Abstract
Background
The purpose of this work is to characterize the anatomy of the intraarticular portion of the long head of the biceps tendon (long head biceps tendon) using magnetic resonance (MR) arthrography by investigating whether anatomical variants may facilitate the onset of a supraequatorial lesion (superior labral anterior to posterior, SLAP).
Materials and methods
In 482 shoulder MR arthrographies, we considered the anatomical variants of the intraarticular portion of the long head of the biceps tendon classified according to Dierickx’s arthroscopic classification; lesions of supraequatorial structures were considered in the data analysis. For each anatomical variant, correlation with SLAP and the odd ratio were statistically evaluated, using Fisher’s exact (or chi-squared) test and logistic regression analysis, respectively.
Results
In the mesotenon-type variant, the SLAP frequency was higher than expected [χ2 (df = 4) = 14.9, p = 0.005] with a higher risk of developing a type I SLAP (p = 0.0003). In the adherent-type variant, the type II SLAP frequency was higher than expected [χ2 (df = 3) = 18.1, p = 0.0004] with a higher risk of developing type II SLAP (p = 0.0001). Two cases of “split” (SPL) long head biceps tendon had III and type IV SLAP, respectively. These patients have a higher risk for type IV SLAP [odds ratio (OR) 19.562, 95% confidence interval (CI) 1.604–238.541, p = 0.001]. An increased risk of developing SLAP type II was calculated for male subjects (OR 3.479, 95% CI 1.013–11.951, p = 0.019).
Conclusions
It is possible that adherence of the long head biceps tendon to the supraspinatus more often predisposes to a lesion of the superior glenoid labrum (SLAP), in view of the close relationships between the fibrocartilage and the bicipital anchor, probably related to the limited excursion of the intraarticular long head biceps tendon.
Highlights
-
Anatomical variants of the intraarticular portion of the long head of the biceps tendon should be studied with MR arthrography.
-
Some anatomical variants are more frequently associated with supraequatorial lesions.
-
The higher frequency of lesions in the presence of certain anatomical variants is probably related to the limited excursion of the intraarticular long head biceps tendon.
Similar content being viewed by others
Introduction
Anatomical background
The upper region of the shoulder is anatomically complex; important structures participate in its stability and proper biomechanical function. Among them, the rotator interval, the coracohumeral ligament (CHL), and the superior glenohumeral ligament (SGHL) are crossed by the intraarticular portion of the long head of the biceps tendon (long head biceps tendon) humeri [1,2,3].
The CHL and SGHL form a sling-like band surrounding the long head of the biceps tendon proximal to the bicipital groove. This biceps reflection pulley plays a key role in the stability of the intraarticular biceps’ tendon [2]. The rotator interval is a complex triangular anatomic space, located in the anterosuperior aspect of the shoulder, bounded by the coracoid process at the base, superiorly by the anterior margin of the supraspinatus muscle tendon and inferiorly by the superior margin of the subscapularis muscle tendon. It plays a major role in shoulder proprioception as it is the intersection point of numerous active and passive stabilizers [2].
The importance of magnetic resonance (MR) arthrography
Evaluation of the superior zone of the shoulder is diagnostically challenging from an imaging standpoint because of the considerable number of normal variations that occur in this location and the difficulty of accurately assessing superior labral injuries. MR arthrography appears to be the best imaging technique currently available for the evaluation of the glenohumeral joint [3].
The sensitivity and specificity of MR arthrography are greater than those of magnetic resonance imaging (MRI) in terms of finding pathologies and anatomic variants of the superior region [4,5,6,7].
Preoperative MRI without contrast medium has often failed to show some lesions later found arthroscopically. The defect in lesion identification has been explained by the need for capsular distension, which makes possible better visualization of all anatomic structures and greater contrast between the glenoid labrum, capsule, capsular recess, glenohumeral ligaments, and articular surface of the rotator cuff [5, 7,8,9,10]. MR arthrography distends the joint and allows better definition of the anatomy [11, 12].
Moreover, SLAP lesions are difficult to diagnose clinically [13,14,15] and are often diagnosed using MR arthrography [14, 15] before proceeding to arthroscopic evaluation.
