Abstract
Pituitary adenylate-cyclase-activating polypeptide (PACAP) is a 27- or 38-amino acid neuropeptide, which belongs to the vasoactive intestinal polypeptide (VIP)/glucagon/secretin family. PACAP shows particularly high homology (~ 68%) to VIP. Because of the high homology of the amino acid sequences of PACAP and VIP, these peptides share three class B-G-protein coupled receptors: the PAC1-Receptor (PAC1-R), the VPAC1-Receptor (VPAC1-R) and VPAC2-Receptor (VPAC2-R). These receptors have high homology to each other, and their high homology is utilized for these discoveries. This review provides mainly an overview of the history of the discovery of PACAP and its three receptors.
Similar content being viewed by others
Review
Introduction
Pituitary adenylate-cyclase-activating polypeptide (PACAP) is a 27- or 38-amino acid neuropeptide, which shows particularly high homology to vasoactive intestinal peptide (VIP). These peptides share two common G protein-coupled receptors (GPCRs), VPAC1-R and VPAC2-R, while PACAP also has an additional specific receptor, PAC1-R. PACAP is primarily expressed in nervous tissues, where its receptors are also widely distributed.
PACAP and its receptors are involved in diverse biological functions. Peripherally, they function in the control of anterior pituitary hormone secretion, vasodilation, adrenaline secretion, insulin secretion and immunosuppression [1]. In the central nervous system, in addition to acting as a neurotransmitter, PACAP exerts a neuroprotective effect in response to cerebral brain ischemia, Parkinson’s disease, traumatic brain injury and spinal injury [2].
More recently, we have reported that PACAP gene-deficient mice develop dry eye-like symptoms such as corneal keratinization and tear reduction, and have shown that PACAP eye drops stimulate tear secretion via an adenylyl cyclase/ cyclic adenylyl cyclase monophosphate/protein kinase A (AC/cAMP/PKA) cascade, which in turn stimulates the translocation of aquaporin 5 from the cytosol to the membrane of lacrimal acinar cells to bring about an increase in water permeability [3]. It has also been demonstrated that injection of PACAP into the footpads of mice significantly promotes sweat secretion at the injection site [4]. These reports suggest that PACAP could also prove clinically useful for the treatment of dry eye disorder and sweating disorder.
A historical table of important discoveries related to effects of PACAP and its receptors in CNS is shown in Table 1.
In this article we provide an overview of the history of the discovery of PACAP and its three receptors.
Overview of PACAP and its receptors
PACAP exists in two forms: PACAP38, which consists of 38 amino acids and PACAP27 which contains 27 amino acids at its N-terminus. These are produced by alternate processing from the PACAP precursor (prepro-PACAP), as discussed below. PACAP belongs to the VIP/glucagon/secretin family, sharing 68% homology of its amino acid sequence with VIP and 37% with secretin. Glucagon-like peptide (GLP)-1, GLP-2, growth hormone secretion hormone (GHRH), glucose-dependent insulinotropic polypeptide (GIP), and peptide histidine-methionine (PHM), which is attracting attention as a potent anti-diabetic drug, also belong to this family. Because of the high homology of the amino acid sequences of PACAP and VIP, these peptides share three class B-G-protein coupled receptors: the PAC1 receptor (PAC1-R), VPAC1 receptor (PVAC1-R) and VPAC2 receptor (VPAC2-R).
PACAP binding sites were studied using a radioreceptor assay with 125I-labelled PACAP in various tissues. The PACAP specific binding sites which were not shared VIP were termed type I, while the binding sites shared with both PACAP and VIP were termed Type II. TypeI binding sites show high affinity for PACAP38 and PACAP27 (Kd = approximately 0.5 nM) and much lower affinity for VIP (Kd > 500 nM) [5,6,7,8]. Type I binding sites are subdivided into the two subtypes named type IA and type IB. Type IA binding sites has an almost equal affinity for PACAP38, whereas type IB binding sites has a considerably greater affinity for PACAP38 than for PACAP27 [9]. Type II binding sites exhibit similar affinity for PACAP and VIP (Kd = approximately 0.5 nM) [6, 7].
According to the affinity for PACAP and VIP, the PAC1-R is classified into type 1 receptor, and VPAC1-R and VPAC2-R are classified into the type II, respectively.
Thus, VPAC1-R and VPAC2-R have comparable affinity for PACAP38, PACAP27 and VIP, whereas the affinity of PAC1-R for PACAP38 and PACAP27 is at least 1000 times greater than that for VIP (Fig. 1).
