Abstract
Background
Boron (B) nutritional disorders, either deficiency or toxicity, may lead to an increase in reactive oxygen species production, causing damage to cells. Oxidative damage in leaves can be attenuated by supplying silicon (Si). The aim of this study was to assess the effect of increasing foliar B accumulation on cotton plants to determine whether adding Si to the spray solution promotes gains to correct deficiency and toxicity of this micronutrient by decreasing oxidative stress via synthetizing proline and glycine-betaine, thereby raising dry matter production.
Results
B deficiency or toxicity increased H2O2 and MDA leaf concentration in cotton plants. H2O2 and MDA leaf concentration declined, with quadratic adjustment, as a function of increased leaf B accumulation. Proline and glycine-betaine leaf concentration increased under B-deficiency and B-toxicity. In addition, production of these nonenzymatic antioxidant compounds was greater in plants under toxicity, in relation to deficient plants. Adding Si to the B spray solution reduced H2O2 and MDA concentration in the plants under nutrient deficiency or toxicity. Si reduced H2O2, primarily in B-deficient plants. Si also increased proline and glycine-betaine concentration, mainly in plants under B toxicity. Dry matter production of B-deficient cotton plants increased up to an application of 1.2 g L− 1 of B. The critical B level in the spray solution for deficiency and toxicity was observed at a concentration of 0.5 and 1.9 g L− 1 of B, respectively, in the presence of Si, and 0.4 and 1.9 g L− 1 of B without it. In addition, the presence of Si in the B solution raised dry matter production in all B concentrations evaluated in this study.
Conclusion
Our findings demonstrated that adding Si to a B solution is important in the foliar spraying of cotton plants because it increases proline and glycine-betaine production and reduces H2O2 and MDA concentration, in addition to mitigating the oxidative stress in cotton plants under B deficiency or toxicity.
Similar content being viewed by others
Background
Boron (B) foliar spraying is a common practice performed by cotton (Gossypium hirsutum L.) growers in different regions worldwide due to B deficiency in different soils [1,2,3]. However, the difference between the adequate boron concentration and toxicity is small [1, 4, 5] and the toxic effect of this element can be verified in cotton plants when it is applied at high concentrations to leaves [3]. During the reproductive stage, plants have a higher B requirement compared to the vegetative stage [6], as in this stage, during the formation of pollen grains, pectins are used to form the cell wall and, in case of B deficiency, the rigidity of the cell wall is compromised [7]. Boron plays a role in pectin synthesis by forming cis-borate ester complexes that are obligatory compounds in cell wall constituents [6]. We believe that the cells in tissues generated in the formation of pollen grains have cells walls with a higher content of complexed boron. Hence, this logically increases B demand for the formation of cell walls. In addition, B deficiency decreases the transport of photoassimilates from leaves to flowers and fruits, impairing flower, seed and fruit formation [8]. Although B plays a vital role in plant reproduction [9], micronutrient deficiency can also impair the vegetative development of cotton plants [10]. This developmental impairment is due to the boron’s function in the cell wall structure, as well as due to its biological function in the metabolism of nucleic acids, proteins, phenols and in the functionality of the plasma membrane and sugar transport [11,12,13,14], cell elongation, and protein synthesis, resulting in increased cell division [15].
Thus, the adequate supply of boron for the cotton crop increases plant development and dry matter production [10]. However, excess B causes toxicity also decreasing the development of cotton plants [10, 15], which may cause metabolic disturbances in the formation of complexes with NAD+ in the ribosomes, affecting the formation of the RNA structure [14]. In addition, boron-related nutritional disorders (either deficiency or toxicity) may lead to an increase in reactive oxygen species (ROS) production, causing oxidative damage to cells, as observed in beets [16] and field peas [17]. Oxidative stress occurs when there is imbalance between ROS production and the antioxidant defense system (enzymatic and nonenzymatic), causing oxidative damage [18, 19]. However, the effects of boron-related nutritional disorder on oxidative stress in cotton plants remains unknown. Oxidative damage in leaves may be attenuated by supplying silicon (Si), which has been widely reported in cotton plants grown under abiotic stresses [20,21,22]. Si supply in cotton plants grown under stress reduces oxidative damage by decreasing the H2O2 content, lipid peroxidation and electrolyte leakage [22]. In addition, the beneficial element plays an important role in the modulation of antioxidant enzymes by increasing the transcription of genes involved in the defense response of plants, which has already been reported for several species [22,23,24]. The role played by B and Si in the enzymatic antioxidant system in leaves has been extensively explored and discussed [20,21,22]. Despite this, the non-enzymatic antioxidant system must be taken into consideration, and studies with this focus on cotton plants grown under B deficiency are scarce. In a review on B toxicity, Hua et al. [25] indicated that the non-enzymatic antioxidant system is important to regulate ROS neutralization in plant cells. Among non-enzymatic antioxidant compounds, the authors highlighted proline and glycine-betaine (GB). Proline is one of the most common osmolytes present in plants, being related to tolerance to various abiotic stresses [26] and to the neutralization of reactive oxygen species, attenuating the damage caused by lipid peroxidation [27]. Similar to proline, GB has an osmotic and antioxidant function related to its contribution to the maintenance of the structural and functional integrity of cells, which is possible due to the interaction between GB and the hydrophilic and hydrophobic domains of protein and membranous complexes in cells [28].
This raises an important question regarding the possibility of increasing the efficiency of B foliar sprayings in order to mitigate the harmful effects of deficiency or toxicity of this nutrients by including Si in the spray solution, although these elements must be chemically compatible. Therefore, the risk of Si polymerization in different nutrient solutions should also be taken into consideration, once this is the main disadvantage of using Si in the solution. The polymerization process of Si lead to the formation of insoluble silicon dioxide (SiO2), which compromises the benefit of this element to the plants. Silanol groups are formed during the polymerization and condensation of monosilicic acid. Then, the supersaturated acid solution is converted into its polymer form, forming insoluble spherical colloidal particles [29] that form long-chain networks, inducing visible changes in the solution from translucid to opaque or murky [30]. Si polymerization in aqueous solution is affected by the pH and Si concentration of the solution [30, 31], as well by the presence of stabilizer [32]. Stabilizers such as sorbitol help to stabilize Si monomers in solution, reducing the polymerization rate of this beneficial element and favoring its absorption [32], enabling Si addition to borate solutions for foliar application [30].
