Abstract
Background
Although pregabalin therapy is beneficial for neuropathic pain (NeP) by targeting the CaVα2δ-1 subunit, its site of action is uncertain. Direct targeting of the central nervous system may be beneficial for the avoidance of systemic side effects.
Results
We used intranasal, intrathecal, and near-nerve chamber forms of delivery of varying concentrations of pregabalin or saline delivered over 14 days in rat models of experimental diabetic peripheral neuropathy and spinal nerve ligation. As well, radiolabelled pregabalin was administered to determine localization with different deliveries. We evaluated tactile allodynia and thermal hyperalgesia at multiple time points, and then analyzed harvested nervous system tissues for molecular and immunohistochemical changes in CaVα2δ-1 protein expression. Both intrathecal and intranasal pregabalin administration at high concentrations relieved NeP behaviors, while near-nerve pregabalin delivery had no effect. NeP was associated with upregulation of CACNA2D1 mRNA and CaVα2δ-1 protein within peripheral nerve, dorsal root ganglia (DRG), and dorsal spinal cord, but not brain. Pregabalin's effect was limited to suppression of CaVα2δ-1 protein (but not CACNA2D1 mRNA) expression at the spinal dorsal horn in neuropathic pain states. Dorsal root ligation prevented CaVα2δ-1 protein trafficking anterograde from the dorsal root ganglia to the dorsal horn after neuropathic pain initiation.
Conclusions
Either intranasal or intrathecal pregabalin relieves neuropathic pain behaviours, perhaps due to pregabalin's effect upon anterograde CaVα2δ-1 protein trafficking from the DRG to the dorsal horn. Intranasal delivery of agents such as pregabalin may be an attractive alternative to systemic therapy for management of neuropathic pain states.
Similar content being viewed by others
Background
Neuropathic pain is a consequence of nerve damage or disease in the central and/or peripheral nervous system such as with diabetes and trauma. The clinical presentation of neuropathic pain includes hyperalgesia, allodynia, and spontaneous pain [1]. Its high prevalence in humans [2–4] has led to the development of a number of animal models of neuropathic pain, including diabetic peripheral neuropathy and spinal nerve ligation.
Changes within the nervous system associated with neuropathic pain include critical upregulation of the calcium channel subunit CaVα2δ-1 [5–7], particularly at the dorsal horn [8, 9]. This is of importance for the clinical utility and potential mechanism of two different pharmacotherapies, gabapentin and pregabalin, both of which are indicated for the management of neuropathic pain due to multiple etiologies. There is uncertainty about the anatomical location of pregabalin's beneficial effect, as CaVα2δ-1 subunit upregulation is multifocal [10]. In addition to the known expression within the DRG, CACNA2D1 mRNA is also localized to brain regions known to be involved in cortical processing, sensory conduction, and arousal [11]. Also, there is also marked CaVα2δ-1 subunit expression in spinal cord regions where DRG projections occur. As a result, the localization of pregabalin's greatest therapeutic effect is uncertain. In addition to changes in CaVα2δ-1, other changes in voltage gated channels [12–14] and microgliosis with associated elevation in cytokines may occur [15–18].
Pregabalin, as with gabapentin, interacts specifically with the CaVα2δ-1 subunit of voltage-gated calcium channels [19–21] providing an antihyperalgesic and antiallodynic effect specific for its action at the CaVα2δ-1 subunit [22]. Recently, Bauer et al [23] demonstrated the importance of trafficking of the CaVα2δ-1 subunit from the dorsal root ganglia to the dorsal horn in the development of neuropathic pain, with its subsequent alleviation with pregabalin treatment. In the present study, we attempted to determine the central and peripheral contributions of pregabalin for relief from neuropathic pain in two separate models: streptozotocin-induced diabetic peripheral neuropathy and traumatic injury (spinal nerve ligation). We also examined the therapeutic potential for intranasal delivery of pregabalin with comparison to more localized delivery systems using implantable pump systems permitting a continuous delivery of pregabalin to specific anatomical locations [10, 24]. Intranasal delivery, first developed to bypass the blood-brain-barrier and directly target therapeutic agents to the central nervous system [25–28] of rodents [29–31] and humans [30, 31], occurs along both the olfactory and trigeminal neural pathways using extracellular pathways rather than axonal transport [30]. Such directed targeting of pregabalin to the brain can avoid gastrointestinal uptake of oral therapy and may permit more potent relief of neuropathic pain behavior while limiting systemic side effects. For neuroinflammatory, neurodegenerative, and neurovascular disorders [28], intranasal delivery is an attractive non-invasive method to target molecules to the central nervous system [32, 33] and even the peripheral nervous system [34].