Pathology background
Anatomical variants of the origin of long head biceps tendon are reported with a frequency of 1.9–7.4% [2, 3], and numerous anatomical variants such as complete absence, split or Y variant, or extracapsular origin have also been described [16]. Superior labral tears from anterior to posterior (SLAP) lesions are a common pathology in overhead-throwing athletes. [17,18,19]. Snyder et al. classified SLAP lesions into four types. Type II SLAP lesions are the most common and involve detachment of both the superior labrum and the biceps anchor from the glenoid [20].
The purpose of this work is to characterize the anatomy of the intraarticular portion of the long head biceps tendon using MR arthrography by investigating whether anatomical variants may facilitate the onset of a SLAP lesion.
Materials and methods
Patients and study design
The present investigation was designed as a monocentric, nonrandomized, retrospective study based on anonymized consecutive data.
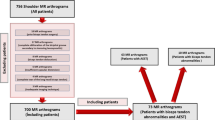
We retrospectively reviewed 482 shoulder MR arthrographies progressively performed in the radiology department of our hospital from January 2017 to December 2020.
The long head biceps tendon anatomical variants of the intraarticular portion were considered and classified according to Dierickx’s arthroscopic classification (Table 1) [21], on the basis of the relationship with the supraspinatus tendon [2]. Anatomical variants and lesions of supraequatorial structures were considered in the data analysis. Both the relative risk (OR) of developing a supraequatorial lesion (SLAP) in the presence of specific anatomical variants and the actual frequency of each of these variants in the study sample were assessed.
Exclusion criteria
The following exclusion criteria were considered: complete tendon injury of the supraspinatus muscle (91 patients), previous shoulder surgery (33 patients), presence of major motion artifacts or inadequate joint distension by the contrast medium (22 patients), adhesive capsulitis (6 patients), and complete inveterate long head biceps tendon injury (4 patients).
Variants related to intraarticular long head biceps tendon morphology and insertion variants on the humeral glenoid were not considered in our series.
MR-arthrography imaging protocol
A 1.5-Tesla MR imaging system (Achieva XR, Philips) was used with a dedicated shoulder array coil. The patients were placed supine with the shoulder in neutral position, the arm placed along the side and the thumb pointing upwards. All patients were asked to give written informed consent before the procedure. MR arthrography was performed immediately after the intraarticular injection of 20 ml of paramagnetic contrast medium (Dotarem 2.5 mmol/l, Guebet). The image acquisition protocol is summarized in Table 2.
Statistical analysis
Statistical analysis was carried out by assessing the correlation between qualitative variables organized in contingency tables, using Fisher’s exact test for 2 × 2 tables and chi-squared test for larger tables. A stepwise logistic regression was then carried out to find significant disease risk factors and their odds ratios. A significance level of 95% (p < 0.05) was applied to all statistical analyses.
Results
In total, 326 MR arthrographies were included in the study, and 156 studies were excluded:
-
91 complete tendon injuries of the supraspinatus muscle
-
33 previous shoulder surgery
-
22 major motion artifacts or inadequate joint distension
-
6 adhesive capsulitis
-
4 complete inveterate long head biceps tendon injury
The selected MR arthrographies were performed in 317 patients, (218 males and 99 females, mean age 46.5 years, age range 18–73 years), with the right shoulder being evaluated in 187 cases and the left in 139.
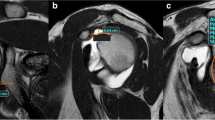
Using Fisher's and chi-squared tests, we calculated the frequency distribution of SLAP-type lesions (Fig. 1) in the different anatomical variants of the intraarticular course of the long head biceps tendon.
The images in a–d are all T1-weighted MR arthrographies, acquired in coronal-oblique section after intraarticular injection of approximately 20 ml of paramagnetic contrast agent. In particular, image a shows a type I SLAP lesion (arrowhead); image b shows a type II SLAP lesion (arrowheads) that does not reach the bicipital anchor (arrow); image c displays a type III SLAP lesion where the lesion (arrowhead) extends in a centrifugal direction alongside the bicipital anchor (curved arrow) delimiting a triangular fragment of fibrocartilage (star); image d displays a type IV SLAP lesion where the lesion (thick arrow) reaches and interrupts part of the bicipital anchor (thin arrow). c and d are reprinted with the permission of E. A. Genovese [3]
The results are summarized in Tables 3 and 4.
