Abstract
Yeast infections have gained significant attention in the field of marine biology in recent years. Among the broad diversity of marine organisms affected by these infections, elasmobranchs (sharks and rays) have emerged as highly susceptible, due to climate change effects, such as increasing water temperatures and pollution, which can alter the composition and abundance of fungal communities. Additionally, injuries, or compromised immune systems resulting from pollution or disease may increase the likelihood of fungal infections in elasmobranchs. Studies are, however, still lacking for this taxonomic group. In this context, this study aimed to screen yeast species in cell cultures obtained from the brain of artisanally captured Pseudobatos horkelii, a cartilaginous fish that, although endangered, is highly captured and consumed worldwide. Fungi were isolated during an attempt to establish primary cultures of elasmobranch neural cells. Culture flasks were swabbed and investigated using morphological, phenotypic, and molecular techniques. Two isolates of the emerging opportunistic pathogen Trichosporon japonicum were identified, with high scores (1.80 and 1.85, respectively) by the MALDI-ToF technique. This is the first report of the basidiomycetous yeast T. japonicum in Pseudobatos horkelii in Brazil. This finding highlights the need for further research to determine the potential impact on elasmobranch health, ecology, as well as on commercial fisheries.
Similar content being viewed by others
Introduction
Significant shifts have increased in the marine environment due to climate change, leading in turn to salinity, light, temperature, sediment, and chemical pollution alterations. These changes result in marine fungi community imbalances, increasing the prevalence and severity of marine animal diseases due to fungal infections [1]. While over 10,000 pathogenic marine fungal species have been identified to date [2], and recently highlighted for their importance concerning marine health [3], their impacts in this regard remain largely understudied.
Certain fungi have been acknowledged as emerging opportunistic pathogens, such as Trichosporon spp. [4]. These organisms are widespread worldwide, with a preference for warm and tropical climates and are found in several environmental compartments, such as soil, decomposing wood and in both freshwater and marine environments, as well as in wildlife, such as bats, cattle and fish [5]. Members belonging to this genus have the potential to induce invasive and life-threatening fungal diseases in immunocompromised individuals [6, 7]. They also pose a risk to humans through the consumption of contaminated foodstuffs [8], including, but not limited, to milk and milk-derived products [9], honey [10], truffles [11] and seafood [12,13,14,15,16,17], as well as through workers exposed to potentially contaminated animals, such as zookeepers and fishers [18, 19].
Several aquatic biota, such as crustaceans and fish are highly vulnerable to fungal pathogens, both pathogenic and zoonotic [20]. Concerning Trichosporon spp. seafood contamination, assessments are mostly limited to crustaceans and bony fish, with reports on elasmobranchs, which encompass sharks and rays, however, still scarce.
Elasmobranchs are currently facing a multitude of threats, including overfishing, chemical pollution, habitat destruction, and climate change effects, resulting in global conservation concerns [21]. In this sense, the above-mentioned shifts in marine microbiota composition and diversity comprise a significant health effect factor that may negatively influence elasmobranch health and immune responses, potentially increasing their susceptibility to infections and diseases, increasing conservation concerns. In addition, many elasmobranch species are highly consumed as a protein source worldwide [22]. This also results in human health risks concerns, with potential human contamination by zoonotic pathogens posing as an important challenge, especially concerning specific vulnerable population strata, such as children and the elderly.
Although rays and skates are more threatened than previously estimated, with a higher percentage of species now being considered at risk compared to sharks [23], elasmobranch microbiome assessments have focused mainly on sharks to date. Furthermore, given their mostly benthic habits and direct association to substrates, many ray and skate species are at extremely high risk for contamination by several pollutants and pathogens, as sediments comprise the ultimate sink for these negative stressors [24,25,26].
In this context, brain samples from a highly endangered elasmobranch, Pseudobatos horkelii, commonly known as the Brazilian guitarfish, were collected as part of an ongoing project that aims to establish neural cell cultures from elasmobranchs. This species is distributed from Brazil to Uruguay, inhabiting coastal waters and occupying a meso-predator position, feeding on crustaceans, cephalopods, polychaetes and small fishes [27]. It is highly caught as bycatch and frequently consumed in Brazil and is categorized as Critically Endangered by the International Union for Conservation of Nature [28]. Although bans and extremely high fines have been implemented regarding this species capture and marketing in Brazil, significant numbers of these rays are sold for human consumption throughout the country (Hauser-Davis, Pers. Comm.).
Given that such cell cultures from this species were consistently contaminated by fungal species and dying at about 7 days post-plating, we decided to further investigate and describe which fungi may be present in the central nervous system of these elasmobranchs. The findings reported herein are highly relevant in describing potential emerging microbial pathogens that may directly affect elasmobranch health and may be transmitted to humans by elasmobranch meat ingestion and handling, also affecting primary studies requiring elasmobranch cell culturing.
Methods
Elasmobranch tissue isolation
P. horkelii specimens were captured by artisanal fishers in the state of Rio de Janeiro, southeastern Brazil. Only samples of recently deceased animals were sampled under a Brazilian Institute of Environment and Renewable Natural Resources Biodiversity Authorization and Information System (SISBIO) authorization, no. 77310–5.
Samples were obtained from two fresh specimens recently landed at an artisanal fisher colony in Rio de Janeiro, Brazil and displayed for sale on stainless steel display tables. Aiming to obtain sterile brain tissue, the animals’ heads were cleaned twice with 70% alcohol followed by cleaning with 2% benzalkonium chloride. Incisions were made on the animals’ heads with the aid of a #15 scalpel and sterile surgical scissors in the presence of a portable Bunsen burner and brain samples were collected. Tissue fragments of 10 by 10 mm2 were dissected from the telencephalon and placed in a transport medium, consisting of Leibovitz L-15 culture medium, 5% penicillin–streptomycin antibiotic solution, 0.25 ug mL−1 of fungizone, 50 ug mL−1 of gentamicin, 50 ug mL−1 neomycin sulfate, 373 mmol L-1 urea and 89 mmol L−1 NaCl (adapted from [29, 30]. Fragments were kept in glass Erlenmeyer flasks on ice throughout transportation to the Laboratory of Structural Biology at the Oswaldo Cruz Foundation.
