Abstract
Background
Calcific aortic valve disease (CAVD) is the second leading cause of adult heart diseases. The purpose of this study is to investigate whether miR-101-3p plays a role in the human aortic valve interstitial cells (HAVICs) calcification and the underlying mechanisms.
Methods
Small RNA deep sequencing and qPCR analysis were used to determine changes in microRNA expression in calcified human aortic valves.
Results
The data showed that miR-101-3p levels were increased in the calcified human aortic valves. Using cultured primary HAVICs, we demonstrated that the miR-101-3p mimic promoted calcification and upregulated the osteogenesis pathway, while anti-miR-101-3p inhibited osteogenic differentiation and prevented calcification in HAVICs treated with the osteogenic conditioned medium. Mechanistically, miR-101-3p directly targeted cadherin-11 (CDH11) and Sry-related high-mobility-group box 9 (SOX9), key factors in the regulation of chondrogenesis and osteogenesis. Both CDH11 and SOX9 expressions were downregulated in the calcified human HAVICs. Inhibition of miR-101-3p restored expression of CDH11, SOX9 and ASPN and prevented osteogenesis in HAVICs under the calcific condition.
Conclusion
miR-101-3p plays an important role in HAVIC calcification through regulation of CDH11/SOX9 expression. The finding is important as it reveals that miR-1013p may be a potential therapeutic target for calcific aortic valve disease.
Similar content being viewed by others
Introduction
Calcific aortic valve disease (CAVD) is a major health problem and the second leading cause of adult heart diseases, especially among the elderly (Freeman and Otto 2005; Baumgartner 2005; Lindroos et al. 1993). More than just a result of degenerative processes, CAVD is now considered to be a complex and active process characterized by multiple pathologic changes (Liu et al. 2007; Miller et al. 2011; Rajamannan et al. 2011). The aortic valve interstitial cell (AVIC) is a fibroblast-like cell type that is multipotent and dominant within the aortic valve to maintain normal valve structure and function throughout life (Monzack and Masters 2011). Aortic valve calcification is believed to involve the differentiation of VICs into either a myofibroblastic or an osteoblast-like phenotype during valve repair and remodeling (Monzack and Masters 2011). Physiological interventions to maintain the balance of VIC differentiation, especially under pathologic stimulations, may thus be beneficial in the prevention or treatment of CAVD. However, due to a lack of therapeutic targets for effective interventions, several medical treatments investigated for slowing or reversing the pathogenic process in CAVD have proven ineffective in clinical trials (Chan et al. 2010; Rossebo et al. 2008; Cowell et al. 2005). Until recently, there is still no medical therapy to prevent or reverse the disease progression; the surgical replacement or implantation of the aortic valve is still the only available treatment and is not suitable for all patients.
Since the first microRNA (miRNA) lin-4 was discovered in 1993 (Lee et al. 1993), miRNAs have been considered to provide an additional level of gene regulation beyond that of transcription factors. miRNAs are a class of small, non-coding RNA molecules that regulate gene expression post-transcriptionally in eukaryotic cells (Bartel 2004; Ambros 2004; Lim et al. 2003). miRNAs are approximately 22 nucleotide, single-stranded RNAs that silence target mRNAs in order to induce target mRNA degradation or translational repression by complementary base-pair binding to the 3′ untranslated regions (3′UTR) (Bartel 2009; Tay et al. 2008). Numerous studies have confirmed that miRNA dysregulation plays important roles in various diseases, such as cancer, cardiovascular diseases, and diabetes, thereby potentiating significant diagnostic and therapeutic approaches (Rossbach 2010; O'Connell et al. 2010; Erson and Petty 2008; Soifer et al. 2007). For example, miRNA mimics and molecules targeted at miRNAs (antimiRs) have shown promise in preclinical development (Rupaimoole and Slack 2017) and over 100 clinical trials treating various diseases (Rupaimoole and Slack 2017; Dai et al. 2015). Several miRNAs have already been found to have important functions in osteo-/chondro-genesis and cartilage/bone formation, such as miR-140 (Le et al. 2013) and miR-101 (Dai et al. 2012, 2015). Recent studies demonstrate that miR-101 plays an active role in the regulation of extracellular matrix (ECM) remodeling including fibrosis (Zhao et al. 2015; Pan et al. 2012), osteoclast differentiation (Lee et al. 2011) and apoptosis (Wu et al. 2015). However, very few studies about the regulatory functions of miRNAs in CAVD have been conducted (Zhang et al. 2014).
miR-101 exacerbated chondrocyte ECM degradation by directly targeting and regulating SOX9 expression, and silencing of miR-101 prevented cartilage degradation in a rat osteoarthritis (OA) model (Dai et al. 2012, 2015). SOX9 is a transcription factor belonging to the Sry-related high-mobility-group box (SOX) protein family (Lefebvre et al. 2007). It is essential for chondrogenesis of MSCs (Guerit et al. 2013; Cairns, et al. 2012; Akiyama 2008), since it is the key transcription factor for BMP2 induced chondrogenesis (Pan et al. 2008). SOX9 is also reported to inhibit the transactivation of Runt-related transcription factor 2 (Runx2) (Cheng and Genever 2010; Yamashita et al. 2009; Zhou et al. 2006), a key transcription factor for osteogenesis, endochondral ossification and aortic valve calcification (Miller et al. 2011; Bruderer et al. 2014; Chen et al. 2014; Towler 2013). Therefore, SOX9 is considered to promote chondrogenesis and prevent osteogenesis. Since both osteogenic and chondrogenic ossification (or bone formation) are present in CAVD (Rajamannan et al. 2011; Mohler et al. 2001), reduced SOX9 promotes aortic valve calcification (Peacock et al. 2010; Lincoln et al. 2007), whereas SOX9 overexpression induces chondrogenesis, which is regulated by the Wnt/β-catenin signaling (Fang et al. 2014).
