Abstract
Background
Genetic variability and liability to develop drug-resistant mutations are the main characteristics of HIV-1, which can not only increase the risk of antiretroviral treatment (ART) failure, but also can lead to the spread of resistant strains. We aim to investigate the distribution of HIV-1 genotypes and prevalence of pretreatment drug resistance (PDR) in ART-naïve HIV-1 infected patients in Shanghai China.
Methods
A cross-sectional study was performed among the newly diagnosed ART-naive HIV-1 infected patients during the period from January 2017 to November 2017 in Shanghai Public Health Clinical Center. The target fragment of 1316 bp in the pol gene spanning the reverse transcriptase and protease regions was amplified using a nested polymerase chain reaction. HIV-1 genotypes were determined by phylogenetic analysis, and PDR associated mutations were determined according to Stanford University HIV Drug Resistance Database (http://hivdb.stanford.edu/).
Results
We successfully amplified pol gene sequences from blood samples of 317 patients, of whom 95.3% were male, and 68.8% were men who have sex with men. The median age was 33 years; and the median CD4 count was 275 cells/μL. The predominant HIV-1 genotype was circulating recombinant form (CRF) 01_AE (53.0%, 168/317), followed by CRF07_BC (29.7%, 94/317), B (7.6%, 24/317), CRF08_BC (1.9%, 6/317), CRF55_01B (1.9%, 6/317), CRF 59_01B (0.9%, 3/317). In addition, 5% (16/317) HIV-1 strains were identified as other subtypes or CRFs/URFs (unique recombinant forms). The overall prevalence of PDR was 17.4% (55/317). PDR frequency to non-nucleoside reverse transcriptase inhibitor (NNRTI, 16.4%) was much higher than that to nucleoside reverse transcriptase inhibitor (NRTI, 4.7%) and protease inhibitor (PI, 0.6%). The most common HIV-1 mutation pattern for NNRTI and NRTI were V179D/E (10.1%, 32/317) and M184 V (2.8%, 9/317), respectively. About half (49.1%, 27/55) of the HIV-1 strains with mutation presented as potential low-level resistant to NNRTI attributed to V179D/E.
Conclusion
The distribution of HIV-1 genotypes in Shanghai China is diverse and complex. The high prevalence of PDR highlights the significance of baseline HIV-1 drug resistance testing. Non-NNRTI-containing regimen may be the preferred initial therapy for newly diagnosed HIV-1 patients in Shanghai in the absence of PDR test results.
Similar content being viewed by others
Background
HIV/AIDS remains a major public health problem in China. According to the official report, since the first HIV-infected case found in 1985, the number of people living with HIV and deaths due to HIV/AIDS had reached to 83,1225 and 255,995 by the end of July 2018, respectively. Furthermore, the number of new HIV infections continue to rise with a total of 134, 512 reported in 2017 [1, 2].
The main characteristics of HIV are its enormous genetic variability and liability to develop drug-resistant mutations along with high rates of virus replication. Based on the phylogenetic analysis data, HIV can be divided into two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is further classified into four distinct groups: M (major), O(outlier), N (non-M, non-O), and P (pending) [3]. Group M viruses are responsible for the AIDS pandemic globally, and nine subtypes, designated by the letters A-D, F-H, J and K, are recognized within this group [4]. Recombinants between subtypes have been designated as circulating recombinant forms (CRFs) if fully sequenced and found in three or more epidemiologically unlinked individuals, and unique recombinant forms (URFs) if not meeting these criteria [4]. A total of 96 CRFs have been identified so far [5]. HIV-1 genotypes are associated with transmission routes, and vary in epidemic size and distribution features [6]. The distribution of HIV-1 genotypes in several regions of China had been described [7,8,9]. However, little data is known about that in Shanghai, which is the economic, financial, trade and shipping center of mainland China. Moreover, with the development of social economy and the rapid growth of migrants, the distribution of HIV-1 genotypes may have become more diverse and complex due to HIV-1 population movements and the ongoing recombination between different HIV-1 subtypes.