At the laboratory, the samples were washed three times in a PBS solution modified for elasmobranchs containing 299 mmol L−1 urea and 68 mmol L−1 NaCl in a sterile penicillin–streptomycin antibiotic solution containing 0.25 ug mL−1 fungizone [30]. The tissue was then fragmented into small pieces of about 1 mm3 each and a 0.05% trypsin/0,02% Ethylenediaminetetraacetic acid (EDTA) solution was added for 10 min at 26 °C. After incubation, mechanical dissociation was performed by pipetting with a sterile glass Pasteur pipette and centrifugation to remove the trypsin solution. The obtained pellet was resuspended in culture medium and cell viability was determined by Trypan blue exclusion. All laboratorial procedures were performed in Biosafety level 2 cell culture hoods.
A total of 500,000 viable cells were then placed in 25 cm2 culture flasks and maintained with a culture medium adapted for elasmobranch cells according to [31], containing 50% Dulbecco’s Modified Eagle Medium (DMEM, ThermoFisher), 35% Leibovitz L-15, 15% Ham’s F-12 (ThermoFisher), 333 mmol L-1 urea, 188 mmol L-1 NaCl, 12% fetal bovine serum (Cultilab, São Paulo), 1% Glutamax, 2% penicillin–streptomycin antibiotic solution, 50 ug mL−1 gentamicin, 50 ug mL−1 neomycin sulfate, 0.25 ug mL-1 fungizone, 2 ng mL−1 Recombinant human epidermal growth factor (ThermoFisher) and 2 ng mL−1 Recombinant Human fibroblast growth factor (ThermoFisher) in an incubator at 26 °C. Occasionally, tissue fragments were also transferred to the cell culture flasks along with dissociated cells, and some adhered as explants in the flasks.
About one week after plating, cultures were consistently noted as contaminated. To identify which microorganisms were most predominant in elasmobranch neural cell cultures, swabs were sampled from the culture flasks after three days in vitro (3 div) and placed in 5 mL tubes containing a 0.9% saline solution. The samples were then taken to the Laboratory of Taxonomy, Biochemistry and Bioprospecting of Fungi in less than 30 min after collection and kept at 4 ºC until the moment of analysis.
Microbiological sampling of elasmobranch cell culture-derived swabs
Samples were streaked onto Sabouraud Dextrose Agar (SDA) and incubated at 30 °C for 48 h for morphological assessments. All samples presenting growth on the SDA medium were then subcultured onto CHROMagar Candida (BD Difco) and colonies were interpreted according to the manufacturer’s instructions.
Proteomic genotyping
In addition to morphologic and phenotypic tests, the obtained isolates were also identified by MALDI-TOF MS following Pinto et al. [32] and Oliveira et al. [33], employing α-cyano-4-hydroxycinnamic acid (CHCA, Fluka, Buchs, Switzerland) used as the matrix. Each sample was analyzed in triplicate from the same culture and from sub-cultures on alternate days. Samples were air-dried at room temperature prior to spectra acquisition. Spectra were obtained on a Microflex mass spectrometer (Bruker Daltonics, Bremen, Germany) using the Flexcontrol v. 3.0 software and spectra were imported and analyzed using the Maldi Biotyper v. 2.0 software (Bruker Daltonics, Bremen, Germany). The Escherichia coli DH5α strain was used for in situ protein extraction employed as the standard for MALDI-TOF MS external calibration, according to [34]. Results are expressed as log values ranging from 0 to 3. Logscore cutoff criteria were applied, where values of 1.7 are employed for reliable genus identification, and values between 1.8 and 2.0 are considered as confirmed species identification [35].
Results And Discussion
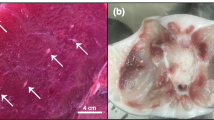
The cell culture samples cultivated onto Sabouraud Dextrose Agar (SDA) as described above resulted in colonies with no bacterial characteristics detected in the SDA cultures. The SDA (Fig. 1A and B) growths were then further investigated by streaking onto CHROMagar Candida (BD Difco) plates. Small light blue-gray colonies (Fig. 1C and D), an atypical color for this substrate according to the manufacturer, were observed, indicating the growth of species other than Candida spp. Biochemical culture characteristics were then examined by conventional microscopy methods [32], and the morphological and phenotypical characteristics of the growths suggested the presence of Trichosporon sp.
A and B Growths observed in Sabouraud Dextrose Agar incubated at 30o C for 48 h in aerobic conditions, samples LTBBF-RHO2 and LTBBF-RHO2; C and D Growths observed in BDTM CHROMagarTM Candida Medium (BD Difco) incubated at 35 oC for 48 h in aerobic conditions, front and back of the petri dishes, samples LTBBF-RHO2 (1) and LTBBF-RHO2(2)
Following microscopy identification, the samples were then analyzed by MALDI-ToF, which attributed high scores of 1.85 and 1.80 to the two isolate growths observed in the Sabouraud Dextrose Agar and in BDTM CHROMagarTM Candida Medium (BD Difco), identifying them at the species level as T. japonicum. MALDI-ToF analyses have been applied with high reliability to identify fungal species from several sources, including in a recent study by our group concerning another elasmobranch species also captured in Rio de Janeiro, Brazil, demonstrating high agreement with conventional identification methods and DNA sequencing [33]. Several studies have, in fact, noted that DNA sequencing is not required when a reliable spectral database is available [36, 37] and that the MALDI-ToF technique is, in fact, superior concerning yeast and mycobacteria isolate identification compared to gene sequencing. This, therefore, makes this technique suitable as a first-line test for yeast and mycobacteria identification [38]. This analytical technique is therefore a proven method for this type of fungal assessment in wildlife.
About 50 Trichosporon species are currently recognized, many of which are associated with human diseases [39]. Trichosporon asahii is the most commonly identified causative agent of trichosporonosis, a disease that exhibits both cutaneous and central nervous system-related manifestations [5], and other species, such as T. cutaneum, T. asteroides, T. mucoides, T. inkin, T. jirovecii, T. dermatis, T. domesticum, T. montevideense, T. coremiiforme, and T. faecale have also shown to be associated with infections [40]. Despite the use of antifungal therapy, the incidence of trichosporonosis in humans has increased over the past decades due to fungal resistance, leading to significant human morbidity and mortality rates ranging from 40 to 90% [5]. Some authors have postulated that this may be due to the increasing use of echinocandins, currently the drugs of choice in many clinical contexts at high-risk for invasive fungal infection [41].