Cell–cell adhesion generated by cadherins plays an important role in multiple aspects of cellular behavior including proliferation, differentiation, apoptosis and the maintenance of tissue integrity (Cavallaro and Dejana 2011; Harris and Tepass 2010). Cadherin-11 (CDH11) is a cell–cell adhesion protein that regulates the differentiation of mesenchymal cells into the osteo- and chondro-lineages (Kii et al. 2004). CDH11 plays an important role in the aortic valve maturation through coordinating cellular migration and extracellular matrix remodeling (Bowen et al. 2015; Zhou et al. 2013); whereas the dysregulation of CDH11 impairs the aortic valve (AV) structural integrity and promotes calcific nodule development on the AVs or in AVICs (Bowen et al. 2015; Sung et al. 2016; Hutcheson et al. 2013).
In this study, we identified a mechanism by which miR-101-3p contributes to human aortic valve interstitial cells (HAVICs) calcification via direct downregulation of SOX9 and CDH11 and activation of osteogenesis. We demonstrated that the expression of miR-101-3p is significantly increased in calcified human aortic valves, suggesting that it is a potential regulator of osteogenesis/chondrogenesis in HAVICs. Using mimic and anti-miR of miR-101-3p, we found that the miR-101-3p mimic promotes HAVICs calcification, but the inhibition of miR-101-3p efficiently protects HAVICs from calcification even in the osteogenic-conditioned medium. We determined that miR-101-3p directly targets CDH11 and SOX9 expression, and that inhibition of miR-101 prevents osteogenic differentiation in HAVICs by restoring SOX9 and CDH11 expressions. These results suggest that inhibition of miR-101 is a potential therapeutic strategy for CAVD.
Materials and methods
Expended methods can be found in Additional file 1.
RNA isolation from human aortic valves, small RNA deep sequencing and qPCR analysis
RNA isolation, small RNA sequencing and target prediction and qPCR were performed as we described recently (Chen et al. 2021a, 2022; Chen and Sun 2019). See Additional file 1: Methods.
Isolation and culture of primary human aortic valve interstitial cells
Human aortic valve interstitial cells (HAVICs) were isolated as described in our previous study (Chen et al. 2016; Song et al. 2015).
miRNA/siRNA transfection and in vitro calcification of HAVICs
The miRNA/siRNA transfection procedure was performed as we described previously (Chen et al. 2021a; Wang et al. 2021) See Additional file 1: Methods.
Western blot analysis
The western blot procedure was performed as described in our recent study (Wang et al. 2021; Chen et al. 2021b; Chen and Sun 2021; Han and Sun 2020, 2022).
Human aortic valve histology and immunohistochemistry staining
The histological and immunohistochemical (IHC) procedures were performed as described in our previous studies (Chen et al. 2016; Lin et al. 2016; Fan et al. 2022a, b).
Statistical analysis
Quantitative data were presented as the Means ± SEM. The unpaired t-test was used for comparisons between two groups. Differences between experimental groups were examined by one-way analysis of variance (ANOVA), followed by the Tukey post-test or two-way ANOVA, followed by the Bonferroni post-test using Prism software (GraphPad). For all analyses, p < 0.05 was considered statistically significant.
Results
The data that support the findings of this study are available upon reasonable request from the corresponding author.
miR-101 expression increased in the calcified human aortic valves
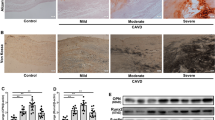
Figure 1A, B illustrate the dramatic structural and morphologic changes in calcified human aortic leaflets vs. the age-matched normal human aortic valve leaflets. Calcified leaflets developed an osseous structure. Alizarin red staining (Fig. 1C, D) displayed a significant calcium deposition on the calcified valves. Masson’s trichrome staining of collagen (blue color) indicated that calcium deposition occurred in the collagen-rich region close to the aortic side of the calcified valves (Fig. 1E, F).
miR-101-3p expression was upregulated in the calcified human aortic valves. A, B Photomicrographs of freshly isolated normal and calcified human aortic valves. C, D Alizarin red staining of normal and calcified human aortic valves, red staining indicating calcification (indicated by arrows). E, F Masson’s Trichrome staining of collagen deposition (blue staining indicated by arrows) of normal and calcified human aortic valves. G Small RNA deep sequencing results of miR-101-3p of normal and calcified human aortic valves. Data = means ± SEM. ***P < 0.001 vs Normal, n = 3. H qPCR analysis of miR-101-3p of normal and calcified human aortic valves. Scale bar = 200 μm. *P < 0.05 vs Normal, n = 3
Using the small RNA-Seq technology we generated microRNA-profiling libraries in normal and calcified aortic valve cells. There are hundreds of mRNA expressions up or down regulated (threshold > 1.5) in the calcified human aortic valves. The small RNA-Seq analysis showed that among these mRNA expressions, miR-101-3p levels in the calcified human aortic valves were almost 3 times higher than that of normal HAVs (Additional file 1: Fig. S1, Fig. 1G). Based on target prediction analysis of top miRs, we found that miR-101-3p may target Cadherin-11 (CDH11) and SOX9 (Additional file 1: Fig. S2) which are involved in chondrogenesis and osteogenesis. The qPCR analysis further confirmed that in the calcified HAVs, the miR-101-3p expression level was increased significantly (Fig. 1H). Therefore, we investigated whether miR-101-3p may contribute to human aortic valvular interstitial cell (HAVIC) calcification in isolated HAVICs.
