Abstract
Background
Neutrophil-lymphocyte count ratio (NLCR) has been reported as better indicator of bacteremia than procalcitonin (PCT), and more precise predictor of mortality than C-reactive protein (CRP) under various medical conditions. However, large controversy remains upon this topic. To address the discrepancy, our group has compared the efficiency of NLCR with conventional inflammatory markers in predicting the prognosis of critical illness.
Methods
We performed a multi-center retrospective cohort study involving 536 ICU patients with outcomes of survival, 28- and 7-day mortality. NLCR was compared with conventional inflammatory markers such as PCT, CRP, serum lactate (LAC), white blood cell, neutrophil and severity score APACHE II (Acute Physiology and Chronic Health Evaluation II) to evaluate the potential outcomes of critical illness. Then, receiver operating characteristics (ROC) curves were constructed to assess and compare each marker’s sensitivity and specificity respectively.
Results
NLCR values were not different between survival and mortality groups. Meanwhile, remarkable differences were observed upon APACHE II score, CRP, PCT and LAC levels between survival and death groups. ROC analysis revealed that NLCR was not competent to predict prognosis of critical illness. The AUROCs of conventional markers such as CRP, PCT, LAC and APACHE II score were more effective in predicting 28- and 7-day mortality.
Conclusions
NLCR is less reliable than conventional markers CRP, PCT, LAC and APACHE II score in assessing severity and in predicting outcomes of critical illness.
Similar content being viewed by others
Background
Systemic inflammation is an integral part of pathophysiological processes in critical illness. The NLCR is a conveniently used marker that is readily calculated according to complete blood count [1]. Previously NLCR has been proved as a marker of infection, but did not obtain wide acceptance. Even though NLCR is a conveniently available marker, it did not obtain wide acceptance in clinical [2]. In contrast, cytokines and some acute phase proteins have been frequently used to assess the inflammatory processes in both clinical and research scenarios. C-reactive protein, white blood cell count and neutrophil percentage have long been recognized as valuable markers of inflammation [3]. Thus, these markers play great roles in recognition of inflammatory status, in assessing the severity of diseases and predicting the following outcomes. However, the sensitivity and specificity has yet to be determined.
The APACHE II scores are still widely accepted as the evaluation tools used for determining the criticality of critically ill patients and for evaluating their prognosis [4]. As in critically ill patients, especially in sepsis patients with infection, the number and proportion of neutrophils are elevated, whereas lymphocytes are decreased. Therefore, Zahorec et al. proposes that the neutrophil lymphocyte count ratio (NLCR) is a rapid, easy-applicable, and cost-effective parameter to evaluate the inflammatory and stress status of critically ill patients [1]. NLCR is biomarker based on proportion of neutrophil count in complete blood cell count, which increases in inflammatory disease. Conversely, lymphocytes usually reflect to the patient’s immune status and decrease as inflammatory disease worsens. Until recently, the diagnostic and prognostic values of NLCR were applied to myriad medical situations such as bacteremia [5], sepsis [6], myocardial infarction [7], aneurysmal subarachnoid hemorrhage [8], community acquired pneumonia [9], acute kidney injury [10], liver transplantation [11] and even colorectal cancer. It also provides a more reliable prediction of patient survival rates [12]. Currently, this remains a hot topic of an open discussion [13].
Systemic inflammation is an unavoidable process of critical disease, and its severity generally associated with the short- and long-term outcomes of critical patients [14]. A great many biomarkers such as CRP and PCT have been applied to assess the severity and progress of systemic inflammation in clinical and research scenarios [14] as well as to predict the prognosis of various diseases. Besides, lactate is another common biomarker to evaluate. However, these biomarkers may have limited use due to the lack of sensitivity and specificity as both infection and stress could lead to remarkable changes of these parameters.
Thus, the objective in our current research was to evaluate the association of NLCR with the outcomes of adult critical ill patients, and to determine whether such marker is superior than conventional biomarkers or not.
