Abstract
Background
This study explores the potential litholytic activity of medicinal plant infusions, specifically Arenaria rubra, Hordeum vulgare, and Zea mays, as well as a combination of these infusions, on various types of urinary calculi. Given the traditional use of these plants in managing kidney stones and the need for alternative treatments, this investigation aims to assess their efficacy in dissolving different types of calculi.
Materials and methods
Samples representing calcium oxalate monohydrate (COM), calcium oxalate dihydrate (COD), cystine (Cy), uric acid (UA), and calcium phosphate (CP) calculi were analyzed using FTIR and optical microscopy. Herbal infusions were prepared at a concentration of 14 g/l, and the dissolution capacity was evaluated by incubating the calculi samples in the infusions for eight weeks. Microscopic examination and statistical analysis were conducted to assess the efficacy of the infusions.
Results and discussion
Limited dissolution of COM and COD calculi was observed with the infusions, consistent with their known resistance to dissolution. Significant dissolution effects were observed for Cy, UA, and CP calculi, especially with infusions of A. rubra and H. vulgare. These findings suggest the potential of plant infusions as complementary therapeutic agents for certain types of kidney stones.
Conclusion
This study highlights the promising litholytic activity of medicinal plant infusions, specifically A. rubra and H. vulgare, which indicates a significant dissolution of various urinary calculi. While further research is needed to validate these findings, integrating plant infusions, extracts, or essential oils into clinical practice could offer additional treatment options for managing kidney stones and preventing recurrence.
Similar content being viewed by others
1 Background
Urolithiasis, a condition with multifactorial origins, is on the rise not only in Algeria but also globally [1]. A study conducted in Algeria involving approximately 3989 cases of urolithiasis assessed the progression of the disease over time. The findings confirmed a consistent evolution of the disease’s characteristics, reflecting shifts in Algeria’s socio-economic landscape [1]. Epidemiological studies worldwide suggest that recurrence rates for this painful ailment can reach up to 50% [2]. Consequently, proactive measures such as diuretic therapy, phytotherapy, and dietary adjustments for predisposed individuals have gained importance [3].
Concurrently, there is a growing trend favoring alternative or complementary phytotherapy over conventional medical treatments for various ailments, including urolithiasis [4]. Traditional medicine, rooted in herbal remedies, is embraced by nearly 80% of the global population. It involves utilizing plants or their extracts to treat various conditions, including urinary stones [5].
Algeria, endowed with diverse climates due to its geographical positioning, boasts a rich phytogenetic reservoir, elevating its stature among Mediterranean nations with a longstanding tradition of herbal medicine. These natural remedies offer both preventive and curative potential [6].
Recognizing the significance of natural resources, the World Health Organization (WHO) advocates for evaluating the safety and efficacy of herbal medicines to integrate them into mainstream healthcare systems [7].
Numerous studies worldwide have explored the therapeutic effects of medicinal plants, yielding promising results and ongoing research endeavors [8, 9]. Plant extracts have primarily been studied for their efficacy in treating oxalocalcic lithiasis, as evidenced by numerous publications [10, 11]. However, there is a scarcity of research on less common lithiasis types, such as cystine, carbapatite, and uric acid stones, largely due to challenges in social care [12, 13].
Dissolution therapy, which includes various approaches such as urinary alkalinization [14], hydration, dietary modifications [15], and specific medications, provides a treatment option for certain types [16, 17]. Yet, some stones, such as cystine, pose considerable challenges and may require alternative interventions like flexible ureterorenoscopy or percutaneous nephrolithotomy [18, 19].
This study aims to analyze samples of various types of kidney stones using FTIR and an optical microscope to elucidate the morpho-constitutional chemical structure of each type. In a subsequent investigation, the study aims to examine the effects of three plant infusions commonly utilized by our population for treating urolithiasis—Arenaria rubra, Hordeum vulgare, and Zea mays—on the dissolution of all types of kidney stones, including calcium oxalate monohydrate (COM), calcium oxalate dihydrate (COD), cystine (Cy), uric acid (UA), and calcium phosphate (CP).
2 Material and methods
2.1 Urinary calculi
Three pure samples of each type of calculus (COM, COD, Cy, UA and CP) originated from a sample library of the pharmacovigilance department at EHU-ORAN in Algeria. Selection was based on attenuated total reflection (FTIR-ATR) spectroscopy in the range of 450–4000 cm−1 (Perkin Elmer) and morphological identification using an educational optical microscope (10×–40×) (IHM) at the Pharmaceutical Inorganic Laboratory of the Faculty of Medicine in Oran [20].