In addition to genetic diversity, another consequence of high variability of HIV is its liability to develop resistance mutation to antiretroviral (ARV) drugs, including “transmitted drug resistance” (TDR), “acquired drug resistance”, and “pretreatment drug resistance (PDR)”, which can not only increase the risk of antiretroviral treatment (ART) failure, but also can lead to the spread of resistant strains, thus posing a major challenge for controlling the HIV epidemic [10]. PDR is detected in ART-naive people initiating ART or people with prior ARV drug exposure initiating or reinitiating first-line ART. However, PDR testing is not routinely performed in China as a developing country with limited conditions. So, we conducted this study to investigate the distribution of HIV-1 genotypes and prevalence of PDR in newly diagnosed HIV-1 infected patients in Shanghai China.
Methods
Study design and participants
A cross-sectional study was performed in Shanghai Public Health Clinical Center (SPHCC), which is the only designated hospital providing the ART and long-term follow-up for HIV/AIDS patients in Shanghai China. A total of 338 patients were enrolled in the study according to the following inclusion criteria: 1) visited SPHCC during the period from Jan 1st, 2017 to Nov 30th, 2017, and had been diagnosed with HIV/AIDS within three months before the visit; 2) had no evidence of receiving ART before the visit; 3) held the Shanghai household registration or residence permit; 4) signed the informed consent for PDR testing, and the plasma sample was successfully collected. Basic epidemiological data such as sex, age, marital status, and self-reported transmission route were recorded upon enrollment. CD4+T cell counts were detected by the flow cytometry. Ethical approval was obtained from the Ethics Committee of SPHCC.
RNA extraction, nested-PCR, and sequencing of viral DNA
The plasma samples were collected and preserved in a − 80 °C freezer until analysis. Viral RNA was extracted from 140 μl plasma using QIAmp Viral RNA Mini Kit (Qiagen, Germany) according to the manufacturer protocol. Then, the target fragment of 1316 bp in the pol gene spanning the reverse transcriptase and protease regions was amplified using a nested polymerase chain reaction (PCR). PrimeScript™ one-step RT-PCR ver. 2.0 (TakaRa, China) was used for the cDNA synthesis and first-round PCR operation. The nested PCR was performed with Ex Taq (TaKaRa, China). The PCR products were sent to BioSune Biotechnology Co. for sequencing (Applied Biosystems, 3730XL). The PCR protocol and primers used were as described previously [8]. The primers for PCR and sequencing are listed in Table 1.
Identification of HIV-1 genotypes and drug-resistance mutations (DRMs)
HIV-1 genotypes were identified by phylogenetic analysis. HIV-1 pol sequences, together with reference sequences of different subtypes and CRFs, were aligned and further edited manually using the BIOEDIT version 7 (http://www.mbio.ncsu.edu/BioEdit/bioedit.html). All the subtyping reference sequences were downloaded from the Los Alamos HIV database (https://www.hiv.lanl.gov/content/sequence/HIV/mainpage.html). The phylogenetic trees were generated using the Neighbor-joining method. DRMs and resistance levels were determined based on Stanford University HIV Drug Resistance Database (HIVDB): HIVdb Program (https://hivdb.stanford.edu/hivdb/by-sequences/). The degree of drug resistance to each ARV was divided into five levels: susceptible, potential low-level resistance, low-level resistance, intermediate resistance, and high-level resistance, according to the HIVDB Genotypic Resistance Test (GRT) Interpretation System (Updated October 2018, https://hivdb.stanford.edu/assets/media/genotypic-resistance-test-interpretation-system-oct2018.57d61710.pdf).
Statistical analysis
Data were analyzed using SPSS 22.0 (IBM Corp., Armonk, NY). Normality of data was assessed by the Kolmogorov–Smirnov test. Quantitative data with normal distribution were expressed as means ± standard deviation (x ± SD) and compared using t tests; quantitative data with skewed distribution were expressed as median (inter-quartile range, IQR) and compared using the Mann-Whitney U test. Categorical variables were expressed as frequencies and percentages and compared using the chi-square (x2) test or Fisher exact test. The logistic regression analysis was used to identify risk factors associated with PDR. All tests were two tailed, and p values < 0.05 were considered significant.
Nucleotide sequence accession numbers
For similar scientific and ethical reasons as explained in Esbjörnsson et al. [11], only a proportion of the 317 sequences is accessible via GenBank (accession numbers, MK573428 - MK573507).