Methods
Study setting and data source
There are six intensive care units (ICU) wards in our hospital which is a tertiary university hospital (The First Affiliated Hospital of Nanjing Medical University),they are Integrated ICU, Geriatric Medicine ICU, Cardiothoracic ICU, Emergency ICU, general surgery ICU, Neurosurgery ICU. We conducted a retrospective study with data collected from these intensive care units. Each patient admitted to ICU has its own focus on diseases, and there are also some critically ill patients with overlapping diagnoses. All the data we extracted are blood tests immediately after entering ICU (including direct admission and transfer from other departments), exported from the hospitalization system to a spreadsheet and used for follow-up analysis. Patients included in this database were admitted in these ICUs from Jan 2018 to Jun 2019. All the physiological and pathophysiological data, microbiological results and survival outcomes were recorded accordingly. We have received ethical approval (2020-SR-055) from the institutional review boards (IRBs) at the First Affiliated Hospital of Nanjing Medical University. Since this study does not contain protected health information and all data were anonymously used, a waiver of the requirement for informed consent was approved by the IRBs.
Patient records
Inclusion criteria of the patients: (1) Adult patients aged > 18 years; (2) Admitted to ICUs in the First Affiliated Hospital of Nanjing Medical University during the period from Jan 2018 to Jun 2019.
Exclusion criteria of the patients: (1) Hematological disease; (2) Chemotherapy; (3) Receiving glucocorticoids; (4) Receiving bone marrow stimulators.
We retrieved the following clinical information for each patient from the database at ICU admission, age, gender, diagnosis, APACHE II score, body temperature, white blood cell count, neutrophil percentage, blood lactate, PCT, CRP, microbiologic results, coexisting diseases and survival records. NLCR was calculated as the ratio of neutrophil and lymphocyte count, as previously described [1].
Patients were divided into three groups according to the survival records: (1) Survival group; (2) 28-day mortality group; (3) 7-day mortality group. The blood samples of the patients in the study were taken within 30 min after admission to the ICU. Generally, the patients admitted into ICUs were more or less on fluid administration, depending on individual circulatory conditions. The timing for blood taking was usually during the early stages of resuscitation.
Statistical analysis
Statistical analysis and graph construction was performed using GraphPad Prism 5.0 and IBM SPSS Statistics 23. Descriptive analysis was conducted for all variables. One way-Anova was applied to evaluate the differences in NLCR, PCT, CRP, LAC levels and APACHE II scores among different groups. Receiver operating characteristic (ROC) curves were built to assess and compare the sensitivity and specificity of the NLCR, PCT, CRP, LAC and APACHE II score in predicting 28-day and 7-day mortality. The area under the ROC curves (AUROCs) varied from 0.5 to 1.0 were accepted, with higher values indicating increased discriminatory ability. Confidence intervals of AUROCs were calculated with non-parametric assumptions. Each biomarker’s discriminant ability was compared according to its individual AUROC. For all the comparison in this study, P < 0.05 was considered the difference to be statistically significant.
Results
General characteristics
Initially, 536 patients were enrolled in this study. Following the flow chart with strict inclusion and exclusion criteria, data from 428 patients were finally analyzed in this study (Fig. 1), among which 310 were medical patients, and the rest were surgical patients. Overall, ICU mortality rate was 24.5% (105 of 428 patients), with 41.0% (43 of 105) deaths occurring during the first 7 days after admission, and 59% (62 of 105) deaths occurring between 7 and 28 days after admission. Table 1 shows the general characteristics of the total enrolled critical ill patients.
Neutrophil count ratio abnormality did not differentiate between survival and mortality groups. Meanwhile, body temperature did not have discriminant potency, either. However, the WBC count in 7-day mortality group was much higher than other groups. Surgery patient’s population in 7-day mortality group presented to be the lowest among all groups, as shown in Table 1, indicating a surgical background may refer a better outcome in critical patients.