miR-101-3p played a role in calcific nodule development in the HAVICs
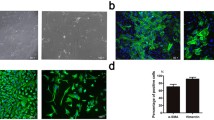
In order to investigate whether miR-101-3p plays a role in HAVIC calcification, we transfected HAVICs with miR-101-3p mimic and inhibitor sequences in the presence of normal and osteogenic-conditioned medium. The osteogenic-conditioned medium induces calcification in vitro. Alizarin red staining indicated that miR-101 mimic alone promoted calcific nodule development in HAVICs even in normal medium (Fig. 2B). In the conditioned medium, calcific nodules developed in the HAVICs transfected with control miR, (Fig. 2D) while the number of calcified nodules were further increased in the HAVICs transfected with miR-101 mimic (Fig. 2E). This result suggests that miR-101 mimic may promote HAVIC calcification. However, the calcified nodule formations were significantly decreased in the HAVICs transfected with miR-101-3p inhibitors (Fig. 2F), suggesting that the inhibition of miR-101 may suppress HAVIC calcification. The quantification of absorbance of alizarin red staining normalized by protein concentration demonstrated that miR-101-3p mimic promoted calcium deposition in the HAVICs in both normal and conditioned medium, and that miR-101-3p inhibitor prevented HAVICs from calcification in the conditioned medium (Fig. 2G). Western blot analysis of Fetuin A, a calcium-binding protein and an early calcification marker, also showed that Fetuin A was significantly increased in the HAVICs in CM (Fig. 2H, I), and that inhibition of miR-101 decreased Fetuin A level in HAVICs (Fig. 2H, J). Collectively, these results suggest that inhibiting miR-101 protects HAVICs from inducible calcification and that miR-101 promotes calcific nodule development within them.
miR-101-3p mimic promoted HAVICs calcification and inhibition of miR-101-3p protected HAVICs from calcification in CM. Alizarin red staining (A–F) of HAVICs cultured in osteogenic-conditioned media (10 mM βGP, 5 mM CaCl2) for 14 days after transfected with miR-101-3p mimic, inhibitor and negative control. G Quantification of alizarin red staining, absorbance normalized by protein concentration. *P < 0.05, ***P < 0.001 and ****P < 0.0001 vs Control; ++++P < 0.0001 vs Mimic; ^P < 0.05, ^^^^P < 0.0001 vs Control, CM; ####P < 0.0001 vs Inhibitor. H Western blot analysis of Fetuin A protein expression in HAVICs cultured in CM for 48 h after miR-101 mimic/inhibitor transfection. β-actin serves as internal loading control. I, J Quantification of Fetuin A protein expression normalized by β-actin. Data = means ± SEM. ****P < 0.0001 vs Control; ^P < 0.05 and ^^^^P < 0.0001 vs Mimic or Inhibitor groups; ++++P < 0.0001 vs Control, CM. n = 3 repeats
miR-101 promoted osteogenesis in HAVICs through regulation of CDH11 and SOX9 expression
We further investigated how miR-101 promotes calcification in HAVICs. Western blot analysis showed that expression of osteogenesis markers BMP2 and Runx2 displayed dose-dependent increases after HAVICs are transfected with various doses of miR-101 mimic (Fig. 3A, D, E). Using TargetScan Human (Release 7.1) (Chen et al. 2021a; Agarwal et al. 2015), we found that mRNAs of BMP2 and Runx2 do not have any predicted binding site of miR-101. Thus, we postulate that there may be upstream factors that are potentially direct targets of miR-101 and that mediate the role of miR-101 in regulating BMP2 and Runx2 expression in HAVICs.
miR-101-3p regulated CDH11 and Sox9 protein expression and osteogenesis markers BMP2/Runx2 in HAVICs. A Western Blots against antibodies of Cadherin-11 (CDH11) (ab147215), Sox9 (AB5535, Millipore), BMP2 (ThermoFisher, 710022), and Runx2 (Santa Cruz sc-12488) in HAVICs at 72 h after transfection with negative control and various doses of miR-101-3p mimic. B–E Quantification of CDH11, Sox9, BMP2, and Runx2 protein expression normalized with β-actin. F Western Blots against antibodies of Cadherin-11 (CDH11) and Sox9 in HAVICs at 72 h after transfection with negative control and various doses of mir-101-3p inhibitor. G, H Quantification of CDH 11 and Sox9 protein expression normalized with β-actin. Data = means ± SEM. *p < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001 vs Control. n = 3 repeats
Among the hundreds of potential mRNA targets of miR-101 (TargetScan Human, Release 7.1) (Chen et al. 2021a; Agarwal et al. 2015), Cadherin-11 (CDH11) and SOX9, which are potentially involved in ECM remodeling, caught our attention. Both of them had two 7-mer miR-101-3p binding sites on their mRNAs (Additional file 1: Fig. S2). Western blot analysis showed the decreased protein expressions of CDH11 and SOX9 in HAVICs after miR-101-3p mimic transfection (Fig. 3A-C), whereas inhibition of miR-101-3p by transfection of miR-101 inhibitor (anti-miRNA-101) increased CDH11 and SOX9 protein levels in HAVICs (Fig. 3F–H). These results provide the first evidence that miR-101 directly regulates CDH11 and SOX9 gene expression in HAVICs. Collectively, all these data suggest that upregulation of miR-101 in the calcified human aortic valves may play an important role in promoting CAVD development via targeting the protein expression of CDH11 and SOX9.
