Abstract
Purpose
Hepatocellular carcinoma (HCC) has a poor prognosis, and alpha-fetoprotein (AFP) is widely used to evaluate HCC. However, the proportion of AFP-negative individuals cannot be disregarded. This study aimed to establish a nomogram of risk factors affecting the prognosis of patients with AFP-negative HCC and to evaluate its diagnostic efficiency.
Patients and methods
Data from patients with AFP-negative initial diagnosis of HCC (ANHC) between 2004 and 2015 were collected from the Surveillance, Epidemiology, and End Results database for model establishment and validation. We randomly divided overall cohort into the training or validation cohort (7:3). Univariate and multivariate Cox regression analysis were used to identify the risk factors. We constructed nomograms with overall survival (OS) and cancer-specific survival (CSS) as clinical endpoint events and constructed survival analysis by using Kaplan-Meier curve. Also, we conducted internal validation with Receiver Operating Characteristic (ROC) analysis and Decision curve analysis (DCA) to validate the clinical value of the model.
Results
This study included 1811 patients (1409 men; 64.7% were Caucasian; the average age was 64 years; 60.7% were married). In the multivariate analysis, the independent risk factors affecting prognosis were age, ethnicity, year of diagnosis, tumor size, tumor grade, surgery, chemotherapy, and radiotherapy. The nomogram-based model related C-indexes were 0.762 (95% confidence interval (CI): 0.752–0.772) and 0.752 (95% CI: 0.740–0.769) for predicting OS, and 0.785 (95% CI: 0.774–0.795) and 0.779 (95% CI: 0.762–0.795) for predicting CSS. The nomogram model showed that the predicted death was consistent with the actual value. The ROC analysis and DCA showed that the nomogram had good clinical value compared with TNM staging.
Conclusion
The age(HR:1.012, 95% CI: 1.006–1.018, P-value < 0.001), ethnicity(African-American: HR:0.946, 95% CI: 0.783–1.212, P-value: 0.66; Others: HR:0.737, 95% CI: 0.613–0.887, P-value: 0.001), tumor diameter(HR:1.006, 95% CI: 1.004–1.008, P-value < 0.001), year of diagnosis (HR:0.852, 95% CI: 0.729–0.997, P-value: 0.046), tumor grade(Grade 2: HR:1.124, 95% CI: 0.953–1.326, P-value: 0.164; Grade 3: HR:1.984, 95% CI: 1.574–2.501, P-value < 0.001; Grade 4: HR:2.119, 95% CI: 1.115–4.027, P-value: 0.022), surgery(Liver Resection: HR:0.193, 95% CI: 0.160–0.234, P-value < 0.001; Liver Transplant: HR:0.102, 95% CI: 0.072–0.145, P-value < 0.001), chemotherapy(HR:0.561, 95% CI: 0.471–0.668, P-value < 0.001), and radiotherapy(HR:0.641, 95% CI: 0.463–0.887, P-value:0.007) were independent prognostic factors for patients with ANHC. We developed a nomogram model for predicting the OS and CSS of patients with ANHC, with a good predictive performance.
Similar content being viewed by others
Introduction
Hepatocellular carcinoma (HCC) is a major end-stage liver disease with the sixth highest incidence rate and third highest cause of cancer-related mortality [1, 2]. According to global cancer statistics for 2020, there were approximately 906,000 new cases and 830,000 HCC deaths. Early and accurate diagnosis and treatment are crucial for mitigating these threats.
Alpha-fetoprotein (AFP) is currently the most widely used biomarker for the diagnosis, monitoring, evaluation of prognosis, and treatment of HCC [2,3,4,5,6]. The elevation of tumor markers, mainly AFP, is independently associated with poor prognostic features, such as shortened survival [7,8,9,10,11]. However, in the HCC patient population, AFP-negative hepatocellular carcinoma (ANHC), i.e., AFP < 20 ng/mL at initial diagnosis, is an important type that causes many patients to lose early diagnosis and treatment and accounts for approximately 30-40% of patients [12]. Therefore, the survival and prognosis of ANHC patients are still a matter of concern. Unfortunately, there are relatively few prognostic studies based on the ANHC cohort [9, 13, 14].