Infection and microorganisms profile
Among total analyzed patients, 280 were detected with infection, including bacteria (total 301 isolates, with 275 gram-negative and 26 gram-positive isolates), fungi (69 isolates), virus (3 isolates), anaerobe (1 isolate) or tuberculosis (1 isolate). On the other hand, 97 cases (survival: 77 cases, 28-day mortality: 14 cases, 7-day mortality: 6 cases) were showed with mixed infection with multiple microorganisms, and other 148 cases (survival: 116 cases, 28-day mortality: 14 cases, 7-day mortality: 18 cases) were not infected. According to infection sites, the patients were categorized with pneumonia (208 cases), bacteremia (19 cases), peritonitis (23 cases), intra-cranial infection (15 cases), and other infections (15 cases). Characteristics of different infection sites were recorded in Table 2. The specific microorganism profile for each group was displayed in supplementary Table 1.
Co-morbid conditions
The incidence of cardiovascular co-morbid conditions on admission to the ICU was higher in patients of both 28- and 7-day mortality groups than in survival group, indicating that cardiovascular disease background could be a major risk factor for negative outcomes. On the other hand, the incidence of malignancies was much lower in mortality groups than that of survival group. Apart from this, the surgery operation incidence in survival group was much higher than that of 7-day mortality group, as described in Table 1. Other co-morbid diseases include diabetes mellitus, hypertension, COPD, liver cirrhosis and renal failure, which did not differentiate between groups. Details were presented in supplementary Table 2.
Diagnostic character of APACHE II and biomarkers
APACHE II, one of several ICU scoring systems, is a severity-of-disease classification system. It is applied within 24 h of admission of a patient to an ICU. Higher APACHE II score negatively correlates with survival rate. In our study, we applied APACHE II to be the comparable reference for the analyzed biomarkers.
NLCR, APACHE II score and other biomarker levels of survival, 28- and 7-day mortality groups were showed in Fig. 2. By studying cohort of 428 critical patients, the APACHE II score at admission in mortality groups were much higher than that of survivors. Both CRP and PCT levels of mortality groups were significantly elevated than those of survival groups (Table 1, Fig. 2). Regarding to LAC, only 28-day mortality group levels were higher than that of survivals, but not 7-day mortality group levels. This is probably because by relatively large deviation and small count of group population. Of note, NLCR did not vary among survivals and non-survival patients (Table 1, Fig. 2).
Discriminant performance of APACHE II and biomarkers
The sensitivity of NLCR, CRP, PCT, LAC and APACHE II score to predict 28- and 7-day mortality was presented in Fig. 2. With group comparison, we found that PCT, CRP and APACHE II score were all discriminant between survival and mortality groups, but not NLCR. Among all biomarkers, both PCT and CRP showed greater differential potency than others with lowest p value at comparison of the survival and mortality groups. On the other hand, LAC showed less differential potency among all groups (Fig. 2).
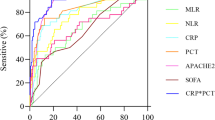
According to area under ROC curves analysis, the AUROCs of NLCR were 0.580 and 0.575 (with P values of 0.045 and 0.111) for 28- and 7-day mortality group, respectively. This suggests that NLCR is not a powerful discriminator to predict mortality. On the other hand, not only APACHE II score, but also CRP and PCT had much higher levels of AUROCs than NLCR (Fig. 3).
Receiver operating characteristic curves and AUROCs of markers for predicting overall 28-day and 7-day mortality. *P < 0.05, ***P < 0.001 compared with APACHE II, &P < 0.05 compared with LAC, #P < 0.05 compared with PCT. The table below showed the area under the ROC curve value with STD, 95% confidence intervals and P values versus survival group
To identify 28-day mortality, APACHE II score presented highest AUROC than all the biomarkers, while PCT and CRP presented to be less potent biomarkers. LAC had relatively higher levels of AUROC, which made it a better indicator to predict 28-day mortality. Moreover, LAC had the lowest value of prediction, while APACHE II score remained the highest value of predicting 7-day mortality. Compared to PCT and CRP, NLCR was a relatively weak biomarker to predict both short and long term mortality according to AUROC levels (data shown in Fig. 3).
In regard to survival analysis, all the biomarkers selected in this study presented good potential to predict outcomes of critical patients. We calculated the cutoff values for each biomarker and graphed the survival curves accordingly. The results showed both PCT and CRP had higher sensitivity and specificity than NLCR and LAC with p values lower than 0.001. Compared to PCT and CRP, NLCR failed to indicate prognosis of critical patients (Fig. 4).