2.2 Medicinal herbal infusions
Through a comprehensive bibliographic investigation, we have ascertained that the predominant plant species employed by the populace for the management of nephrolithiasis include A. rubra, H. vulgare, and Z. mays’s stigma (Fig. 1) [21]. The botanical specimens were obtained from a local herbalist drug store located in the city of Oran, Algeria. The investigated plants were identified at the LRDP research laboratory of the University of Oran 1, and their authentication was conducted by the university’s plant identification and validation authority [21].
2.3 Litholytic activity
We begin by weighing kidney stone samples and then transferring them into Petri dishes. Next, using the same scale, we measure out 14 g each of powdered complete dried herbs (A. rubra and H. vulgare) and corn stigmat (Z. mays). Boiling 1 L of distilled water is poured over the herbal powder, which is left to steep for 30 min at 100 °C. After infusion, the herbal mixture is filtered using a funnel and filter paper. Subsequently, the kidney stones in the Petri dishes are completely submerged in the herbal infusions and left in contact for two weeks. The stones are then rinsed with distilled water and dried in an oven at 40 °C for 24 h [20]. The following day, their weights are recorded. This entire process is repeated for 8 weeks. Each experience was performed in triplicate (Fig. 2). We also prepared a mixture of the three infusions, with distilled water as a negative control and potassium citrate (Sigma-Aldrich, Germany) as a positive witness. The herbal infusions were retrieved for microscopic observation (magnification 40×, 10×) to search for any possible presence of crystals that have dissolved from the stones [22].
The effectiveness of the infusion was assessed by determining the rate of dissolution of calculi after exposure to the experimental environment. This was done by comparing the residual weight of the calculi after incubation with the infusion to their initial weight (Fig. 2). The percentage of dissolution was then calculated using the following formula (12, 20):
L%: is the rate of calculi dissolution; W initial and W final are the weights of the calculus before and after the incubation with the plants infusion.
2.4 Data analysis
Data were expressed as mean ± standard deviation (SD). Statistical comparisons between all groups were performed by a one-way ANOVA. The difference is considered as significant for p ≤ 0.05.
3 Results
3.1 FTIR analysis
The infrared spectra of certain lithic constituents and specific mixtures analyzed in the laboratory are presented in Fig. 3. The absorption spectrum obtained is specific to the molecular composition and the potential crystalline structure of the sample. Integrating morphological data with the results of infrared spectrophotometry has led to several observations. The identified lithic constituents are of mineral or organic origin, with the absence of iatrogenic molecules (medications).
The FTIR spectra analysis covered the range of 450–4000 cm−1 for all sample classes. COM exhibits a peak at 3427 cm−1 corresponding to O–H stretching bonds from the water molecule in the monohydrate form. A strong, sharp peak around 1613 cm−1 corresponds to the stretching vibration of the carbonyl (C=O) bonds in the oxalate ion. Another peak in the region of 1314 cm−1 corresponds to the stretching vibration of C–O bonds in the oxalate ion. The absorption peak at 511 cm−1 confirms the presence of bonds with the metal (calcium) [23].
COD exhibits a peak at 3331 cm−1 corresponding to the stretching vibration of O–H bonds from the water molecule in the monohydrate form. A sharp peak around 1613 cm−1 corresponds to the stretching vibration of the carbonyl (C=O) bonds in the oxalate ion. Peak around 1317 cm−1 corresponds to the stretching vibration of C–O bonds in the oxalate ion. Peaks in the region of 1027 cm−1 corresponds to various stretching vibrations of carbon–carbon bonds. The peak at 777 cm−1 can be attributed to the bending vibration of the C–C–O bonds within the oxalate ion. The absorption peak at 513 cm−1 confirms the presence the bonds with oxygen-metal (calcium) [24].
Cy exhibits absorption bands around 2500–2600 cm−1 may indicate the stretching vibration of the S–H bonds present in cysteine. A peak around 1700 cm−1 corresponds to the stretching vibration of the carbonyl (C=O) bonds present in the molecule. Peaks in the region of 1200–1400 cm−1 correspond to the stretching vibrations of the carbon–nitrogen (C–N) bonds. A strong peak around 500–600 cm−1 corresponds to the stretching vibration of the disulfide (S–S) bond, which is characteristic of cystine. The bands around 1500–1700 cm−1 are attributed to amide I (C=O stretching coupled with N–H bending) and amide II (N–H bending coupled with C–N stretching) vibrations, respectively [25].