The prevalence of fungal infections has, in fact, increased substantially worldwide, particularly among high-risk populations such as individuals with HIV/AIDS, transplant recipients, and those with weakened immune systems [42,43,44], although certain fungi, including Cryptococcus, Coccidioides, and Histoplasma, can also infect people with healthy immune systems [45,46,47]. For example, in the USA, Candida spp. caused 72 to 228 infections per million inhabitants annually, while Cryptococcus neoformans was responsible for 30–66 infections per million inhabitants, and Aspergillus spp., for around 12–34 infections per million inhabitants in the late 1990s and early 2000s [48]. More recently, the average occurrence of new Candida spp. infections was around 9 per 100,000 individuals between 2013 and 2017 and, according to the CDC, an estimated 25,000 cases of candidemia are reported annually throughout the United States [49]. The extent of central nervous system damage, however, varies based on the specific fungal forms present in the human body, such as blastopores or hyphae [44, 50, 51]. Fungi like Histoplasma, Blastomyces, Coccidioides, Candida, Paracoccidioides and Cryptococcus can enter capillaries and subarachnoid spaces, leading to conditions such as meningitis and subpial ischemic lesions. Candida, in contrast, has the potential to infiltrate blood vessels, causing localized necrotic lesions, while larger vessels may be breached by Aspergillus, Cladosporium, and Mucoromycetes, which could potentially result in strokes [52].
Conversely, the source of Trichosporon spp. infections remains uncertain, even though the incidence of infection by this pathogenic agent has increased globally in recent decades [5], especially due to increased immunosuppressive conditions and malignant hematological diseases [39, 53, 54]. Trichosporon spp. have been frequently detected in hospital environments, with a notable association between trichosporonosis and invasive clinical procedures, such as the use of probes and catheters [55]. For example, one study involving 17 patients revealed that 10 experienced fungemia with approximately 41% of these developing trichosporonosis after catheter usage [56]. Most suffered from acute leukemia and neutropenia, underwent chemotherapy and were receiving high doses of corticoids, which the authors indicate may have potentially influenced the development of trichosporonosis. Another study assessed the species distribution and antifungal susceptibilities of 22 bloodstream Trichosporon isolates recovered from patients hospitalized in five medical centers between 1995 and 2007 in São Paulo, Brazil [57]. The samples were obtained from both pediatric (44%) and adult patients presenting a diverse range of underlying conditions, such as premature birth, surgery, organ failures, inflammatory gastrointestinal disease, and cancer. Most patients (67%) developed systemic bacterial infections either before or concurrently with fungemia and had a central venous catheter in place when fungemia occurred. The authors indicate that these findings support the idea that invasive Trichosporon infections can occur in non-cancer patients with chronic illnesses and disruptions to skin and mucous membranes. The predominant species was T. asahii, followed by T. larkias, T. coremiiforme, T. dermatis, T. inkin, T. ovoides, and T. mucoides. Another investigation evaluated 24 clinical isolates of Trichosporon species isolated from blood, samples, pleural fluid and nails from 2005–2016 in a tertiary hospital in North India [58]. The isolates were identified as T. asahii, followed by T. dermatis (8.33%), T. japonicum (4.17%), T. ovoides (4.17%) and T. mucoides (4.17%). The authors indicated isolate resistance to fluconazole, voriconazole and itraconazole and susceptibility to ketoconazole, which is of concern due to the increasing number of cases of disseminated trichosporonosis being noted worldwide.
Trichosporon spp. are also, nevertheless, considered a normal inhabitant of the digestive tract and may be present on healthy patient skin [54, 59, 60]. In newborns, natural protective barriers, such as the skin and digestive mucosa, may become more permeable, potentially facilitating exposure to various pathogens, such as Trichosporon spp. In fact, Trichosporon spp. have already been detected on the skin of premature infants [61]. The mechanisms through which fungi can invade the central nervous system (CNS), though, have not yet been fully elucidated [62]. The blood brain barrier (BBB) is a critical component of the mammalian brain, formed primarily by the brain capillary endothelium under the influence of neighboring astrocytic glia [63]. It establishes a specialized microenvironment for optimal neuronal function and plays a crucial role in preventing harmful substances from entering the brain. Infection by C. albicans causes BBB disruption that can lead to transient encephalitis and cognitive impairment [64, 65], probably by crossing endothelial cells through the paracellular pathway while also being internalized by cells before exiting the basal surface through the transcellular pathway [66, 67]. Cryptococcus spp., in turn, employs several mechanisms to break through the BBB and access the CNS, also through facilitated paracellular passage. In vitro experiments, for example, have demonstrated that phagocytes containing viable C. neoformans can cross a layer of brain endothelial cells, indicating that Cryptococcus spp. can employ a “Trojan horse” strategy to infiltrate the brain [68].
Despite the fact that elasmobranch brains possess several similarities to mammalian brains [69], they differ concerning the BBB. While the endothelium of elasmobranchs does not act as a barrier to macromolecule entry, a glial BBB is suggested to exist [70]. Studies involving skates, dogfish, sharks, and rays have demonstrated that dyes are prevented from entering the brain but can pass through the endothelial layer [71]. These differences concerning mammalian and elasmobranch BBB probably interfere with brain permeability and may comprise an important factor in the invasiveness of pathogens like fungi, and in the establishment of CNS infections.
Although considered an exceedingly rare representative within the Trichosporon genus, clinical cases of contamination by T. japonicum have been reported in several regions, including Japan, where this species was initially discovered in 1971 from the atmosphere of a microbiological laboratory, as well as other countries [72,73,74]. The first documented clinical case of T. japonicum contamination occurred in 2008, involving a child diagnosed with acute myeloid leukemia (AML) who experienced fungemia associated with T. japonicum [75]. In Brazil, only two T. japonicum isolates have been isolated from humans [76] and one from artisanal cheese [77], making this only the third report of this fungal pathogen in the country, and the first in wildlife consumed by humans.