Our primary objective was to study the central delivery of pregabalin using either intranasal or intrathecal therapy and compare to near-nerve delivery for impact upon neuropathic pain behaviors. Previously, intranasal delivery targeting the central nervous system has been used as a method of delivery for neuropathic pain states [34–36]. Intranasal delivery is dependent upon the olfactory and trigeminal nerves connecting the nose to the brain, the rostral migratory stream, and less so upon vasculature and lymphatic pathways [28, 37]. The benefit of intranasal delivery is the concentration of effect in the central nervous system; systemic delivery to some extent will occur with small, lipophilic molecules delivered intranasally but is otherwise quite limited [28]. A small, lipophilic molecule such as pregabalin would be anticipated to have greater systemic distribution following intranasal delivery than other compounds [38]. Rarely used in human clinical management at present [35], the non-invasive nature of intranasal delivery along with limited systemic exposure make it potentially attractive for use in the clinic with human patients. Intrathecal delivery of compounds with various mechanisms [10, 39, 40] has also led to amelioration of neuropathic pain states. Already used in the clinic for delivery of opioids, baclofen and conotoxins, intrathecal delivery is an invasive technique used in specific situations of refractoriness or intolerance of oral delivery, but its use depends upon the agent used, its site of action, and its ability to cross the blood-brain barrier. Finally, near nerve delivery has also been used to modulate sensory nerve regeneration and impact upon pain behaviors [24, 41, 42]. Our secondary objectives of this study were to evaluate changes in expression of the CaVα2δ-1 subunit in the dorsal spinal cord, DRG peripheral nerve and brain, evaluate potential changes in other voltage-gated calcium and sodium channels in the spinal cord, DRG and nerve, evaluate for changes in accumulation of microglia, and to evaluate the safety and tolerability of pregabalin using the various delivery routes in rodents, including the use of intranasal delivery. We also examined the previously demonstrated role of CaVα2δ-1 subunit trafficking [23] in a dynamic and progressive model of neuropathic pain, diabetic peripheral neuropathy. The determination of impact of pregabalin upon the CaVα2δ-1 subunit within nervous system tissues depending upon the mode of delivery would assist in the understanding of the pharmacological action of pregabalin upon the CaVα2δ-1 subunit and its effects upon pathways relevant in the development of neuropathic pain.
Results
Radiolabelled Detection of Pregabalin Localization
At 73 hours of intrathecal or intranasal delivery, pregabalin concentrations were detected throughout the nervous system (Figure 1). Intranasal delivery gave consistently higher elevated pregabalin levels throughout the central nervous system structures at and above the cephalad portion of the cervical spinal cord. In contrast, intrathecal delivery was associated with higher pregabalin penetration into the lumbar spinal cord and much higher levels within lumbar dorsal root ganglia. Systemic presence of pregabalin was much higher at 73 hours with intranasal pregabalin delivery, but was similar to concentrations achieved with intrathecal delivery at 74 and 77 hours.