In the subgroup of the mesotenon-type variant (MESO) [31/326 (9.5%)], 6/31 (19.4%) had a SLAP lesion. Of these, 4/6 had a type I SLAP, 1/6 had a type II SLAP, and 1/6 had a type III SLAP. No patients with the MESO variant had a type IV SLAP lesion.
A higher-than-expected frequency was found in the group of patients with SLAP 1 and MESO-SB [χ2 (df = 4) = 14.9, p = 0.005]. The risk of developing a type I SLAP lesion was higher in the MESO-PA anatomical variant group (p = 0.0003) (Fig. 2). The OR of the subgroups could not be evaluated owing to the small number of patients for each group.
a MR arthrography, sagittal SE T1-weighted. In MESO-PA anatomical variant, long head biceps tendon is partially fused to the inferior surface of the supraspinatus tendon (arrow). Contrast agent outlines acute angle between the anterior portion of the long head biceps tendon and the inferior surface of the supraspinatus (curved arrow). b Same patients as in a. MR arthrography, coronal SE T1-weighted. MESO-PA anatomical variant is associated with SLAP lesion type I. MESO-PA anatomical variant is associated with SLAP lesion type I (arrow)
In the subgroup with adherent long head biceps tendon (ADH) [40/326 (12.3%)], 15/40 (37.5%) had a SLAP lesion. Of these, 4/15 had a type I SLAP, 5/15 had a type II SLAP, 5/15 had a type III SLAP, and 2/15 had a type IV SLAP.
Compared with expectation, we found a higher frequency of type II SLAP in patients with adherent ADH-PM long head biceps tendon [χ2 (df = 3) = 18.1, p = 0.0004]. In addition, patients with the ADH-PM long head biceps tendon variant also have an increased risk of developing type II SLAP (p = 0.0001) (Fig. 3). Again, the small number of subgroups did not allow the OR to be calculated.
a MR arthrography, sagittal SE T1-weighted. In ADH-PM anatomical variant, long head biceps tendon is partially medially to the inferior surface of the supraspinatus (arrow). b Same patients as in a. MR arthrography, coronal SE T1-weighted. ADH-PM anatomical variant is associated with SLAP lesion type II (arrow)
Both the cases of split (SPL) long head biceps tendon [2/326 (0.6%)] had SLAP lesions, types III and IV, respectively. We also found an increased risk of developing type IV SLAP in those patients who presented with a split long head biceps tendon as an anatomical variant with OR 19.562 (95% CI 1.604–238.541, p = 0.001) (Fig. 4).
a MR arthrography, T1-weighted with fat signal saturation (FAT-SAT) imaging on axial plane. SPL-DO anatomical variant (long head biceps tendon has double band) comes from supraspinatus tendon (white curved arrow) and from glenoid (white arrow). b MR arthrography, sagittal SE T1-weighted. SPL-DO anatomical variant (double beams of long head biceps tendon; black arrows). c MR-arthrography, coronal SE T1-weighted. SPL-DO anatomical variant is associated with SLAP lesion type IV (white arrow)
Among patients with SLAP type II, 88.9% were male and 11.1% were female; an increased risk of developing SLAP type II was calculated for male subjects, with OR 3.479 (95% CI 1.013–11.951, p = 0.019).
Discussion
The main result emerging from the obtained data is that, indeed, there is a correlation between the presence of specific anatomical variants of the intraarticular portion of the long head biceps tendon and the SLAP lesion. Until now, many of these conditions have been considered insignificant and not responsible for shoulder pathology.
Only a few authors have looked for a relationship between anatomic variants and upper shoulder pathologies in adults [22,23,24]. Studies in the literature show an association between distinct types of long head biceps tendon anchorage and shoulder instability secondary to post-traumatic biceps anchor injury [14]. In particular, an association between posterior anchorage and SLAP lesion is demonstrated. [14]
Many authors have described pathologies and anatomic variants of the superior region of the shoulder [1, 3, 7, 14, 21, 25,26,27,28,29,30,31,32,33,34]. However, few authors classified or analyzed the variants systematically with a large group of participants [21, 25]. Some studies have classified or analyzed the anatomical variants of long head biceps tendon [21], while others have examined the diagnostic accuracy of MRI in diagnosing pathological patterns [35]. However, these studies were mainly conducted in small groups of patients and not systematically performed.