In this scenario, it is important to test the hypothesis that the a) deficiency and toxicity of B increases oxidative stress, with a consequent decrease in cotton plant development; and that b) Si addition to B solutions for foliar application, without polymerization, reduces the oxidative stress in cotton plants by increasing the production of proline and glycine-betaine. In this context, a study was conducted to evaluate the effect of increasing foliar B accumulation in the leaves of cotton plants to determine whether adding Si to the spray solution promotes gains to correct the deficiency and toxicity of this micronutrient by decreasing oxidative stress through the synthetization of proline and glycine-betaine, thereby increasing dry matter production.
Results
Control x factorial
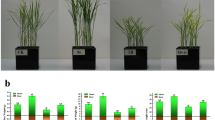
Cotton plants grown without B deficiency (control) exhibited greater B accumulation, higher shoot dry matter production and lower B use efficiency when compared to those from the factorial treatment. In addition, Si accumulation, oxidative stress (measured by H2O2 and MDA production), and the induction of the proline nonenzymatic antioxidant system and GB systems were the lowest in control plants (Table 1).
Turbidity test of the spray solution and B and Si accumulation in cotton plants leaves
Adding 1.00 g L− 1 of Si in the form of SiKE to the B spray solution (2.50 g L− 1) did not increase the turbidity index of the solution, with an average value of 0.80 NDU (Fig. 1a). Adding 1.25 g L− 1 of Si to the B solution (2.50 g L− 1) caused an exponential rise in the turbidity index, reaching 1.73 NDU, 360 minutes after preparation (Fig. 1a). Increasing the Si concentration to 1.50 g L− 1 resulted in the immediate polymerization of the solution, precluding readings for turbidity index. Therefore, the ideal Si concentration in the spray solution was between 1.00 and 1.25 g L− 1, in order to avoid polymerization.
To improve Si concentration accuracy in the solution, a second assessment was carried out, indicating that the maximum amount of Si (SiKE) to be added to the B solution (2.50 g L− 1 of B) is 1.00 g L− 1 of Si, without changing the turbidity index, with an average value of 0.80 NDU (Fig. 1b). The increase in Si concentration from 1.05 to 1.25 g L− 1 raised the turbidity index exponentially, with a maximum of 1.51; 1.55; 1.58; 1.69 and 1.73 NDU after 360 minutes, for following Si concentrations 1.05; 1.10; 1.15; 1.20 and 1.25 g L− 1, respectively.
With respect to the solution color, the solution mixture with Si (1.00 g L− 1) and B (2.50 g L− 1) showed visible changes during the period assessed (Fig. 2). Adding Si (1.25 g L− 1) resulted in the formation of polymers immediately after solution preparation (Fig. 2a), evolving to a murky whitish color and gelatinous appearance after 360 minutes (Fig. 2b). Adding Si (1.50 g L− 1) to the B solution (2.50 g L− 1 of B) also resulted in polymerization, immediately forming a gel after solution preparation (Fig. 2a) and at 360 minutes (Fig. 2b).
Assessment of the visual colorimetric change in the boron (2.50 g L− 1) + silicon mixture (1.00; 1.25 and 1.50 g L− 1) immediately (a) and 360 min after solution preparation (b); and boron (2.50 g L− 1) + silicon mixture (1.00; 1.05; 1.10; 1.15; 1.20 and 1.25 g L− 1) at 0 (c); 60 (d); 120 (e); 180 (f); 300 (g) and 360 (h) min after solution preparation
The second turbidity index assessment, with a higher number of Si concentrations, showed no change in the color of the B spray solution (2.50 g L− 1) with the addition of Si between 1.00 and 1.25 g L− 1, except for polymer formation at a concentration of 1.25 g L− 1, in the first 60 min after solution preparation (Fig. 2c,d). At 120 minutes after B spray solution preparation (Fig. 2e), adding Si (1.25 and 1.20 g L− 1) to the B solution changed the color of the solution, evolving to a whitish color.
Gelatinization started 180 minutes (Fig. 2f) after spray solution preparation using the B solution mixed with Si (1.25 g L− 1); at 300 minutes (Fig. 2g) the B solution exhibited a gelatinous appearance when Si (1.20 g L− 1) was added, with a change in solution color with Si addition at the concentrations of 1.15 and 1.10 g L− 1.
At the end of the visual assessment, after 360 minutes (Fig. 2h) the B solution added with 1.25 and 1.20 g L− 1 of Si exhibited a thick gelatinous appearance. The solutions with 1.15 and 1.10 g L− 1 of Si showed a whitish color, and for those with 1.05 g L− 1 of Si it was possible to see a slight color change, indicating the onset of visible polymerization.
Boron foliar spraying, in the absence or presence of Si (1.00 g L− 1), caused a linear increase in B accumulation in cotton plants shoots, reaching a maximum of 906.8 and 901.0 mg of B per plant, in the presence and absence of Si, respectively (Fig. 3a). In addition to that, at low B concentrations (0.5 and 1.00 g L− 1), adding Si to the B solution increased micronutrient absorption. Silicon accumulation in the leaves of cotton plants also increased with the increasing in B concentration, reaching 39,11 mg of Si per plant at 2,04 g L− 1 of B (Fig. 3b).
Oxidative stress, proline and glycine-betaine
B deficiency (0.0 and 0.5 g L− 1 of B) or toxicity (2.5 g L− 1 of B) increased H2O2 (Fig. 4a) and MDA concentration (Fig. 4b).