Radiolabelled pregabalin detection. The harvested tissues are displayed on the X axis for the systemic (A, D) and for nervous system tissues (B, C, E, F). Systemic (A) or nervous system (B, C) pregabalin concentrations are demonstrated for 73-74 hours after intrathecal (white bars) or intranasal (black bars) or near nerve (gray bars) pregabalin delivery. Note that the 73 and 74 hour timepoints were analyzed together for these timepoints early after completion of 72 hours of delivery. At 77 hours, at 5 hours after the most recent intranasal delivery, systemic (D), brain (E), and cord (F) pregabalin concentrations started to fall. Near nerve delivery led to sciatic nerve pregabalin concentrations several fold higher than achieved with either intrathecal or intranasal delivery, but near nerve delivery was not associated with detectable pregabalin elsewhere. Significant differences between concentrations achieved with intervention methods were determined by matched ANOVA testing, with * indicating significant differences (p < 0.05) between the intranasal or intrathecal pregabalin delivery and saline delivery techniques for each tissue and respective delivery method. The doses provided through either intranasal or intrathecal delivery were selected to be similar to doses of pregabalin used in later subexperiments [n = 4-6 rats in each rat cohort for each time point]. Note that data are not shown for locations other than the sciatic nerve for near nerve pregabalin delivery, as other locations had no measureable pregabalin found.
After 74 hours of chronic delivery, concentrations with intranasally delivered pregabalin began to fall while intrathecal levels were remaining stable throughout the nervous system and systemically (Additional File 1). At 77 hours after intranasal delivery, both systemic and nervous system levels of intranasally delivered pregabalin began to fall while intrathecal dosing provided relatively stable pregabalin concentrations.
Near nerve delivery of pregabalin only led to detection at the proximal sciatic nerve, with zero detection of meaningful radioactive pregabalin at other systemic or nervous system sites. The concentrations of pregabalin received at the proximal sciatic nerve were several fold higher with near nerve delivery than could be achieved with intrathecal or intranasal delivery (Figure 1).
Maintenance of Diabetes in the Diabetic Peripheral Neuropathy Model
After streptozotocin injection, rats developed diabetes within 1 week in greater than 80% of animals, and in each case, diabetes was maintained over the length of the study. Diabetic rats were smaller than non-diabetic rats at endpoint (201.8 ± 14.7 grams as compared to 234.5 ± 15.2 grams). Diabetic rat glycemia was increased as compared to non-diabetic rats at endpoint (26.4 ± 3.1 mmol/L as compared to 6.1 ± 2.2 mmol/L).
Neuropathic Pain Behavior
Despite different methods and dosing of pregabalin delivery, there was no impact upon locomotor functioning in rat cohorts studied with Rotarod testing at 72-74 hours post-pregabalin delivery (data not shown). Diabetic rats and rats subjected to spinal nerve ligation both developed tactile allodynia and thermal hyperalgesia during experimentation (Figure 2). Both intrathecal and intranasal pregabalin provision led to improvement in tactile allodynia and thermal hyperalgesia at high doses (2.04 mg/kg/d) in both diabetic peripheral neuropathy and spinal nerve ligation models. Medium (0.51 mg/kg/d) doses of pregabalin decreased thermal hyperalgesia due to diabetic peripheral neuropathy and spinal nerve ligation without impact upon tactile allodynia. Lower doses (0.051 mg/kg/d) of intrathecal or intranasal pregabalin had no impact upon either tactile allodynia or thermal hyperalgesia in either model. Near-nerve delivery of pregabalin had no impact upon tactile allodynia or thermal hyperalgesia with any dose (data not shown) in either model of neuropathic pain.