Discussion
In this study, we compared the discriminatory ability of NLCR with CRP, PCT and APACHE II score on prediction of critical illness. The results indicates that PCT, CRP and APACHE II score were all discriminant between survival and mortality group, not NLCR. The ROC analysis also revealed APACHE II score, CRP, PCT and LAC had higher levels of AUROCs than NLCR, which made NLCR a relatively weak predictor of mortality. For prediction of 28-day mortality, APACHE II score and LAC presented higher efficacy than all other markers. Meanwhile, for prediction of 7-day mortality, both CRP and PCT displayed comparable efficacy as well as APACHE II score.
NLCR, as a relatively new biomarker, numerous groups have demonstrated it was able to predict outcomes of various oncology patients and served as prognostic pre-operatively in patients with colorectal cancer [1, 15,16,17,18]. In addition, there are also current studies showing an association between NLCR and the prognosis and mortality in infectious diseases. Previous investigations had already presented that NLCR was a more sensitive parameter in the prediction of appendicitis [19]. Zahorec and colleagues have observed lymphocytopenia in ICU patients following major surgery and sepsis, and noticed higher levels of NLCR related with severity of the clinical courses [1]. Huang and colleagues analyzed previous studies and concluded that the NLCR was associated with the prognosis of sepsis and that a higher NLCR indicate unfavorable prognosis [20]. In this study, we demonstrated that NLCR did not have high discriminant ability to predict outcomes of critical illness. Compared to conventional biomarkers and APACHE II score, AUROCs of NLCR did not show advantages of differentiation but borderline predictive capability of 28-day mortality. On the other hand, not only APACHE II score, but also CRP and PCT showed better potential of prognostic value on mortality outcomes of critical illness.
Regarding to conventional biomarkers, Wyllie and colleagues have determined that CRP alone could not precisely predict bacterial infection than lymphocytopenia alone or a combination of lymphocytopenia and neutrophilia [21]. Although procalcitonin has been evaluated and proved to be prognostic with critical illness, especially of septic inflammation, its implementation has been hampered due to the high costs and lacking of accessibility in developing countries.
Elevated LAC is often considered a marker of circulatory ischemia and hypoxia and is also directly related to the prognosis of sepsis. In our study, LAC levels were less powerful in predicting 7-day mortality than 28-day mortality. These results were in accordance with some of previous conclusions [6, 22], but contradicted with others [5]. This may cause by insufficient population or tumor background, which may not be able to applied to patients with inflammatory background. Our study enrolled 428 cases into final analysis, which presented a relatively large study population and hence improved its reliability.
Meanwhile, most recent investigations demonstrated that NLCR, a simple and easily obtainable marker, had higher predictive value in bacterial infection, and can be integrated into daily practice without extra costs [5]. With these characteristics, NLCR was even suggested to be widely applied to the surveillance protocols of clinical scenarios especially in developing countries. However, based on AUROCs calculation and comparison, NLCR was proved to be less valuable than any of the above-mentioned conventional biomarkers and APACHE II score to predict prognosis or to evaluate the severity of a disease. Moreover, NLCR levels did not distinguish between survival and mortality groups, which suggested it may not capable to be a reliable indicator to evaluate severity or to predict prognosis of critical illness.
Conclusion
NLCR as a non-specific biomarker, was associated with both 7- and 28-day mortality in adult critical patients. However, both CRP and PCT were more sensitive and specific to predict prognosis in critical ill patients. Compared to traditional predictive indicators, NLCR shows no advantages over PCT, CRP, and APACHE II score. Thus, NLCR could not be an ideal substitute to conventional markers in evaluation of severity of critical illness.
Limitations of this study
Indeed, the present study does have several limitations. First, although this study involves a relatively large population, it is a retrospective study, thus the potential of this study is to be further determined by prospective researches in much larger population in other centers. Second, this study generally compared the potential of NLCR and other traditional inflammatory markers such as CRP, PCT, white blood cell count, neutrophil count and APACHE II severity score, but not the more potentially predictive markers. Further studies should be conducted to evaluate such predictive potentials and compare them with those short- and long-term bio-reactive proteins, such as acute phase proteins in this context. Third, this study did not separately demonstrate relationships between NLCR and risk of death in infected patients. At this point, it may contradict with the conclusions of other literatures.