Congenital variants of the relationship of the long head biceps tendon with the rotator cuff have been reported in the literature; some have described absence of long head biceps tendon [22, 30,31,32,33], others the intracapsular and extracapsular course of the tendon [22, 23, 36]. Anatomical variants of long head biceps tendon origin are reported to occur with a frequency of 1.9–7.4%.
Our data show that split tendon is a rare finding, found in 0.9% of cases, while the variant characterized by adherent long head biceps tendon with the supraspinatus was seen in 12.3% of cases, followed by the variant with the mesotenon, in 9.5% of cases.
Many studies have shown that these conditions are inherited and are the result of partial detachment from the mesothelium or synovial fusion with the inferior surface of the capsule [37,38,39].
With respect to the association between certain anatomic variants and the presence of SLAP lesions, in our case series, it appears that the long head biceps tendon mesotenon (MESO-PA) variant may predispose to superficial shoulder pathology; specifically, we found a higher-than-expected frequency of SLAP 1 in the group of patients with MESO-PA anatomical variant. The OR for this variant, though, could not be calculated.
To the best of our knowledge, there are no studies in the literature that can confirm or disprove this finding. The reason for this apparent association could be found in a decreased range of movement of the intraarticular portion of the long head biceps tendon.
In concordance with the literature, we found significant correlation between the adherent long head biceps tendon group and the free long head biceps tendon group for SLAP lesion [12, 13, 40, 41]. In fact, compared with expectation, we found a higher frequency of type II SLAP in patients with adherent (ADH-PM) long head biceps tendon.
This result could also be in line with the association of type I SLAPs in the presence of mesotenon-type anatomic variants. In this case, because there is an additional limitation to the range of motion of the long head biceps tendon intraarticular portion, the association is with a type II SLAP. As the degree of fixation increases, the degree of injury to the glenoid fibrocartilage seems to be higher.
Finally, we found an increased risk of developing SLAP type IV in those patients who presented with a SPL-long head biceps tendon as an anatomical variant. In this case, the presence of this anatomical variant seems to act as a risk factor itself in developing a type IV SLAP lesion.
Again, to the best of our knowledge, there are no studies in the literature that can confirm or debunk this observation. The reasons for this result would appear to belong to a different order than the previously described variants and are likely to be due mainly to biomechanical reasons similar to those leading to a greater association of SLAP in cases of posterior CLB tendon insertion described by Jakanani et al. [14].
In our case series, we did not find any cases of long head biceps tendon agenesis. Four anatomical variants, from hypoplasia of the biceps to its complete absence, have been described in the literature [31]. Other authors have described three variants of the proximal extracapsular portion of the long head biceps tendon [42]. Cases of split tendon and complete fusion with the inferior surface of the rotator cuff have been described in three cases [24, 34, 37].
The strengths of our study are the large initial sample size, the systematic use of MR arthrography, and the single-center design of the study. Other authors evaluated the sensitivity and specificity of MRI versus MR arthrography in 150 shoulders. The sensitivity and specificity values of MRI without contrast medium were respectively 83% and 100% for anterior labrum lesions, 85% and 100% for SLAP lesions, and 92% and 100% for supraspinatus tendon lesions.
Compared to arthroscopy, MR arthrography reaches sensitivity and specificity values respectively of 98% and 100% for anterior labral lesions, specifically 98% and 99% for SLAP lesions and 100% sensitivity and specificity for cuff lesions [7, 13].
A limitation of our study is the absence of systematic comparison between MR-arthrography and arthroscopy, but data in the literature consider MR arthrography the gold standard in the evaluation of the upper portion of the shoulder [5, 43].
Conclusions
Our findings suggest that the diagnosis of anatomical variants of upper shoulder structures should be performed with MR arthrography.