Behavioral testing results for both models of neuropathic pain. Tactile (A) and thermal (B) sensory testing data for rats with or without (control) diabetes and diabetic peripheral neuropathy (DPN) receiving intranasal pregabalin are presented for multiple doses of pregabalin or saline. Intrathecal pregabalin delivery was also followed by measurements of tactile allodynia (C) and thermal hyperalgesia in diabetic rats (D). High doses of either intranasal or intrathecal pregabalin impacted upon these neuropathic pain behaviours in DPN. For rats with or without spinal nerve ligation receiving intranasal pregabalin, tactile (E) and thermal (F) sensory testing data are presented; intranasal pregabalin at high dose impacted upon both measures of neuropathic pain behaviour. Finally, intrathecal pregabalin also impacted upon both tactile allodynia (G) and thermal hyperalgesia (H) due to spinal nerve ligation when compared to saline delivery. Significant differences were detected between the diabetic rats receiving high dose (2.04 mg/kg/d) (*) and medium dose (0.51 mg/kg/d) (θ) pregabalin as compared to diabetic rats receiving saline delivery (non-matched ANOVA tests, F-values range between 1.98-22.86 for indicated groups and time points, n ≥ 5, p < 0.0125 after Bonferroni correction). Significant differences were detected between rats with spinal nerve ligation receiving high dose (2.04 mg/kg/d) (*) and medium dose (0.51 mg/kg/d) (θ) pregabalin as compared to rats with spinal nerve ligation receiving saline delivery (non-matched ANOVA tests, F-values range between 2.25-8.11 for indicated groups and time points, n ≥ 5, p < 0.0125 after Bonferroni correction) [n = 5-6 rats in each cohort for each time point]. Data for rats receiving low dose pregabalin were similar to data for rats receiving saline and are not shown.
mRNA Expression for CACNA2D1 and Other Voltage Gated Channels in Diabetic Peripheral Neuropathy
Both diabetic peripheral neuropathy and spinal nerve ligation were associated with elevation of CACNA2D1 mRNA expression within both the DRG and dorsal spinal cord (Figure 3). However, there was no observed impact of pregabalin delivery upon CACNA2D1 mRNA expression within either location when compared to saline delivery in either model.
Expression of CACNA2D1 mRNA. Expression of CACNA2D1 mRNA was upregulated in both dorsal spinal cord (A) and lumbar DRGs (B) in the presence of either diabetic peripheral neuropathy (DPN) or spinal nerve ligation (SNL). Dorsal horn mRNA expression was unchanged by presence of high dose pregabalin (2.04 mg/kg/d) provided either through intrathecal or intranasal delivery when compared to saline delivery (A). There was no impact of pregabalin delivered in any manner upon mRNA expression in lumbar DRGs when compared to saline delivery, although the presence of spinal nerve ligation or diabetic peripheral neuropathy was associated with elevated CACNA2D1 mRNA expression when compared to control rat specimens (B), indicated by the * above the Control rat data (non-matched ANOVA tests, F-values range between 0.42-1.00 for indicated groups and time points, n ≥ 3, * p < 0.0125) [n = 6-8 specimens for each cohort].
Protein Expression for α2δ-1 and other Voltage Gated Channels
Immunohistochemical detection of CaVα2δ-1 protein in nerves exposed to diabetes or spinal nerve ligation demonstrated an increased number of CaVα2δ-1 positive profiles near the site of spinal nerve ligation, but without impact of pregabalin delivery, irrespective of site of delivery (Additional File 2). The presence of diabetes or the provision of spinal nerve ligation was also associated with a shift to a greater number of CaVα2δ-1 positive DRG neurons within the DRG, but again there was no impact with delivery of pregabalin regardless of method of delivery or form of neuropathic pain (Additional File 2). Both diabetes and spinal nerve ligation led to greater expression of CaVα2δ-1 protein at the dorsal horn; at this location, however, receipt of high dose pregabalin via intrathecal or intranasal delivery was associated with relatively diminished expression of CaVα2δ-1 protein (Figure 4) at the dorsal horn.
CaVα 2 δ-1 protein expression. CaVα2δ-1 protein expression was detected by immunohistochemistry at the dorsal horn regions of rats subjected to spinal nerve ligation or diabetes (A). Rats exposed to high dose (2.04 mg/kg/d) pregabalin provided intrathecally or intranasally had less expression of CaVα2δ-1 at the dorsal horn as compared to rats receiving near-nerve delivery of pregabalin or with saline delivery for both models of diabetic peripheral neuropathy (DPN) (B) and spinal nerve ligation (SNL) (C). As compared to control rat specimens, the presence of either spinal nerve ligation or diabetes uniformly led to upregulation of CaVα2δ-1 at the dorsal horn. Unmatched ANOVA tests were performed between cohorts receiving pregabalin and saline for each intervention and condition, and between intervention locations, with * indicating significant difference (p < 0.0125 after Bonferroni corrections) between cohorts [n = 3-5 specimens for each cohort].