Fungal pathogens cause significant economic losses to aquaculture activities, increasingly posing risks to both cultured and wild fish populations worldwide [78], as farmed fisheries often drain their residues into rivers [79], and subsequently, the oceans. This may, therefore, potentially lead to local and pan-continental extinctions [80], presenting broader implications for global health, biodiversity, and conservation [81, 82]. This indicates the need for continuous monitoring of both freshwater and marine fungal fauna associations. Specifically concerning Trichosporon spp. fish contamination, one study reported the presence of Trichosporon beigelii, a novel fungal pathogen, in the cuticle of the freshwater crayfish Astacus astacus, raising concerns that crayfish may serve as a vector for this pathogen, significantly [83], while another study isolated T. jirovecii form the exoskeleton as well as eyestalks, gills, muscle and haemolymph of red swamp crayfish (Procambarus larkia) from the River Nile [16]. In another study, T. mucoides was isolated from the gills and intestine of Tilapia (Oreochromis sp., 4 positive isolates out of 27, 5.40%), the gills and intestine of African catfish (Clarias gariepinus, 3 positive isolates out of 22, 3.89%) and from the gills of gray mullet (Mugil cephalus, 2 positive isolates out of 21, 3.12%) [17]. Interestingly, fish feed has also been noted as contaminated with Trichosporon asahii [84], comprising another concern for aquaculture activities in this regard. As, however, bans are in place concerning P. horkeili marketing and consumption in Brazil, this report may also comprise an indirect conservation tool, as increased awareness of the potential contamination of these fish with zoonotic fungi may lead to reduced consumption of the critically endangered Pseudobatos horkeili in Brazil.
People routinely exposed to fungus-contaminated animals are at heightened risk for contracting fungal infections. One report, for example, indicated mycobacteriosis due to a finger wound caused by the dorsal fin spines of a fish (Tilapia sp.) netted during fishing activities by a fisher, which the authors postulated as being caused by either Mycobacterium marinuum or Sporothrix shenckii [85]. Other studies have reported the presence of several pathogenic fungi in the traumatogenic structures of freshwater fish, such as stings, rayed fins and teeth, and high incidence of fungal conditions in artisanal fishers [86], indicating occupation exposure concerns. As elasmobranchs are manipulated by fishers with no gloves, and usually sold decharacterized worldwide, i.e., with no caudal fin or head, potential fisher fungal exposure is increased, causing further concerns regarding the detection of the rare Trichosporon japonicum in the P. horkeili specimens analyzed herein. Finally, this report may also comprise an indirect conservation tool, as increased awareness of the potential contamination of these fish with zoonotic fungi may lead to reduced consumption of the critically endangered Pseudobatos horkeili in Brazil.
Conclusion
The method applied herein comprises an adequate tool to identify atypical and emerging fungal species in wildlife that may not be recognizable by conventional methods which is especially valuable in emerging fungal infections or cases where traditional methods might fail. The detected Pseudobatos horkelii contamination by Trichosporon japonicum indicate the possible ubiquitous contamination of Brazilian coastal waters by this rare fungus, suggesting that Pseudobatos horkelii may be an interesting sentinel species for emerging fungal pathogens in coastal marine waters. Furthermore, potential consumer and fisher contaminations may also occur. The limited understanding of elasmobranch associations with fungal pathogens remains and the significant lack of research on fungal biodiversity, prevalence, and their physiological impacts is concerning given the crucial role fungi may play in addressing climate change effects, the vulnerability of elasmobranchs, and the potential zoonotic nature of various fungal species. This clearly indicates the need for further assessments on the fungal diversity of elasmobranchs. This report also served as an indirect conservation tool by raising awareness about potential zoonotic fungi contamination, potentially reducing consumption of the critically endangered Pseudobatos horkeili in Brazil.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Garcia-Solache MA, Casadevall A. Global warming will bring new fungal diseases for mammals. Bio. 2010;1(1):e00061-10. https://doi.org/10.1128/mBio.00061-10.
Jones EBG. Are there more marine fungi to be described? Bot Mar. 2011;54(4):343–54.
Gonçalves MFM, Esteves AC, Alves A. Marine fungi: opportunities and challenges. Encyclopedia. 2022;2:559–77.
Mehta V, Nayyar C, Gulati N, Singla N, Rai S, Chandar JA. Comprehensive review of trichosporon spp.: an invasive and emerging fungus. Cureus. 2021;13(8):e17345. https://doi.org/10.7759/cureus.17345.
Castano G, Yarrarapu SNS, Mada PK. Trichosporonosis. In StatPearls: StatPearls Publishing; 2023.
Mariné M, Brown NA, Riaño-Pachón DM, Goldman GH. On and under the skin: emerging basidiomycetous yeast infections caused by trichosporon species. PloS Pathog. 2015;11(7):e1004982.
Mulè A, Rossini F, Sollima A, Lenzi A, Fumarola B, Amadasi S, Chiari E, Lorenzotti S, Saccani B, Van Hauwermeiren E, et al. Trichosporon asahii infective endocarditis of prosthetic valve: a case report and literature review. Antibiotics. 2023;12:1181.
Gujjari P, Suh SO, Coumes K, Zhou JJ. Characterization of oleaginous yeasts revealed two novel species: trichosporon cacaoliposimilis sp nov and trichosporon oleaginosus sp Nov. Mycologia. 2011;103(5):1110–8. https://doi.org/10.3852/10-403.
Ider S, Belguesmia Y, Cazals G, Boukherroub R, Coucheney F, Kihal M, Enjalbal C, Drider D. The antimicrobial peptide oranicin P16 isolated from trichosporon asahii ICVY021, found in camel milk’s, inhibits Kocuria rhizophila. Food Bioscience. 2020;36(100670):ISSN 2212-4292. https://doi.org/10.1016/j.fbio.2020.100670.
Xue SJ, Liu J, Li XC, Zhang XT, Xin ZZ, Jiang WW, Zhang JY. First natural yeast strain trichosporon asahii hz10 with robust flavonoid productivity and its potential biosynthetic pathway. J Agric Food Chem. 2023. https://doi.org/10.1021/acs.jafc.3c05188.
Buzzini P, Gasparetti C, Turchetti B, Cramarossa MR, Vaughan-Martini A, Martini A, Pagnoni UM, Forti L. Production of volatile organic compounds (VOCs) by yeasts isolated from the ascocarps of black (Tuber melanosporum Vitt.) and white (Tuber magnatum Pico) truffles. Arch Microbiol. 2005;184(3):187–93.