Results
Demographic and clinical characteristics of the participants
A total of 338 HIV-1 infected patients were enrolled in the study, and pol gene sequences were successfully amplified and analyzed from the plasma samples of 317 participants, including 302 (95.3%) male and 15 (4.7%) female. The median age was 33 years (IQR: 29–44 years), ranging from 20 to 79 years. Most of the participants (68.5%) were single, 28.1% were married, and 3.5% were divorced/widowed. The self-reported risk factors for HIV-1 infection were mainly homosexual contact (68.8%) and heterosexual contact (14.8%); two (0.6%) cases via intravenous drug use, and one case (0.3%) via blood transfusion; however, 49 (15.5%) patients did not know how they got infected. The median CD4 count was 275 cells/μL (IQR: 168–368 cells/μL), ranging from 1 to 1814 cells/μL. A total of 98 patients (30.9%) had a CD4 count < 200 cells/μL, 124 patients (39.1%) had a CD4 count between 200 and 350 cells/μL, and 95 patients (30.0%) had a CD4 count ≥350 cells/μL. There were no significant differences in age, sex, marital status, self-reported risk factor for HIV-1 infection, and CD4 counts among the groups divided by HIV-1 genotypes (Table 2).
Distribution of HIV-1 genotypes
Among the 317 successfully amplified samples, CRF01_AE (53.0%, 168/317) was the predominant genotype, followed by CRF07_BC (29.7%, 94/317), B (7.6%, 24/317), CRF08_BC (1.9%, 6/317), CRF55_01B (1.9%, 6/317), CRF59_01B (0.9%, 3/317), G (0.6%, 2/317). In addition, 4 strains were identified as other CRFs, including CRF02_AG (0.3%, 1/317), CRF67_01B (0.3%, 1/317), CRF68_01B (0.3%, 1/317), CRF65_cpx (0.3%, 1/317). However, another 10 (3.2%) HIV-1 strains did not cluster with any present known reference sequences, and were determined as URFs. Further analysis using REGA HIV-1 Subtyping Tool – Version 3.0 (http://dbpartners.stanford.edu:8080/RegaSubtyping/stanford-hiv/typingtool/) and HIV blast (https://www.hiv.lanl.gov/content/sequence/BASIC_BLAST/basic_blast.html), these strains were likely recombinants of CRF01_AE/B (Ho-01B-94,758, Ho-01B-94,223, Ho-01B-94,812), CRF01_AE/B/C (He-01 BC-95961, Ho-01 BC-94675, Ho-01 BC-95927, un-01 BC-94292), CRF01_AE/C (Ho-01C-94,192), and CRF01_AE/CRF07_BC (Ho-0107-94,199, Ho-0107-95,109) (Fig. 1 and Fig. 2).
(a and b) Phylogenetic trees of HIV-1 pol genes were constructed using MEGA 7 based on neighbor-joining methods. a (total tree) shows clusters of all the sequences, whereas b (subtree) indicates mainly clusters of different subtypes. The strain name of the reference sequences are shown in green. The bootstrap values greater than 70% based on 1000 replicates are shown on the major branches. The scale bar indicates 0.01 for the total tree and 0.005 for the subtree. The newly identified strains are named in “transmission route-subtypes-sample code”. Ho represents homosexual; He represents heterosexual; un represents unknown; IDU represents intravenous drug use. In front of strain name, newly identified unique recombinant forms (URFs) are color-coded in red dots
Prevalence and risk factors of PDR associated mutations
The overall prevalence of PDR mutation among the 317 participants was 17.4% (55/317). Twelve nucleoside reverse transcriptase inhibitor (NRTI) resistance associated mutation patterns, 21 non-nucleoside reverse transcriptase inhibitor (NNRTI) mutation patterns, and two protease inhibitor (PI) mutation patterns were identified, respectively. The most frequent NNRTI associated mutation was V179D/E, which was observed in 10.1% (32/317) of patients, followed by Y181C (2.5%, 8/55) and K103 N (1.9%, 6/317). M184 V (2.8%, 9/317) was the most prevalent NRTI associated mutation, followed by K65R (2.2%, 7/317). The majority (72.7%, 40/55) of HIV-1 variants with PDR mutation displayed a single drug class resistance mutation. Fifteen (27.3%, 15/55) strains harbored both NRTI and NNRTI mutations. No HIV-1 strain with PDR mutations to triple classes of drugs was found in this study. Moreover, most (69.1%, 38/55) of the HIV-1 variants with mutation exhibited single mutation; two base mutation were found in 4 (7.3%) samples, and multiple (> = 3) base mutations in 13 (23.6%) samples (Fig. 3).
