buffy coat sample with high protein content.
Conclusions
Both the commercial and in house RT-PCR tests can be used to investigate T. gondii RE gene in various clinical specimens with their high sensitivity and specificity. In house RT-PCR assay can be favorable due to cost savings compared to using the commercial test.
Similar content being viewed by others
Background
Toxoplasma gondii is an opportunistic protozoan parasite that can infect all warm-blooded animals including birds and can cause serious clinical manifestations. T. gondii causes congenital toxoplasmosis leading to fetal anomalies in newborns, retinochoroiditis causing blindness, deadly toxoplasmic encephalitis in immunocompromised patients or transplant recipients [1, 2].
Toxoplasmosis can be diagnosed using histological, serological, and molecular methods [1, 3,4,5,6]. In recent years, serological and molecular diagnostic methods are frequently used. Serological screening for toxoplasmosis is performed in pregnant women in most of the countries during the first trimester. When IgM positivity and low IgG avidity are detected in the pregnant woman, there is a risk of congenital toxoplasmosis in the fetus [1]. In order to rule out this suspicion, amniocentesis is recommended during the 16-18th weeks of gestation and amniotic fluid is investigated with Polymerase chain reaction (PCR) to detect T. gondii DNA [1, 7]. In addition, molecular techniques become important due to decreased levels of antibodies in immune suppressed patients [8,9,10].
The majority of T. gondii strains from most parts of the world belong to three different clonal clade, called type I, type II and type III. There are also atypical and recombinant strains present [11,12,13,14,15]. The T. gondii B1 gene (GenBank no: AF179871) containing 35 copies, the RE gene (GenBank no: AF146527) containing 200–300 copies and the SAG1 (GenBank: M23658) gene consisting of single copy were used in molecular diagnostic tests [16,17,18,19]. In a recent study, the copy numbers for the B1 gene were found to be 5, 12 and 7 times lower than the previous estimations of 35 copies for type I, type II and Type III strains, respectively. For T. gondii RE gene were found to be 8, 4 and 4 times lower than the previous estimations of 200–300 copies for type I, type II and Type III strains, respectively [17]. PCR studies performed in various clinical samples showed that T. gondii RE gene was more sensitive and specific than B1 gene [20,21,22,23,24,25,26,27,28,29]. Moreover Toxoplasma Reference centers in Europe are using PCR reactions targeting T. gondii RE gene more frequently compared to T. gondii PCR targeting B1 gene [30, 31].
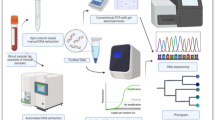
In this study, two hybridization probe based methods which are an in house Real Time PCR and a commercial Real Time PCR targeting T. gondii RE gene were compared in terms of sensitivity and specificity using different clinical samples.
Methods
Clinical samples
In this study, 38 specimens [12 peripheral blood sample, 11 amniotic fluid, 9 tissue, 5 cerebrospinal fluid (CSF) and 1 intraocular fluid] obtained from 38 clinically toxoplasmosis suspected seropositive patients admitted to Ege University Faculty of Medicine, were investigated for the presence of T. gondii using an in house Real Time PCR and a commercial Real Time PCR targeting RE gene.