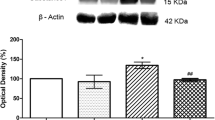
Dorsal spinal cord protein expression revealed by Western blotting for CaVα2δ-1 protein was elevated in the presence of diabetes or after spinal nerve ligation (Figure 5). In either model of neuropathic pain, delivery of intranasal or intrathecal high dose pregabalin was associated with less significant elevation of CaVα2δ-1 protein at the dorsal horn; meanwhile, near-nerve pregabalin delivery had no impact upon dorsal horn expression of CaVα2δ-1 protein. Similarly, diabetes and spinal nerve ligation led to elevation of CaVα2δ-1 protein at the dorsal root ganglia, greatest in smaller nociceptive neurons. However, no method of pregabalin delivery impacted upon CaVα2δ-1 cellular or axonal patterns of expression in the dorsal root ganglia or in peripheral nerve (Additional Files 2, 3).
Protein Quantification. Western blotting identified increased levels of CaVα2δ-1 protein in the dorsal horn and lumbar DRGs for rats exposed to either diabetic peripheral neuropathy (DPN) or spinal nerve ligation (SNL) when compared to control rats (A). In the dorsal spinal cord, either intrathecal or intranasal delivery of high dose (2.04 mg/kg/d) pregabalin was associated with less upregulation of CaVα2δ-1 protein than with saline delivery (B). The near nerve delivery of pregabalin failed to impact upon CaVα2δ-1 protein expression in the dorsal spinal cord following SNL or DPN (A, B). Finally, pregabalin delivered in any manner was not associated with impact upon CaVα2δ-1 protein expression in the lumbar DRGs exposed to either DPN or SNL when compared to saline delivery (intrathecal delivery results shown) (A). Multiple unmatched ANOVA tests were performed between cohorts receiving pregabalin and saline for each intervention and condition, with * indicating significant difference (p < 0.0125 after Bonferroni corrections) between cohorts. β-actin protein levels were used for quantification of ratios of CaVα2δ-1/β-actin demonstrated in B.
CaVα2δ-1 protein in the thalamus and primary sensory cortex of the brain from rats exposed to diabetic peripheral neuropathy, as well as in the contralateral thalamus and primary sensory cortex, was unchanged in the presence of neuropathic pain when compared to control rats without diabetes or traumatic nerve injury (Additional File 4).
Development of Increased Microglial Density and Activation in Diabetic Peripheral Neuropathy
Microglial quantification was performed in the dorsal thoracic and lumbar spinal cords in rats subjected to diabetes and spinal nerve ligation. There was an increased density of activated microglia in the dorsal spinal cord regions in rats in either neuropathic pain model when compared to control rat spinal cord specimens (Figure 6). There was no difference in microglial quantification in rats receiving saline or pregabalin with any method of delivery in either model of neuropathic pain.
Microglia Assessment. Microglia accumulation was assessed in the dorsal regions of thoracic and lumbar spinal cord for control rat spinal cord (A), and with either spinal nerve ligation (B) or diabetic peripheral neuropathy (C) in rats. Greater immunohistochemically-identified accumulation and activation of microglia in the dorsal spinal cord of rats was seen with spinal nerve ligation or diabetic peripheral neuropathy (D) without impact of pregabalin delivery.* indicates significant differences between control (sham SNL surgery or citrate-injected non-diabetic rat respectively) and all SNL and DPN rat values using individual unmatched ANOVA testing (p < 0.0125 after Bonferroni corrections). Bar = 50 μm.