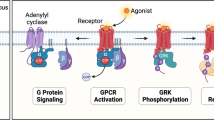
These receptors are associated with AC through cAMP, and activate PKA, which in turn can activate the mitogen-activated protein kinase (MAPK) pathway. PAC1-R is also coupled to phospholipase C, the activation of which stimulates Ca2+ mobilization and protein kinase C activation [10].
Discovery of PACAP
In the 1980s, many of the hypothalamic releasing hormones that had been isolated, including growth hormone releasing hormone (GHRH) and corticotropin releasing hormone (CRH), had been reported to stimulate pituitary AC and increase cAMP accumulation [11, 12]. To isolate novel hypophysiotropic neuropeptides, Miyata, Arimura and colleagues screened fractions from an extract of ovine hypothalamic tissue (4370 pieces; 2400 g) by monitoring their AC activity in cultured rat anterior pituitary cells. Based on this approach, they isolated a novel neuropeptide comprising 38 amino acid residues. This peptide was named PACAP38 [13], which, based on the homology (68%) of its N-terminus amino acid sequence (amino acids 1-28) with that of ovine VIP, a 28 amino acid peptide hormone, was considered to belong to the VIP/glucagon/secretin family.
As the amino acid sequence of PACAP38 has a cleavage-amidation site (Gly28-Arg29-Arg30), it was anticipated that it would be possible to generate a shorter form of the peptide. Less than a year later, this proved to be the case when PACAP27, a C-terminally truncated form of PACAP38 with 27 residues was isolated. The shorter peptide was shown to display similar biological activity in terms of AC stimulation to PACAP38, at a level about 1000 times greater than that of VIP [14]. The amino acid sequence of PACAP is identical in all mammals, and in species such as the chicken, frog, salmon, and tunicate, only a few amino acids are substituted. This suggests that PACAP is highly conserved and has remained almost unchanged during an evolutionary period of ∼700 million years, indicating that it must play an important role in physiological function [15].
At around the same time, in 1990, the cDNA encoding the precursor of PACAP38 was successfully cloned from an ovine hypothalamus cDNA library by Kimura and colleagues [16]. In this study, they used a chemically synthesized oligonucleotide coding the 1-27 amino acid sequence of PACAP38 as a probe, together with a cDNA-encoded precursor of human PACAP38 from a human testis cDNA library, using a synthetic oligonucleotide probe [16]. Another group subsequently reported cloning of the precursor of human PACAP38 and the human PACAP gene [17]. The former cDNA clone encodes a protein of 176 amino acids, currently known as “prepro-PACAP”. Prepro-PACAP (amino acids 1-176) is initially metabolized by signal proteases to generate the signal peptide (amino acids 1-25) and pro-PACAP (amino acids 26-176). Pro-PACAP is metabolized by pro-hormone convertases and carboxypeptidases to produce a small fragment (amino acids 26-79), a large PACAP-related peptide (amino acids 82-129; PRP, the physiological function of which remains unclear) and C-terminal peptides (amino acids 132-170). The C-terminal peptides (amino acids 132-170 and 132-159) are metabolized by peptidylglycine alpha-amidating monooxygenase enzymes to PACAP38 and PACAP27, respectively, which have amidated C-terminals [18, 19] (Fig. 2). In the case of PACAP 27, analysis of the genomic structure of human PACAP has revealed that it is not generated by alternative splicing mechanisms, and PACAP precursor splice variants have not been reported.
Discovery of PAC1-R
In 1993, Pisegna and Wank reported the cloning of a PACAP-selective receptor termed PACAP type I receptor (PAC1-R) which encodes a 495-amino acid protein with seven putative transmembrane domains and shows high homology with the VIP receptor, secretin receptor, GLP-1, PTH-PTHrP (parathyroid hormone and parathyroid hormone-related peptide) receptor, and calcitonin receptor. This novel receptor was screened using a rat VIP receptor cDNA as a probe by cross-hybridization from a cDNA library which was constructed from the rat pancreatic acinar carcinoma cell line AR4-2 J. The results of a binding assay with 125I-labeled PACAP and stimulation of cellular cAMP accumulation by PACAP in COS-7 cells transfected with the novel receptor cDNA were characteristic of a PACAP-specific receptor [20].
Within a short period, several other groups independently reported the existence of this receptor. For example, Hashimoto and colleagues isolated it as a PACAP receptor from a rat brain cDNA library by cross-hybridization with rat VIP receptor cDNA under low stringency conditions [21]. Using a similar approach, Hosoya and colleagues also cloned the receptor [22], as did Morrow and colleagues based on RT-PCR. The latter group first performed RT-PCR using rat anterior pituitary gland RNA as a template with a pair of degenerate oligonucleotide primers, corresponding to conserved regions in the third and seventh transmembrane domains of the rat secretin, pig calcitonin, and opossum PTH receptors. They then screened a rat olfactory bulb cDNA library with one of the PCR products as the probe, and isolated the receptor [23]. This receptor was termed the PAC1-Receptor by the International Union of Pharmacology [24] (Table 2).