Specimen processing and DNA extraction
DNA extraction from amniotic fluid, peripheral blood, tissue, CSF, or intraocular fluid sample was performed using the Qiagen Mini Kit according to the manufacturer’s protocol. Initially, specimens were processed. During the DNA extraction of blood sample, the buffy coat part was used [31,32,33]. For this purpose, 2–4 ml whole blood in EDTA was centrifuged at 3000 rpm for 15 min. After centrifugation, plasma was discharged until ~ 300 μl plasma remained. Then, 500 μl of sample was used for DNA extraction [~ 300 μl plasma (+) ~ 100 μl buffy coat (+) ~ 100 μl of erythrocyte cluster]. During the extraction of DNA from amniotic fluid, at least 6 ml sample was used [34]. For a sample more than 6 ml, whole specimen was transferred to a sterile 15 ml tube and centrifuged at 3000 rpm for 15 min. Next, the supernatant was discarded until ~ 6 ml amniotic fluid remained. For the tissue, 50 mg sample was finely chopped and added to tube containing ATL buffer, 80 μl Proteinase K, 0,1 mm glass beads, and 2 mm zirconia beads (BioSpec Products, U.S.A.) and incubated for 15 min at 56 °C with 1400 rpm in a heat shaker (Lab4You) until the sample melted. Next, the tissue samples were incubated at 95 °C for 5 min. After incubation, samples were vortexed for 2 min on a Disruptor Genie (Scientific Industries, U.S.A.) [35, 36]. CSF and intraocular fluid samples were used directly when the volume of sample was ~ 200 μl, and in the case of excess, the sample was centrifuging at 3000 rpm and the supernatant was discarded until ~ 200 μl sample remained.
Real time PCR
In house Real Time PCR to detect 134 bp region of T. gondii RE gene (GenBank no: AF146527) was performed as described with some minor modifications [20]. The analysis of results was performed by 1.5 LightCycler Real Time instrument using LightCycler software, Version 3.5 according to the manufacturers protocol (Roche). Twenty microliter final volume PCR reaction included 1x LightCycler Fast Start DNA Master HybProbe mix with MgCl2 final concentration adjusted to 5 mM (Roche), the primers 5′-AGGCGAGGGTGAGGATGA-3′ (18 nt, TOX-SE forward primer; final concentration: 0.5 μM) and 5′-TCGTCTCGTCTGGATCGCAT-3′ (20 nt, TOX-AS reverse primer; final concentration: 0.5 μM), the hybridization probes 5′-GCCGGAAACATCTTCTCCCTCTCC-3′-FL (24 nt, TOX FLU, labeled at the 3′ end with fluorescein; final concentration: 0.1 μM) and 5′-640-CTCTCGTCGCTTCCCAACCACG-3′ (22 nt, TOX LCR labeled at the 5′ end with LC-Red 640; final concentration: 0.5 μM) (IDT), DNA template or controls. The PCR amplification reactions were performed by the following calculated protocol: 10 min initial denaturation step at 95 °C, followed by 50 cycles of 5 s at 95 °C, 10 s at 60 °C, and 15 s at 72 °C. PCR reactions were performed in duplicate.
As positive and negative controls, T. gondii genomic DNA serially 10-fold diluted ranging from 106 to 101 parasites/μl and distilled water were used, respectively. In clinical samples and positive controls, crossing point (Cp) values were used to assess the amount of T. gondii DNA. Melting curve analysis was performed using the following calculated protocol: 20 s denaturation step at 95 °C followed by 20 s annealing step at 40 °C and extension step gradually increasing temperature to 85 °C.
To analyze the PCR inhibition attributable to the specimen matrix, PCR reactions were prepared in duplicate for each sample. One of the reactions contained only purified patient DNA sample and the other reaction contained 1 T. gondii tachyzoite spiked into the purified patient sample. The sample is accepted as inhibited when the spiked control did not show any crossing point (Cp) value.
The commercial T. gondii Real Time PCR kit (TIBMolBiol) targeting 134 bp fragment of T. gondii RE gene was applied according to the manufacturer’s protocol. The analysis of results was performed by 1.5 LightCycler Real Time instrument using LightCycler software, Version 3.5 according to the manufacturers protocol (Roche). In this test, the internal control target was investigated as multiplex. The control reaction mix contained the primer and the probe mix that determines the internal control, while the reaction also included the lambda phage as the extraction control target. Twenty microliter final volume PCR reaction included 1x LightCycler Fast Start DNA Master HybProbe mix with MgCl2 final concentration adjusted to 4.5 mM (Roche), 2 μl PSR mix (parameter specific reagents; containing T. gondii RE gene specific primers and hybridization probes), DNA template or controls, 2 μl control target (lambda phage) and 0.5 μl control reaction mix (contains primers and probe for lambda phage). The PCR amplification reactions were performed by the following calculated protocol: 10 min initial denaturation step at 95 °C, followed by 50 cycles of 5 s at 95 °C, 5 s at 62 °C, and 15 s at 72 °C. Moreover, a single cycle of 55 °C with 0.5 step size was included to the amplification step. PCR reactions were performed in duplicate.