We analyzed the impact of the patient’s general condition, tumor indications, and treatment methods on survival, identified relevant risk factors, and constructed a nomogram to guide prognostic evaluation and early diagnosis of ANHC.
Materials and methods
Research design and patients
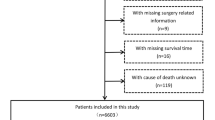
This study collected the clinical data of patients with ANHC from the Surveillance, Epidemiology, and End Results (SEER) database. The inclusion criteria for this study were patients diagnosed between 2004 and 2015 with a primary tumor in the liver, and the third edition of the International Classification of Tumor Diseases was 8170/3: hepatocellular carcinoma, NOS. The confirmed cases included in this study required HCC to be confirmed and diagnosed through pathological examination and were initially diagnosed as AFP-negative (< 20 ng/mL). The exclusion criteria were AFP positivity (> 20 ng/mL) or unknown status at initial diagnosis, unknown tumor size, unknown marital status at diagnosis, unknown surgical treatment, and lack of complete survival time.
Data collection and statistical variable definition
The following clinical information was collected for further analysis: baseline demographic data, including age, ethnicity, gender, year of diagnosis, marital status, survival time, cancer-specific survival rate, and survival status; tumor characteristics, including tumor size (maximum diameter, in cm), pathological tumor grading; and TNM stage according to the American Joint Committee on Cancer 6th TNM stage; and treatment strategies, including surgery (liver resection and liver transplantation), chemotherapy, and radiotherapy at the primary site.
Gender was categorized as male or female. The year of diagnosis was categorized according to before and after 2009. Ethnicity was categorized into three racial groups: Caucasians, African Americans, and others. Marital status at the time of diagnosis was divided into married and unmarried/divorced/widowed groups. Surgery types were divided into no surgery, liver resection, and liver transplantation. For radiotherapy and chemotherapy, patients were classified as having, not having, or unknown.
Statistical analysis
We randomly divided all eligible patients with ANHC into two groups at 7:3: training cohort (n = 1267) and validation cohort (n = 544). Univariate COX analysis was conducted in the training cohort with overall survival (OS) as the endpoint, and statistically significant individuals (p < 0.1) were selected for the multivariate COX analysis. Furthermore, a nomogram was constructed using OS and cancer-specific survival (CSS) as endpoints (P < 0.05). The two nomogram groups were validated using a validation cohort. A nomogram model for independent prognostic factors was established based on multivariate Cox regression analysis (p < 0.05), and the fit was evaluated using a consistency index (C-index).
In the training and validation cohorts, calibration curves for the first, third, and fifth years were established based on the nomogram model by comparing the predicted and actual observed values of the nomogram in the OS and CSS groups. Calibration curves of the validation cohort were established in the same way. The Receiver Operating Characteristic (ROC) table and Decision Curve Analysis (DCA) evaluation model was fitted, under which diagnostic efficacy was compared with that of TNM staging. Clinical information extraction was performed using SEER*Stat software version 8.3.8 (www.seer.cancer.gov/seerstat). Data analysis was carried out using R software version 4.2.2(R-Project, https://www.r-project.org/). A P-value < 0.05 was considered statistically significant.
Results
Patient characteristics
Based on the selection criteria, 1811 patients from the SEER database diagnosed with ANHC (1409 men; average age, 64 years; age range 13–96 years) were selected for inclusion in this study. 1054 (58.6%) patients were diagnosed after 2009. The most common ethnicity was Caucasian, accounting for 64.7% of the study population. In total, 1099 patients (60.7%) were married. In terms of tumor characteristics, the median tumor diameter was 4.5 cm (IQR, 2.6–7.5 cm). Pathological grades I and II were observed in a total of 1584 individuals, representing 87.5% of the total population. TNM stages I and II were identified in 1350 individuals, accounting for 74.6% of the total. In terms of treatment, the majority of patients (1075, 59.4%) received surgical treatment, while liver transplantation was performed in 259 patients (14.3%). Chemotherapy was administered to 560 patients (30.9%) and 114 patients (6.3%) received radiotherapy. 70% of the patients were allocated to the training cohort and 30% to the validation cohort. The baseline characteristics of the total, training, and validation cohorts are shown in Table 1.