We next assessed CDH11 and Sox9 expression in the calcified human aortic valves (HAVs). Immunohistochemical analysis indicated that CDH11 expression was decreased in calcified HAVs (Fig. 4A, B). Western blot analysis further confirmed downregulation of CDH11 protein expression in calcified human aortic valves (Fig. 4C, D), as well as SOX9 protein expression (Fig. 4E, F).
CDH11 and SOX9 protein expression were decreased in calcified human aortic valves (HAVs). A Immunohistochemical staining against the antibody of Cadherin-11 (CDH11) (ab147215) on calcified human aortic valves and age-matched normal human aortic valves (Positive staining indicated by red arrows). Negative control staining without the secondary antibody. B Semi-quantification of area fraction of CDH11 staining on human aortic valves. ***P < 0.001 vs Normal. C, D Western blot analysis of CDH11 protein expression in human aortic valves normalized with β-actin. E, F Western blot analysis of SOX9 protein expression in human aortic valves normalized with β-actin. In A, scale bar = 500 μm for the upper panel and 200 μm for the lower panel. Data = means ± SEM. **P < 0.01, and ****P < 0.0001 vs Normal. n = 3
CDH11 and Sox9 regulated osteogenesis
Knockdown of CDH11 upregulated SOX9 expression in HAVICs (Additional file 1: Fig. S3A–C). Interestingly, knockdown of SOX9 increased CDH11 expression (Additional file 1: Fig. S3D–F). These results suggest that CDH11 and SOX9 may cross-regulate each other. Knockdown of CDH11 or SOX9 alone was insufficient to induce osteogenesis (Additional file 1: Fig. S4A–C). In contrast, knockdown of both CDH11 and SOX9 together promoted osteogenesis as evidenced by upregulation of Runx2, BMP2 and OCN expression in HAVICs (Additional file 1: Fig. S5).
miR-101-3p regulated osteogenesis in HAVICs treated with osteogenic-conditioned medium (CM)
In order to induce in vitro calcification in HAVICs, cells are usually treated with conditioned medium (10 mM βGP and 5 mM CaCl2 in M199) (Yang et al. 2009). To investigate the functional role of miR-101-3p in the regulation of calcification in HAVICs, we transfected HAVICs with miR-101 mimic and inhibitor for 48 h and then treated cells with CM for another 48 h. Western blot analysis showed significant decreases in CDH11 and SOX9 protein expression in miR-101 mimic-treated cells cultured in CM; HAVICs transfected with mimic and cultured in CM had lowest protein expression levels of CDH11 and SOX9 in all groups (Fig. 5A–C). Most importantly, transfection with miR-101 mimic upregulated osteogenesis markers Runx2 and OPN in HAVICs. Culturing with CM further increased protein expression of Runx2 and OPN in HAVICs (Fig. 5A, D, E). Overall, these data suggest that miR-101 promoted osteogenesis and calcification likely through direct targeting CDH11 and SOX9 mRNAs.
Mimic of miR-101 downregulated CDH-11 and SOX9 expression and promoted osteogenesis in HAVICs treated by CM after transfection. A Western blot against antibodies of CDH11, SOX9, Runx2, and OPN in HAVICs transfected with miR-101-3p mimic for 48 h and then treated with CM for additional 48 h. β-actin serves as internal loading control. B–E Quantification of CDH11, SOX9, Runx2, and OPN normalized with β-actin. Data = means ± SEM. *p < 0.05, **P < 0.01 and ****P < 0.0001 vs Control; +++P < 0.001 vs Control, CM; ^P < 0.05 and ^^^P < 0.001 vs Mimic. n = 3 repeats
Conversely, transfection with miR-101 inhibitor (anti-miR-101) rescued the downregulation of CDH11 and SOX9 expression levels in the CM, suggesting the inhibition of miR-101-3p in CM protected CDH11 and SOX9 expression from in vitro osteogenic induction (Fig. 6A–C). Transfection with miR-101 inhibitor also abolished upregulation of Runx2 and OPN protein expression (Fig. 6A, D, E) in CM. Overall, these results suggest that miR-101-3p is actively involved in the regulation of osteogenesis in HAVICs and that inhibition of miR-101-3p is efficient in protecting HAVICs from osteogenic differentiation and calcification in vitro.
Inhibition of miR-101 rescued downregulation of CDH-11 and SOX9 expression and prevented osteogenesis in CM-treated HAVICs. A Western blot against antibodies of CDH11, SOX9, Runx2, and OPN in HAVICs transfected with miR-101-3p inhibitor for 48 h and then treated with CM for additional 48 h. β-actin serves as internal loading control. B–E Quantification of CDH11, Sox9, Runx2 and OPN protein expression normalized with β-actin. Data = means ± SEM. **P < 0.01 and ***P < 0.001 vs Control; +P < 0.05, ++P < 0.001 and ++++P < 0.0001 vs Control, CM. n = 3 repeats
By theory, the miR-101-3p inhibitor should inhibit miR-101-3p which increases SOX9 protein expression. However, the miR-101-3p inhibitor did not affect the basal SOX9 protein expression level. This is likely because the basal SOX9 protein expression is high (Fig. 6C), inhibition of miR-101-3p did not further increase its expression. As a result, the basal protein expression of RUNX2, a target of SOX9, was not affected by miR-101-3p inhibitor (Fig. 6D). Nevertheless, the miR-101-3p inhibitor prevented CM-induced downregulation of SOX9 protein expression and, consequently, upregulation of RUNX2 protein expression. In contrast, Fig. 5C, D showed that miR-101-3p mimic downregulated the basal level of SOX9 protein expression which resulted in an increase in Runx2 expression.