H2O2 concentration declined as a function of increased leaf B accumulation, with a minimum point at B concentrations of 1.37 and 1.49 g L− 1 in the presence and absence of Si, respectively, (Fig. 4a); and B concentrations of 1.66 and 1.5 g L− 1 in the presence and absence of Si, respectively, for MDA (Fig. 4b). Boron deficiency stood out in H2O2 (Fig. 4a) and MDA production (Fig. 4b) when compared to its toxicity. In the 0.0 g L− 1 of B treatment, 539.1 and 767.3 μmol g− 1 of H2O2 was produced in the presence and absence of Si, respectively. These values represent an increase of 15 and 38% in H2O2 produced by plants grown under B toxicity (2.5 g L− 1), which obtained 467.3 and 555.4 μmol g− 1, in the presence and absence of Si, respectively (Fig. 5a). In the same treatment (0.0 g L− 1 of B), MDA concentration was 4.2 and 5.1 g g− 1, representing an increase of 50 and 30% compared to the 2.5 g L− 1 of B treatment, which produced 2.8 and 3.9 g g− 1 of MDA in the presence and absence of Si, respectively (Fig. 4b).
Proline (Fig. 4c) and glycine-betaine (Fig. 4d) concentration declined with a quadratic adjustment as a function of the rise in B concentration. The minimum points for proline were observed with the addition of 1.14 and 1.03 g L− 1 of B, and a production of 42.6 and 39.7 μmol g− 1 of proline, in the presence and absence of Si, respectively (Fig. 4c). The minimum points for glycine-betaine occurred with 1.0 and 1.3 g L− 1 of B, and a production of 5.2 and 5.5 g g− 1, in the presence and absence of Si, respectively (Fig. 4d).
In addition, production of these nonenzymatic antioxidant compounds was greater in plants under toxicity, with a proline concentration of 108.5 and 86.89 g g− 1, an increase of 34 and 56% in relation to deficient plants, which produced 81.0 and 55.8 g g− 1 in the presence and absence of Si, respectively (Fig. 4c). Glycine-betaine concentration was also higher in plants under toxicity, primarily in the presence of Si, with a production of 6.96 g g− 1 of glycine-betaine. This represents a 21% increase in relation to nutrient-deficient plants, which produced 5.74 g g− 1 of glycine-betaine (Fig. 4d). In the absence of Si, the increase was 4% in plants under toxicity (6.42 g g− 1) when compared to those with nutrient deficiency (6.18 g g− 1).
Adding Si to the B spray solution reduced H2O2 (Fig. 4a) and MDA concentration (Fig. 4b) in the plants under nutrient deficiency (0.0 and 0.5 g L− 1 of B) and those under toxicity (2.5 g L− 1 of B). Si reduced H2O2, primarily in B-deficient plants, with a production of 539.1 and 402.2 μmol g− 1 for 0.0 and 0.5 g L− 1 of B, respectively (Fig. 4a). These values are 29 and 24% lower compared to H2O2 production in the absence of the element, which was 767.3 and 532.9 μmol g− 1 for 0.0 and 0.5 g L− 1 of B, respectively (Fig. 4b). Si also increased proline (Fig. 4c) and glycine-betaine concentration (Fig. 4d), mainly in plants under B toxicity (2.5 g L− 1 of B).
Dry matter production
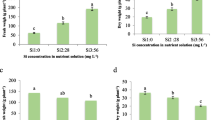
Dry matter production in B-deficient cotton plants increased with B applications of 1.2 and 1.1 g L− 1, with maximum production of 6.1 and 5.4 g in the presence and absence of Si, respectively (Fig. 5a).
The critical B level in the spray solution was established in cotton plants for deficiency and toxicity, with a 10% decline in total dry matter produced at B concentrations of 0.5 and 1.9 g L− 1, respectively, in the presence of Si, and 0.4 and 1.9 g L− 1 of B without it. In addition, the presence of Si in the B solution raised dry matter production in all B concentrations evaluated in this study (Fig. 5a).
The efficiency of shoot B applications in the absence or presence of Si (Fig. 5b) caused a linear reduction with an increase in the B concentration applied. The presence of Si in the B solution raised B use efficiency at concentrations of 1.5 and 2.5 g L− 1.
Discussion
Nutritional disorders are related to several losses in plant metabolism [6], and this effect occurs due to an increase in ROS (Table 1) in cotton plants, as also observed in beet [16] and field peas [17]. This effect is known for its B toxicity, being considered as the cause of oxidative damage to cells [25]. The adequate supply of B attenuates oxidative stress (Fig. 4a,b) by, first stimulating stress defense mechanisms [33]. Micronutrient deficiency, in turn, reduces the antioxidant system by decreasing the metabolic pathways for the biosynthesis of antioxidant compounds [13, 34] or by using the antioxidant system for other metabolic pathways.
A plant defense mechanism against B deficiency was evidenced in cotton grown under deficiency of this nutrient, as there was an increase in proline content (Fig. 4c). Proline has an important function in enhancing plant tolerance to abiotic stresses and reducing oxidative damages, since it increases the redox potential, which is essential to the antioxidant defense mechanism of plants, replenishing the supply of NADP+ [35]. NADP+ is generated during proline synthesis and its oxidation results in the production of NADPH, which acts as a buffering agent of the redox potential inside the cell [36] by binding to ROS and easily neutralizing them [37]. Moreover, plants under stressful conditions exhibit an increase in proline concentration, given the excess production of low molecular weight compounds that are highly soluble as compatible organic solutes.
Another important non-enzymatic antioxidant component is the GB content, which increased both B deficiency and B toxicity in cotton plants (Fig. 4d). This probably occurs as GB has an antioxidant effect, contributing to the maintenance of the structural integrity and to the functioning of cells, which is possible due to its interaction with the hydrophilic and hydrophobic domains of protein and membrane complex [28]. In addition, GB reduces ROS contents, contributing to their homeostasis [38] and inducing gene expression related to stress defense mechanisms through the octadecanoid pathway [39]. Therefore, this increase in GB activates several antioxidant mechanisms in cotton plants grown under stress (B deficiency and toxicity), which was verified by Hamani et al. [40], who observed that exogenous application of GB in this species under stress (saline) decreased oxidative stress.