As positive and negative controls, T. gondii plasmid containing 134 bp fragment of RE gene serially 10-fold diluted ranging from 106 to 101 copy plasmids/reaction and distilled water were used. In clinical samples and positive controls, crossing point (Cp) values were used to assess the amount of T. gondii DNA. Melting curve analysis was performed using the following calculated protocol: 20 s denaturation step at 95 °C followed by 20 s annealing step at 40 °C and extension step gradually increasing temperature to 85 °C.
Results
According to the results, among the 38 clinical samples analyzed, T. gondii RE gene was not detected in 30 of them and T. gondii RE gene was detected in 6 samples by both in house Real Time PCR and commercial Real Time PCR (Table 1). Regarding the remaining two samples; one sample was partly compatible in both assays in which T. gondii RE gene was detected during the first run and not detected in the subsequent using in house Real Time PCR, whereas the commercial Real Time PCR was positive in both runs for this sample. In the remaining sample, the results were completely incompatible between both assays (Table 1). Both of these samples were buffy coat samples. As a result, the compatibility rate of both tests was 97.4% (37/38) after two tests.
The sensitivity of the in house Real Time PCR was 87.5% (7/8) and the specificity was 100% (30/30) when we take commercial Real Time PCR as the golden standard. Conversely, if we take in house Real Time PCR as the golden standard, the sensitivity and specificity of the commercial Real Time PCR was 100% (7/7) and 96.8% (30/31), respectively. Due to low sample size, individual sensitivity and specificity values were not calculated.
As the results of the melting curve analysis of positive control and positive clinical samples were examined, it was found that Tm of all samples peaked around 67.5 ± 1 °C using in house and commercial Real Time PCRs. When the internal control data were examined, any inhibition was not detected in both tests. As the positive controls were examined, it was found that the analytical sensitivity of the commercial Real Time PCR was 10 copy plasmid/reaction and the analytical sensitivity of the in house Real Time PCR was 1 parasite/reaction. No signal was detected in the distilled water negative control samples.
Discussion
Molecular diagnostic tests have high sensitivity and specificity in diagnosing congenital toxoplasmosis, toxoplasmic retinochoroiditis, as well as organ transplantation and AIDS patients [37]. T. gondii B1 and RE genes are the most frequently targeted markers in the molecular diagnosis of toxoplasmosis due to their high copy numbers [16, 17]. Some studies have shown that the T. gondii RE gene is more sensitive and specific than the B1 gene [20,21,22,23,24,25,26,27,28,29,30]. In East Africa, blood samples of 63 patients were investigated with Real Time PCR methods targeting the B1 and RE genes of T. gondii. Interestingly, B1 gene was detected in all cases known to be positive for T. gondii, whereas in 3 samples (4.8%), RE gene could not be detected. The authors stated that the G/C mutation at position 275 of the RE gene inhibited the binding of the primer to the RE gene and affected real time PCR results. In addition, the genotypes detected in Africa are different from Europe. The higher ratio of RE/B1 copy number detected in type II strains which is more common in Europe has been shown to be a cause of the high specificity and success of real time PCR targeting the RE gene in Europe [29].