Univariate and Multivariate Analysis for OS
In the total cohort, the median OS was 38.0 months (95% CI:33.6–42.4 months), with 1-year, 3-year, and 5-year OS rates of 73.8%, 50.9%, and 39.3%, respectively. We analyzed risk factors affecting the early prognosis of patients with ANHC through univariate and multivariate COX regression analysis. In the analysis of overall survival, univariate COX regression analysis showed that age, marital status, ethnicity, year of diagnosis, tumor pathological grade, tumor diameter, surgery, chemotherapy, and radiotherapy were significant indicators affecting the overall survival of these patients. Further multivariate COX regression analysis was performed on significant individuals with independent predictors, including age(HR:1.012, 95% CI: 1.006–1.018, P-value < 0.001), ethnicity(African-American: HR:0.946, 95% CI: 0.783–1.212, P-value: 0.66; Others: HR:0.737, 95% CI: 0.613–0.887, P-value: 0.001), tumor diameter(HR:1.006, 95% CI: 1.004–1.008, P-value < 0.001), year of diagnosis (HR:0.852, 95% CI: 0.729–0.997, P-value: 0.046), tumor grade(Grade 2: HR:1.124, 95% CI: 0.953–1.326, P-value: 0.164; Grade 3: HR:1.984, 95% CI: 1.574–2.501, P-value < 0.001; Grade 4: HR:2.119, 95% CI: 1.115–4.027, P-value: 0.022), surgery(Liver Resection: HR:0.193, 95% CI: 0.160–0.234, P-value < 0.001; Liver Transplant: HR:0.102, 95% CI: 0.072–0.145, P-value < 0.001), chemotherapy(HR:0.561, 95% CI: 0.471–0.668, P-value < 0.001), and radiotherapy(HR:0.641, 95% CI: 0.463–0.887, P-value:0.007). The hazard ratios and P-values are presented in Table 2.
Nomogram for Predicting OS
A prognostic evaluation of the nomogram for predicting OS in patients with ANHC has been developed and validated based on the independent prognostic variables (age, tumor diameter, ethnicity, pathological grade, surgical, chemotherapy, and radiotherapy conditions) identified in the multivariate analysis (Fig. 1A). Considering that the year of diagnosis has no clinical significance as a predictor variable, we did not include it in the nomogram. In the training and validation cohorts, the nomogram showed satisfactory performance in predicting OS with C-index of 0.762 (95% CI: 0.752–0.772) and 0.752 (95% CI: 0.740–0.769), respectively. In the training and validation cohorts, the calibration curves of 1-year, 3-year and 5-year OS probabilities showed the best consistency between the actual observations and the model predictions based on the nomogram (Supplementary Fig. 1).
By applying the best cutoff value of the nomogram in the training cohort, we developed risk stratification for OS. According to the nomogram-based model score, the whole patients with ANHC were divided into low-risk group (≤ 177.6 score) and high-risk group (> 177.6 score). Kaplan-Meier analysis showed that the median OS of the low-risk and high-risk groups were 65 months (95% CI: 60–70 months) and 8 months (7–10 months), respectively (P < 0.001, Fig. 2A). In the training cohort, the median OS values of the low-risk group and the high-risk group were 45 months (95% CI:43–55 months) and 5 months (4–7 months), respectively (P < 0.001, Fig. 2C). In the validation cohort, the median OS values of the low-risk and high-risk groups were 40 months (95% CI:35–56 months) and 2 months (1–6 months), respectively (P < 0.001, Fig. 2E).
Kaplan–Meier curves for risk classification based on the nomogram scores. (A) In all cohort of OS group; (B) In all cohort of CSS group; (C) In the training cohort of OS group; (D) In the training cohort of CSS group; (E) In the validation cohort of OS group; (F) In the validation cohort of CSS group
Nomogram for predicting CSS
The nomogram for predicting CSS in patients with ANHC has also been developed and validated based on all independent prognostic variables identified in the multivariate analysis (Fig. 1B). The C-index was 0.785 (95% CI: 0.774–0.795) and 0.779 (95% CI: 0.762–0.795) in the training and validation cohorts, respectively. The calibration curves of 1-year, 3-year and 5-year CSS was also been formed (Supplementary Fig. 2).