In human, PAC1-Receptor was isolated from a human pituitary cDNA library. and the amino acid sequences of the human PAC1-R shows 92.5% homology with that of rat PAC1-R protein [25].
Discovery of VPAC1-R
In 1991, Ishihara and colleagues isolated the rat secretin receptor based on a direct expression cloning strategy. In this method, a CDM8 expression library from the cDNA of NG108-15 cells, a hybrid cell line of mouse N18TG2 neuroblastoma and rat C6Bu-1 glioma, was transfected into COS cells, and the cells were assayed for their ability to bind secretin [26]. In the following year, using the secretin receptor cDNA as a probe, the same group cloned a novel receptor which encoded a 459-amino acid protein for VIP from a rat lung cDNA library by cross-hybridization. This novel receptor, which exhibited 50% amino acid sequence identity with rat PAC1-R, was initially designated the VIP receptor [27].
Subsequently, Lutz and colleagues also cloned the receptor using the same methods employed in the isolation of PAC1-R [23], but termed it the VIP1 receptor [28]. Reports of this novel receptor continued thereafter, variously referring to it as the VIP-PACAP type II receptor [29], or PVR2 [30].
However it has now been termed the VPAC1-Receptor by the International Union of Pharmacology [24] (Table 2).
The human VPAC1-R has been cloned from a HT29 human colonic adenocarcinoma cell line cDNA library using rat secretin cDNA sequence as a probe. The human VPAC1-R comprises 457 amino acids, and human and rat VPAC2-R proteins ehhibt 84% amino acids identity [31].
Discovery of VPAC2-R
In 1993, Lutz and colleagues also cloned another novel VIP receptor which they referred to as the VIP2 receptor. First, they isolated a cDNA fragment from rat pituitary gland mRNA by RT-PCR using degenerate oligonucleotide primers corresponding to the third and seventh transmembrane domains of the secretin/calcitonin/parathyroid hormone family of GPCRs. Using this cDNA fragment, they then cloned the VIP2 receptor which encodes 437 amino acids protein, from a rat olfactory bulb cDNA library. [28].
In another study, Inagaki and colleagues isolated a subtype of the PACAP receptor [32]. They began by isolating the cDNA of putative receptors by RT-PCR with primers which were selected from a region of homology among GPCRs of the VIP/glucagon/secretin family from rat pancreatic islet RNA. Using the cDNA of the putative receptors as a probe, a novel receptor was then cloned by hybridization from a MIN6 cDNA library which was generated from a mouse insulin-secreting cell line. This receptor was designated PACAPR-3.
Usdin and colleagues also later reported cloning of the same receptor from a rat cerebral cortex cDNA library [33], and Rawlings and colleagues adopted the term PVR3 [30]. Other groups also independently reported the existence of the same novel receptor within a short period [34], and it was subsequently termed the VPAC2-Receptor by the International Union of Pharmacology [24] (Table 2).
The human VPAC2-Receptor cloned from a human placenta cDNA library using a combination of techniques including PCR with primer corresponding to conserved regions in the first-intracellular loop and the seventh transmembrane domains of the secretin, PTH, glucagon, and VIP receptors, and cDNA licrary screening. This receptor comprises 438 amino acids and possesses 87% sequence identity with rat VPAC2-R [35].
Distribution of PACAP and its receptors in the CNS
A few years after isolation of PACAP, the distribution of PACAP and its receptor has been thoroughly studied in the rat brain.
In the rat barin, the precursor of the PACAP mRNA is present in the olfactory bulb, the cingulate cortex and the cortex extract in cerebral cortex, the hippocampus, the thalamus, and the hypothalamus, and the highest concentrations of PACAP are located in hypothalamic area [36, 37].
The PAC1-Receptor mRNA are observed over wide area such as the olfactory bulb, the cingulate cortex, piriform cortex, the dentate gyrus of the hippocampus, the paraventricular nucleus, the ventromedial nucleus and the supraoptic nucleus of the hypothalamus, the cerebellum, the lateral paragigantocellular, the pontine nuclei and the vagal complex of the brainstem, and the spinal cord [38,39,40,41].
In these areas, the localization of PAC1-R mRNA correlates well with the distribution of typeI (PACAP-specific) binding sites which examined by autoradiography [9, 41,42,43,44].