UA spectrum, there is a prominent, sharp peak at around 1680 cm−1 corresponds to the stretching vibration of the carbonyl (C=O) bonds within in the molecule. A peak around 1600–1660 cm−1 corresponds to the amide I band, which arises from the C=O stretching vibration coupled with N–H bending. A peak around 1530–1560 cm−1 corresponds to the amide II band, which arises from the N–H bending coupled with C–N stretching. Peaks around 700–900 cm−1 correspond to the out-of-plane bending vibrations of the aromatic C–H bonds in the benzene ring. Various peaks in the region of 2800–3000 cm−1 correspond to stretching and bending vibrations of carbon–hydrogen (C–H) bonds in the molecule [26].
CP exhibits peaks of ν1 PO4 and ν4 PO4 at 1000 cm−1 and 560–610 cm−1 respectively. Peaks at 1430 cm−1 and 871 cm−1 corresponding to B-Type Carbonate and A-Type Carbonate, respectively, in carbonate apatite, carbonate ions replace some of the phosphate ions in the crystal lattice. ν1 OH Stretching observed as a broad band around 2900 cm−1 [27].
3.2 Microscopical morphology identification
Microscopical morphology findings elucidate physical traits such as surface roughness and crystal size, which influence stones’ reactivity to treatments (Fig. 4). Smoother surfaces may hinder dissolution compared to rougher ones, while crystal size affects susceptibility to litholytic agents. Structural defects and impurities revealed by microscopy also affect the stability and dissolution potential of stones.
COM has a smooth, spherical, mammillary, or mulberry-shaped dark-brown surface (Fig. 4). Frequent single umbilication is indicative of a papillary origin. The section has a compact, concentric structure with radial crystallization [28].
COD has a spiculated surface showing aggregated bipyramidal crystals with blunt angles and ridges, and pale yellow–brown (Fig. 4). The section is a compact unorganized crystallization, pale yellow–brown [28].
Cy has a homogeneous, microcrystalline surface that is smooth with a very fine roughness and a whitish to yellowish color (Fig. 4). The section is heterogeneous, compact, with microcrystalline whitish thin concentric layers in the periphery. Crystalline, unorganized core. Yellowish to pale brown–yellow [28].
UA has a homogeneous, crystalline, smooth, or slightly embossed surface in ocher or orange (Fig. 4). The section consists of compact concentric layers with a radiating organization around a well-defined nucleus, forming a concentric structure with radial crystallization. Ocher to red–orange [28].
CP has heterogeneous surface, which is heterogeneous, both embossed rough, surface with confluent superficial sometimes appearing porous and ranging in color from porous cream to dark brown (Fig. 4). The section is also heterogeneous, concentric, made of irregularly alternating thick, whitish, and thin, brown–yellow layers [28].
3.3 The infusion crystals microscopical identification
Crystals identification in different infusion samples were conducted using an optical microscope at a magnification of 40×, 10×. COM and COD crystals appear as envelope-shaped or elongated structures with pointed ends. Cy crystals present as hexagonal or rectangular plates. UA crystals appear as rhomboid or diamond-shaped structures with smooth edges. CP crystals appear as needle-like or prismatic structures (Fig. 5) [28].
3.4 Litholytic activity
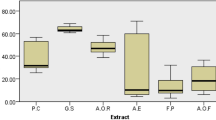
The dissolution capacity of different types of calculi by various infusions, distilled water, and citrate over 8 weeks is illustrated in (Fig. 6).
COM calculis showed a low percentage of dissolution in the three infusion plants, with a high percentage of dissolution observed with A. rubra (29%), and (25%, 28% and 27%) for H. vulgare, Z. mays and the mixture, respectively. However, it was still better than the positive control (20%). While the dissolution was better observed with distilled water (33%) (Fig. 6).
COD calculis showed a significantly lower percentage of dissolution in the three infusion plants (6.78%, 7.42%, 8.85%, 7.19%) for A. rubra, H. vulgare, Z. mays and the mixture, respectively. These percentages were lower than the positive control (9%) (Fig. 6). While the dissolution was significantly better with distilled water samples (20.14%).
Cy calculis showed a very high percentage of dissolution in the three infusion plants starting from the second week of the experiment with (85.61%, 83.00%, 84.85%) for A. rubra, Z. mays and the mixture, respectively. These values were higher than both the positive and negative controls (58% and 17.49%, respectively) (Fig. 6).