miR-101 regulated asporin (ASPN) expression in HAVICs treated with osteogenic-conditioned medium
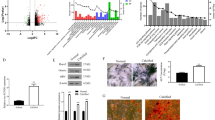
Asporin was reported as a regulator of osteoblast mineralization (Kalamajski et al. 2009; Tomoeda et al. 2008) and played regulatory roles in the chondrogenesis (Nakajima et al. 2007). Interestingly, a significant increase of ASPN expression was found in the HAVICs cultured in the CM as well as in HAVICs treated with miR-101-3p mimic (Fig. 7A, B). ASPN expression levels in the mimic-treated HAVICs cultured in CM were almost threefold higher than those in the control HAVICs. On the other hand, inhibition of miR-101 in the CM-treated HAVICs decreased the ASPN expression level to the normal level (Fig. 7A–C). These results suggest that miR-101 regulates ASPN in HAVIC calcification. Interestingly, siRNAs knockdown of CDH11 or SOX9 separately in the HAVICs did not affect ASPN expression (Additional file 1: Fig. S4C). However, knockdown of both proteins via co-transfection of CDH11 and SOX9 SiRNA led to a significant increase of ASPN protein expression in HAVICs (Fig. 7D–G, Additional file 1: Fig. S5A, B). This result suggests that miR-101 upregulates ASPN expression in HAVICs likely through CDH11 and SOX9. These results provided new mechanistic insights into the regulation of ASPN, a regulator of osteogenesis and calcification (Kalamajski et al. 2009; Tomoeda et al. 2008).
miR-101 mediated ASPN expression in CM treated HAVICs through regulation of CDH11 and Sox9. A Western Blot against antibodies of ASPN in HAVICs transfected with miR-101-3p mimic or inhibitor for 48 h and then treated with CM for 48 h. β-Actin serves as internal loading control. B, C Quantifications of ASPN protein expression normalized with β-actin. *P < 0.05 and ****P < 0.0001 vs Control; +P < 0.05 and ++P < 0.01 vs Control, CM; ^^P < 0.01 vs Mimic. D Western Blot against CDH11, Sox9 and ASPN in HAVICs co-transfected with SiRNAs of CDH11 (10 nM) and Sox9 (1 nM) for 72 h. E–G Quantifications of WBs normalized by β-actin. Data = means ± SEM. ***P < 0.001 and ****P < 0.0001 vs Control. n = 3 repeats
Discussion
CAVD is considered a progressive process, and the aortic valve interstitial cells (AVICs) are closely regulated by multiple levels of biological factors in various physiologic scenarios, facing numerous stresses and challenges during an entire lifespan to keep the structural and functional integrity of the aortic valves. However, the mechanisms of the development of CAVD remain elusive due to the complicated cell potency of AVICs, which have the ability to differentiate into other type-like cells, such as osteoblast-like and osteoclast-like cells under various circumstances.
The present study demonstrates that increased expressions of miR-101-3p plays a crucial role in the regulation of HAVIC calcification. To the best of our knowledge, this is the first report showing that miR-101 directly promotes HAVIC calcification. More importantly, the inhibition of miR-101-3p effectively prevented or attenuated the calcification in HAVICs even under osteogenic conditions (Fig. 2). This finding not only provides new therapeutic insights into CAVD, but also fundamentally advances the current understanding of the molecular regulation of CAVD. Furthermore, the current study reveals novel mechanisms through which miR-101-3p regulates HAVIC calcification. Specifically, miR-101-3p directly binds to CDH11 and SOX9, which promotes osteogenesis. All these data suggest that inhibition of miR-101-3p in the HAVICs may offer a useful strategy for effective protection of aortic valves from calcification under osteogenic stress.
Aortic valve fibrosis and calcification share distinct processes in ECM remodeling which involves regulations through different signaling pathways. However, there are crosstalk and autoregulation of these pathways, which are mediated by unknown regulators (proteins). It has been reported that CDH11 and SOX9 are associated with ECM remodeling in the aortic valves; therefore, dysregulation of these proteins may promote osteogenesis and calcification of AVs (Peacock et al. 2010; Lincoln et al. 2007; Kii et al. 2004; Bowen et al. 2015; Sung et al. 2016). We have observed that in HAVICs, miR-101-3p activated the osteogenic pathway and promoted calcification (Fig. 5). Our data also demonstrate that the miR-101-3p mimic downregulated expression of CDH11 and SOX9 probably through targeting their mRNAs. Furthermore, our data suggests that siRNA knockdown of CDH11 increased SOX9 expression, and vice versa (Additional file 1: Fig. S3). Thus, CHD11 and SOX9 may cross-regulate each other in mediating HAVIC calcification. We also found that knockdown of either of these two genes alone using SiRNAs was insufficient to promote osteogenesis in the HAVICs (Additional file 1: Fig. S4). Interestingly, knockdown of both CDH11 and SOX9 simultaneously induced osteogenic responses (e.g., upregulation of BMP2, Runx2, ASPN and OCN) (Additional file 1: Fig. S5). Although the mechanism underlying the regulation of SOX9 expression by CHD11 is unclear, it appears that downregulation of both is required for promoting osteogenesis in HAVICs. A limitation of this study is that it lacks mechanistic investigation into the crosstalk of CDH11 and SOX9. A further study is warranted for investigating whether the cell adhesion molecule CDH11 inhibits SOX9 transcription or promotes degradation of SOX9 protein in HAVICs. Collectively, all these findings suggest that the miR-101-3p promotes osteogenesis and calcification in the HAVICs likely via downregulation of CDH11 and SOX9, the major regulators of ECM remodeling, calcification and ossification in aortic valves.