It is possible that adherence of the long head biceps tendon to the supraspinatus more often predisposes to a lesion of the superior glenoid labrum (SLAP), in view of the close relationships between the fibrocartilage and the bicipital anchor, probably related to the limited excursion of the intraarticular long head biceps tendon.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- SLAP:
-
Superior labral anterior to posterior
- long head biceps tendon:
-
Long head of the biceps tendon
- MESO-VI:
-
Mesotenon variant with vinculum
- MESO-SB:
-
Mesotenon variant with small band
- MESO-PU:
-
Mesotenon variant with pulley-like sling
- MESO-PA:
-
Partial mesotenon
- MESO-CO:
-
Complete mesotenon
- ADH-PM:
-
Partially medially adherent to the supraspinatus
- ADH-PL:
-
Partially laterally adherent to the supraspinatus
- ADH-CL:
-
Completely adherent to the supraspinatus attaching to the labrum
- ADH-CO:
-
Completely adherent to supraspinatus not attaching to the labrum
- SPL-DO:
-
Split biceps double origin
- SPL-RE:
-
Split biceps reversed type
- ABS:
-
Absence of tendon
References
Hunt SA, Kwon YW, Zuckerman JD (2007) The rotator interval: anatomy, pathology, and strategies for treatment. J Am Acad Orthop Surg 15:218–227. https://doi.org/10.5435/00124635-200704000-00005
Petchprapa CN, Beltran LS, Jazrawi LM et al (2010) The rotator interval: a review of anatomy, function, and normal and abnormal MRI appearance. Am J Roentgenol 195:567–576. https://doi.org/10.2214/AJR.10.4406
Genovese E, Spanò E, Castagna A et al (2013) MR-arthrography in superior instability of the shoulder: correlation with arthroscopy. Radiol Med 118:1022–1033. https://doi.org/10.1007/s11547-013-0942-y
Morag Y, Jacobson JA, Shields G et al (2005) MR arthrography of rotator interval, long head of the biceps brachii, and biceps pulley of the shoulder. Radiology 235:21–30. https://doi.org/10.1148/radiol.2351031455
Van der Woude HJ, Vanhoenacker FM (2007) MR arthrography in glenohumeral instability. JBR-BTR 90:377–383
Le Huec JC, Schaeverbeke T, Moinard M et al (1996) Traumatic tear of the rotator interval. J Shoulder Elbow Surg 5:41–46. https://doi.org/10.1016/S1058-2746(96)80029-2
Tarallo N, Morgano MC, Curti M et al (2021) Intra-articular long head of the biceps tendon: magnetic resonance-arthrography classification and review of literature. Pol J Radiol 86:e93–e101. https://doi.org/10.5114/pjr.2021.104206
Albrecht C, Tichy B, Zak L et al (2014) Influence of cell differentiation and IL-1β expression on clinical outcomes after matrix-associated chondrocyte transplantation. Am J Sports Med 42:59–69. https://doi.org/10.1177/0363546513507543
Kreitner KF, Botchen K, Rude J et al (1998) Superior labrum and labral–bicipital complex: MR imaging with pathologic–anatomic and histologic correlation. AJR Am J Roentgenol 170:599–605. https://doi.org/10.2214/ajr.170.3.9490937
Beltran J, Bencardino J, Mellado J et al (1997) MR arthrography of the shoulder: variants and pitfalls. RadioGraphics 17:1403–1412. https://doi.org/10.1148/radiographics.17.6.9397454 (discussion 1412–15)
Smith DK, Chopp TM, Aufdemorte TB et al (1996) Sublabral recess of the superior glenoid labrum: study of cadavers with conventional nonenhanced MR imaging, MR arthrography, anatomic dissection, and limited histologic examination. Radiology 201:251–256. https://doi.org/10.1148/radiology.201.1.8816553
Magee T (2009) 3-T MRI of the shoulder: is MR arthrography necessary? Am J Roentgenol 192:86–92. https://doi.org/10.2214/ajr.08.1097
Chang D, Mohana-Borges A, Borso M, Chung CB (2008) SLAP lesions: anatomy, clinical presentation, MR imaging diagnosis and characterization. Eur J Radiol 68:72–87. https://doi.org/10.1016/j.ejrad.2008.02.026
Jakanani GC, Botchu R, Rennie WJ (2012) The MR arthrographic anatomy of the biceps labral insertion and its morphological significance with labral tears in patients with shoulder instability. Eur J Radiol 81:3390–3393. https://doi.org/10.1016/j.ejrad.2012.03.009