Given this context, it is pertinent to highlight that the objective of this study was to evaluate the effect of increasing B accumulation in the leaves of cotton plants to determine whether adding Si to the spray solution promotes gains to correct the deficiency and toxicity of this micronutrient by decreasing oxidative stress through the synthetization of proline and glycine-betaine, thereby increasing dry matter production. The addition of Si to the borate solution contributes to reduce oxidative damage in cotton leaves (Fig. 4a, b), increasing both proline (Fig. 4c) and GB (Fig. 4d) under stress conditions caused by B-related nutritional disorders. It is important to note that, for Si to have a beneficial effect, there was no spray polymerization for foliar application, which is important as Souza Júnior et al. [32] indicated that polymerization is a limiting factor in the foliar application of Si. The evaluation of the turbidity index enabled to detect soluble changes in the silicon solution (Fig. 2) before the visible phase (Fig. 1). It was evident that even at a high B concentration (2.5 g L− 1), a maximum of 1.00 g L− 1 Si can be added to the B solution without significant polymerization. The stability of this silicon solution is partially explained by the stabilizing effect of the sorbitol present in the product to ensure successful Si foliar application [32]. This is because adding stabilizers is an efficient alternative to reduce the polymerization of the silicon solution, thereby increasing Si leaf absorption in several crops [17, 40, 41], including cotton [10, 30, 32].
The low polymerization of Si in the B solution (2.50 g L− 1 B) can be confirmed in the present study by the efficiency of B foliar spraying, which was evidenced by the increased B and Si accumulation in the shoots of cotton plants (Fig. 3). Sorbitol has another effect in Si leaf absorption, since it decreases the deliquescence point of the drops deposited onto the leaf surface, thereby lowering the rate of water evaporation (Kubicki and Heaney, 2003), and favoring leaf absorption [6].
Adding Si to the B solution reduced H2O2 (Fig. 4a) and MDA production (Fig. 4b). This beneficial effect of Si is demonstrated by its role in increasing the activity of the nonenzymatic defense system, such as increasing proline (Fig. 4c) and GB (Fig. 4d). Similar results have been reported by other authors, indicating that Si applications activate enzymatic defense mechanisms [22] and, especially their non-enzymatic counterparts [42], given the increased production of proline and GB due to the protective/activating function of Si. However, there are reports that there is a reduction in the production of proline [43,44,45] and GB [45] after the application of Si, thus decreasing cell damage due to the protective effect of Si. It is possible that these differences are caused by genotypic differences and/or by the activation of other metabolic pathways where these compounds are degraded to form new compounds with greater antioxidant power, although this needs to be proven.
In the present study, we showed that Si modulates the nonenzymatic antioxidant system, thereby attenuating nutritional stress and favoring an increase in the dry matter production of cotton plants grown under B deficiency or toxicity (Fig. 5a). Thus, the antioxidant role played by Si could explain its benefits in B-deficient cotton plants, as reported by Souza Júnior et al. [10] and Barros et al. [46], who attributed it to the increased chlorophyll concentration and photosynthetic efficiency.
It is known that the toxicity of a chemical element promoted by foliar application occurs relatively fast and that there are few strategies to reverse leaf damage [43]. Therefore, our findings showed that leaf damage can be mitigated by adding Si to the B solution during spraying, demonstrating that Si has a high potential in reducing oxidative stress. This likely favored the plant metabolism, thereby increasing its capacity to convert the micronutrient into biomass, since Si increased the B use efficiency at the highest concentrations applied to the leaves (Fig. 5b).
The present study revealed the relationship between B and Si in cotton plants by proposing foliar spraying in B-deficient areas at the critical concentration of 0.5 g L− 1 of B with the addition of 1.0 g L− 1 Si, and by demonstrating that a B concentration above 1.9 g L− 1 may be toxic to cotton plants. This information regarding the interaction of these elements for better efficiency in foliar spraying in cotton plants has global implications, given that several growing regions of this crop have reported B deficiency.
Conclusions
In conclusion, adding Si to a B solution for foliar spraying in cotton plants increases proline and glycine-betaine production and reduces H2O2 and MDA concentration, mitigating the oxidative stress in cotton plants under B deficiency or toxicity.
Methods
Growing conditions
The experiment was carried out between january and may (2019) in a greenhouse at the São Paulo State University (UNESP), Jaboticabal, Brazil. Seeds of cotton (Gossypium hirsutum L.) plants, cultivar 954 GLT, were used, commercially acquired from BASF, as it is a cultivar that presents consistent results in different regions and is highly adaptable to cotton producing regions in Brazil. Plants were grown in a soilless cultivation system. Relative humidity (78.4 ± 4.4%), maximum (35.1 ± 6.3 °C) and minimum temperature (22.0 ± 3.2 °C) were recorded with a thermometer hydrometer in the greenhouse during the entire experimental period (Fig. 6).
Cotton seeds were sown into trays containing sand previously washed with water and HCl solution (0.1 M). After germination, four seedlings were transplanted to plastic pots with a 7 dm3 capacity (upper diameter: 16 cm; lower diameter: 11 cm; height: 33 cm). Pots were filled with 6 dm3 of sand previously washed, as above. Thinning was performed with the plants in the vegetative growth stage F2 (two fully developed leaves), maintaining one plant per pot, which was considered as the experimental unit.
A complete nutrient solution was applied to the plants in the vegetative stage F4 (four fully developed leaves) [44]. The nutrient solution was prepared with Fe-EDDHMA as the iron source; pH between 5.5 and 6.5, adjusted with NaOH (1 M) or HCl (1 M) solution, and with a reduction only in B concentration (from 46.2 to 33.7 μmol L− 1), in order to cause moderate B deficiency in cotton plants grown in a soilless system [10].
The nutrient solution was applied for 1 days at 20% of its total concentration, as indicated by Hoagland and Arnon [44]. After this period, the concentration was increased to 40% for 1 week and then to 60%, which was maintained until the end of the experimental.