Asporin (ASPN) plays an important role in the regulation of cartilage development and osteoarthritis (OA) pathology (Kalamajski et al. 2009; Tomoeda et al. 2008). ASPN was present in the HAVICs and the expression level was upregulated in the HAVICs treated with osteogenic-conditioned medium (Fig. 7). Knockdown of both CDH11 and SOX9 increased ASPN expression. Thus, mR-101 regulates ASPN expression, likely via CDH11/SOX9.
It was reported that miR-101-3p regulates the PTEN/AKT axis in trophoblast cells in the placenta (Zhang et al. 2022). Metformin can inhibit AVICs calcification in vitro by activating the PI3K/AKT signaling pathway (En et al. 2021). Yang et al. (2022) found that LEVs might inhibit neuron apoptosis via the miR-101a-3p/c-Fos/TGF-β axis, and has-miR-101-3p is a potential marker of neurological recovery in IS patients. TGF-beta1 is characteristically present within calcific aortic stenosis cusps, and mediates the calcification of aortic valve interstitial cells in culture through mechanisms involving apoptosis (Jian et al. 2003). A further study is needed to investigate whether these pathways regulate CDH11, SOX9 and ASPN in AVICs and mediate the role of miR-101-3P in AVIC calcification.
Conclusion
The present study demonstrates that the level of miR-101-3p was remarkably increased in the calcified human aortic valves, and the miR-101-3p mimic promotes osteogenesis and calcification in the HAVICs (Additional file 1: Fig. S6). Inhibition of miR-101-3p showed strong therapeutic potential to prevent or attenuate the development of HAVIC calcification. Furthermore, the data indicated that miR-101-3p directly targeted CDH11 and SOX9 to regulate ASPN and the downstream osteogenesis pathway. These findings not only advance our current understanding of the mechanism of human CAVD but also provide new therapeutic insights into CAVD. Whether ASPN is directly and actively involved in HAVIC calcification induced by CM and/or miR-101 treatment warrants further investigation.
Availability of data and materials
The data that support the findings of this study are available upon reasonable request from the corresponding author.
Abbreviations
- CAVD:
-
Calcific aortic valve disease
- HAVICs:
-
Human aortic valve interstitial cells
- CDH11:
-
Cadherin-11
- CHAVs:
-
Calcified human aortic valves
- ASPN:
-
Asporin
- Runx2:
-
Runt-related transcription factor 2
- BMP2:
-
Bone morphogenetic protein 2
- OPN:
-
Osteopontin
- OCN:
-
Osteocalcin
References
Agarwal V, Bell GW, Nam JW, Bartel DP. Predicting effective microRNA target sites in mammalian mRNAs. Elife. 2015;4: e05005.
Akiyama H. Control of chondrogenesis by the transcription factor Sox9. Mod Rheumatol. 2008;18:213–9.
Ambros V. The functions of animal microRNAs. Nature. 2004;431:350–5.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–97.
Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–33.
Baumgartner H. Aortic stenosis: medical and surgical management. Heart. 2005;91:1483–8.
Bowen CJ, Zhou J, Sung DC, Butcher JT. Cadherin-11 coordinates cellular migration and extracellular matrix remodeling during aortic valve maturation. Dev Biol. 2015;407:145–57.
Bruderer M, Richards RG, Alini M, Stoddart MJ. Role and regulation of RUNX2 in osteogenesis. Eur Cell Mater. 2014;28:269–86.
Cairns DM, et al. Interplay of Nkx3.2, Sox9 and Pax3 regulates chondrogenic differentiation of muscle progenitor cells. PLoS ONE. 2012;7: e39642.
Cavallaro U, Dejana E. Adhesion molecule signalling: not always a sticky business. Nat Rev Mol Cell Biol. 2011;12:189–97.
Chan KL, et al. Effect of Lipid lowering with rosuvastatin on progression of aortic stenosis: results of the aortic stenosis progression observation: measuring effects of rosuvastatin (ASTRONOMER) trial. Circulation. 2010;121:306–14.
Chen K, Sun Z. Autophagy plays a critical role in Klotho gene deficiency-induced arterial stiffening and hypertension. J Mol Med. 2019;97:1615–25.
Chen K, Sun Z. Estrogen inhibits renal Na-Pi Co-transporters and improves klotho deficiency-induced acute heart failure. Redox Biol. 2021;47: 102173.
Chen H, et al. Runx2 regulates endochondral ossification through control of chondrocyte proliferation and differentiation. J Bone Miner Res. 2014;29:2653–65.
Chen J, Lin Y, Sun Z. Deficiency in the anti-aging gene Klotho promotes aortic valve fibrosis through AMPKalpha-mediated activation of RUNX2. Aging Cell. 2016;15(5):853–60.
Chen K, Zhang B, Sun Z. MicroRNA 379 regulates klotho deficiency-induced cardiomyocyte apoptosis via repression of Smurf1. Hypertension. 2021a;78:342–52.
Chen K, et al. Klotho deficiency causes heart aging via impairing the Nrf2-GR pathway. Circ Res. 2021b;128:492–507.
Chen K, Wang S, Sun Z. In vivo cardiac-specific expression of adenylyl cyclase 4 gene protects against Klotho deficiency-induced heart failure. Transl Res. 2022;244:101–13.
Cheng A, Genever PG. SOX9 determines RUNX2 transactivity by directing intracellular degradation. J Bone Miner Res. 2010;25:2680–9.