By applying the same method, all ANHC patients were divided into low-risk group (≤ 145.4 score) and high-risk group (> 145.4 score) under the condition of setting CSS as terminal event. Kaplan-Meier analysis showed that the median CSS of the low-risk and high-risk groups were 104 months (95% CI: 79–128 months) and 8 months (7–10 months), respectively (P < 0.001, Fig. 2B). In the training cohort, the median CSS values of the low-risk group and the high-risk group were 96 months (95% CI: 74–106 months) and 7 months (6–10 months), respectively (P < 0.001, Fig. 2D). In the validation cohort, the median CSS values of the low-risk and high-risk groups were 105 months (95% CI:67–114 months) and 10 months (7–17 months), respectively (P < 0.001, Fig. 2F).
Discussion
Based on SEER database, our study collected the largest cohort of AFP-negative patients available and constructed a new nomogram through data analysis. Compared with traditional TNM staging, it has improved effectiveness in evaluating patient prognosis. The independent risk factors included in our final nomogram were age, tumor diameter, ethnicity, pathological grade, surgical, chemotherapy, and radiotherapy conditions.
Patients with HCC constitute a substantial population characterized by a poor prognosis and a relatively short survival period. The 2022 Barcelona Clinic Liver Cancer guidelines propose that AFP is an important indicator that affects the treatment, prognosis, and clinical decision-making for HCC [15]. The concentration of AFP in adults is about 5–10 ng/mL, and the commonly used optimal screening threshold is 16–20 ng/mL [6, 7, 16, 17]. Nearly half of HCC patients are AFP-negative, most of which are early and small HCC; in advanced patients, 15-30% of serum have AFP values at the normal range (< 20 ng/mL) [18, 19]. Therefore, the ANHC population should be non-negligible.
Usually, OS, with all-cause mortality as the endpoint, is widely used in clinical trials because of its better operability and excellent effectiveness. CSS considers tumor mortality as the endpoint event and has better specificity in evaluating specific diseases. However, sampling and follow-up collection are more difficult in clinical trials, and the SEER database compensates for this difficulty. Previous studies have analyzed HCC based on SEER databases, which extensively include AFP factors. The conclusion revolves around the correlation between AFP positivity and adverse prognosis, tumor progression and metastasis, premature death, and other outcomes [8, 20,21,22]. However, few studies have been conducted in AFP-negative patients [9, 13, 14], and most of which did not involve treatment. Our study collected data from all AFP-negative patients in the SEER database covering various treatment options, including surgery, radiotherapy, and chemotherapy.
The overall conclusions drawn by the two groups were similar but with slight differences. Various sociological factors influence the occurrence and prognosis of HCC [23,24,25]. Our study showed that the CSS group was less affected by social factors and that the age and ethnicity factors included in the OS group were not statistically significant in the CSS group. It is worth mentioning that Chen et al. pointed out the impact of marital status on the prognosis of patients with HCC of TNM stage Ia in that positive marital status is an independent predictor of longer OS and reviewed previous relevant studies to support it [26]. The overlap between the target populations of the two studies was relatively low. Therefore, the impact of marital factors may require a clear understanding of the patients’ scope of application. Tumor burden factors play a prominent role in predicting tumor prognosis. Tumor size and grade are independent prognostic indicators of OS [13, 27, 28], and their impact on survival time is consistent with common sense. The difference between the two groups was relatively small. ANHC is more common in the early stages of the disease and has a better overall prognosis. Currently, the most beneficial treatment methods include surgery and radiofrequency ablation [8, 11, 23, 29]. Particularly for stage 0 disease or patients without surgical contraindications, resection and transplantation are the preferred treatment methods and have a better long-term prognosis than radiofrequency ablation [29,30,31]. Our results draw the same conclusion that the benefits of resection, especially liver transplantation, are significant. For example, a 60-year-old patient with stage a ANHC who undergoes resection can benefit from approximately 0.25 to 0.75 years of overall survival compared to refusing treatment; the five-year cancer-specific survival rate will increase from 0.3 to 0.8 in this scenario. Chemotherapy and radiotherapy have relatively little impact on the prognosis and survival time. The median survival period for symptomatic late-stage cases treated with systemic therapy is 1-1.5 years [23]. Considering that both are mostly used for patients with late-stage disease, the extension of survival time is relatively low. However, it is still an important treatment method for unresectable tumors.