The multinomial logistic regression analysis for risk factors of PDR showed that the HIV-1 genotype was a potential influencing factor associated with PDR. CRF07_BC strains had a lower risk of PDR (odds ratio, 0.139; 95% CI, 0.030–0.639; p = 0.011) (Table 3).
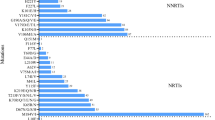
Resistance level to different ARV drugs (Fig. 4)
We analyzed HIV-1 resistance level to a total of 13 commonly used ARV drugs, including lamivudine (3TC), abacavir (ABC), emtricitabine (FTC), tenofovir disoproxil fumarate (TDF), zidovudine (AZT), doravirine (DOR), efavirenz (EFV), nevirapine (NVP), etravirine (ETR), rilpivirine (RPV), atazanavir/r (ATV/r), lopinavir/r (LPV/r) and darunavir/r (DRV/r). The overall prevalence of HIV-1 PDR to NNRTI (16.4%, 52/317) was much higher than that to NRTI (4.7%, 15/317) and PI (0.6%, 2/317) (X2 = 63.10, p < 0.0001). In addition, nearly half of (49.1%, 27/55) HIV-1 strains with PDR mutation exhibited the degree of potential low-level resistance to NNRTI ascribed to V179D/E.
Frequency and levels of HIV-1 pretreatment drug resistance to different antiretroviral drugs among 317 newly diagnosed HIV/AIDS patients in Shanghai. 3TC: lamivudine; ABC: abacavir; FTC: emtricitabine; TDF: tenofovir disoproxil fumarate; AZT: zidovudine; EFV: efavirenz; DOR: doravirine; NVP: nevirapine; ETR: etravirine; RPV: rilpivirine; ATV: atazanavir/r; LPV: lopinavir/r
All the 15 HIV-1 strains with NRTI resistance mutations were simultaneously resistant to 3TC, ABC, and FTC. PDR frequency to AZT (0.3%, 1/317) was significantly lower than that to TDF (2.5%, 8/317) (X2 = 5.523, p = 0.019) and 3TC/ABC/FTC (4.7%, 15/317) (X2 = 12.567, p < 0.0001). There was no significantly difference between the prevalence of PDR to TDF and 3TC/ABC/FTC (X2 = 2.211, p = 0.137).
For NNRTI, DOR showed the lowest PDR frequency (4.4%, 14/317) (x2 = 27.522, p < 0.0001). The PDR frequency to EFV (16.1%, 51/317), NVP (16.1%, 51/317), ETR (15.1%, 48/317) and RPV (14.8%, 47/317) was similar (x2 = 0.307, p = 0.959). The degree of resistance was mainly at a potential low level, accounting for 0.3% (1/317), 9.5% (30/317), 9.5% (30/317), 12.6% (40/317), and 9.1% (29/317) for DOR, EFV, NVP, ETR, and RPV, respectively.
PI resistance were detected in only two samples, being simultaneously resistant to ATV/r and LPV/r at a potential low level caused by M46I/L mutation. There was no HIV-1 strain resistant to DRV/r found in our study.
Discussion
This study showed that CRF01_AE (53.0%) was the predominant HIV-1 genotype in Shanghai, followed by CRF07_BC (29.7%) and B (7.6%). There was a high prevalence (17.4%) of PDR mutation in the newly diagnosed treatment-naive HIV/AIDS patients in Shanghai. The PDR mutation frequency to NNRTIs (16.4%) was much higher than NRTIs (4.7%) and PIs (0.6%). Moreover, about half (49.1%, 27/55) of the virus strains with mutations presented as potential low-level resistant to NNRTI ascribed to V179D/E.