Roth FJ Jr, Ahearn DG, Fell JW, Meyers SP, Meyer SA. Ecology and taxonomy of yeasts isolated from various marine substrates. Limnol Oceanogr. 1962;7(1962):178–85.
Eklund MW, Spinelli J, Miyauchi D, Groninger H. Characteristics of yeasts isolated from Pacific crab meat. Appl Microbiol. 1965;13(6):985–90. https://doi.org/10.1128/am.13.6.985-990.1965.
Bruce J, Morris EO. Psychrophilic yeasts isolated from marine fish. Antonie Van Leeuwenhoek. 1973;39(2):331–9. https://doi.org/10.1007/BF02578865.
Aubin J, Gatesoupe FJ, Labbé L, Lebrun L. Trial of probiotics to prevent the vertebral column compression syndrome in rainbow trout (Oncorhynchus mykiss Walbaum). Aquac Res. 2005;36:758–67.
Abdallah ESH, Mahmoud MM, Abdel-Rahim IR. Trichosporon jirovecii infection of red swamp crayfish (Procambarus larkia). J Fish Dis. 2018;41(11):1719–32. https://doi.org/10.1111/jfd.12879.
Tartor Y, Taha M, Mahboub H, El Ghamery M. Yeast species associated with diseased fish: Occurrence, identification, experimental challenges and antifungal susceptibility testing. Aquaculture. 2018;488:134–44.
Adebiyi AI, Oluwayelu DO. Zoonotic fungal diseases and animal ownership in Nigeria. Alexandria J Med. 2018;54(4):397–402. https://doi.org/10.1016/j.ajme.2017.11.007.
Le F, Liu B, Si Z, Li S, Qiao J. Prevalence of dermatitis and superficial fungal infection of the hands in seafood workers: an investigation from food markets in Ningbo, China. Risk Manag Healthcare Policy. 2020;13:427–31. https://doi.org/10.2147/RMHP.S240327.
Ziarati M, Zorriehzahra MJ, Hassantabar F, Mehrabi Z, Dhawan M, Sharun K, Emran TB, Dhama K, Chaicumpa W, Shamsi S. Zoonotic diseases of fish and their prevention and control. Vet Q. 2022;42(1):95–118. https://doi.org/10.1080/01652176.2022.2080298.
Dulvy NK, Fowler SL, Musick JA, Cavanagh RD, Kyne PM, Harrison LR, Carlson JK, Davidson LN, Fordham SV, Francis MP, Pollock CM, Simpfendorfer CA, Burgess GH, Carpenter KE, Compagno LJ, Ebert DA, Gibson C, Heupel MR, Livingstone SR, Sanciangco JC, Stevens JD, Valenti S, White WT. Extinction risk and conservation of the world’s sharks and rays. Elife. 2014;3:e00590. https://doi.org/10.7554/eLife.00590.
Seidu I, Brobbey L, Danquah E, Oppong S, Beuningen D, Seidu M, Dulvy NK. Fishing for survival: importance of shark fisheries for the livelihoods of coastal communities in Western Ghana. Fish Res. 2021;246:106157. https://doi.org/10.1016/j.fishres.2021.106157.
Dulvy NK, Pacoureau N, Rigby CL, Pollom RA, Jabado RW, Ebert DA, Finucci B, Pollock CM, Cheok J, Derrick DH, Herman KB, Sherman CS, VanderWright WJ, Lawson JM, Walls RHL, Carlson JK, Charvet P, Bineesh KK, Fernando D, Ralph GM, Matsushiba JH, Hilton-Taylor C, Fordham SV, Simpfendorfer CA. Overfishing drives over one-third of all sharks and rays toward a global extinction crisis. Curr Biol. 2021;31(22):5118–9. https://doi.org/10.1016/j.cub.2021.11.008.
Raghukumar C, Raghukumar S, Sheelu G, Gupta SM, Nath BN, Rao BR. Buried in time: culturable fungi in a deep-sea sediment core from the chagos Trench, Indian Ocean. Deep-Sea Res I. 2004;51:1759–68.
Rojas-Jimenez K, Grossart HP, Cordes E, Cortés J. Fungal communities in sediments along a depth gradient in the Eastern tropical pacific. Front Microbiol. 2020;6(11):575207. https://doi.org/10.3389/fmicb.2020.575207.
Marie Booth J, Fusi M, Marasco R, Michoud G, Fodelianakis S, Merlino G, Daffonchio D. The role of fungi in heterogeneous sediment microbial networks. Sci Rep. 2019;9(1):7537. https://doi.org/10.1038/s41598-019-43980-3.
Michael, SW (1993) Reef sharks and rays of the world. A guide to their identification, behavior, and ecology. Sea Challengers, Monterey, California. 107 p.
Pollom, R, Barreto, R, Charvet, P, Chiaramonte, GE, Cuevas, JM, Herman, K, Martins, MF, Montealegre-Quijano, S, Motta, F, Paesch, L & Rincon, G (2020) Pseudobatos horkelii. The IUCN Red List of Threatened Species 2020: e.T41064A2951089. https://doi.org/10.2305/IUCN.UK.2020-3.RLTS.T41064A2951089.en. Accessed on 15 November 2023.
Hartmann JX, Bissoon LM, Poyer JC. Routine establishment of primary elasmobranch cell cultures. In vitro Cell Develop Biol. 1992;28A(2):77–9. https://doi.org/10.1007/BF02631008.
Uno Y, Nozu R, Kiyatake I, Higashiguchi N, Sodeyama S, Murakumo K, Sato K, Kuraku S. Cell culture-based karyotyping of orectolobiform sharks for chromosome-scale genome analysis. Commun Biology. 2020;3(1):652. https://doi.org/10.1038/s42003-020-01373-7.
Poyer JC, Hartmann JX. Establishment of a cell line from brain tissue of the silky shark, Carcharhinus falciformis. In Vitro Cell Dev Biol – Animal. 1992;28:682–4. https://doi.org/10.1007/BF02631047.
Pinto TN, Kohn A, da Costa GL, Oliveira LMA, Pinto TCA, Oliveira MME. Candida guilliermondii as an agent of postpartum subacute mastitis in Rio de Janeiro, Brazil: case report. Front Microbiol. 2022;23(13):964685. https://doi.org/10.3389/fmicb.2022.964685.