Amin MF, Youssef AO (2012) The diagnostic value of magnetic resonance arthrography of the shoulder in detection and grading of SLAP lesions: comparison with arthroscopic findings. Eur J Radiol 81:2343–2347. https://doi.org/10.1016/j.ejrad.2011.07.006
Jeong JY, Park SM, Park YE, Yoo JC (2017) Morphological classification of anatomical variants of the intra-articular portion of the long head of the biceps brachii tendon and analysis of the incidence and the relationship with shoulder disease for each subtype. J Orthop Surg 25:2309499017742207. https://doi.org/10.1177/2309499017742207
Phornphutkul C, Tahwang S, Settakorn J (2021) Effects of type II SLAP lesion repair techniques on the vascular supply of the long head of the biceps tendon: a cadaveric injection study. J Shoulder Elbow Surg 30:772–778. https://doi.org/10.1016/j.jse.2020.07.014
Andrews JR, Carson WG, McLeod WD (1985) Glenoid labrum tears related to the long head of the biceps. Am J Sports Med 13:337–341. https://doi.org/10.1177/036354658501300508
Morgan CD, Burkhart SS, Palmeri M, Gillespie M (1998) Type II SLAP lesions: three subtypes and their relationships to superior instability and rotator cuff tears. Arthroscopy 14:553–565. https://doi.org/10.1016/s0749-8063(98)70049-0
Snyder SJ, Karzel RP, Del Pizzo W et al (1990) SLAP lesions of the shoulder. Arthroscopy 6:274–279. https://doi.org/10.1016/0749-8063(90)90056-j
Dierickx C, Ceccarelli E, Conti M et al (2009) Variations of the intra-articular portion of the long head of the biceps tendon: a classification of embryologically explained variations. J Shoulder Elbow Surg 18:556–565. https://doi.org/10.1016/j.jse.2009.03.006
Ogawa K, Naniwa T (1998) A rare variation of the biceps: a possible cause of degeneration of the rotator cuff. J Shoulder Elbow Surg 7:295–297. https://doi.org/10.1016/s1058-2746(98)90059-3
Yeh L, Pedowitz R, Kwak S et al (1999) Intracapsular origin of the long head of the biceps tendon. Skeletal Radiol 28:178–181. https://doi.org/10.1007/s002560050497
Hyman JL, Warren RF (2001) Extra-articular origin of biceps brachii. Arthroscopy 17:E29. https://doi.org/10.1053/jars.2001.24691
Kanatli U, Ozturk BY, Esen E, Bolukbasi S (2011) Intra-articular variations of the long head of the biceps tendon. Knee Surg Sports Traumatol Arthrosc 19:1576–1581. https://doi.org/10.1007/s00167-010-1384-x
Cooper DE, O’Brien SJ, Warren RF (1993) Supporting layers of the glenohumeral joint. An anatomic study. Clin Orthop. https://doi.org/10.1097/00003086-199304000-00019
Ferrari DA (1990) Capsular ligaments of the shoulder. Anatomical and functional study of the anterior superior capsule. Am J Sports Med 18:20–24. https://doi.org/10.1177/036354659001800103
Tuite MJ, Blankenbaker DG, Seifert M et al (2002) Sublabral foramen and Buford complex: inferior extent of the unattached or absent labrum in 50 patients. Radiology 223:137–142. https://doi.org/10.1148/radiol.2231010896
Vangsness CT, Jorgenson SS, Watson T, Johnson DL (1994) The origin of the long head of the biceps from the scapula and glenoid labrum. An anatomical study of 100 shoulders. J Bone Joint Surg Br 76:951–954
Franco JC, Knapp TP, Mandelbaum BR (2005) Congenital absence of the long head of the biceps tendon. A case report. J Bone Joint Surg Am 87:1584–1586. https://doi.org/10.2106/JBJS.D.02459
Ghalayini SRA, Board TN, Srinivasan MS (2007) Anatomic variations in the long head of biceps: contribution to shoulder dysfunction. Arthroscopy 23:1012–1018. https://doi.org/10.1016/j.arthro.2007.05.007
Glueck DA, Mair SD, Johnson DL (2003) Shoulder instability with absence of the long head of the biceps tendon. Arthroscopy 19:787–789. https://doi.org/10.1016/s0749-8063(03)00392-x