HIV-1 is characterized by its vast genetic variability caused by high-rate but error-prone replication and recombination of the virus. The global distribution of HIV-1genotypes is highly heterogeneous and varies geographically [6]. Furthermore, the HIV-1 epidemic may evolve along with the change in transmission risk behaviours, lifestyle and the patterns of human mobility. Various aspects of HIV infection may be affected by its diversity, including disease progression [12], response to ART, transmission routes, vaccine development, immune response and escape, etc. [6]. It is of great significance to know the epidemiological characteristics of AIDS in a certain area, so as to better control the epidemic. Our study found a total of 11 HIV-1 genotypes and 10 HIV-1 strains of URFs in 317 newly diagnosed HIV/AIDS patients in 2017, indicating that the distribution of HIV-1 genotypes in Shanghai is diverse and complex. The results were similar to that of the study conducted in Jiangsu province (a close neighbor of Shanghai), showing that the prevalence of genotype CRF01_AE, CRF07_BC, and B were 60.06, 22.29, and 5.88%, respectively [13]. Similarly, the surveys carried out in Hebei, Fujian, and regions across China also reported that CRF01_AE, CRF07_BC, and B were the predominant subtypes [9, 14, 15]. However, some other studies displayed different results. For example, a study performed in 205 HIV (+) blood donors from five Chinese blood centers showed that CRF07_BC (61.5%) was the most prevalent, followed by CRF01_AE (20%) and CRF08_BC (8.3%) [16]. Besides, a recent investigation on HIV-1 genetic characteristics in Yunnan province revealed that CRF08_BC (47.4%) was the most common, followed by URFs (18.2%), CRF01_AE (15.8%) and CRF07_BC (14.4%) [17]. The reasons for the discrepancy may be attributed to the difference in the transmission routes. According to the national official report, heterosexual contact has become the predominant risk factor for HIV epidemic in China [2]. While, in our study, homosexual contact accounted for 68.8% of HIV transmission factors for Shanghai cases. Chen’s study revealed that CRF07_BC was increasing among men who have sex with men (MSM) in Fujian; our findings also indicated that CRF07_BC was one of the most prevalent genotypes among MSM in Shanghai [9].
Another serious consequence of HIV-1 high variability is drug resistance - an emerging threat to epidemic control, which can cause treatment failure and further spread of drug resistant HIV. According to WHO surveys of PDR between 2014 and 2016, seven Latin American and African countries estimated a prevalence of PDR greater than 10% in adults initiating ART [18]. Our study in Shanghai also showed a high prevalence of PDR among the newly diagnosed HIV-1 infected patients in 2017, which is higher than that in Hebei, Beijing and Yunnan province [8, 19, 20]. ART has been widely used in China since 2006, when the government’s “Four Free and One Care” policy - free antiretroviral drugs to AIDS patients who are rural residents or people without insurance living in urban areas; free voluntary counselling and testing; free drugs to HIV-infected pregnant women to prevent mother-to-child transmission, and HIV testing of newborn babies; free schooling for AIDS orphans; care and economic assistance to the households of people living with HIV/AIDS - for AIDS control was introduced. Because of the limited availability of drugs in China, the regimen composed of TDF, 3TC and EFV is currently the most commonly used free first-line therapy. According to our study, PDR mutation frequency to NNRTIs (16.4%) was much higher than NRTIs (4.7%) and PIs (0.6%) in Shanghai, and the above three kinds of drugs exhibited a PDR mutation frequency of 2.5% (TDF), 4.7% (3TC) and 16.1% (EFV), respectively, which highlights the importance of routinely PDR testing. WHO’s new recommendations on the public health response to PDR indicate that in countries where population-levels of PDR to NNRTI reach the threshold of 10%, a change in the first-line ART regimen (from NNRTI-based to non-NNRTI based, such as integrase inhibitors) should be urgently considered [21]. Therefore, PI/integrase inhibitor-containing regimen may be preferred initial therapy for the newly diagnosed HIV/AIDS patients in Shanghai.
However, it should be noted that for about half (49.1%) of HIV-1 strains with drug resistant mutations, the degree of resistance was classified as potential low-level due to the single V179D/E mutation, which is selected by NNRTI and contribute low-levels reductions in susceptibility to each of the NNRTIs. However, whether this single mutation can lead to clinical treatment failure needs to be further studied and confirmed. The trend of increasing V179D/E mutation in genotype CRF01_AE among MSM population was also reported in other study [22].
In addition, our study showed that HIV-1 genotype was a potential influencing factor of PDR. CRF07_BC strains had a lower risk for PDR. However, another study revealed that genotype C was associated with high risk for resistance [8]. The reason for this difference need further study to demonstrate. However, due to the small sample size further research in a larger population is necessary to confirm that.
We have to point out that there are some limitations of this study. First, sample selection bias may exist, because the newly diagnosed AIDS patients who didn’t sign the consent form for the pretreatment HIV-1 drug resistance testing were not included in the study. The majority of the participants were male and MSM. Second, our analysis about PDR concentrated only on NRTI, NNRTI and PI, not containing integrase inhibitors, which are a newer kind of ARV drugs. It is expected that resistance to integrase inhibitors will invariably emerge with their increasingly widely used in ART-naive and ART-experienced patients. Third, the participants enrolled in the study were newly diagnosed with HIV-1 infection in 2017. However, many patients didn’t know when they had got HIV-1 infection. Some patients might have been infected for a relatively long time. Therefore, the finding may not really reflect the current HIV epidemic in Shanghai.