In Turkey, two cases of congenital toxoplasmosis detected in 1972 and 2007 have been isolated from two infants and has been shown to be similar to the isolates of African genotype 1 [38]. In addition, two studies conducted in big groups of cats and wild birds of Turkey, Type II and III strains are found to be more frequent like in Europe [14, 15]. In our country, more frequent detection of type II strains in animals suggests that PCR targeting the T. gondii RE gene may be more sensitive. Thus, in this study, two very similar hybridization probe based methods which are an in house Real Time PCR described by Cassaign et al., 2006 and a commercial Real Time PCR kit targeting the same region of T. gondii RE gene were first time compared in terms of sensitivity and specificity using various clinical samples.
A total of 38 suspected cases of toxoplasmosis were included to the study. The reason for low sample size can be attributed to presence of T. gondii in blood approximately for only 2 weeks during acute infection of immunocompetent patients [1, 2]. Besides, in a patient infected with T. gondii, seroconversion (i.e. IgG positivity) takes place around 15 days and as the clinicians order to perform T. gondii PCR, tachyzoites may become latent in tissues (also called chronic toxoplasmosis) and not found in blood circulation [1, 2]. In addition, an important clinical presentation called congenital toxoplasmosis is a rare disease according to the Oprhanet report series [39]. Overall, T. gondii DNA positivity may not be frequently detected in blood or amniotic fluid samples of toxoplasmosis cases. Thus, although we aimed to evaluate more T. gondii DNA positive samples in this study, we couldn’t achieve it and the low T. gondii DNA positive sample size can affect the interpretation of the sensitivity ratio which is the limitation of this study.
The results showed that the compatibility rate of the in house Real Time PCR and commercial Real Time PCR targeting the T. gondii RE gene was 97.4% using 38 different clinical samples. Among the 38 samples analyzed in this study, 36 of them (six positive and 30 negative) were found to be fully compatible, while one buffy coat sample was partly compatible and the other was completely incompatible (Table 1). In these two samples, the UV260/280 ratio was> 1.8 and no inhibition was observed. In one sample, the in house Real Time PCR was one time negative and the other time positive which can be attributed to a low T. gondii DNA in the sample (Table 1). In this sample, the Cp value of the in house Real Time PCR was 36, supporting the difference among the results. Similarly, with the commercial Real Time PCR, the Cp value of 34.6 supports the fact that the concentration of T. gondii RE gene was low. Consequently, the commercial Real Time PCR kit can be considered to be more sensitive in detecting the T. gondii RE gene. In this situation, the problem of hypersensitivity of the PCR may occur leading to false positivity due to forced cross-linking of primers and probes to primer dimers at the last cycles of the PCR. In this study, the Cp value of the buffy coat sample (which was found two times negative with in house Real Time PCR and two times positive with commercial Real Time PCR kit) as detected by commercial Real Time PCR kit were 34.3 and 34 and can be evaluated as hypersensitivity. Conversely, the inaccurate negative result as detected by in house Real Time PCR may be due to the loss of sensitivity because of high protein content in buffy coat. In a study, the buffy coat samples of 44 patients with clinically proven acute toxoplasmosis was investigated in two different centers by hybridization probe based Real Time PCR assay targeting the T. gondii RE gene. Both centers found 25% (9/44) positivity. Only four of these nine patients were found to be positive in both centers, while four samples were positive in one center and one sample positive in another center [33]. As a result, buffy coat samples can be considered as challenging samples for the detection of T. gondii DNA.
Conclusions
Both tests were compatible with amniotic fluid, tissue, CSF or intraocular fluid samples, and showed complete incompatibility only in a buffy coat sample with a dense protein content. In particular, it may be useful to analyze high protein content samples such as buffy coat twice to detect low amount of T. gondii DNA. As a result, the high compatibility, analytical sensitivity, sensitivity, and specificity of both tests suggest that the Real Time PCR methods targeting T. gondii RE gene can be used to investigate T. gondii DNA in various clinical samples. In house real time PCR can be preferred by developing laboratories due to cost savings.
Availability of data and materials
The dataset analyzed during the current study is available from the corresponding author on reasonable request.