The VPAC1-R mRNA is present mainly in the cerebral cortex and the hippocampus, whereas VPAC2-R mRNA is predominantly expressed in the central nucleus of amygdala, the hippocampus, thalamus, hypothalamus, and the pontine nuclei of brainstem [27, 33, 45].
The distribution of TypeII (PACAP/VIP) binding sites is more restricted than that of typeI binding sites, and are located in the olfactory bulb, the cerebral cortex, the dentate gyrus of the hippocampus, thalamus, the locus coeruleus of the pons, area postrema, and the spinal cord [46,47,48,49,50]. For more details on distribution, see the excellent review in [1].
As described above, the distribution of PACAP and its receptor is well established in the rodents, little information is available regarding its distribution of human brain. Concentrations of pituitary adenylate cyclase activating polypeptide (PACAP) in microdissected human brain regions were measured by radioimmunoassay. The highest concentrations of PACAP were observed in the dorsal vagal complex, the bed nucleus of the stria terminalis, the median eminence-pituitary stalk, and in the periventricular and paraventricular hypothalamic nuclei. In addition, they were found in some hypothalamic (supraoptic and ventromedial), preoptic and brainstem nuclei. High concentrations were also measured in the septum pellucidum, periaqueductal and spinal gray matters, the motor facial, and in the spinal nucleus of the trigeminal nerve [51].
About the PACAP receptors, the distribution in the human cerebellum during development was reported [52].
In human fetuses, PAC1-R mRNA is associated with the external granule cell layer (EGL), a germinative neuroepithelium, and with the internal granule cell layer. The distribution pattern of VPAC1-R mRNA during development was very similar to that of PAC1-R mRNA, whereas VPAC2-R mRNA was visualized only in adults. The distribution of 125I-PACAP27 binding sites was consistent with that of PAC1-R and VPAC1-R mRNA [52].
Conclusion
This review introduce an overview of the history of the discovery of PACAP and its three receptors. PACAP and its receptors are involved in diverse biological functions. An understanding of their signaling pathways may lead to the development of new therapeutic drugs.
References
Vaudry D, Falluel-Morel A, Bourgault S, Basille M, Burel D, Wurtz O, Fournier A, Chow BK, Hashimoto H, Galas L, Vaudry H (2009) Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol Rev 61:283–357
Shioda S, Nakamachi T (2015) PACAP as a neuroprotective factor in ischemic neuronal injuries. Peptides 72:202–207
Nakamachi T, Ohtaki H, Seki T, Yofu S, Kagami N, Hashimoto H, Shintani N, Baba A, Mark L, Lanekoff I, Kiss P, Farkas J, Reglodi D, Shioda S (2016) PACAP suppresses dry eye signs by stimulating tear secretion. Nat Commun 7:12034
Sasaki S, Watanabe J, Ohtaki H, Matsumoto M, Murai N, Nakamachi T, Hannibal J, Fahrenkrug J, Hashimoto H, Watanabe H, Sueki H, Honda K, Miyazaki A, Shioda S (2017) Pituitary adenylate cyclase-activating polypeptide promotes eccrine gland sweat secretion. Br J Dermatol 176:413–422
Cauvin A, Buscail L, Gourlet P, De Neef P, Gossen D, Arimura A, Miyata A, Coy DH, Robberecht P, Christophe J (1990) The novel VIP-like hypothalamic polypeptide PACAP interacts with high affinity receptors in the human neuroblastoma cell line NB-OK. Peptides 11:773–777
Gottschall PE, Tatsuno I, Miyata A, Arimura A (1990) Characterization and distribution of binding sites for the hypothalamic peptide, pituitary adenylate cyclase-activating polypeptide. Endocrinology 127:272–277
Lam HC, Takahashi K, Ghatei MA, Kanse SM, Polak JM, Bloom SR (1990) Binding sites of a novel neuropeptide pituitary-adenylate-cyclase-activating polypeptide in the rat brain and lung. Eur J Biochem 193:725–729
Suda K, Smith DM, Ghatei MA, Bloom SR (1992) Investigation of the interaction of VIP binding sites with VIP and PACAP in human brain. Neurosci Lett 137:19–23