Conclusions
In summary, our study provides the knowledge of molecular epidemiological characteristics of HIV-1 and the prevalence of PDR among the newly diagnosed AIDS patients in Shanghai in 2017. The distribution of HIV-1 genotypes in Shanghai is diverse and complex, with CRF01_AE and CRF07_BC as the predominant genotypes. The high PDR mutation frequency in ART-naive patients highlights the significance of routine testing of pretreatment HIV-1 drug resistance, and non-NNRTI-containing regimen, such as PI/integrase inhibitor-based regimen may be the preferred initial therapy for this population by reason of high prevalence of PDR to NNRTI.
Abbreviations
- 3TC:
-
Lamivudine
- ABC:
-
Abacavir
- ART:
-
Antiretroviral treatment
- ARV:
-
Antiretroviral
- ATV/r:
-
Atazanavir/r
- AZT:
-
Zidovudine
- CRF:
-
Circulating recombinant form
- DOR:
-
Doravirine
- DRMs:
-
Drug-resistance mutations
- EFV:
-
Efavirenz
- ETR:
-
Etravirine
- FTC:
-
Emtricitabine
- IDU:
-
Intravenous drug use
- IQR:
-
Inter-quartile range
- LPV/r:
-
Lopinavir/r
- NNRTI:
-
Non-nucleoside reverse transcriptase inhibitor
- NRTI:
-
Nucleoside reverse transcriptase inhibitor
- NVP:
-
Nevirapine
- PCR:
-
Polymerase chain reaction
- PDR:
-
Pretreatment drug resistance
- PI:
-
Protease inhibitor
- RPV:
-
Rilpivirine
- SPHCC:
-
Shanghai Public Health Clinical Center
- TDF:
-
Tenofovir disoproxil fumarate
- TDR:
-
Transmitted drug resistance
- URFs:
-
Unique recombinant forms
References
National Center for AIDS/STD Control and Prevention, China CDC. Update on the AIDS/STD epidemic in China in December 2017. Chin J AIDS STD. 2018;24:111.
National Center for AIDS/STD Control and Prevention, China CDC. Update on the AIDS/STD epidemic in China in July, 2018. Chin J AIDS STD. 2018;24:865.
Plantier JC, Leoz M, Dickerson JE, De Oliveira F, Cordonnier F, Lemée V, et al. A new human immunodeficiency virus derived from gorillas. Nat Med. 2009;15:871–2.
Robertson DL, Anderson JP, Bradac JA, Carr JK, Foley B, Funkhouser RK, et al. HIV-1 nomenclature proposal. Science. 2000;288:55–6.
Los Alamos National Laboratory. http://www.hiv.lanl.gov/content/sequence/HIV/CRFs/CRFs.html. Accessed 14 Oct 2018.
Hemelaar J. Implications of HIV diversity for the HIV-1 pandemic. J Inf Secur. 2013;66:391–400.
Chen M, Ma Y, Chen H, Dai J, Dong L, Yang C, et al. HIV-1 genetic transmission networks among men who have sex with men in Kunming, China. PLoS One. 2018;13:e0196548.
Song YX, Xin RL, Li ZC, Yu HW, Lun WH, Ye J, et al. Prevalence of transmitted drug resistance among HIV-1 treatment-naive patients in Beijing. Epidemiol Infect. 2018;146:339–44.
Chen ZW, Liu L, Chen G, Cheung KW, Du Y, Yao X, et al. Surging HIV-1 CRF07_BC epidemic among recently infected men who have sex with men in Fujian, China. J Med Virol. 2018;90:1210–21.
Global action plan on HIV drug resistance 2017–2021. Geneva: World Health Organization; 2017. Licence: CC BY-NC-SA 3.0 IGO.
Esbjörnsson J, Mild M, Audelin A, Fonager J, Skar H, Bruun Jørgensen L, et al. HIV-1 transmission between MSM and heterosexuals, and increasing proportions of circulating recombinant forms in the Nordic countries. Virus Evol. 2016;2(1):vew010.