UA calculis showed a 100% dissolution in the mixture of the three infusions, which was higher than both the positive and negative controls (53% and 49.36%, respectively). The dissolution rates were also high for the other infusions separately (60.42%; 67.08%; 52.81%) for A. rubra, H. vulgare, Z. mays respectively (Fig. 6).
CP calculis showed a modest dissolution (36.75%; 33.92%; 27.79%; 30.86%) for A. rubra, H. vulgare, Z. mays and the mixture, respectively, showed higher values than both positive and negative controls (18%; 11.18%) (Fig. 6).
4 Discussion
Studying the litholytic activity of plant infusions is crucial for exploring potential natural remedies for kidney stone management. This research offers safer and more accessible treatment options, especially in regions with limited healthcare resources. This study employed two approaches: Firstly, analyzing calculus samples of different types using FTIR and optical microscopy to elucidate their morpho-constitutional chemical structure; and secondly, investigating the effects of three commonly used plant infusions (A. rubra, H. vulgare, Z. mays) on the dissolution of various kidney stone types (COM, COD, Cy, UA, CP).
Crystal identification under an optical microscope revealed characteristic morphologies of urinary calculi in various infusion samples. (COM) and (COD) crystals displayed envelope-shaped or elongated structures with pointed ends. (Cy) crystals exhibited hexagonal or rectangular plates, while (UA) crystals appeared rhomboid or diamond-shaped with smooth edges. (CP) crystals were identified as needle-like or prismatic structures. These findings provide valuable insights into the composition and potential litholytic activity of urinary calculi influenced by plant infusions.
Our results showed no significant dissolution effect on COM and COD calculi with three infusions (Table 1). We may explain this result by the fact that calcium oxalate calculi are notoriously challenging to dissolve due to their insoluble nature, high stability, and low solubility product [29]. Their dense crystal structure and tendency to form hard coatings also hinder dissolution efforts [30].
Although, for Cy calculi, our results show a significant dissolution effect of the three plants and the mixture infusions (p < 0.001) (Table 1). Arenaria rubra showed the highest dissolution effect on Cy after eight weeks of experimentation (85.61%) [31].
Furthermore, UA calculi exhibit a significant dissolution effect with H. vulgare (67.08%) (p < 0.05) and complete dissolution of calculi (100%) in the mixture (p < 0.01) (Table 1). We can explain the dissolution effect of H. vulgare infusion by its high content of certain compounds, such as phytosterols, polyphenols, and dietary fibers. These bioactive compounds may have beneficial effects on stone dissolution [32].
Regarding CP calculi, we observed that the dissolution effect was significant with all three plant infusions (p < 0.01) compared to the negative control (Table 1). The highest dissolution effect was observed with A. rubra infusion (36.75%) [33]. Studies that have been conducted and published, examining the effects of medicinal plant extracts on urinary calculi, have frequently focused on cystine lithiasis [20, 34, 35]. The Moroccan study [20] showed that the extract of the seeds of T. foenum-graecum has a better effect on the dissolution of Cy and CP stones.
Limitations in employing DRX, TEM, and MEB for characterization and incomplete extraction of plant biomolecules constrain the depth of understanding regarding litholytic activity. Nonetheless, the study still offers valuable insights into the potential therapeutic effects of the plant infusions on kidney stone dissolution.
5 Conclusion
In conclusion, this study investigated the dissolution effects of three plant infusions—A. rubra, H. vulgare, and Z. mays—along with a mixture of these infusions on various types of kidney stones. FTIR analysis provided insights into the molecular composition and crystalline structure of the stones, while microscopic examination elucidated their morphological characteristics. The results revealed that COM and COD calculi exhibited limited dissolution with the infusions, consistent with their known resistance to dissolution because of their dense and stable crystal structures. However, Cy calculi exhibited a significantly high dissolution effect with all infusions, especially with A. rubra. UA calculi showed significant dissolution, particularly with H. vulgare infusion and the combination of infusions, attributed to the bioactive compounds found in H. vulgare. CP calculi showed significant dissolution with all infusions, with the most pronounced effect seen with A. rubra infusion. These findings suggest that plant infusions, particularly from A. rubra and H. vulgare, show promise as potential therapeutic agents for certain types of kidney stones, providing a natural and complementary alternative to conventional treatments. Further experimentation will be necessary to provide empirical evidence of the beneficial effects of these tested infusions on the dissolution of such calculi. Due to the time required to observe any significant effects in vitro, it appears challenging to advocate for the use of these plants in patients for the direct dissolution of calculi in situ. However, there may still be potential benefits yet to be fully evaluated for the prevention of recurrence using these plants. The absence of standardized preparation protocols and long-term efficacy data necessitates further investigation for clinical translation and recurrence prevention. Additionally, the safety profile and potential adverse effects of prolonged use of these herbal remedies remain to be fully elucidated.