Cauvin A, Robberecht P, De Neef P, Gourlet P, Vandermeers A, Vandermeers-Piret MC, Christophe J (1991) Properties and distribution of receptors for pituitary adenylate cyclase activating peptide (PACAP) in rat brain and spinal cord. Regul Pept 35:161–173
Shioda S, Takenoya F, Wada N, Hirabayashi T, Seki T, Nakamachi T (2016) Pleiotropic and retinoprotective functions of PACAP. Anat Sci Int 91:313–324
Vale W, Spiess J, Rivier C, Rivier J (1981) Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science 213:1394–1397
Guillemin R, Brazeau P, Böhlen P, Esch F, Ling N, Wehrenberg WB (1982) Growth hormone-releasing factor from a human pancreatic tumor that caused acromegaly. Science 218:585–587
Miyata A, Arimura A, Dahl RR, Minamino N, Uehara A, Jiang L, Culler MD, Coy DH (1989) Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem Biophys Res Commun 161:567–574
Miyata A, Jiang L, Dahl RD, Kitada C, Kubo K, Fujino M, Minamino N, Arimura A (1990) Isolation of a neuropeptide corresponding to the N-terminal 27 residues of the pituitary adenylate cyclase activating polypeptide with 38 residues (PACAP38). Biochem Biophys Res Commun 170:643–648
Arimura A (1998) Perspectives on pituitary adenylate cyclase activating polypeptide (PACAP) in the neuroendocrine, endocrine, and nervous systems. Jpn J Physiol 48:301–331
Kimura C, Ohkubo S, Ogi K, Hosoya M, Itoh Y, Onda H, Miyata A, Jiang L, Dahl RR, Stibbs HH, Arimura A, Fujino M (1990) A novel peptide which stimulates adenylate cyclase: molecular cloning and characterization of the ovine and human cDNAs. Biochem Biophys Res Commun 166:81–89
Hosoya M, Kimura C, Ogi K, Ohkubo S, Miyamoto Y, Kugoh H, Shimizu M, Onda H, Oshimura M, Arimura A, Fujino M (1992) Structure of the human pituitary adenylate cyclase activating polypeptide (PACAP) gene. Biochim Biophys Acta 1129:199–206
Li M, Nakayama K, Shuto Y, Somogyvari-Vigh A, Arimura A (1998) Testis-specific prohormone convertase PC4 processes the precursor of pituitary adenylate cyclase-activating polypeptide (PACAP). Peptides 19:259–268
Li M, Shuto Y, Somogyvári-Vigh A, Arimura A (1999) Prohormone convertases 1 and 2 process ProPACAP and generate matured, bioactive PACAP38 and PACAP27 in transfected rat pituitary GH4C1 cells. Neuroendocrinology 69:217–226
Pisegna JR, Wank SA (1993) Molecular cloning and functional expression of the pituitary adenylate cyclase-activating polypeptide type I receptor. Proc Natl Acad Sci U S A 90:6345–6349
Hashimoto H, Ishihara T, Shigemoto R, Mori K, Nagata S (1993) Molecular cloning and tissue distribution of a receptor for pituitary adenylate cyclase-activating polypeptide. Neuron 11:333–342
Hosoya M, Onda H, Ogi K, Masuda Y, Miyamoto Y, Ohtaki T, Okazaki H, Arimura A, Fujino M (1993) Molecular cloning and functional expression of rat cDNAs encoding the receptor for pituitary adenylate cyclase activating polypeptide (PACAP). Biochem Biophys Res Commun 194:133–143
Morrow JA, Lutz EM, West KM, Fink G, Harmar AJ (1993) Molecular cloning and expression of a cDNA encoding a receptor for pituitary adenylate cyclase activating polypeptide (PACAP). FEBS Lett 329:99–105
Harmar AJ, Arimura A, Gozes I, Journot L, Laburthe M, Pisegna JR, Rawlings SR, Robberecht P, Said SI, Sreedharan SP, Wank SA, Waschek JA (1998) International Union of Pharmacology. XVIII. Nomenclature of receptors for vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide. Pharmacol Rev 50:265–270
Ogi K, Miyamoto Y, Masuda Y, Habata Y, Hosoya M, Ohtaki T, Masuo Y, Onda H, Fujino M (1993) Molecular cloning and functional expression of a cDNA encoding a human pituitary adenylate cyclase activating polypeptide receptor. Biochem Biophys Res Commun 196:1511–1521
Ishihara T, Nakamura S, Kaziro Y, Takahashi T, Takahashi K, Nagata S (1991) Molecular cloning and expression of a cDNA encoding the secretin receptor. EMBO J 10:1635–1641