Tumor TNM stage is an independent risk factor for OS in patients with HCC [28]. and it’s widely used in clinical evaluation in the therapy of various kinds of carcinoma. The DCA results indicated that the net benefit of the nomogram in prognostic evaluation was superior to that of TNM staging. And we analyzed the area under the curve (AUC) of ROC, which also indicates that our nomogram has better diagnostic efficiency than TNM staging. With improvements in surgical technologies and the development of new treatment modalities, the prognosis and overall survival of patients with HCC will gradually improve. Over time, the proportion of patients with HCC with lower AFP and earlier BCLC stage has increased, and overall survival has improved [32].
Improvements in overall survival increased the number of potential patients and led to significant improvements in systemic therapy management [32]. The cut-off point for the year of diagnosis in our study was 2009, which was the beginning of the advent of tyrosine kinase inhibitors (TKIs) led by sorafenib [33]. Targeted medicines play an important role in improving patient survival. The application of neoadjuvant or conversion therapy effectively reduced the tumor burden before surgery [34, 35]. It can also be administered after surgery or ablation to reduce the risk of tumor recurrence or progression [36].
Starting in 2015, immunotherapy was introduced at this stage, and patient survival after this time point improved [32]. For example, the IMbrave-150 study established the effectiveness of the classic treatment regimen of atezolizumab combined with bevacizumab, which can be used as the first-line treatment for most advanced HCC [37]. Considering that the application of systemic therapy depends on excellent liver function, there is room for its application in patients with ANHC [37, 38]. An important part of the ANHC patient population has BCLC stage B disease, which can also benefit from systemic therapy [32, 39]. Unfortunately, the data of this study were obtained from the SEER database, and the immunotherapy or targeted therapy status of relevant patients was not collected.
There are still shortcomings in this study. We did not obtain other widely used treatment methods for HCC, such as ablation and interventional surgery, and the prognostic evaluation of these treatment methods may not be comprehensive. The main population in the SEER database comprises Caucasians, and its applicability to other countries or regions may be limited. Third, although T staging is determined based on tumor size, number, and extent of tumor invasion, the SEER database lacks key data on portal vessel invasion status, residual liver function, and underlying liver disease, etc. Therefore, some clinically critical tumor-related variable information, such as Child-Pugh class and MELD score, may be missing in our model. In addition, due to the retrospective nature of the study, we were unable to control the treatment of all patients, and their treatment may be susceptible to differences in residential location and time. More importantly, our prognostic model incorporated treatments, which may limit the utility of our study. In the future, a prospective multi-center clinical study on patients with AFP-negative HCC is still needed to incorporate more clinical parameters and further clarify the impact of these parameters on prognosis.
Conclusion
Our study demonstrated that age, ethnicity, tumor diameter, tumor grade, surgery, chemotherapy, and radiotherapy were independent prognostic factors for patients with ANHC. Surgical treatment has the most significant benefits among the various treatment methods. We designed a reliable nomogram targeting the ANHC population to evaluate prognosis. Our nomogram model shows good predictive performance for both OS and CSS, and can divide ANHC patients into subgroups with completely different prognosis.
Data availability
The data that support the findings of this study are available from SEER database (https://seer.cancer.gov/) and from the corresponding authors upon reasonable request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and Mortality Worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. PubMed PMID: 33538338. Epub 2021/02/05.
Debruyne EN, Delanghe JR. Diagnosing and monitoring hepatocellular carcinoma with alpha-fetoprotein: new aspects and applications. Clin Chim Acta. 2008;395(1–2):19–26. PubMed PMID: 18538135. Epub 2008/06/10.
Qian X, Liu Y, Wu F, Zhang S, Gong J, Nan Y et al. The performance of serum alpha-fetoprotein for detecting early-stage Hepatocellular Carcinoma is influenced by antiviral therapy and serum aspartate aminotransferase: a study in a large cohort of Hepatitis B Virus-infected patients. Viruses. 2022;14(8). PubMed PMID: 36016291. Pubmed Central PMCID: PMC9416230. Epub 2022/08/27.
Marrero JA, Feng Z, Wang Y, Nguyen MH, Befeler AS, Roberts LR, et al. Alpha-fetoprotein, des-gamma carboxyprothrombin, and lectin-bound alpha-fetoprotein in early hepatocellular carcinoma. Gastroenterology. 2009;137(1):110–8. PubMed PMID: 19362088. Pubmed Central PMCID: PMC2704256. Epub 2009/04/14.