Mariani PP, Bellelli A, Botticella C (1997) Arthroscopic absence of the long head of the biceps tendon. Arthroscopy 13:499–501. https://doi.org/10.1016/s0749-8063(97)90130-4
Kim KC, Rhee KJ, Shin HD (2009) A long head of the biceps tendon confluent with the intra-articular rotator cuff: arthroscopic and MR arthrographic findings. Arch Orthop Trauma Surg 129:311–314. https://doi.org/10.1007/s00402-007-0559-5
Kim JY, Rhee S-M, Rhee YG (2019) Accuracy of MRI in diagnosing intra-articular pathology of the long head of the biceps tendon: results with a large cohort of patients. BMC Musculoskelet Disord 20:270. https://doi.org/10.1186/s12891-019-2654-5
MacDonald PB (1998) Congenital anomaly of the biceps tendon and anatomy within the shoulder joint. Arthroscopy 14:741–742. https://doi.org/10.1016/s0749-8063(98)70101-x
Audenaert EA, Barbaix EJ, Van Hoonacker P, Berghs BM (2008) Extraarticular variants of the long head of the biceps brachii: a reminder of embryology. J Shoulder Elbow Surg 17:114S-117S. https://doi.org/10.1016/j.jse.2007.06.014
Tena-Arregui J, Barrio-Asensio C, Puerta-Fonollá J, Murillo-González J (2005) Arthroscopic study of the shoulder joint in fetuses. Arthroscopy 21:1114–1119. https://doi.org/10.1016/j.arthro.2005.05.013
Herold T, Hente R, Zorger N et al (2003) Indirect MR-arthrography of the shoulder-value in the detection of SLAP-lesions. ROFO 175:1508–1514. https://doi.org/10.1055/s-2003-43408
Waldt S, Burkart A, Lange P et al (2004) Diagnostic performance of MR arthrography in the assessment of superior labral anteroposterior lesions of the shoulder. AJR Am J Roentgenol 182:1271–1278. https://doi.org/10.2214/ajr.182.5.1821271
Funk L, Snow M (2007) SLAP tears of the glenoid labrum in contact athletes. Clin J Sport Med 17:1–4. https://doi.org/10.1097/JSM.0b013e31802ede87
Burkart AC, Debski RE (2002) Anatomy and function of the glenohumeral ligaments in anterior shoulder instability. Clin Orthop. https://doi.org/10.1097/00003086-200207000-00005
Tuite MJ, Rutkowski A, Enright T et al (2005) Width of high signal and extension posterior to biceps tendon as signs of superior labrum anterior to posterior tears on MRI and MR arthrography. Am J Roentgenol 185:1422–1428. https://doi.org/10.2214/AJR.04.1684
Acknowledgements
Not applicable.
Funding
None declared.
Author information
Authors and Affiliations
Contributions
MC, MCM, NT, and EAG contributed equally in conception and design, acquisition of data, or analysis and interpretation of data. LC, GB, and GMC contributed in drafting the article or revising it critically for important intellectual content. EAG agrees to be accountable for all aspects of the work if questions arise related to its accuracy or integrity. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The procedure performed in the study involving human participants was conducted according to the ethical standards of the national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The approval for this specific study was obtained according to the National Policy in the matter of Privacy Act on a retrospective analysis of anonymized data; informed consent, as stated by Legge 22 Dicembre 2017 n. 219 Gazzetta Ufficiale Della Repubblica Italiana, was signed by each patient.
Ethical approval was waived by the local ethics committee of University of Insubria in view of the retrospective nature of the study, and all the procedures being performed were part of the routine care.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Calvi, M., Morgano, M.C., Tarallo, N. et al. MR arthrography: correlation between anatomic intraarticular variants of the long head of the biceps tendon (long head biceps tendon) and superior labral anterior to posterior (SLAP) lesions. J Orthop Traumatol 23, 13 (2022). https://doi.org/10.1186/s10195-022-00631-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s10195-022-00631-0