Verification of Anterograde Trafficking of CaVα2δ-1 in Dorsal Root Ligation and Spinal Nerve Ligation
For rats undergoing spinal nerve ligation +/- dorsal root ligation, immunohistochemistry identified accumulation of CaVα2δ-1 at sites proximal to the ligatures placed at the dorsal root but not spinal nerve at 7 days after procedures were performed (Figure 7). Dorsal horn expression of CaVα2δ-1 was elevated with immunohistochemistry or with Western blotting with the absence of dorsal root ligation regardless of delivery of intrathecal pregabalin or saline; however, CaVα2δ-1 protein levels at the dorsal horn were suppressed in the presence of a dorsal root ligature. When dorsal root ligature was present, CaVα2δ-1 protein accumulation occurred at the portion of dorsal root distal (but not proximal) to dorsal root ligature, with partial suppression due to intrathecal pregabalin delivery. At the spinal root, the presence or absence of spinal nerve ligation did not influence expression of CaVα2δ-1 protein. These findings demonstrate the anterograde transport of CaVα2δ-1 protein away from the dorsal root ganglia after traumatic nerve injury.
Dorsal and Spinal Root Ligation Experiments. A cartoon diagram of the procedures performed in spinal nerve ligation and dorsal root ligation is provided. Tissues obtained after 7 days of spinal nerve ligation and/or dorsal root ligation (or neither in control rats) were examined using immunohistochemistry for CaVα2δ-1 protein and with Western blotting for CaVα2δ-1 protein and β-actin, used as a loading control. At the level of the dorsal horn (1), the presence of a dorsal root ligature prevented upregulation of CaVα2δ-1 protein regardless of delivery of intrathecal pregabalin or intrathecal saline. At the dorsal root proximal to dorsal root ligature (2), there was a similar lack of upregulation of CaVα2δ-1 protein regardless of delivery of intrathecal pregabalin or intrathecal saline, unless dorsal root ligature was absent. At the level of the dorsal root distal to spinal nerve ligation (3), there was an accumulation of CaVα2δ-1 protein, with less significant CaVα2δ-1 protein expression when intrathecal pregabalin was delivered or if spinal nerve ligation was absent. Distal to the dorsal root ganglia and proximal to spinal nerve ligation (4) or at the spinal root distal to spinal nerve ligation (5), there was no change in expression of CaVα2δ-1 protein irregardless of delivery of intrathecal pregabalin or intrathecal saline, or even if spinal nerve ligation was absent. Multiple unmatched ANOVA tests were performed between cohorts receiving intrathecal pregabalin and saline as compared to control rat samples and samples without placement of either spinal or dorsal root ligatures, with * indicating significant difference (p < 0.017 after Bonferroni corrections) between cohorts. Bars = 10 μm.
Discussion
Sufficient doses of pregabalin provided through intranasal or intrathecal methods ameliorated tactile allodynia and thermal hypersensitivity due to spinal nerve ligation or diabetic peripheral neuropathy, but there was no benefit of providing pregabalin at the level of the peripheral nerve in either condition, suggesting that pregabalin's benefit is localized to the dorsal root ganglia or its central projections. In particular, pregabalin provision intranasally or intrathecally led to partial reversal of upregulation of CaVα2δ-1 at the pre-synaptic nerve terminals in the dorsal horn of the spinal cord [7, 8]. This may relate to postulated central trafficking of the CaVα2δ-1 subunit from the dorsal root ganglia [23], and its potential block with pregabalin [23]. Pregabalin's effect appeared to be limited to impact upon trafficking of CaVα2δ-1 protein away from the dorsal root ganglia centrally.