Cowell SJ, et al. A randomized trial of intensive lipid-lowering therapy in calcific aortic stenosis. N Engl J Med. 2005;352:2389–97.
Dai L, Zhang X, Hu X, Zhou C, Ao Y. Silencing of microRNA-101 prevents IL-1beta-induced extracellular matrix degradation in chondrocytes. Arthritis Res Ther. 2012;14:R268.
Dai L, et al. Silencing of miR-101 prevents cartilage degradation by regulating extracellular matrix-related genes in a rat model of osteoarthritis. Mol Ther. 2015;23:1331–40.
En Q, Zeping H, Yuetang W, Xu W, Wei W. Metformin alleviates the calcification of aortic valve interstitial cells through activating the PI3K/AKT pathway in an AMPK dependent way. Mol Med. 2021;27:156.
Erson AE, Petty EM. MicroRNAs in development and disease. Clin Genet. 2008;74:296–306.
Fan J, Wang S, Chen K, Sun Z. Aging impairs arterial compliance via Klotho-mediated downregulation of B-cell population and IgG levels. Cell Mol Life Sci. 2022a;79:494.
Fan J, Wang S, Lu X, Sun Z. Transplantation of bone marrow cells from miR150 knockout mice improves senescence-associated humoral immune dysfunction and arterial stiffness. Metabolism. 2022b;134: 155249.
Fang M, Alfieri CM, Hulin A, Conway SJ, Yutzey KE. Loss of beta-catenin promotes chondrogenic differentiation of aortic valve interstitial cells. Arterioscler Thromb Vasc Biol. 2014;34:2601–8.
Freeman RV, Otto CM. Spectrum of calcific aortic valve disease: pathogenesis, disease progression, and treatment strategies. Circulation. 2005;111:3316–26.
Guerit D, et al. Sox9-regulated miRNA-574-3p inhibits chondrogenic differentiation of mesenchymal stem cells. PLoS ONE. 2013;8: e62582.
Han X, Sun Z. Epigenetic regulation of KL (Klotho) via H3K27me3 (histone 3 lysine [K] 27 trimethylation) in renal tubule cells. Hypertension. 2020;75:1233–41.
Han X, Sun Z. Adult mouse kidney stem cells orchestrate the de novo assembly of a nephron via Sirt2-modulated canonical Wnt/β-catenin signaling. Adv Sci. 2022;9: e2104034.
Harris TJ, Tepass U. Adherens junctions: from molecules to morphogenesis. Nat Rev Mol Cell Biol. 2010;11:502–14.
Hutcheson JD, et al. Cadherin-11 regulates cell-cell tension necessary for calcific nodule formation by valvular myofibroblasts. Arterioscler Thromb Vasc Biol. 2013;33:114–20.
Jian B, Narula N, Li QY, Mohler ER 3rd, Levy RJ. Progression of aortic valve stenosis: TGF-beta1 is present in calcified aortic valve cusps and promotes aortic valve interstitial cell calcification via apoptosis. Ann Thorac Surg. 2003;75:457–65 (discussion 465–456).
Kalamajski S, Aspberg A, Lindblom K, Heinegard D, Oldberg A. Asporin competes with decorin for collagen binding, binds calcium and promotes osteoblast collagen mineralization. Biochem J. 2009;423:53–9.
Kii I, Amizuka N, Shimomura J, Saga Y, Kudo A. Cell-cell interaction mediated by cadherin-11 directly regulates the differentiation of mesenchymal cells into the cells of the osteo-lineage and the chondro-lineage. J Bone Miner Res. 2004;19:1840–9.
Le LT, Swingler TE, Clark IM. Review: the role of microRNAs in osteoarthritis and chondrogenesis. Arthritis Rheum. 2013;65:1963–74.
Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993;75:843–54.
Lee SC, et al. A non-coding transcript of nephronectin promotes osteoblast differentiation by modulating microRNA functions. FEBS Lett. 2011;585:2610–6.
Lefebvre V, Dumitriu B, Penzo-Mendez A, Han Y, Pallavi B. Control of cell fate and differentiation by Sry-related high-mobility-group box (Sox) transcription factors. Int J Biochem Cell Biol. 2007;39:2195–214.
Lim LP, Glasner ME, Yekta S, Burge CB, Bartel DP. Vertebrate microRNA genes. Science. 2003;299:1540.
Lin Y, Chen J, Sun Z. Antiaging gene Klotho deficiency promoted high-fat diet-induced arterial stiffening via inactivation of AMP-activated protein kinase. Hypertension. 2016;67(3):564–73.
Lincoln J, Kist R, Scherer G, Yutzey KE. Sox9 is required for precursor cell expansion and extracellular matrix organization during mouse heart valve development. Dev Biol. 2007;305:120–32.
Lindroos M, Kupari M, Heikkila J, Tilvis R. Prevalence of aortic valve abnormalities in the elderly: an echocardiographic study of a random population sample. J Am Coll Cardiol. 1993;21:1220–5.
Liu AC, Joag VR, Gotlieb AI. The emerging role of valve interstitial cell phenotypes in regulating heart valve pathobiology. Am J Pathol. 2007;171:1407–18.
Miller JD, Weiss RM, Heistad DD. Calcific aortic valve stenosis: methods, models, and mechanisms. Circ Res. 2011;108:1392–412.
Mohler ER 3rd, et al. Bone formation and inflammation in cardiac valves. Circulation. 2001;103:1522–8.
Monzack EL, Masters KS. Can valvular interstitial cells become true osteoblasts? A side-by-side comparison. J Heart Valve Dis. 2011;20:449–63.