Oliveira MME, Lopes AP, Pinto TN, da Costa GL, Goes-Neto A, Hauser-Davis RA. A Novel One Health Approach concerning Yeast Present in the Oral Microbiome of the Endangered Rio Skate (Rioraja agassizii) from Southeastern Brazil. Microorganisms. 2023;11(8):1969. https://doi.org/10.3390/microorganisms11081969.
Oliveira MM, Santos C, Sampaio P, Romeo O, Almeida-Paes R, Pais C, Lima N, Zancopé-Oliveira RM. Development and optimization of a new MALDI-TOF protocol for identification of the Sporothrix species complex. Res Microbiol. 2015;166(2):102–10. https://doi.org/10.1016/j.resmic.2014.12.008.
Stevenson LG, Drake SK, Murray PR. Rapid identification of bacteria in positive blood culture broths by matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol. 2010;48(2):444–7. https://doi.org/10.1128/JCM.01541-09.
Matos AMF, Moreira LM, Barczewski BF, de Matos LX, Oliveira JBV, Pimentel MIF, Almeida-Paes R, Oliveira MG, Pinto TCA, Lima N, Matos MO, Costa LGM, Santos C, Oliveira MME. Identification by MALDI-TOF MS of Sporothrix brasiliensis Isolated from a subconjunctival infiltrative lesion in an immunocompetent patient. Microorganisms. 2019;8(1):22. https://doi.org/10.3390/microorganisms8010022.
Flórez-Muñoz SV, Gómez-Velásquez JC, Loaiza-Díaz N, Soares C, Santos C, Lima N, Mesa-Arango AC. ITS rDNA Gene Analysis Versus MALDI-TOF MS for identification of neoscytalidium dimidiatum isolated from onychomycosis and dermatomycosis cases in medellin (Colombia). Microorganisms. 2019;7(9):306. https://doi.org/10.3390/microorganisms7090306.
Chen JH, Yam WC, Ngan AH, Fung AM, Woo WL, Yan MK, Choi GK, Ho PL, Cheng VC, Yuen KY. Advantages of using matrix-assisted laser desorption ionization-time of flight mass spectrometry as a rapid diagnostic tool for identification of yeasts and mycobacteria in the clinical microbiological laboratory. J Clin Microbiol. 2013;51(12):3981–7. https://doi.org/10.1128/JCM.01437-13.
Montoya AM, González GM. Trichosporon spp.: an emerging fungal pathogen. Medicina Universitaria. 2014;16:37–43.
Liu XZ, Wang QM, Theelen B, Groenewald M, Bai FY, Boekhout T. Phylogeny of tremellomycetous yeasts and related dimorphic and filamentous basidiomycetes reconstructed from multiple gene sequence analyses. Stud Mycol. 2015;81:1–26. https://doi.org/10.1016/j.simyco.2015.08.001.
Liao Y, Lu X, Yang S, Luo Y, Chen Q, Yang R. Epidemiology and outcome of trichosporon fungemia: a review of 185 reported cases from. Open Forum Infect Dis. 2015;2(4):ofv141. https://doi.org/10.1093/ofid/ofv141.
Bongomin F, Gago S, Oladele RO, Denning DW. Global and multi-national prevalence of fungal diseases—estimate precision. J Fungi. 2017;3(4):57.
McCarthy M, Rosengart A, Schuetz AN, Kontoyiannis DP, Walsh TJ. Mold infections of the central nervous system. N Engl J Med. 2014;371(2):150–60. https://doi.org/10.1056/NEJMra1216008.
Góralska K, Blaszkowska J, Dzikowiec M. Neuroinfections caused by fungi. Infection. 2018;46(4):443–59. https://doi.org/10.1007/s15010-018-1152-2.
Yamaguchi H, Komase Y, Ikehara M, Yamamoto T, Shinagawa T. Disseminated cryptococcal infection with eosinophilia in a healthy person. J Infect Chemother. 2008;14(4):319–24. https://doi.org/10.1007/s10156-008-0618-z.
Martinez A, McClaskey D. A case of extra-articular coccidioidomycosis in the knee of a healthy patient. J Am Board Fam Med. 2023;36(2):376–9. https://doi.org/10.3122/jabfm.2022.220234R2.
Albandak M, Azar J, Ayyad M, Salah Q, Toqan A, Giacaman N, Marzouqa N, Al-Tawil M, Wishah B, Barabrah A. Chronic Cavitary Pulmonary Histoplasmosis in an Immunocompetent Patient. Cureus. 2023;15(4):e37095. https://doi.org/10.7759/cureus.37095.
Pfaller MA, Pappas PG, Wingard JR. Invasive fungal pathogens: current epidemiological trends. Clin Infect Dis. 2006;43(Issue Supplement_1):S3–14.
Tsay S, Williams S, Mu Y, Epson E, Johnston H, Farley MM, Harrison LH, Vonbank B, Shrum S, Dumyati G, Zhang A, Schaffner W, Magill S, Vallabhaneni S. 363. National Burden of Candidemia, United States, 2017. Open Forum Infect Dis. 2018;5(Suppl 1):S142-3. https://doi.org/10.1093/ofid/ofy210.374.
Das S, Bhuyan R, Bagchi A, Saha T. Network analysis of hyphae forming proteins in Candida albicans identifies important proteins responsible for pathovirulence in the organism. Heliyon. 2019;5(6):01916. https://doi.org/10.1016/j.heliyon.2019.e01916.
Panackal AA, Williamson PR. Fungal infections of the central nervous system. Continuum (Minneap Minn). 2015;21(6):1662–78. https://doi.org/10.1212/CON.0000000000000241.
Shankar SK, Mahadevan A, Sundaram C, Sarkar C, Chacko G, Lanjewar DN, Santosh V, Yasha TC, Radhakrishnan VV. Pathobiology of fungal infections of the central nervous system with special reference to the Indian scenario. Neurol India. 2007;55(3):198–215. https://doi.org/10.4103/0028-3886.35680.