Ishihara T, Shigemoto R, Mori K, Takahashi K, Nagata S (1992) Functional expression and tissue distribution of a novel receptor for vasoactive intestinal polypeptide. Neuron 8:811–819
Lutz EM, Sheward WJ, West KM, Morrow JA, Fink G, Harmar AJ (1993) The VIP2 receptor: molecular characterisation of a cDNA encoding a novel receptor for vasoactive intestinal peptide. FEBS Lett 334:3–8
Ciccarelli E, Vilardaga JP, De Neef P, Di Paolo E, Waelbroeck M, Bollen A, Robberecht P (1994) Properties of the VIP-PACAP type II receptor stably expressed in CHO cells. Regul Pept 54:397–407
Rawlings SR, Piuz I, Schlegel W, Bockaert J, Journot L (1995) Differential expression of pituitary adenylate cyclase-activating polypeptide/vasoactive intestinal polypeptide receptor subtypes in clonal pituitary somatotrophs and gonadotrophs. Endocrinology 136:2088–2098
Sreedharan SP, Patel DR, Huang JX, Goetzl EJ (1993) Cloning and functional expression of a human neuroendocrine vasoactive intestinal peptide receptor. Biochem Biophys Res Commun 193:546–553
Inagaki N, Yoshida H, Mizuta M, Mizuno N, Fujii Y, Gonoi T, Miyazaki J, Seino S (1994) Cloning and functional characterization of a third pituitary adenylate cyclase-activating polypeptide receptor subtype expressed in insulin-secreting cells. Proc Natl Acad Sci U S A 91:2679–2683
Usdin TB, Bonner TI, Mezey E (1994) Two receptors for vasoactive intestinal polypeptide with similar specificity and complementary distributions. Endocrinology 135:2662–2680
Svoboda M, Tastenoy M, Van Rampelbergh J, Goossens JF, De Neef P, Waelbroeck M, Robberecht P (1994) Molecular cloning and functional characterization of a human VIP receptor from SUP-T1 lymphoblasts. Biochem Biophys Res Commun 205:1617–1624
Adamou JE, Aiyar N, Van Horn S, Elshourbagy NA (1995) Cloning and functional characterization of the human vasoactive intestinal peptide (VIP)-2 receptor. Biochem Biophys Res Commun 209:385–392
Arimura A, Somogyvári-Vigh A, Miyata A, Mizuno K, Coy DH, Kitada C (1991) Tissue distribution of PACAP as determined by RIA: highly abundant in the rat brain and testes. Endocrinology 129:2787–2789
Ghatei MA, Takahashi K, Suzuki Y, Gardiner J, Jones PM, Bloom SR (1993) Distribution, molecular characterization of pituitary adenylate cyclase-activating polypeptide and its precursor encoding messenger RNA in human and rat tissues. J Endocrinol 136:159–166
Hashimoto H, Nogi H, Mori K, Ohishi H, Shigemoto R, Yamamoto K, Matsuda T, Mizuno N, Nagata S, Baba A (1996) Distribution of the mRNA for a pituitary adenylate cyclase-activating polypeptide receptor in the rat brain: an in situ hybridization study. J Comp Neurol 371:567–577
Nomura M, Ueta Y, Serino R, Kabashima N, Shibuya I, Yamashita H (1996) PACAP type I receptor gene expression in the paraventricular and supraoptic nuclei of rats. Neuroreport 8:67–70
Shioda S, Nakai Y, Nakajo S, Nakaya K, Arimura A (1996) Localization of pituitary adenylate cyclase-activating polypeptide and its type I receptors in the rat ovary: immunohistochemistry and in situ hybridization. Ann N Y Acad Sci 805:677–683
Shioda S, Shuto Y, Somogyvari-Vigh A, Legradi G, Onda H, Coy DH, Nakajo S, Arimura A (1997a) Localization and gene expression of the receptor for pituitary adenylate cyclase-activating polypeptide in the rat brain. Neurosci Res 28:345–354
Masuo Y, Ohtaki T, Masuda Y, Nagai Y, Suno M, Tsuda M, Fujino M (1991) Autoradiographic distribution of pituitary adenylate cyclase activating polypeptide (PACAP) binding sites in the rat brain. Neurosci Lett 126:103–106
Masuo Y, Ohtaki T, Masuda Y, Tsuda M, Fujino M (1992) Binding sites for pituitary adenylate cyclase activating polypeptide (PACAP): comparison with vasoactive intestinal polypeptide (VIP) binding site localization in rat brain sections. Brain Res 575:113–123