Nakajima M, et al. Mechanisms for asporin function and regulation in articular cartilage. J Biol Chem. 2007;282:32185–92.
O’Connell RM, Rao DS, Chaudhuri AA, Baltimore D. Physiological and pathological roles for microRNAs in the immune system. Nat Rev Immunol. 2010;10:111–22.
Pan Q, et al. Sox9, a key transcription factor of bone morphogenetic protein-2-induced chondrogenesis, is activated through BMP pathway and a CCAAT box in the proximal promoter. J Cell Physiol. 2008;217:228–41.
Pan Z, et al. MicroRNA-101 inhibited postinfarct cardiac fibrosis and improved left ventricular compliance via the FBJ osteosarcoma oncogene/transforming growth factor-beta1 pathway. Circulation. 2012;126:840–50.
Peacock JD, Levay AK, Gillaspie DB, Tao G, Lincoln J. Reduced sox9 function promotes heart valve calcification phenotypes in vivo. Circ Res. 2010;106:712–9.
Rajamannan NM, et al. Calcific aortic valve disease: not simply a degenerative process: a review and agenda for research from the National Heart and Lung and Blood Institute Aortic Stenosis Working Group. Executive summary: calcific aortic valve disease-2011 update. Circulation. 2011;124:1783–91.
Rossbach M. Small non-coding RNAs as novel therapeutics. Curr Mol Med. 2010;10:361–8.
Rossebo AB, et al. Intensive lipid lowering with simvastatin and ezetimibe in aortic stenosis. N Engl J Med. 2008;359:1343–56.
Rupaimoole R, Slack FJ. MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov. 2017;16:203–22.
Soifer HS, Rossi JJ, Saetrom P. MicroRNAs in disease and potential therapeutic applications. Mol Ther. 2007;15:2070–9.
Song R, et al. BMP-2 and TGF-beta1 mediate biglycan-induced pro-osteogenic reprogramming in aortic valve interstitial cells. J Mol Med. 2015;93:403–12.
Sung DC, et al. Cadherin-11 overexpression induces extracellular matrix remodeling and calcification in mature aortic valves. Arterioscler Thromb Vasc Biol. 2016;36:1627–37.
Tay Y, Zhang J, Thomson AM, Lim B, Rigoutsos I. MicroRNAs to Nanog, Oct4 and Sox2 coding regions modulate embryonic stem cell differentiation. Nature. 2008;455:1124–8.
Tomoeda M, et al. PLAP-1/asporin inhibits activation of BMP receptor via its leucine-rich repeat motif. Biochem Biophys Res Commun. 2008;371:191–6.
Towler DA. Molecular and cellular aspects of calcific aortic valve disease. Circ Res. 2013;113:198–208.
Wang Q, Wang S, Sun Z. Kidney-specific Klotho gene deletion causes aortic aneurysm via hyperphosphatemia. Hypertension. 2021;78:308–19.
Wu D, Jiang H, Chen S, Zhang H. Inhibition of microRNA-101 attenuates hypoxia/reoxygenationinduced apoptosis through induction of autophagy in H9c2 cardiomyocytes. Mol Med Rep. 2015;11:3988–94.
Yamashita S, et al. Sox9 directly promotes Bapx1 gene expression to repress Runx2 in chondrocytes. Exp Cell Res. 2009;315:2231–40.
Yang X, et al. Pro-osteogenic phenotype of human aortic valve interstitial cells is associated with higher levels of Toll-like receptors 2 and 4 and enhanced expression of bone morphogenetic protein 2. J Am Coll Cardiol. 2009;53:491–500.
Yang Z, et al. Lactobacillus plantarum-derived extracellular vesicles protect against ischemic brain injury via the microRNA-101a-3p/c-Fos/TGF-β axis. Pharmacol Res. 2022;182: 106332.
Zhang M, et al. MicroRNA-30b is a multifunctional regulator of aortic valve interstitial cells. J Thorac Cardiovasc Surg. 2014;147:1073-1080.e1072.
Zhang X, et al. Long non-coding RNA LINC01347 suppresses trophoblast cell migration, invasion and EMT by regulating miR-101-3p/PTEN/AKT axis. Reprod Biol. 2022;22: 100670.
Zhao X, et al. MicroRNA-101a inhibits cardiac fibrosis induced by hypoxia via targeting TGFbetaRI on cardiac fibroblasts. Cell Physiol Biochem. 2015;35:213–26.
Zhou G, et al. Dominance of SOX9 function over RUNX2 during skeletogenesis. Proc Natl Acad Sci USA. 2006;103:19004–9.
Zhou J, et al. Cadherin-11 expression patterns in heart valves associate with key functions during embryonic cushion formation, valve maturation and calcification. Cells Tissues Organs. 2013;198:300–10.
Acknowledgements
Not applicable.
Disclosures
All authors have nothing to disclose.
Funding
This work was supported by National Heart, Lung, and Blood Institute (HL118558); National Institutes of Health (HL154147, AG062375 and AG049780).
Author information
Authors and Affiliations
Contributions
Conceptualization: ZS. Methodology: JC and YL. Investigation: JC. Visualization: JC. Writing original draft: JC. Writing review and editing: ZS. Funding acquisition: ZS. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable (no human or animal subjects).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Online Supplemental Methods and Data.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, J., Lin, Y. & Sun, Z. Inhibition of miR-101-3p prevents human aortic valve interstitial cell calcification through regulation of CDH11/SOX9 expression. Mol Med 29, 24 (2023). https://doi.org/10.1186/s10020-023-00619-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s10020-023-00619-4