Fagundes-Junior, AAP, de Carvalho RT, Focaccia R, Fernandez JG, de Araujo HBN, Strabelli TMV, Kopel L, Lage SG. (2008). Trichosporon asahii an Emerging Etiologic Agent of Fungal Infection and Colonization in Heart Failure Patients in Intensive Care Unit. Case Report and Literature Review. Revista Brasileira de Terapia Intensiva. Vol 20, (1) 106–109. https://www.scielo.br/j/rbti/a/fYPjYRynZcm3RDNtX4ffszz/?format=pdf&lang=pt.
Colombo AL, Padovan AC, Chaves GM. Current knowledge of trichosporon spp. And trichosporonosis. Clin Microbiol Rev. 2011;24(4):682–700. https://doi.org/10.1128/CMR.00003-11.
Fanfair RN, Heslop O, Etienne K, Rainford L, Roy M, Gade L, et al. Trichosporon asahii among intensive care unit patients at a medical center in Jamaica. Infect Control Hosp Epidemiol. 2013;34:638–41. https://doi.org/10.1086/670633.
Kontoyiannis DP, Torres HA, Chagua M, Hachem R, Tarrand JJ, Bodey GP, Raad II. Trichosporonosis in a tertiary care cancer center: risk factors, changing spectrum and determinants of outcome. Scand J Infect Dis. 2004;36(8):564–9. https://doi.org/10.1080/00365540410017563.
Chagas-Neto TC, Chaves GM, Melo AS, Colombo AL. Bloodstream infections due to Trichosporon spp.: species distribution, Trichosporon asahii genotypes determined on the basis of ribosomal DNA intergenic spacer 1 sequencing, and antifungal susceptibility testing. J Clin Microbiol. 2009;47(4):1074–81. https://doi.org/10.1128/JCM.01614-08.
Singh S, Capoor MR, Varshney S, Gupta DK, Verma PK, Ramesh V. Epidemiology and antifungal susceptibility of infections caused by trichosporon species : an emerging non-candida and non-cryptococcus yeast worldwide. Indian J Med Microbiol. 2019;37(4):536–41. https://doi.org/10.4103/ijmm.IJMM_19_146.
Zhang E, Sugita T, Tsuboi R, Yamazaki T, Makimura K. The opportunistic yeast pathogen trichosporon asahii colonizes the skin of healthy individuals: analysis of 380 healthy individuals by age and gender using a nested polymerase chain reaction assay. Microbiol Immunol. 2011;55:483–8. https://doi.org/10.1111/j.1348-0421.2011.00341.x.
Cho O, Matsukura M, Sugita T. Molecular evidence that the opportunistic fungal pathogen Trichosporon asahii is part of the normal fungal microbiota of the human gut based on rRNA genotyping. Int J Infect Dis. 2015;39:87–8. https://doi.org/10.1016/j.ijid.2015.09.009.
Kaufman D, Boyle R, Hazen KC, Patrie JT, Robinson M, Donowitz LG. Fluconazole prophylaxis against fungal colonization and infection in preterm infants. N Engl J Med. 2001;345:1660–6. https://doi.org/10.1056/NEJMoa010494.
Snarr BD, Drummond RA, Lionakis MS. It’s all in your head: antifungal immunity in the brain. Curr Opin Microbiol. 2020;58:41–6. https://doi.org/10.1016/j.mib.2020.07.011.
Kadry H, Noorani B, Cucullo L. A blood-brain barrier overview on structure, function, impairment, and biomarkers of integrity. Fluids Barriers CNS. 2020;17(1):69. https://doi.org/10.1186/s12987-020-00230-3.
Navarathna DH, Munasinghe J, Lizak MJ, Nayak D, McGavern DB, Roberts DD. MRI confirms loss of blood-brain barrier integrity in a mouse model of disseminated candidiasis. NMR Biomed. 2013;26(9):1125–34. https://doi.org/10.1002/nbm.2926.
Wu Y, Du S, Johnson JL, Tung HY, Landers CT, Liu Y, Seman BG, Wheeler RT, Costa-Mattioli M, Kheradmand F, Zheng H, Corry DB. Microglia and amyloid precursor protein coordinate control of transient Candida cerebritis with memory deficits. Nat Commun. 2019;10(1):58. https://doi.org/10.1038/s41467-018-07991-4.
Sheppard DC, Filler SG. Host cell invasion by medically important fungi. Cold Spring Harb Perspect Med. 2014;5(1):a019687. https://doi.org/10.1101/cshperspect.a019687.
Liu Y, Mittal R, Solis NV, Prasadarao NV, Filler SG. Mechanisms of Candida albicans trafficking to the brain. PloS Pathog. 2011;7(10):e1002305. https://doi.org/10.1371/journal.ppat.1002305.
Santiago-Tirado FH, Onken MD, Cooper JA, Klein RS, Doering TL. Trojan horse transit contributes to blood-brain barrier crossing of a eukaryotic pathogen. mBio. 2017;8(1):e02183-16. https://doi.org/10.1128/mBio.02183-16.
Kreps E. Brain lipids of elasmobranchs (an essay on comparative neurobiology), comparative biochemistry and physiology part B: comparative biochemistry, 68(3). ISSN. 1981;363–367:0305–491.
Bundgaard M, Cserr H. A glial blood-brain barrier in elasmobranchs. Brain Res. 1981;226(1–2):61–73. https://doi.org/10.1016/0006-8993(81)91083-0. (PMID: 7296300).
Cserr HF, Fenstermacher J, Rall D. Brain-barrier systems in sharks. Comp Biochem Physiol A Physiol. 1972;42(1):73–8. https://doi.org/10.1016/0300-9629(72)90368-4.
Menu E, Kabtani J, Roubin J, Ranque S, L’Ollivier C. Pericardial effusion due to trichosporon japonicum: a case report and review of the literature. Pathogens. 2022;11(5):598. https://doi.org/10.3390/pathogens11050598.
Albitar-Nehme S, Agosta M, Kowalska AH, Mancinelli L, Onori M, Lucignano B, Mattana G, Quagliarella F, Cefalo MG, Merli P, Locatelli F, Perno CF, Bernaschi P. Case report: trichosporon japonicum fungemia in a pediatric patient with refractory acute b cell lymphoblastic leukemia. Front Pediatr. 2022;3(10):861476. https://doi.org/10.3389/fped.2022.861476.