Abbreviations
- CSF:
-
Cerebrospinal fluid
- DNA:
-
Deoxyribonucleic acid
- IgG:
-
Immune globulin G
- IgM:
-
Immune globulin M
- min:
-
minutes; rpm: rounds per minute
- PCR:
-
Polymerase chain reaction
- RE:
-
Repeat element
- RT-PCR:
-
Real-time polymerase chain reaction
- Toxoplasma gondii :
-
T. gondii
References
Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363:1965–76.
Weiss LM, Kim K. Toxoplasma gondii, the model Apicomplexan: perspectives and methods. Great Britain: Elsevier Ltd; 2007.
Smit GSA, Vu TLB, Do TD, Speybroeck N, Devleesschauwer B, Padalko E, et al. Prenatal diagnosis and prevention of toxoplasmosis in pregnant women in northern Vietnam: study protocol. BMC Infect Dis. 2017;17:364.
Robert-Gangneux F, Dardé ML. Epidemiology of and diagnostic strategies for toxoplasmosis. Clin Microbiol Rev. 2012;25:264–96.
Dogan N, Akda İ, Essizoglu A, Güleç G. Bipolar afektif bozukluk ve şizofreni hastalarinda Toxoplasma gondii varlığının ve ilişkisinin serolojik ve moleküler yöntemlerle araştirilmasi. Flora. 2018;23(3):142–9.
Gashout A, Amro A, Erhuma M, Al-Dwibe H, Elmaihub E, Babba H, Nattah N, Abudher A. Molecular diagnosis of toxoplasma gondii infection in Libya. BMC Infect Dis. 2016;16:157.
Thangarajah P, Hajissa K, Wong WK, Abdullah MA, Ismail N, Mohamed Z. Usefulness of paired samples for the Serodiagnosis of toxoplasmosis infection in a tertiary teaching Hospital in Malaysia. BMC Infect Dis. 2019;19:202.
Derouin F, Pelloux H. ESCMID study group on clinical parasitology. Prevention of toxoplasmosis in transplant patients. Clin Microbiol Infect. 2008;14:1089–101.
Martino R, Maertens J, Bretagne S, Rovira M, Deconinck E, Ullmann AJ, et al. Toxoplasmosis after hematopoietic stem cell transplantation. Clin Infect Dis. 2000;31:1188–95.
Fricker-Hidalgo H, Bulabois CE, Brenier–Pinchart MP, Hamidfar R, Garban F, Brion JP, et al. Diagnosis of toxoplasmosis after allogeneic stem cell transplantation: results of DNA detection and serological techniques. Clin Infect Dis. 2009;48:e9–15.
Dardé ML. Toxoplasma gondii, “new” genotypes and virulence. Parasite. 2008;15:366–71.
Ajzenberg D, Yera H, Marty P, Paris L, Dalle F, Menotti J, et al. Genotype of 88 Toxoplasma gondii isolates associated with toxoplasmosis in immunocompromised patients and correlation with clinical findings. J Infect Dis. 2009;199:1155–67.
Delhaes L, Ajzenberg D, Sicot B, Bourgeot P, Dardé ML, Dei-Cas E, et al. Severe congenital toxoplasmosis due to a Toxoplasma gondii strain with an atypical genotype: case report and review. Prenat Diagn. 2010;30:902–5.
Can H, Döşkaya M, Ajzenberg D, Özdemir HG, Caner A, İz SG, et al. Genetic characterization of Toxoplasma gondii isolates and toxoplasmosis seroprevalence in stray cats of İzmir, Turkey. PLoS One. 2014;9:e104930.
Karakavuk M, Aldemir D, Mercier A, Atalay Şahar E, Can H, Murat JB, et al. Prevalence of toxoplasmosis and genetic characterization of Toxoplasma gondii strains isolated in wild birds of prey and their relation with previously isolated strains from Turkey. PLoS One. 2018;13:e0196159.