Availability of data and materials
The datasets produced and/or analyzed in this study remain the property of the authors and the university, primarily because they contain ongoing other studies. However, they can be obtained from the corresponding author upon reasonable request.
References
Sbahi K, Kacem B (2021) Retrospective epidemiological study of urolithiasis in western Algeria over a period of 50 years. Afr J Urol 27:146
Taype-Huamaní W, Ayala-García R, Rodríguez-Gonzales R, Amado-Tineo J (2020) Characteristics and evolution of patients with urinary lithiasis in emergency of a tertiary hospital. Fac Med Hum 20:608–613
Corbo J, Wang J (2019) Kidney and Ureteral Stones. Emerg Med Clin North Am. 37(4):637–648
Ridwan HS, Megantara S, Levita J (2024) Review of the phytotherapy for nephrolithiasis. JIFB 15(1):92
Wachtel-Galor S, Benzie IFF (2011) Herbal medicine: an introduction to its history, usage, regulation, current trends, and research needs. In: Benzie IFF, Wachtel-Galor S (eds) Herbal medicine: biomolecular and clinical aspects, vol 45. CRC Press/Taylor & Francis, Boca Raton (FL), pp 1249–1259
Djahafi A, Taïbi K, Abderrahim LA (2021) Aromatic and medicinal plants used in traditional medicine in the region of Tiaret, North West of Algeria. Mediterr. Bot. 42:2603–9109
The World Health Report (2000) Geneva, World Health Organisation
Villa G, Boarin M, Rosa D, Togni S, Manara DF, Bonetti L, Terzoni S (2022) Phytotherapy in urological benign disease: a systematic review. Int J Uroligical Nurs 16:174–195
Caione P, Salerno A, Collura G (2022) Phytotherapy as ancillary treatment after urinary stone lithotripsy in pediatric age. Ann Ital Chir 93(3):313–318
Bouanani S, Henchiri C, Migianu-Griffoni E, Aouf N (2010) Pharmacological and toxicological effects of paronychia argentea in experimental calcium oxalate nephrolithiasis in rats. J Ethnopharmacol 129:38–45
Belmamoun AR, Ammam A, Chadli R, Mhamdia C, Bakdi A (2022) Phytotherapy of urinary calculi: the mass reduction of calcium oxalate stones in vitro by the aqueous extract of Urtica dioica L. Int J Minor Fruits Med Aromat Plants 8(2):36–42
Saso L, Valentini G, Leone MG, Grippa E, Silvestrini B (1998) Development of an in vitro assay for the screening of substances capable of dissolving calcium oxalate crystals. Urol Int 61:210–214
Valencia Herrera AR et al (2023) Diagnostic and therapeutic approach to renal lithiasis: current progress and perspectives. J Adv Zool 44:469
Tamborino F, Cicchetti R, Mascitti M, Litterio G, Orsini A, Ferretti S, Basconi M, De Palma A, Ferro M, Marchioni M, Schips L (2024) Pathophysiology and main molecular mechanisms of urinary stone formation and recurrence. Int J Mol Sci 25(5):3075
Cicerello E, Merlo F, Maccatrozzo L (2010) Urinary alkalization for the treatment of uric acid nephrolithiasis. Arch Ital Urol Androl 82(3):145–148
Oswal M, Varghese R, Zagade T, Dhatrak C, Sharma R, Kumar D (2023) Dietary supplements and medicinal plants in urolithiasis: diet, prevention, and cure. J Pharm Pharmacol 75(6):719–745
Moussa M, Papatsoris AG, Abou Chakra M, Moussa Y (2020) Update on cystine stones: current and future concepts in treatment. Intractable Rare Dis Res 9:71–78
Lutegha M, Michael AM, Fred HJ, Moïse MV, Magloire DI, Jean S (2021) Urinary lithiasis: epidemiological, clinic and therapeutic aspects at CNHU-HKM. J Med Res 7(1):11–14
Ngo TC, Assimos DG (2007) Uric acid nephrolithiasis: recent progress and future directions. Rev Urol 9(1):17–27
Yachi L, Bennis S, Aliat Z (2018) In vitro litholytic activity of some medicinal plants on urinary stones. Afr J Urol 24:197–201
Taibi K, Abderrahim LA, Boussaid M, Taibi F, Achir M, Souana K, Benaissa T, Farhi KH, Naamani FZ, Said KN (2021) Unraveling the ethnopharmacological potential of medicinal plants used in Algerian traditional medicine for urinary diseases. Eur J Integr Med 44:101339