Bongomin F, out A, Calisti G, Richardson MD, Barnard J, Venkateswaran R, Vergidis P. Trichosporon japonicum fungemia and ventricular assist device infection in an immunocompetent patient. Open Forum Infect Dis. 2019;6(9):ofz343. https://doi.org/10.1093/ofid/ofz343.
Ağirbasli H, Bilgen H, Ozcan SK, Otlu B, Sinik G, Cerikçioğlu N, Durmaz R, Can E, Yalman N, Gedikoğlu G, Sugita T. Two possible cases of trichosporon infections in bone-marrow-transplanted children: the first case of T. japonicum isolated from clinical specimens. Jpn J Infect Dis. 2008;61(2):130–2.
de Andrade IB, Figueiredo-Carvalho MHG, Chaves ALDS, Coelho RA, Almeida-Silva F, Zancopé-Oliveira RM, Frases S, Brito-Santos F, Almeida-Paes R. Metabolic and phenotypic plasticity may contribute for the higher virulence of Trichosporon asahii over other Trichosporonaceae members. Mycoses. 2023;66(5):430–40. https://doi.org/10.1111/myc.13562.
Aragão MO, Evangelista SR, Passamani FRF, Guimarães JPM, Abreu LR, Batista LR. Fungal community and physicochemical profiles of ripened cheeses from the Canastra of Minas Gerais. Brazil J Food Sci Technol. 2022;59(12):4685–94. https://doi.org/10.1007/s13197-022-05548-4.
Shabir U, Dar JS, Bhat AH, Nazir R, Ameen F. The hidden world of fish fungal pathogens: molecular identification and phylogenetic analysis in common carp. Cyprinus Carpio Arch Microbiol. 2023;205(9):311. https://doi.org/10.1007/s00203-023-03651-4.
Andreou D, Arkush KD, Guégan JF, Gozlan RE. Introduced pathogens and native freshwater biodiversity: a case study of Sphaerothecum destruens. PLoS ONE. 2012;7(5):e36998. https://doi.org/10.1371/journal.pone.0036998.
Gozlan RE, Marshall WL, Lilje O, Jessop CN, Gleason FH, Andreou D. Current ecological understanding of fungal-like pathogens of fish: what lies beneath? Front Microbiol. 2014;5:62. https://doi.org/10.3389/fmicb.2014.00062.
Van West P. Saprolegnia parasitica, an oomycete pathogen with a fishy appetite: new challenges for an old problem. Mycologist. 2006;20:99–104.
Derome N, Gauthier J, Boutin S, Llewellyn M (2016) Fungal secondary invaders of fish. The rasputin effect: when commensals and symbionts become parasitic. Springer, pp 109–126.
Soderhall K, Rantamaki J, Constantinescu O. Isolation of trichosporon beigelii from the freshwater crayfish astacus astacus. Aquaculture. 1993;116(1):25–31.
Pinheiro REE, Rodrigues AMD, Santos JTO, Costa JA, Pereyra CM, Torres AM, Rosa CA, Santos ARO, Muratori MCS. Occurrence and diversity of yeast species isolated from fish feed and tambatinga gut. Lat Am J Aquat Res. 2018;46(4):837–42. https://doi.org/10.3856/vol46-issue4-fulltext-22.
Haddad VJ, Miot HA, Bartoli LD, Cardoso Ade C, de Camargo RM. Localized lymphatic sporotrichosis after fish-induced injury (Tilapia sp.). Med Mycol. 2002;40(4):425–7. https://doi.org/10.1080/mmy.40.4.425.427.
Leme FC, Negreiros MM, Koga FA, Bosco Sde M, Bagagli E, Haddad JV. Evaluation of pathogenic fungi occurrence in traumatogenic structures of freshwater fish. Rev Soc Bras Med Trop. 2011;44(2):182–5. https://doi.org/10.1590/s0037-86822011005000007.
Acknowledgements
The implementation of the Projeto Pesquisa Marinha e Pesqueira is a compensatory measure established by the Conduct Adjustment Agreement under the responsibility of the PRIO company, conducted by the Federal Public Ministry—MPF/RJ.
Funding
This research received support from the Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro (FAPERJ) (DA, RAHD and MMEO), through a Jovem Cientista do Nosso Estado 2021–2024 grant (process number E-26/201.270/20211 for RAHD and E-26/201.336/2021 for DA), Jovem Pesquisador Fluminense com Vínculo em ICTS do Estado do Rio de Janeiro grant (process number E-26/2010.300/2022 for RAHD) and the Brazilian National Council of Scientific and Technological Development (CNPq), through a productivity grant (process number 308811/2021–6 for RAHD and 307007/2022–7 for MMEO). Daniel Adesse is supported by a special grant from a Federal Deputy Fund (Marcelo Freixo 2022-2023), award number IOC-008-FIO-23.
Author information
Authors and Affiliations
Contributions
MCPR: data curation, visualization, investigation, formal analysis, writing—original draft and writing—draft reviewing. GLdC.: validation, data curation, visualization, investigation, formal analysis. TNP: validation, data curation, visualization, investigation, formal analysis. DA: conceptualization, resources, investigation, validation, data curation, visualization, investigation, formal analysis, funding acquisition, project administration, supervision, writing—original draft and writing—draft reviewing. MMEO: conceptualization, resources, investigation, validation, data curation, visualization, investigation, formal analysis, funding acquisition, project administration, super-vision, writing—original draft and writing—draft reviewing. RAHD.: conceptualization, resources, investigation, validation, data curation, visualization, investigation, formal analysis, funding acquisition, project administration, supervision, writing—original draft and writing—draft reviewing. All authors contributed to the article and approved the submitted version. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Only samples of recently deceased animals were sampled under a Brazilian Institute of Environment and Renewable Natural Resources Biodiversity Authorization and Information System (SISBIO) authorization, no. 77310–5.
Consent for publication
Not Applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Peixoto-Rodrigues, M.C., da Costa, G.L., Pinto, T.N. et al. A novel report on the emerging and zoonotic neurotropic fungus Trichosporon japonicum in the brain tissue of the endangered Brazilian guitarfish (Pseudobatos horkelii) off the southeastern coast of Brazil. BMC Microbiol 23, 367 (2023). https://doi.org/10.1186/s12866-023-03128-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12866-023-03128-w