Availability of data and materials
The data-sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- APACHE II:
-
Acute Physiology and Chronic Health Evaluation II
- AUROC:
-
Area under the ROC curve
- CI:
-
Confidence interval
- CRP:
-
C-reactive protein
- ICU:
-
Intensive care unit
- IRB:
-
Institutional review boards
- LAC:
-
Lactate
- NE:
-
Neutrophil
- NLCR:
-
Neutrophil-lymphocyte count ratio
- PCT:
-
Procalcitonin
- ROC:
-
Receiver operating characteristic
- WBC:
-
White blood cell
References
Zahorec R. Ratio of neutrophil to lymphocyte counts--rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy. 2001;102(1):5–14.
Salciccioli JD, Marshall DC, Pimentel MA, Santos MD, Pollard T, Celi LA, et al. The association between the neutrophil-to-lymphocyte ratio and mortality in critical illness: an observational cohort study. Crit Care. 2015;19:13.
Torres A, Artigas A, Ferrer R. Biomarkers in the ICU: less is more? No. Intensive Care Med. 2021;47(1):97–100.
Rhodes A, Evans L, Alhazzani E, Levy W, Antonelli MM, et al. Surviving Sepsis campaign: international guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017;43(3):304–7.
de Jager CP, van Wijk PT, Mathoera RB, de Jongh-Leuvenink J, van der Poll T, Wever PC. Lymphocytopenia and neutrophil-lymphocyte count ratio predict bacteremia better than conventional infection markers in an emergency care unit. Crit Care. 2010;14(5):R192.
Westerdijk K, Simons KS, Zegers M, Wever PC, Pickkers P, de Jager CPC. The value of the neutrophil-lymphocyte count ratio in the diagnosis of sepsis in patients admitted to the intensive care unit: a retrospective cohort study. PLoS One. 2019;14(2):e0212861.
Hong D, Choi KH, Song YB, Lee JM, Park TK, Yang JH, Hahn JY, Choi JH, Choi SH, Kim SM, Choe Y, Kim EK, Chang SA, Lee SC, Oh JK, Gwon HC. Prognostic implications of post-percutaneous coronary intervention neutrophil-to-lymphocyte ratio on infarct size and clinical outcomes in patients with acute myocardial infarction. Sci Rep. 2019;9(1):9646.
Giede-Jeppe A, Reichl J, Sprügel MI, Lücking H, Hoelter P, Eyüpoglu IY, Kuramatsu JB, Huttner HB, Gerner ST. Neutrophil-to-lymphocyte ratio as an independent predictor for unfavorable functional outcome in aneurysmal subarachnoid hemorrhage. J Neurosurg. 2019;132:400–7.
Curbelo J, Rajas O, Arnalich B, Galván-Román JM, Luquero-Bueno S, Ortega-Gómez M, Lancho A, Roy E, Sánchez Azofra A, Mateo Jiménez G, Gómez M, Moldenhauer F, Aspa J. Neutrophil count percentage and neutrophil-lymphocyte ratio as prognostic markers in patients hospitalized for community-acquired pneumonia. Arch Bronconeumol. 2019;55(9):472–7.
Zhou X, Liu J, Ji X, Yang X, Duan M. Predictive value of inflammatory markers for acute kidney injury in sepsis patients: analysis of 753 cases in 7 years. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2018;30(4):346–50.
Kwon HM, Moon YJ, Jung KW, Park YS, Jun IG, Kim SO, Song JG, Hwang GS. Neutrophil-to-lymphocyte ratio is a predictor of early graft dysfunction following living donor liver transplantation. Liver Int. 2019;39(8):1545–56.
Kumarasamy C, Sabarimurugan S, Madurantakam RM, Lakhotiya K, Samiappan S, Baxi S, Nachimuthu R, Gothandam KM, Jayaraj R. Prognostic significance of blood inflammatory biomarkers NLR, PLR, and LMR in cancer-A protocol for systematic review and meta-analysis. Medicine. 2019;98(24):e14834.