Homan WL, Vercammen M, De Braekeleer J, Verschueren H. Identification of a 200- to 300-fold repetitive 529 bp DNA fragment in Toxoplasma gondii, and its use for diagnostic and quantitative PCR. Int J Parasitol. 2000;30:69–75.
Costa JM, Bretagne S. Variation of B1 gene and AF146527 repeat element copy numbers according to Toxoplasma gondii strains assessed using real-time quantitative PCR. Clin Microbiol. 2012;50:1452–4.
Burg JL, Perelman D, Kasper LH, Ware PL, Boothroyd JC. Molecular analysis of the gene encoding the major surface antigen of Toxoplasma gondii. J Immunol. 1988;141:3584–91.
Burg JL, Grover CM, Pouletty P, Boothroyd JC. Direct and sensitive detection of a pathogenic protozoan, Toxoplasma gondii, by polymerase chain reaction. J Clin Microbiol. 1989;27:1787–92.
Cassaing S, Bessières MH, Berry A, Berrebi A, Fabre R, Magnaval JF. Comparison between two amplification sets for molecular diagnosis of toxoplasmosis by real-time PCR. J Clin Microbiol. 2006;44:720–4.
Kasper DC, Sadeghi K, Prusa AR, Reischer GH, Kratochwill K, Förster-Waldl E, et al. Quantitative real-time polymerase chain reaction for the accurate detection of Toxoplasma gondii in amniotic fluid. Diagn Microbiol Infect Dis. 2009;63:10–5.
Menotti J, Garin YJ, Thulliez P, Sérugue MC, Stanislawiak J, Ribaud P, et al. Evaluation of a new 5′-nuclease real-time PCR assay targeting the Toxoplasma gondii AF146527 genomic repeat. Clin Microbiol Infect. 2010;16:363–8.
Filisetti D, Gorcii M, Pernot-Marino E, Villard O, Candolfi E. Diagnosis of congenital toxoplasmosis: comparison of targets for detection of Toxoplasma gondii by PCR. J Clin Microbiol. 2003;41:4826–8.
Reischl U, Bretagne S, Krüger D, Ernault P, Costa JM. Comparison of two DNA targets for the diagnosis of toxoplasmosis by real-time PCR using fluorescence resonance energy transfer hybridization probes. BMC Infect Dis. 2003;3:7.
Edvinsson B, Lappalainen M, Evengård B. ESCMID study Group for Toxoplasmosis. Real-time PCR targeting a 529-bp repeat element for diagnosis of toxoplasmosis. Clin Microbiol Infect. 2006;12:131–6.
Sterkers Y, Varlet-Marie E, Cassaing S, Brenier-Pinchart MP, Brun S, Dalle F, et al. Multicentric comparative analytical performance study for molecular detection of low amounts of Toxoplasma gondii from simulated specimens. J Clin Microbiol. 2010;48:3216–22.
Belaz S, Gangneux JP, Dupretz P, Guiguen C, Robert-Gangneux F. A 10-year retrospective comparison of two target sequences, REP-529 and B1, for Toxoplasma gondii detection by quantitative PCR. J Clin Microbiol. 2015;53:1294–300.
Filisetti D, Sterkers Y, Brenier-Pinchart MP, Cassaing S, Dalle F, Delhaes L, et al. Multicentric comparative assessment of the bio-evolution Toxoplasma gondii detection kit with eight laboratory-developed PCR assays for molecular diagnosis of congenital toxoplasmosis. J Clin Microbiol. 2015;53:29–34.
Wahab T, Edvinsson B, Palm D, Lindh J. Comparison of the AF146527 and B1 repeated elements, two real-time PCR targets used for detection of Toxoplasma gondii. J Clin Microbiol. 2010;48:591–2.
Roux G, Varlet-Marie E, Bastien P, Sterkers Y. French National Reference Center for toxoplasmosis network. Evolution of Toxoplasma-PCR methods and practices: a French national survey and proposal for technical guidelines. Int J Parasitol. 2018;48:701–7.