Basille M, Gonzalez BJ, Leroux P, Jeandel L, Fournier A, Vaudry H (1993) Localization and characterization of PACAP receptors in the rat cerebellum during development: evidence for a stimulatory effect of PACAP on immature cerebellar granule cells. Neuroscience 57:329–338
Sheward WJ, Lutz EM, Harmar AJ (1995) The distribution of vasoactive intestinal peptide2 receptor messenger RNA in the rat brain and pituitary gland as assessed by in situ hybridization. Neuroscience 67:409–418
Besson J, Dussaillant M, Marie JC, Rostene W, Rosselin G (1984) In vitro autoradiographic localization of vasoactive intestinal peptide (VIP) binding sites in the rat central nervous system. Peptides 5:339–340
Besson J, Sarrieau A, Vial M, Marie JC, Rosselin G, Rostene W (1986) Characterization and autoradiographic distribution of vasoactive intestinal peptide binding sites in the rat central nervous system. Brain Res 398:329–336
Martin JL, Dietl MM, Hof PR, Palacios JM, Magistretti PJ (1987) Autoradiographic mapping of [mono[125I]iodo-Tyr10, MetO17]vasoactive intestinal peptide binding sites in the rat brain. Neuroscience 23:539–565
Yashpal K, Sarrieau A, Quirion R (1991) [125I]vasoactive intestinal polypeptide binding sites: quantitative autoradiographic distribution in the rat spinal cord. J Chem Neuroanat 4:439–446
Vertongen P, Schiffmann SN, Gourlet P, Robberecht P (1998) Autoradiographic visualization of the receptor subclasses for vasoactive intestinal polypeptide (VIP) in rat brain. Ann N Y Acad Sci 865:412–415
Palkovits M, Somogyvári-Vigh A, Arimura A (1995) Concentrations of pituitary adenylate cyclase activating polypeptide (PACAP) in human brain nuclei. Brain Res 699:116–120
Basille M, Cartier D, Vaudry D, Lihrmann I, Fournier A, Freger P, Gallo-Payet N, Vaudry H, Gonzalez B (2006) Localization and characterization of pituitary adenylate cyclase-activating polypeptide receptors in the human cerebellum during development. J Comp Neurol 496:468–478
Morley JE, Horowitz M, Morley PM, Flood JF (1992) Pituitary adenylate cyclase activating polypeptide (PACAP) reduces food intake in mice. Peptides 13:1133–1135
Murase T, Kondo K, Otake K, Oiso Y (1993) Pituitary adenylate cyclase-activating polypeptide stimulates arginine vasopressin release in conscious rats. Neuroendocrinology 57:1092–1096
Puig de Parada M, Parada MA, Hernández L (1995) Dipsogenic effect of pituitary adenylate cyclase activating polypeptide (PACAP38) injected into the lateral hypothalamus. Brain Res 696:254–257
Li S, Grinevich V, Fournier A, Pelletier G (1996) Effects of pituitary adenylate cyclase-activating polypeptide (PACAP) on gonadotropin-releasing hormone and somatostatin gene expression in the rat brain. Brain Res Mol Brain Res 41:157–162
Anderson ST, Sawangjaroen K, Curlewis JD (1996) Pituitary adenylate cyclase-activating polypeptide acts within the medial basal hypothalamus to inhibit prolactin and luteinizing hormone secretion. Endocrinology 137:3424–3429
Cavallaro S, Copani A, D'Agata V, Musco S, Petralia S, Ventra C, Stivala F, Travali S, Canonico PL (1996) Pituitary adenylate cyclase activating polypeptide prevents apoptosis in cultured cerebellar granule neurons. Mol Pharmacol 50:60–66
Chang JY, Korolev VV, Wang JZ (1996) Cyclic AMP and pituitary adenylate cyclase-activating polypeptide (PACAP) prevent programmed cell death of cultured rat cerebellar granule cells. Neurosci Lett 206:181–184
Uchida D, Arimura A, Somogyvári-Vigh A, Shioda S, Banks WA (1996) Prevention of ischemia-induced death of hippocampal neurons by pituitary adenylate cyclase activating polypeptide. Brain Res 736:280–286
Cagampang FR, Piggins HD, Sheward WJ, Harmar AJ, Coen CW (1998) Circadian changes in PACAP type 1 (PAC1) receptor mRNA in the rat suprachiasmatic and supraoptic nuclei. Brain Res 813:218–222
Sauvage M, Brabet P, Holsboer F, Bockaert J, Steckler T (2000) Mild deficits in mice lacking pituitary adenylate cyclase-activating polypeptide receptor type 1 (PAC1) performing on memory tasks. Brain Res Mol Brain Res 84:79–89