Ammor K, Mahjoubi F, Bousta D, Elhabbani R, Chaqroune A (2020) In vitro litholytic activity of extracts and phenolic fractions of some medicinal plants on urinary stones. Phytochemistry 9(6):468
Maruyama M, Sawada KP, Tanaka Y, Okada A, Momma K, Nakamura M, Mori R, Furukawa Y, Sugiura Y, Tajiri R, Taguchi K (2023) Quantitative analysis of calcium oxalate monohydrate and dihydrate for elucidating the formation mechanism of calcium oxalate kidney stones. PLoS ONE 18(3):282743
Shaltout AA, Dabi MM, Said DA, Allam MA, Ahmed SI (2021) EDXRF, FTIR, and XRD characterization of low calcium oxalate urinary stones collected from arid area. X-Ray Spectrom 51:214–229
Su Y, Hessou EP, Colombo E, Belletti G, Moussadik A, Lucas IT, Frochot V, Daudon M, Rouzière S, Bazin D, Li K (2022) Crystalline structures of L-cysteine and L-cystine: a combined theoretical and experimental characterization. Amino Acids 54:1123–1133
Pradhane AP, Methekar RN, Agrawal SG (2023) Investigations on melamine-based uric acid kidney stone formation and its prevention by inhibitors. Urolithiasis 51:68
Cruz-May TN, Herrera A, Rodríguez-Hernández J, Basulto-Martínez M, Flores-Tapia JP, Quintana P (2021) Structural and morphological characterization of kidney stones in patients from the Yucatan Maya population. J Mol Struct 1235(5):130267
Belhachem A, Lansari A, Azzi F, Bengueddach A, Yousfi MJ, Toumi H (2023) HANDBOOK OF UROLITHIASIS First edition Pharmacovigilance and Urologic Surgery Departments of EHU-ORAN. Lambert Academic Publishing, Saarbrucken
Taguchi K, Okada A, Unno R, Hamamoto S, Yasui T (2021) Macrophage function in calcium oxalate kidney stone formation: a systematic review of literature. Front Immunol. https://doi.org/10.3389/fimmu.2021.673690
Robinson TE, Hughes EA, Wiseman OJ, Stapley SA, Cox SC, Grover LM (2020) Hexametaphosphate as a potential therapy for the dissolution and prevention of kidney stones. R Soc Chem 8:5215–5224
Daga S, Palit V, Forster JA, Biyani CS, Joyce AD, Dimitrova AB (2021) An update on evaluation and management in cystinuria. Urology 149:70–75
Moore J, Nevo A, Salih S, Abdul-Muhsin H, Keddis M, Stern K, Humphreys M (2021) Outcomes and rates of dissolution therapy for uric acid stones. J Nephrol 35:665–669
Reddi AS (2022) Calcium, phosphorus, and magnesium disorders and kidney stones. In: Absolute nephrology review, pp 173–209
Alluru S Reddi (2022) Absolute Nephrology Review, Springer edition, pp 173–209
Hannache B, Bazin D, Boutefnouchet A, Daudon M (2012) Effet des extraits de plantes médicinales sur la dissolution des calculs rénaux de cystine in vitro: étude à l’échelle mésoscopique. Prog Urol 10:577–582
Acknowledgements
Authors would like to thank the team of Pharmacovigilance and Urological surgery departments of EHU-ORAN, and the pharmaceutical development research laboratory, University Oran 1-Algeria.
Funding
No funding was obtained for this research.
Author information
Authors and Affiliations
Contributions
AAB, SM, RL, DM and ANB carried out the study, designed and conducted all laboratory experiments; analyzed and interpreted experimental results. HT, MJY and FB supervised the study. AB, AA, WB prepared the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Since this is a retrospective and non-interventional study, it is not mandatory to seek the advice of an ethics committee according to Algerian regulations.
Consent for publication
All authors read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Belhachem, A., Makhloufi, S., Lecheheb, R. et al. Morpho-constitutional analysis of urolithiasis and in vitro litholytic evaluation of select plants against diverse renal calculi variants. Afr J Urol 30, 31 (2024). https://doi.org/10.1186/s12301-024-00434-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12301-024-00434-1