Robert-Gangneux F, Brenier-Pinchart MP, Yera H, Belaz S, Varlet-Marie E. Bastien P; molecular biology study Group of the French National Reference Center for toxoplasmosis. Evaluation of Toxoplasma ELITe MGB real-time PCR assay for diagnosis of toxoplasmosis. J Clin Microbiol. 2017;55:1369–76.
Brenier-Pinchart MP, Capderou E, Bertini RL, Bailly S, Fricker-Hidalgo H, Varlet-Marie E, et al. Molecular diagnosis of toxoplasmosis: value of the buffy coat for the detection of circulating Toxoplasma gondii. Diagn Microbiol Infect Dis. 2015;82:289–91.
Ajzenberg D, Lamaury I, Demar M, Vautrin C, Cabié A, Simon S, et al. Performance testing of PCR assay in blood samples for the diagnosis of Toxoplasmic encephalitis in AIDS patients from the French Departments of America and genetic diversity of Toxoplasma gondii: a prospective and multicentric study. PLoS Negl Trop Dis. 2016;10:e0004790.
Morelle C, Varlet-Marie E, Brenier-Pinchart MP, Cassaing S, Pelloux H, Bastien P, et al. Comparative assessment of a commercial kit and two laboratory-developed PCR assays for molecular diagnosis of congenital toxoplasmosis. J Clin Microbiol. 2012;50:3977–82.
Döskaya M, Caner A, Degirmenci A, Wengenack NL, Yolasigmaz A, Turgay N, et al. Degree and frequency of inhibition in a routine real-time PCR detecting Pneumocystis jirovecii for the diagnosis of Pneumocystis pneumonia in Turkey. J Med Microbiol. 2011;60:937–44.
Karakavuk M, Aykur M, Şahar EA, Karakuş M, Aldemir D, Döndüren Ö, et al. First time identification of Acanthamoeba genotypes in the cornea samples of wild birds; is Acanthamoeba keratitis making the predatory birds a target? Exp Parasitol. 2017;183:137–42.
Remington JS, Thulliez P, Montoya JG. Recent developments for diagnosis of toxoplasmosis. J Clin Microbiol. 2004;42:941–5.
Döşkaya M, Caner A, Ajzenberg D, Değirmenci A, Dardé ML, Can H, et al. Isolation of Toxoplasma gondii strains similar to Africa 1 genotype in Turkey. Parasitol Int. 2013;62:471–4.
List of rare diseases and synonyms - Orphanet. https://www.orpha.net/orphacom/cahiers/docs/GB/List_of_rare_diseases_in_alphabetical_order.pdf. Accessed 23 Oct 2019.
Acknowledgements
The authors would like to acknowledge Toxoplasma Biological Resource Center (BRC)/Centre National de Référence (CNR) Toxoplasmose for their technical help in development of the in house Real Time PCR method.
Funding
This study was partly supported by the grant given by the Scientific Research Projects Branch Directorate of Ege University, Turkey (Grant No: 2013-TIP-050) to Y.G. The funding body does not have any role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
MD, EAŞ, MK, HC, ADD, AYG conceived and designed the study. HP, MST, MIT, MY worked in acquisition of the clinical samples. MD, HP, MK, EAŞ, MST, MY, ADD, HC helped in interpretation of data. MD, AYG performed the statistical analyses. MD, ADD, MK, EAŞ, MIT, HC interpreted the results and worked in drafting the manuscript. MD and ADD participated in revising the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All samples were collected with patient written consent as approved by the Research Ethics Committee of the Ege University Faculty of Medicine (Approval number: 13–3/3; 18–5.1/29).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Döşkaya, M., Pullukçu, H., Karakavuk, M. et al. Comparison of an in house and a commercial real-time polymerase chain reaction targeting Toxoplasma gondii RE gene using various samples collected from patients in Turkey. BMC Infect Dis 19, 1042 (2019). https://doi.org/10.1186/s12879-019-4666-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12879-019-4666-z