Telegdy G, Kokavszky K (2000) The action of pituitary adenylate cyclase activating polypeptide (PACAP) on passive avoidance learning. The role of transmitters. Brain Res 874:194–199
Gray SL, Cummings KJ, Jirik FR, Sherwood NM (2001) Targeted disruption of the pituitary adenylate cyclase-activating polypeptide gene results in early postnatal death associated with dysfunction of lipid and carbohydrate metabolism. Mol Endocrinol 15:1739–1747
Hashimoto H, Shintani N, Tanaka K, Mori W, Hirose M, Matsuda T, Sakaue M, Miyazaki J, Niwa H, Tashiro F, Yamamoto K, Koga K, Tomimoto S, Kunugi A, Suetake S, Baba A (2001) Altered psychomotor behaviors in mice lacking pituitary adenylate cyclase-activating polypeptide (PACAP). Proc Natl Acad Sci U S A 98:13355–13360
Hashimoto H, Kunugi A, Arakawa N, Shintani N, Fujita T, Kasai A, Kawaguchi C, Morita Y, Hirose M, Sakai Y, Baba A (2003) Biochem Biophys Res Commun 311:337–343
Cazillis M, Gonzalez BJ, Billardon C, Lombet A, Fraichard A, Samarut J, Gressens P, Vaudry H, Rostène W (2004) VIP and PACAP induce selective neuronal differentiation of mouse embryonic stem cells. Eur J Neurosci 19:798–808
Freson K, Hashimoto H, Thys C, Wittevrongel C, Danloy S, Morita Y, Shintani N, Tomiyama Y, Vermylen J, Hoylaerts MF, Baba A, Van Geet C (2004) The pituitary adenylate cyclase-activating polypeptide is a physiological inhibitor of platelet activation. J Clin Invest 113:905–912
Hirose M, Hashimoto H, Iga J, Shintani N, Nakanishi M, Arakawa N, Shimada T, Baba A (2006) Inhibition of self-renewal and induction of neural differentiation by PACAP in neural progenitor cells. Ann N Y Acad Sci 1070:342–347
Hashimoto R, Hashimoto H, Shintani N, Chiba S, Hattori S, Okada T, Nakajima M, Tanaka K, Kawagishi N, Nemoto K, Mori T, Ohnishi T, Noguchi H, Hori H, Suzuki T, Iwata N, Ozaki N, Nakabayashi T, Saitoh O, Kosuga A, Tatsumi M, Kamijima K, Weinberger DR, Kunugi H, Baba A (2007) Pituitary adenylate cyclase-activating polypeptide is associated with schizophrenia. Mol Psychiatry 12(11):1026–1032
Hashimoto R, Hashimoto H, Shintani N, Ohi K, Hori H, Saitoh O, Kosuga A, Tatsumi M, Iwata N, Ozaki N, Kamijima K, Baba A, Takeda M, Kunugi H (2010) Possible association between the pituitary adenylate cyclase-activating polypeptide (PACAP) gene and major depressive disorder. Neurosci Lett 468:300–302
Ressler KJ, Mercer KB, Bradley B, Jovanovic T, Mahan A, Kerley K, Norrholm SD, Kilaru V, Smith AK, Myers AJ, Ramirez M, Engel A, Hammack SE, Toufexis D, Braas KM, Binder EB, May V (2011) Post-traumatic stress disorder is associated with PACAP and the PAC1-receptor. Nature 470:492–497
Vacic V, McCarthy S, Malhotra D, Murray F, Chou HH, Peoples A, Makarov V, Yoon S, Bhandari A, Corominas R, Iakoucheva LM, Krastoshevsky O, Krause V, Larach-Walters V, Welsh DK, Craig D, Kelsoe JR, Gershon ES, Leal SM, Dell Aquila M, Morris DW, Gill M, Corvin A, Insel PA, McClellan J, King MC, Karayiorgou M, Levy DL, DeLisi LE, Sebat J (2011) Duplications of the neuropeptide receptor gene VIPR2 confer significant risk for schizophrenia. Nature 471:499–503
Miyamoto K, Tsumuraya T, Ohtaki H, Dohi K, Satoh K, Xu Z, Tanaka S, Murai N, Watanabe J, Sugiyama K, Aruga T, Shioda S (2014) PACAP38 suppresses cortical damage in mice with traumatic brain injury by enhancing antioxidant activity. J Mol Neurosci 54:370–379
Acknowledgements
This work was supported by Japan Society for the Promotion of Science (JSPS).
Funding
This work was supported by JSPS KAKENHI Grant Number JP15K15670, 16H02684 and 17 K11597.
Author information
Authors and Affiliations
Contributions
All authors designed the review and drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Hirabayashi, T., Nakamachi, T. & Shioda, S. Discovery of PACAP and its receptors in the brain. J Headache Pain 19, 28 (2018). https://doi.org/10.1186/s10194-018-0855-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s10194-018-0855-1