Terentiev AA, Moldogazieva NT. Alpha-fetoprotein: a renaissance. Tumour Biol. 2013;34(4):2075–91. PubMed PMID: 23765762. Epub 2013/06/15.
Trevisani F, D’Intino PE, Morselli-Labate AM, Mazzella G, Accogli E, Caraceni P, et al. Serum alpha-fetoprotein for diagnosis of hepatocellular carcinoma in patients with chronic liver disease: influence of HBsAg and anti-HCV status. J Hepatol. 2001;34(4):570–5. PubMed PMID: 11394657. Epub 2001/06/08.
Bruix J, Reig M, Sherman M. Evidence-based diagnosis, staging, and treatment of patients with Hepatocellular Carcinoma. Gastroenterology. 2016;150(4):835–53. PubMed PMID: 26795574. Epub 2016/01/23.
Bai DS, Zhang C, Chen P, Jin SJ, Jiang GQ. The prognostic correlation of AFP level at diagnosis with pathological grade, progression, and survival of patients with hepatocellular carcinoma. Sci Rep. 2017;7(1):12870. PubMed PMID: 28993684. Pubmed Central PMCID: PMC5634482. Epub 2017/10/11.
Lin K, Huang Q, Zeng J, Ding Z, Wang L, Chen Z, et al. Clinical significance of alpha-fetoprotein in alpha-fetoprotein negative Hepatocellular Carcinoma underwent curative resection. Dig Dis Sci. 2021;66(12):4545–56. PubMed PMID: 33723698. Pubmed Central PMCID: PMC8589766. Epub 2021/03/17.
Wang W, Wei C. Advances in the early diagnosis of hepatocellular carcinoma. Genes Dis. 2020;7(3):308–19. PubMed PMID: 32884985. Pubmed Central PMCID: PMC7452544. Epub 2020/09/05.
Hu X, Chen R, Wei Q, Xu X. The Landscape of Alpha Fetoprotein in Hepatocellular Carcinoma: where are we? Int J Biol Sci. 2022;18(2):536–51. PubMed PMID: 35002508. Pubmed Central PMCID: PMC8741863. Epub 2022/01/11.
Zhou JM, Wang T, Zhang KH. AFP-L3 for the diagnosis of early hepatocellular carcinoma: a meta-analysis. Med (Baltim). 2021;100(43):e27673. PubMed PMID: 34713864. Pubmed Central PMCID: PMC8556013. Epub 2021/10/30.
Zhou D, Liu X, Wang X, Yan F, Wang P, Yan H, et al. A prognostic nomogram based on LASSO Cox regression in patients with alpha-fetoprotein-negative hepatocellular carcinoma following non-surgical therapy. BMC Cancer. 2021;21(1):246. PubMed PMID: 33685417. Pubmed Central PMCID: PMC7938545. Epub 2021/03/10.
Li W, Han L, Xiao B, Li X, Ye Z. A predictive nomogram of early recurrence for patients with AFP-Negative Hepatocellular Carcinoma underwent curative resection. Diagnostics (Basel). 2022;12(5). PubMed PMID: 35626229. Pubmed Central PMCID: PMC9140180. Epub 2022/05/29.
Reig M, Forner A, Rimola J, Ferrer-Fabrega J, Burrel M, Garcia-Criado A, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022;76(3):681–93. PubMed PMID: 34801630. Pubmed Central PMCID: PMC8866082. Epub 2021/11/22.
Gupta S, Bent S, Kohlwes J. Test characteristics of alpha-fetoprotein for detecting hepatocellular carcinoma in patients with hepatitis C. A systematic review and critical analysis. Ann Intern Med. 2003;139(1):46–50. PubMed PMID: 12834318. Epub 2003/07/02.
Feng H, Li B, Li Z, Wei Q, Ren L. PIVKA-II serves as a potential biomarker that complements AFP for the diagnosis of hepatocellular carcinoma. BMC Cancer. 2021;21(1):401. PubMed PMID: 33849479. Pubmed Central PMCID: PMC8045263. Epub 2021/04/15.