Pots were drained once a week to eliminate excess of salts, with 700 mL of deionized water being added to the substrate (sand) of each pot to drain the nutrient solution, which was then discarded. After 2 hours, a new nutrient solution was provided to the plants. This was performed for the entire duration of the experiment (rest of the growth cycle), with the nutrients required for plant development being regularly supplied.
Experimental design and treatments
The experiment was carried out under a completely randomized block design (5 × 2 + 1) with four repetitions. The following treatments were applied: five foliar B concentrations (0.0; 0.5; 1.0; 1.5 and 2.5 g L− 1), absence and presence of Si (1.00 g L− 1); and one control treatment with no micronutrient deficiency, adding 46.2 μmol L− 1 of B to the nutrient solution during the entire experimental period.
Boron concentrations in the spray solution were 0%; 33%; 66%; 100 and 166% of the concentration recommended by Görmüs [45], where the author indicates B foliar spraying of 1.5 g L− 1 for cotton plants grown in a micronutrient-deficient environment.
The Si concentration to be mixed with the B solution and to be applied itself was defined based on the visual evaluation of the final solution to avoid using it with evidence of polymerization, which was measured by using the turbidity index.
Turbidity index of the B + Si spray solution
To prepare the solution, boric acid (B: 175 g kg− 1; density: 1.43 g cm− 3; solubility in water at 20 °C: 47.2 g L− 1) was used as the B source, with the pH of the B spray solution adjusted to 9.0 using an NaOH solution (1 M). The Si source consisted of potassium silicate stabilized with sorbitol (SiKE; Si: 115 g L− 1; K2O: 113.85 g L− 1; sorbitol: 100 mL L− 1; pH:12.0). A potassium solution was also prepared with 47 mg L− 1 of K, in the form of KCl, to balance the macronutrient between treatments, since the Si source used in this study contained potassium.
Two turbidity index assessments of the B + Si spray solution were performed to determine the homogeneity of the Si solution, since an increase in its value would most likely increase the chance of Si polymerization. For this test, the B concentration of the solution was fixed at 2.5 g L− 1, the highest concentration used in this experiment. Assessment I consisted of adding Si to the B solution at concentrations of 1.00; 1.25 and 1.50 g L− 1 of Si; assessment II used a B mixture (2.5 g L− 1) with smaller intervals between Si concentrations (1.00; 1.05; 1.10; 1.15; 1.20 and 1.25 g L− 1) in order to increase the accuracy of the concentration in the spray solution. In both assessments, SiKE was used as Si source and H3BO4 as B source in the solution with a final volume of 50 mL, and pH adjusted to 9.05 ± 0.02 using a solution of HCl (1 M) or NaOH (1 M) at a temperature of 20 °C, with three repetitions. After the spray solution was prepared, the turbidity index was measured with a Tecnopon® microprocessed turbidity meter (model TB1000) at 0; 30; 60; 90; 120; 180; 240; 300 and 360 minutes, and photographs were taken using a camera with 9238 × 6928-pixel resolution.
All beakers with the different solutions were placed in front of a completely black background and were then photographed. The images were adjusted using Adobe Photoshop® in order to improve the contrast with the black background without changing B + Si solution color.
Foliar application in the treatments
The solution was prepared according to the different treatments and immediately applied to the plants with a manual sprayer until run-off, in order to cover the entire plant shoot (all leaves). A 5.0 mL volume of the solution was applied to each pot. Sprayings occurred between 7 and 8 am, starting with cotton plants in the reproductive stage B1 (first completely developed flower bud), with four foliar applications 4 days apart. Temperature and relative humidity were measured by a thermometer hydrometer during applications, with temperature ranging between 20 and 23 °C, and relative humidity above 85%, conditions favorable for foliar spraying [6].
During foliar applications, all pots were covered with cotton to avoid any dropping or spillage from the plant shoot after being sprayed with the B + Si solution to the substrate (sand), in order to guarantee that B and Si elements absorption was totally foliar.
Non-enzymatic antioxidant system (proline and glycine-betaine)
Leaves were collected 3 weeks after the B + Si mixture foliar application, being immediately frozen in liquid nitrogen and then placed in a freezer at − 80 °C to further assessment of the oxidative stress.
The rest of the shoot (branches and stem) was also collected, washed with water, detergent solution (0.1%), HCl solution (0.1%) and deionized water, being then placed in a forced air circulation oven at 65 ± 5 °C until constant mass. Further, branches and stems samples were weighed, grinded in a Wiley mill and stored for subsequent assessments.
In order to assess the oxidative stress, malondialdehyde (MDA) and hydrogen peroxide (H2O2) concentrations were determined on the leaves.
Initially, 0.4 g of the frozen plant material was weighed and grinded with 20% (m/v) polyvinylpyrrolidone and 0.1% trichloroacetic acid (TCA). Samples were centrifuged at 11,000 rotations per minute at 4 °C for 10 minutes. The supernatant was separated into Eppendorf tubes containing a 20% TCA solution and 5% thiobarbituric (TBA) acid. All samples were then incubated in a water bath for 30 minutes at 95 °C, and transferred to an ice bath for 10 minutes to stop the reaction, being centrifuged again at 11,000 rotations per minute at 4 °C for 10 minutes. Next, the samples were read in a spectrophotometer at wavelengths between 535 and 600 nm and MDA calculated using an extinction coefficient of 1.55 10− 5 mol− 1 cm− 1 [42].
H2O2 concentration was determined by homogenizing the frozen grinded plant tissue with 0.1% TBA, followed by centrifugation at 11,000 rotations per minute at 4 °C for 10 minutes. The supernatant was also transferred to Eppendorf tubes containing a buffer solution (pH 7.5) and potassium iode and then incubated for 1 h in an ice bath. Spectrophotometric readings were conducted at 390 nm wavelength, in line with the methodology described by Alexieva et al. [47].