Li Y, Han Y, Xie J, Gu L, Li W, Wang H, et al. CRF01_AE subtype is associated with X4 tropism and fast HIV progression in Chinese patients infected through sexual transmission. AIDS. 2014;28:521–30.
Yang Y, Zhao XP, Zou HC, Chu MJ, Zhong P, Li XS, et al. Phylogenetic and temporal danamics of human immunodeficiency virus type 1 CRF01_AE and CRF07_BC among recently infected antiretroviral therapy-naive men who have sex with men in Jiangsu province, China, 2012 to 2015: a molecular epidemiology-based study. Medicine (Baltimore). 2018;97:e9826.
Lu X, Kang X, Liu Y, Cui Z, Guo W, Zhao C, et al. HIV-1 molecular epidemiology among newly diagnosed HIV-1 individuals in Hebei, a low HIV prevalence province in China. PLoS One. 2017;12:e0171481.
He X, Xing H, Ruan Y, Hong K, Cheng C, Hu Y, et al. A comprehensive mapping of HIV-1 genotypes in various risk groups and regions across China based on a nationwide molecular epidemiologic survey. PLoS One. 2012;7:e47289.
Zeng P, Liu Y, He M, Wang J, Keating S, Mao W, et al. The infection staging and profile of genotypic distribution and drug resistance mutation among the human immunodeficiency virus-1 infected blood donors from five Chinese blood centers, 2012-2014. PLoS One. 2017;12:e0179328.
Chen M, Jia MH, Ma YL, Luo HB, Chen HC, Yang CJ, et al. The changing HIV-1 genetic characteristics and transmitted drug resistance among recently infected population in Yunnan, China. Epidemiol Infect. 2018;146:775–81.
HIV drug resistance report 2017. Geneva: World Health Organization; 2017. Licence: CC BY-NC-SA 3.0 IGO.
Xuan Q, Liang S, Qin W, Yang S, Zhang AM, Zhao T, et al. High prevalence of HIV-1 transmitted drug resistance among therapy-naïve Burmese entering travelers at Dehong ports in Yunnan, China. BMC Infect Dis. 2018;18:211–9.
Lu X, Zhao H, Zhang Y, Wang W, Zhao C, Li Y, et al. HIV-1 drug-resistant mutations and related risk factors among HIV-1-positive individuals experiencing treatment failure in Hebei Province, China. AIDS Res Ther. 2017;14:4.
Guidelines on the public health response to pretreatment HIV drug resistance. Geneva: World Health Organization; 2017. http://www.who.int/hiv/topics/drugresistance/en. Accessed 7 July 2017.
Li X, Xue Y, Lin Y, Gai J, Zhang L, Cheng H, et al. Evolutionary dynamics and complicated genetic transmission network patterns of HIV-1 CRF01_ AE among MSM in Shanghai, China. Sci Rep. 2016;6:34729.
Acknowledgements
We wish to thank all the patients who participated in the study. We are grateful for Tangkai Qi, Jun Chen, Wei Song, Yang Tang and Jianjun Sun for coordination.
Funding
This study was supported by the China’s Thirteenth Five-Year major national science and technology projects: AIDS and hepatitis, and other major infectious disease control and prevention (No.2017ZX10202101), Outstanding Youth Grant from Shanghai Municipal Commission of Health and Family Planning of China (No.20144Y0074), and Shanghai Municipal Key Clinical Specialty. The finding body had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Availability of data and materials
The datasets analyzed during the current study are available from the corresponding author on reasonable request. A proportion of sequences is accessible via GenBank (accession numbers, MK573428 - MK573507).
Author information
Authors and Affiliations
Contributions
WZY, ZM and LHZ conceived of the study, participated in the design and coordination. ZM performed RNA extraction and nested-PCR, provided the data on HIV-1 sequences. WZY, LL, SYZ, ZRF, WJR participated in the data collection. WZY analyzed the data and drafted the manuscript. LHZ reviewed this manuscript. All authors read and approved the final draft of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study protocols were approved by the Research Ethics Committee of the Shanghai Public Health Clinical Center and all subjects provided written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing interests associated with this study.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Wang, Z., Zhang, M., Zhang, R. et al. Diversity of HIV-1 genotypes and high prevalence of pretreatment drug resistance in newly diagnosed HIV-infected patients in Shanghai, China. BMC Infect Dis 19, 313 (2019). https://doi.org/10.1186/s12879-019-3927-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12879-019-3927-1