Wang M, Devarajan K, Singal AG, Marrero JA, Dai J, Feng Z, et al. The Doylestown Algorithm: a test to improve the performance of AFP in the detection of Hepatocellular Carcinoma. Cancer Prev Res (Phila). 2016;9(2):172–9. PubMed PMID: 26712941. Pubmed Central PMCID: PMC4740237. Epub 2015/12/30.
Wang T, Zhang KH. New blood biomarkers for the diagnosis of AFP-Negative Hepatocellular Carcinoma. Front Oncol. 2020;10:1316. PubMed PMID: 32923383. Pubmed Central PMCID: PMC7456927. Epub 2020/09/15.
Zhang H, Du X, Dong H, Xu W, Zhou P, Liu S, et al. Risk factors and predictive nomograms for early death of patients with advanced hepatocellular carcinoma: a large retrospective study based on the SEER database. BMC Gastroenterol. 2022;22(1):348. PubMed PMID: 35854221. Pubmed Central PMCID: PMC9297630. Epub 2022/07/20.
Xie J, Zheng C, Xie J, Wang F, Liu D, Zeng R, et al. No significant relationship exists between tumor size and prognosis in distant metastatic hepatocellular carcinoma: a propensity score matching analysis based on SEER database. BMC Gastroenterol. 2022;22(1):274. PubMed PMID: 35655184. Pubmed Central PMCID: PMC9161599. Epub 2022/06/03.
Liu M, Xu M, Tang T. Association between chemotherapy and prognostic factors of survival in hepatocellular carcinoma: a SEER population-based cohort study. Sci Rep. 2021;11(1):23754. PubMed PMID: 34887446. Pubmed Central PMCID: PMC8660869. Epub 2021/12/11.
Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S, et al. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7(1):6. PubMed PMID: 33479224. Epub 2021/01/23.
Nephew LD, Gupta D, Carter A, Desai AP, Ghabril M, Patidar KR, et al. Social determinants of health impact mortality from HCC and Cholangiocarcinoma: a population-based cohort study. Hepatol Commun. 2023;7(3):e0058. PubMed PMID: 36757397. Epub 2023/02/10.
Lu W, Zheng F, Li Z, Zhou R, Deng L, Xiao W, et al. Association between Environmental and Socioeconomic Risk Factors and Hepatocellular Carcinoma: a Meta-analysis. Front Public Health. 2022;10:741490. PubMed PMID: 35252078. Pubmed Central PMCID: PMC8893961. Epub 2022/03/08.
Chen F, Wu Y, Xu H, Song T, Yan S. Impact of marital status on overall survival in patients with early-stage hepatocellular carcinoma. Sci Rep. 2022;12(1):19923. PubMed PMID: 36402820. Pubmed Central PMCID: PMC9675859. Epub 2022/11/20.
Zhang L, Chen J, Ning D, Liu Q, Wang C, Zhang Z, et al. FBXO22 promotes the development of hepatocellular carcinoma by regulating the ubiquitination and degradation of p21. J Exp Clin Cancer Res. 2019;38(1):101. PubMed PMID: 30808376. Pubmed Central PMCID: PMC6390379. Epub 2019/02/28.
Wang X, Mao M, He Z, Zhang L, Li H, Lin J, et al. Development and validation of a Prognostic Nomogram in AFP-negative hepatocellular carcinoma. Int J Biol Sci. 2019;15(1):221–8. PubMed PMID: 30662361. Pubmed Central PMCID: PMC6329916. Epub 2019/01/22.
Couri T, Pillai A. Goals and targets for personalized therapy for HCC. Hepatol Int. 2019;13(2):125–37. PubMed PMID: 30600478. Epub 2019/01/03.
Mao S, Yu X, Shan Y, Fan R, Wu S, Lu C. Albumin-Bilirubin (ALBI) and Monocyte to lymphocyte ratio (MLR)-Based Nomogram Model to Predict Tumor recurrence of AFP-Negative Hepatocellular Carcinoma. J Hepatocell Carcinoma. 2021;8:1355–65. PubMed PMID: 34805014. Pubmed Central PMCID: PMC8594894. Epub 2021/11/23.
Xie DY, Ren ZG, Zhou J, Fan J, Gao Q. Critical appraisal of Chinese 2017 guideline on the management of hepatocellular carcinoma. Hepatobiliary Surg Nutr. 2017;6(6):387–96. PubMed PMID: 29312973. Pubmed Central PMCID: PMC5756765. Epub 2018/01/10.