Proline concentration was determined using the method proposed by Bates et al. [48]. Plant material was defrosted at ambient temperature and 0.5 g of fresh matter was macerated in liquid nitrogen and subsequently 2 mL of sulfosalicylic acid was added, followed by adding more 8 mL of the same reagent. The grinded material was doubly filtered in a glass funnel using filter paper. After filtering, 1 mL of glacial acetic acid, 1 mL of ninhydrin acid and 1 mL of plant extract were pipetted into a glass test tube. The tubes were then agitated and placed in a water bath at 100 °C for 1 hour and then into an ice bath to stop the reaction. Toluene was added (2 mL), followed by a 20-second agitation. Spectrophotometric readings were performed at 520 nm (adding 0.5 mL of the supernatant in a quartz cuvette).
Glycine-betaine concentration was determined according to the methodology proposed by Grieve and Grattan [49]. For that, 1 g of frozen plant material was placed in paper bags and dried in a forced air circulation oven at 80 °C for 4 days, then manually grinded in a crucible. Extracts were prepared by adding 5 mL of deionized water to 0.125 g of the macerated material, which remained under agitation for 24 hours at 25 °C. The extracts were mixed at 1:1 with H2SO4 (2 M), then maintained in an ice bath for 1 hour to stop the reaction. Further, 0.1 mL of KI-I2 was added to the tubes, which were then agitated and kept at 4 °C for 16 hours. The KI-I2 solution was previously prepared by diluting 15.7 g of iodine and 20 g of K in 100 mL of distilled water. The tubes were then centrifuged at 3500 rotations per minute for 15 minutes at 0 °C. The supernatant was discarded, leaving periodate crystals, which were dissolved in 4.5 mL of 1,2-dichloroethane. Two hours and 30 minutes later absorbance was read at a wavelength of 365 nm in a Beckman DU 640 spectrophotometer and the glycine-betaine concentration was calculated.
Nutritional analysis of B and Si accumulation and plant shoot dry matter production
After analysis of the oxidative stress and nonenzymatic antioxidant compounds, leaves were defrosted at ambient temperature and then washed with water, neutral detergent solution (0.1%), HCl solution (0.1%) and deionized water. After decontamination, leaves were placed in a forced air circulation oven at 65 ± 5 °C until constant mass and then weighed. Samples were then manually grinded in a crucible and mixed with the previously collected macerated plant shoot samples (branches and stems).
Shoot dry matter consisted of the leaf dry matter left over from dry matter analysis of branches and stems collected initially. After the plant material was mixed, B concentration was determined by a dry digestion of the samples, burning in a muffle furnace at 400 °C, followed by colorimetric reaction with azomethine-H and colorimetric spectrophotometric reading [50]. Si concentration was determined from an alkaline digestion of the plant material with H2O2 and NaOH in an oven at 90 °C for 4 h [51], followed by colorimetric reaction with ammonium molybdate in acid medium (oxalic acid and hydrochloric acid), being then determined by colorimetric spectrophotometric readings [52].
B and Si accumulation were calculated as the product of B or Si concentration and shoot dry matter. In addition, B use efficiency was calculated as the ratio between the square of plant shoot dry matter values and plant shoot B accumulation, as described by Siddiqi and Glass [53].
Statistical analysis
The data obtained were submitted to analysis of variance (F-test), and when significant, to polynomial regression or exponential growth.
The exponential growth model was used to study polymerization. Singular models with one or two parameters were tested, and the best fit models applied (those with the highest regression coefficient at 5% using the T-test).
The other variables were submitted to polynomial regression, the linear and quadratic mathematical models tested, and those with the best fit were applied. The model selection criterion established was the magnitude of significant polynomial regression coefficients at 5% probability using the T-test. When significant, the maximum and minimum points were obtained by the derivation of equations.
Statistical analyses were conducted with the Sisvar® software [54] and all graphs were created with Sigmaplot®.
Availability of data and materials
The datasets generated and/or analyzed during the current study are available from the corresponding author upon request.
Abbreviations
- B:
-
Boron
- GB:
-
glycine-betaine
- H2O2 :
-
Hydrogen peroxide
- H3BO4 :
-
Boric acid
- K2O:
-
Potassium oxide
- KCl:
-
Potassium chloride
- MDA:
-
Malondialdehyde
- NaOH:
-
Sodium hydroxide
- ROS:
-
Reactive oxygen species
- Si:
-
Silicon
- SiKE:
-
Potassium silicate stabilized with sorbitol
- SiO2 :
-
Silicon dioxide
References
Atique-ur-Rehman QR, Hussain A, Sardar H, Sarwar N, Javeed HMR, et al. Soil applied boron (B) improves growth, yield and fiber quality traits of cotton grown on calcareous saline soil. Plos One. 2020;15:e0231805. https://doi.org/10.1371/journal.pone.0231805.
Khan MA, Wahid A, Ahmad M, Tahir MT, Ahmed M, Ahmad S, et al. World cotton production and consumption: an overview. In: Ahmad S, Hasanuzzaman M, editors. Cotton Production and Uses. Singapore: Springer; 2020. p. 1–7. https://doi.org/10.1007/978-981-15-1472-2_1.
Wahid MA, Saleem M, Irshad S, Khan S, Cheema MA, Saleem MF, et al. Foliar feeding of boron improves the productivity of cotton cultivars with enhanced boll retention percentage. J Plant Nutr. 2020;43:2411–24. https://doi.org/10.1080/01904167.2020.1783300.
Brdar-Jokanović M. Boron toxicity and deficiency in agricultural plants. Int J Mol Sci. 2020;21:1424. https://doi.org/10.3390/ijms21041424.
Wang G, DiTusa SF, Oh D-H, Herrmann AD, Mendoza-Cozatl DG, O’Neill MA, et al. Cross species multi-omics reveals cell wall sequestration and elevated global transcript abundance as mechanisms of boron tolerance in plants. New Phytol. 2021;230:1985–2000. https://doi.org/10.1111/NPH.17295.
de M Prado R. Mineral nutrition of tropical plants. Switzerland: Springer Nature; 2021.
Aouali N, Laporte P, Clément C. Pectin secretion and distribution in the anther during pollen development in Lilium. Planta. 2001;213:71–9. https://doi.org/10.1007/s004250000469.