Voltage-gated calcium channels are heterogenous multimeric complexes composed of several different subunits including α1, β, α2δ, and γ [43, 44]. The CaVα2δ-1 subunit is an auxiliary subunit which facilitates targeting and assembly of channels at the cell surface [45]. CACNA2D1 is transcribed as a single mRNA from a single gene, with the translated CaVα2δ-1 subunit protein cleaved to yield α2 and δ proteins; these are subsequently disulfide-linked and glycosylated, leading to functional CaVα2δ-1 subunits [46]. There are four different CaVα2δ-1 subunits: the CaVα2δ-1 subunit has been investigated most, due to its ability to serve as a ligand for the gabapentinoids [19]. Expression of CaVα2δ-1 in normal situations is essentially confined to neurons withn the brain, spinal cord, and DRG [11]. In the brain, CACNA2D1 mRNA is localized to regions important for cortical processing, primary sensory transmission, and arousal; however, we discovered no measurable changes in CaVα2δ-1 protein expression in the neuropathic pain states studied, regardless of mode of pregabalin delivery. Instead, the observed changes in CaVα2δ-1 protein expression at the dorsal horn appear to be of greater importance.
Upregulation of CaVα2δ-1 in the spinal cord is essential for both initiation and maintenance of neuropathic pain in many conditions [47]. Although upregulation of CaVα2δ-1 occurred in the DRG and at the dorsal root as well, there was no impact of pregabalin upon CaVα2δ-1 protein levels at these locations. In addition, elevation of CACNA2D1 mRNA in neuropathic pain states at the dorsal horn and dorsal root ganglia was not impacted by forms of pregabalin delivery. The isolated impact of pregabalin with lowering of elevated CaVα2δ-1 protein levels at the dorsal horn suggests a pregabalin-mediated prevention of an anterograde trafficking process [23]. However, no such distal anterograde trafficking of CaVα2δ-1 protein towards the periphery could be detected, and there was no accumulation of CaVα2δ-1 protein at the spinal nerve ligation as occurred at the ligature around the dorsal nerve root. The presence of a dorsal root ligature prevented the upregulation of CaVα2δ-1 protein levels at the dorsal horn, leading to accumulation of CaVα2δ-1 protein proximal to the dorsal root ligature, implicating CaVα2δ-1 protein trafficking away centrally from the dorsal root ganglia as an important factor in establishment of neuropathic pain, confirming the results of Bauer et al [23]. These same unmyelinated DRG sensory neurons important for nociception (unmyelinated C-fibers for transmission of thermal hyperalgesia and large-diameter Aδ afferent fibers for transmission of tactile allodynia [48, 49]) synapse in laminae I and II of the dorsal horn or at Lissauer's tract. We determined that CaVα2δ-1 immunoreactivity was elevated unilaterally with spinal nerve ligation and bilaterally with diabetic peripheral neuropathy in the dorsal column at thoracolumbar levels above the spinal nerve ligation injury, as well as in DRG neurons [23]. Most importantly, decreases in CaVα2δ-1 immunoreactivity at the dorsal horn were associated with successful analgesia of neuropathic pain with pregabalin. As a result, we hypothesize that pregabalin acts to slow or prevent the trafficking of the CaVα2δ-1 protein from the dorsal root ganglia to the dorsal horn.
The upregulation of CaVα2δ-1 within the spinal cord leads to increased presynaptic Ca2+ influx as well as neurotransmitter release, contributing to sensitization and neuropathic pain [50]. In models of neuropathic pain, the gabapentinoids gabapentin and pregabalin reduce neurotransmitter release and excitatory synaptic transmission at the spinal cord [51, 52]. Controversies regarding the mechanism of action of the gabapentinoids have occurred, with some studies demonstrating no acute effects of gabapentin upon spontaneous synaptic currents in lamina II neurons [53], while other studies have shown gabapentin's ability to reduce both inhibitory and excitatory neurotransmission at the dorsal horn by preferentially blocking only P/Q-type Ca2+ channels [54] and through prevention of intracellular endosome recycling of CaVα2δ-1 protein [55]. There has been no effect upon the levels of CaVα2δ-1 protein or CACNA2D1 mRNA elsewhere, though, such as at the DRG (despite the presence of all three CaVα2δ subunits at the DRG) [11] and in the peripheral nerve. This may signify that trafficking, but not expression or endocytosis, of CaVα2δ-1 from DRG to presynaptic terminals is impacted upon by pregabalin [23]. Other potential mechanisms for gabapentinoids include a more acute inhibitory effect upon neurotransmitter release [56] which may relate to upregulation of protein kinase C activation [57, 58].