Garuti F, Neri A, Avanzato F, Gramenzi A, Rampoldi D, Rucci P, et al. The changing scenario of hepatocellular carcinoma in Italy: an update. Liver Int. 2020;41(3):585–97.
Ielasi L, Tovoli F, Tonnini M, Stefanini B, Tortora R, Magini G et al. Prognostic impact of metastatic site in patients receiving first-line Sorafenib Therapy for Advanced Hepatocellular Carcinoma. Cancers (Basel). 2023;15(5). PubMed PMID: 36900314. Pubmed Central PMCID: PMC10000514. Epub 2023/03/12.
Zhang S, Yuan L, Danilova L, Mo G, Zhu Q, Deshpande A, et al. Spatial transcriptomics analysis of neoadjuvant cabozantinib and nivolumab in advanced hepatocellular carcinoma identifies independent mechanisms of resistance and recurrence. Genome Med. 2023;15(1):72. PubMed PMID: 37723590. Pubmed Central PMCID: PMC10506285. Epub 2023/09/19.
Wu XK, Yang LF, Chen YF, Chen ZW, Lu H, Shen XY, et al. Transcatheter arterial chemoembolisation combined with lenvatinib plus camrelizumab as conversion therapy for unresectable hepatocellular carcinoma: a single-arm, multicentre, prospective study. EClinicalMedicine. 2024;67:102367. PubMed PMID: 38169778. Pubmed Central PMCID: PMC10758712. Epub 2024/01/04.
Wang X, Cao W, Qiu Y, Ji H, Yuan J, Wu W et al. Clinical efficacy and safety evaluation of camrelizumab plus lenvatinib in adjuvant therapy after hepatocellular carcinoma surgery. Front Oncol. 2023;13:1174999. PubMed PMID: 38162488. Pubmed Central PMCID: PMC10755294. Epub 2024/01/02.
Stefanini B, Ielasi L, Chen R, Abbati C, Tonnini M, Tovoli F, et al. TKIs in combination with immunotherapy for hepatocellular carcinoma. Expert Rev Anticancer Ther. 2023;23(3):279–91.
D’Avola D, Granito A, Torre-Aláez Mdl, Piscaglia F. The importance of liver functional reserve in the non-surgical treatment of hepatocellular carcinoma. J Hepatol. 2022;76(5):1185–98.
Ielasi L, Stefanini B, Conti F, Tonnini M, Tortora R, Magini G, et al. Comparative Analysis of Subclassification Systems in patients with Intermediate-Stage Hepatocellular Carcinoma (Barcelona Clinic Liver classification B) receiving systemic therapy. Curr Oncol. 2024;31(1):547–57.
Acknowledgements
Not applicable.
Funding
The present study was funded by the Hebei Provincial Key R&D Program Project (grant no.22377745D), the Hebei Natural Science Foundation Project (grant no.H2022108003), and the Xingtai City Key R&D Program Project (grant no.2020ZC376).
This study was supported by grants from 2023 Hebei Provincial Medical Science Research Project (20230117).
Author information
Authors and Affiliations
Contributions
Conceptualization and Methodology: Chengyu Liu, Li Peng. Data Curation: Chengyu Liu, Jitao Wang. Formal analysis: Chengyu Liu, Jinlong Li, Jitao Wang. Writing - Original Draft: Chengyu Liu, Jitao Wang. Writing - Review & Editing: Zhilei Zhang, Yuming Jia, Chong Zhang, Wuhan Yang, Wenchuan Wang, Kuopeng Liang, Xiaojuan Wang, Congxi Xu, Zikang Li. Visualization: Chengyu Liu, Jinlong Li. Supervision: Jitao Wang, Li Peng. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The SEER database is publicly available and provides de-identified case data. Because of the analysis used anonymous clinical data, written informed consent from subjects was waived.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Liu, C., Li, Z., Zhang, Z. et al. Prediction of survival and analysis of prognostic factors for patients with AFP negative hepatocellular carcinoma: a population-based study. BMC Gastroenterol 24, 93 (2024). https://doi.org/10.1186/s12876-024-03185-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12876-024-03185-z