Oosterhuis DM, Zhao D. Effect of boron deficiency on the growth and carbohydrate metabolism of cotton. In: Plant Nutrition. Netherlands: Springer; 2001. p. 166–7. https://doi.org/10.1007/0-306-47624-x_80.
Leonard A, Holloway B, Guo M, Rupe M, Yu G, Beatty M, et al. Tassel-less1 encodes a boron channel protein required for inflorescence development in maize. Plant Cell Physiol. 2014;55:1044–54. https://doi.org/10.1093/pcp/pcu036.
de Souza Junior JP, de Prado RM, Dos S Sarah MM, Felisberto G. Silicon mitigates boron deficiency and toxicity in cotton cultivated in nutrient solution. J Plant Nutr Soil Sci. 2019;182:805–14. https://doi.org/10.1002/jpln.201800398.
Mengel K, Kirkby EA. Principles of plant nutrition. Berlin: Springer Science and Business Media; 2012.
Goldbach HE, Wimmer MA. Boron in plants and animals: is there a role beyond cell-wall structure? J Plant Nutr Soil Sci. 2007;170:39–48. https://doi.org/10.1002/jpln.200625161.
Brown PH, Bellaloui N, Wimmer MA, Bassil ES, Ruiz J, Hu H, et al. Boron in plant biology. Plant Biol. 2002;4:205–23. https://doi.org/10.1055/s-2002-25740.
Camacho-Cristóbal JJ, Rexach J, González-Fontes A. Boron in plants: deficiency and toxicity. J Integr Plant Biol. 2008;50:1247–55. https://doi.org/10.1111/j.1744-7909.2008.00742.x.
Ahmed N, Abid M, Rashid A, Ali MA, Ammanullah M. Boron requirement of irrigated cotton in a typic Haplocambid for optimum productivity and seed composition. Commun Soil Sci Plant Anal. 2013;44:1293–309. https://doi.org/10.1080/00103624.2012.756508.
Song B, Hao X, Wang X, Yang S, Dong Y, Ding Y, et al. Boron stress inhibits beet (Beta vulgaris L.) growth through influencing endogenous hormones and oxidative stress response. Soil Sci Plant Nutr. 2019;65:346–52. https://doi.org/10.1080/00380768.2019.1617641.
Oliveira KR, Souza Junior JP, Bennett SJ, Checchio MV, de Alves RC, Felisberto G, et al. Exogenous silicon and salicylic acid applications improve tolerance to boron toxicity in field pea cultivars by intensifying antioxidant defence systems. Ecotoxicol Environ Saf. 2020;201:110778. https://doi.org/10.1016/j.ecoenv.2020.110778.
Kim Y-H, Khan AL, Waqas M, Lee I-J. Silicon regulates antioxidant activities of crop plants under abiotic-induced oxidative stress: a review. Front Plant Sci. 2017;8:510.
Fryzova R, Pohanka M, Martinkova P, Cihlarova H, Brtnicky M, Hladky J, et al. Oxidative stress and heavy metals in plants. In: Reviews of Environmental Contamination and Toxicology. New YorkLLC: Springer; 2018. p. 129–56. https://doi.org/10.1007/398_2017_7.
Bharwana SA, Ali S, Farooq MA, Abbas F, Bioremed BJ. Alleviation of lead toxicity by silicon is related to elevated photosynthesis, antioxidant enzymes suppressed lead uptake and oxidative stress in cotton. J Bioremed Biodeg. 2013;4:4. https://doi.org/10.4172/2155-6199.1000187.
Farooq MA, Ali S, Hameed A, Ishaque W, Mahmood K, Iqbal Z. Alleviation of cadmium toxicity by silicon is related to elevated photosynthesis, antioxidant enzymes; suppressed cadmium uptake and oxidative stress in cotton. Ecotoxicol Environ Saf. 2013;96:242–9.
Moldes CA, Lima Filho OF, Manuel Camiña J, Gabriela Kiriachek S, Lia Molas M, Mui TS. Assessment of the effect of silicon on antioxidant enzymes in cotton plants by multivariate analysis. J Agric Food Chem. 2013;61:11243–9. https://doi.org/10.1021/jf4039088.
Liang Y, Sun W. Mechanisms of silicon-mediated alleviation of abiotic stresses in higher plants: a review. Environ Pollut. 2007;147:422–8.
Khandekar S, Leisner S. Soluble silicon modulates expression of Arabidopsis thaliana genes involved in copper stress. J Plant Physiol. 2011;168:699–705.
Hua T, Zhang R, Sun H, Liu C. Alleviation of boron toxicity in plants: mechanisms and approaches. Crit Rev Environ Sci Technol. 2020. https://doi.org/10.1080/10643389.2020.1807451.
Gill SS, Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem. 2010;48:909–30.
Das K, Roychoudhury A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Frontiers in environmental. Science. 2014;2:53. https://doi.org/10.3389/fenvs.2014.00053.
Sun H, Luo M, Zhou X, Zhou Q, Sun Y, Ge W, et al. Exogenous glycine betaine treatment alleviates low temperature-induced pericarp browning of ‘Nanguo’ pears by regulating antioxidant enzymes and proline metabolism. Food Chem. 2020;306:125626.
Zhuravlev LT. The surface chemistry of amorphous silica. Zhuravlev model. Colloids Surfaces A Physicochem Eng Asp. 2000;173:1–38 www.elsevier.nl/locate/colsurfa. Accessed 1 Nov 2020.
Souza Júnior JP, de Mello PR, Campos CNS, Oliveira DF, Cazetta JO, Detoni JA. Silicon foliar spraying in the reproductive stage of cotton plays an equivalent role to boron in increasing yield, and combined boron-silicon application, without polymerization, increases fiber quality. Ind Crop Prod. 2022;182:114888. https://doi.org/10.1016/j.indcrop.2022.114888.
Haynes RJ. What effect does liming have on silicon availability in agricultural soils? Geoderma. 2019;337:375–83.