Povsic MK, Beovic B, Ihan A. Perioperative increase in neutrophil CD64 expression is an Indicator for intra-abdominal infection after colorectal Cancer surgery. Radiol Oncol. 2016;51(2):211–20.
Cheval C, Timsit JF, Garrouste-Orgeas M, Assicot M, De Jonghe B, Misset B, Bohuon C, Carlet J. Procalcitonin (PCT) is useful in predicting the bacterial origin of an acute circulatory failure in critically ill patients. Intensive Care Med. 2000;26(Suppl 2):S153–8.
Sarraf KM, Belcher E, Raevsky E, Nicholson AG, Goldstraw P, Lim E. Neutrophil / lymphocyte ratio and its association with survival after complete resection in non-small cell lung cancer. J Thorac Cardiovasc Surg. 2009;137(2):425–8.
Cho H, Hur HW, Kim SW, Kim SH, Kim JH, Kim YT, Lee K. Pre-treatment neutrophil to lymphocyte ratio is elevated in epithelial ovarian cancer and predicts survival after treatment. Cancer Immunol Immunother. 2009;58(1):15–23.
Azab B, Bhatt VR, Phookan J, Murukutla S, Kohn N, Terjanian T, Widmann WD. Usefulness of the neutrophil-to-lymphocyte ratio in predicting short- and long-term mortality in breast cancer patients. Ann Surg Oncol. 2012;19(1):217–24.
Walsh SR, Cook EJ, Goulder F, Justin TA, Keeling NJ. Neutrophil-lymphocyte ratio as a prognostic factor in colorectal cancer. J Surg Oncol. 2005;91(3):181–4.
Goodman DA, Goodman CB, Monk JS. Use of the neutrophil:lymphocyte ratio in the diagnosis of appendicitis. Am Surg. 1995;61(3):257–9.
Huang Z, Fu Z, Huang W, Huang K. Prognostic value of neutrophil-to-lymphocyte ratio in sepsis: a meta-analysis. Am J Emerg Med. 2020;38(3):641–7.
Wyllie DH, Bowler IC, Peto TE. Bacteraemia prediction in emergency medical admissions: role of C reactive protein. J Clin Pathol. 2005;58(4):352–6.
Riché F, Gayat E, Barthélémy R, Le Dorze M, Matéo J, Payen D. Reversal of neutrophil-to-lymphocyte count ratio in early versus late death from septic shock. Crit Care. 2015;19:439.
Acknowledgments
The statistical analysis of this study received help from Dr. Qiulun Lu and Dr. Yifeng Wang from Nanjing Medical University, Nanjing, China.
Funding
This study was financed by National Natural Science Foundation of China (NNSFC), No.81770287 and No. 82070475. The corresponding author Dr. Han received the fundings in 2017 and 2020 and initiated a sequential researches regarding to biomarker analysis and multiple organ dysfunction in sepsis.
Author information
Authors and Affiliations
Contributions
TZ, DZ and XL retrieved the data of patients according to the background of critical illness, survival and mortality. YH initiated the study, analyzed and interpreted the data. NZ and YH constructed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
We have received ethical approval (2020-SR-055) from the institutional review boards (IRBs) at the First Affiliated Hospital of Nanjing Medical University. Since this study does not contain protected health information and all data were anonymously used, a waiver of the requirement for informed consent was approved by the IRBs. Individual patients consent was not obtained since all data used in this study were acquired retrospectively from the laboratory information system without any additional sampling or laboratory analysis.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1.
Microorganisms isolated from the patients in the study cohort. Table S2. Coexisting disease of the study population stratified by survival and mortality
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhou, T., Zheng, N., Li, X. et al. Prognostic value of neutrophil- lymphocyte count ratio (NLCR) among adult ICU patients in comparison to APACHE II score and conventional inflammatory markers: a multi center retrospective cohort study. BMC Emerg Med 21, 24 (2021). https://doi.org/10.1186/s12873-021-00418-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12873-021-00418-2