Souza Junior JP, de Mello PR, Soares MB, da Silva JLF, de Farias Guedes VH, dos Santos Sarah MM, et al. Effect of different foliar silicon sources on cotton plants. J Soil Sci Plant Nutr. 2020:1–9. https://doi.org/10.1007/s42729-020-00345-4.
Aydin M, Tombuloglu G, Sakcali MS, Hakeem KR, Tombuloglu H. Boron alleviates drought stress by enhancing gene expression and antioxidant enzyme activity. J Soil Sci Plant Nutr. 2019;19:545–55. https://doi.org/10.1007/s42729-019-00053-8.
Cakmak I, Römheld V. Boron deficiency-induced impairments of cellular functions in plants. Plant Soil. 1997;193:71–83. https://doi.org/10.1023/A:1004259808322.
Hassine AB, Ghanem ME, Bouzid S, Lutts S. An inland and a coastal population of the Mediterranean xero-halophyte species Atriplex halimus L. differ in their ability to accumulate proline and glycinebetaine in response to salinity and water stress. J Exp Bot. 2008;59:1315–26. https://doi.org/10.1093/jxb/ern040.
Anwar Hossain M, Hoque MA, Burritt DJ, Fujita M. Proline protects plants against abiotic oxidative stress: biochemical and molecular mechanisms. In: Oxidative damage to plants: antioxidant networks and signaling. Amsterdam: Elsevier Inc.; 2014. p. 477–522.
Liang X, Zhang L, Natarajan SK, Becker DF. Proline mechanisms of stress survival. Antioxid Redox Signal. 2013;19:998–1011. https://doi.org/10.1089/ars.2012.5074.
Badran EG, Abogadallah GM, Nada RM, Nemat Alla MM. Role of glycine in improving the ionic and ROS homeostasis during NaCl stress in wheat. Protoplasma. 2015;252:835–44. https://doi.org/10.1007/s00709-014-0720-2.
Karabudak T, Bor M, Özdemir F, Türkan I. Glycine betaine protects tomato (Solanum lycopersicum) plants at low temperature by inducing fatty acid desaturase7 and lipoxygenase gene expression. Mol Biol Rep. 2014;41:1401–10. https://doi.org/10.1007/s11033-013-2984-6.
de Peixoto MM, Flores RA, do Couto CA, HDN P, de Mello Prado R, Souza-Junior JP, et al. Silicon application increases biomass yield in sunflower by improving the photosynthesizing leaf area. Silicon. 2020:1–6. https://doi.org/10.1007/s12633-020-00818-2.
Moraes DHM, Mesquita M, Magalhães Bueno A, Alves Flores R, Elias de Oliveira HF, Raimundo de Lima FS, et al. Combined effects of induced water deficit and foliar application of silicon on the gas exchange of tomatoes for processing. Agronomy. 2020;10:1715. https://doi.org/10.3390/agronomy10111715.
Gratão PL, Monteiro CC, Carvalho RF, Tezotto T, Piotto FA, Peres LEP, et al. Biochemical dissection of diageotropica and never ripe tomato mutants to cd-stressful conditions. Plant Physiol Biochem. 2012;56:79–96.
Noreen S, Fatima Z, Ahmad S, Athar H-R, Ashraf M. Foliar application of micronutrients in mitigating abiotic stress in crop plants. Plant Nutr Abiotic Stress Toler. 2018:95–117. https://doi.org/10.1007/978-981-10-9044-8_3.
Hoagland DR, Arnon DI. The water-culture method for growing plants without soil; 1950.
Görmüs Ö. Interactive effect of nitrogen and boron on cotton yield and fiber quality. Turk J Agric For. 2005;29:51–9.
Barros TC, de Mello PR, Roque CG, Arf MV, Vilela RG. Silicon and salicylic acid in the physiology and yield of cotton. J Plant Nutr. 2019;42:458–65.
Alexieva V, Sergiev I, Mapelli S, Karanov E. The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ. 2001;24:1337–44.
Bates LS, Waldren RP, Teare ID. Rapid determination of free proline for water-stress studies. Plant Soil. 1973;39:205–7.
Grieve CM, Grattan SR. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil. 1983;70:303–7. https://doi.org/10.1007/BF02374789.
Bataglia OC, Teixeira JPF, Furlani PR, Furlani AMC, Gallo JR. Métodos de análise química de plantas. Campinas: Instituto agronômico de Campinas; 1978.
Kraska JE, Breitenbeck GA. Simple, robust method for quantifying silicon in plant tissue. Commun Soil Sci Plant Anal. 2010;41:2075–85.
Kondörfer GH, Pereira HS, Nola A. Análise de silício: solo, planta e fertilizante. Uberlândia: Universidade Federal de Uberlândia; 2004.
Siddiqi MY, Glass ADM. Utilization index: a modified approach to the estimation and comparison of nutrient utilization efficiency in plants. J Plant Nutr. 1981;4:289–302. https://doi.org/10.1080/01904168109362919.
Ferreira DF. Sisvar: a guide for its bootstrap procedures in multiple comparisons. Ciência e Agrotecnologia. 2014;38:109–13.
Acknowledgements
The authors would like to thank the Study Group of Plant Nutrition at Unesp (Genplant) and to Leonardo Henrique Sória for the help with the graph mathematic settings.
Funding
This study was funded by the Brazilian Coordination for the Improvement of Higher Education Personnel (CAPES), Brazil, Code 001; and by National Counsel of Technological and Scientific Development (CNPa), Brazil, Code163008/2020–5.
The funding body played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
All authors have read and approved the final manuscript. JPSJ, PLG, RMP and CNSC planed and designed of the research; JPSJ, JOC and CNSC performed experiments and conducted fieldwork. PLG, JOC, KRO and GSSJ performed laboratorial analyses. JPSJ wrote the manuscript. RMP, PLG, JOC and KRO reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
de Souza Júnior, J.P., de M Prado, R., Campos, C.N.S. et al. Addition of silicon to boron foliar spray in cotton plants modulates the antioxidative system attenuating boron deficiency and toxicity. BMC Plant Biol 22, 338 (2022). https://doi.org/10.1186/s12870-022-03721-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12870-022-03721-7