Chronic, and not acute, pregabalin delivery reduces the neuronal expression of CaVα2δ-1 in vitro without impact upon constitutive endocytosis, leading to high levels of CaVα2δ-1 being present in intracellular vesicles [23]. This effect may be a consequence of calcium channel trafficking through the Von Willebrand factor-A domain in the α2 subunit of CaVα2δ-1 [59], as well as blocked CaVα2δ-1 trafficking [23, 60]. Transgenic mutants for CaVα2δ-1 unable to bind gabapentinoids have reduced trafficking, supporting this hypothesis [60, 61]. This hypothesized mechanism of disrupted trafficking would be unique, distinct from that of other pharmacotherapies with a direct neuronal surface action, and may relate to CaVα2δ-1 recycling at the endosome [55]. We postulate that the effects of pregabalin upon CaVα2δ-1 trafficking occurs peripherally, either at the dorsal root ganglia or the dorsal root.
There was no measureable impact of pregabalin delivered by any method upon the activation and accumulation of microglia in the spinal cord either. Ectopic activity contributing to sensitization due to excessive activity in voltage-gated calcium channels and voltage-gated sodium channel may occur within both A or C fibres [62–64], relating to neuronal hyperexcitability [14]. Microglia and their activation, leading to cytokine production important in neuropathic pain, are apparent and play a key role in pathophysiological pain due to both nerve injury [15–17] and diabetes [18]. Pregabalin's lack of impact upon these other established mechanisms for induction of neuropathic pain suggest its specificity at the CaVα2δ-1 subunit of the voltage-dependent calcium channel. However, other pathological changes associated with neuropathic pain continue abated despite pregabalin delivery.
There are limitations to the present study that require discussion. There is overlap between the tissues that receive pregabalin from intranasal and intrathecal delivery-it is probable that both forms of delivery impacted upon the dorsal horn region, but there may have been some undefined impact of intranasal pregabalin delivery at supraspinal locations as well (even with the lack of impact upon CaVα2δ-1 expression in brain). The absence of heightened CaVα2δ-1 expression at the supraspinal locations does not exclude the possibility of pregabalin possessing a role in supraspinal pain processing-it is possible that pregabalin may have important effects at the medulla and cortex [65–68]; this may explain the effects of intranasal pregabalin delivery, which only resulted in small concentrations of pregabalin in the lumbar spinal cord and dorsal root ganglia. Pregabalin's effects within the cerebrum may be contributory, especially after demonstrations of high cerebral concentratons of pregabalin following its intranasal delivery. Although we did not demonstrate impact of forms of delivery of pregabalin upon microglia accumulation in the spinal cord, it is possible that a longer duration of pregabalin delivery, or earlier onset of pregabalin delivery, is required before an anti-neuroinflammatory effect may be demonstrated, as shown in prior studies [36, 69, 70]. For example, in a model of diabetic peripheral neuropathy with daily gabapentin delivery over 5 days followed by immediate tissue harvesting, amelioration of spinal microglial activation occurred [71]. The levels of pregabalin achieved within the lumbar spinal cord and dorsal root ganglia were measured to be low after 74 hours-we hypothesize that pregabalin's effects may be summated over time rather than related to an absolute concentration achieved at one time point, although we cannot verify this. We did not study a systemic application such as with gastric/oral or intravenous forms of delivery due to the more widespread distribution of gabepentinoids with these methods and the presence of prior data assessing these delivery methods [72, 73]. The timing of pregabalin/saline delivery in the spinal nerve ligation experiments was initiated 7 days after the spinal nerve ligation occurred in order to provide intervention during the time of greatest neuropathic pain behavior; however, we selected the first 7 days after double ligation to study CACNA2D1 mRNA and CaVα2δ-1 protein expression during the time period of neuropathic pain behavior initiation-this difference in timing may have led to inconsistencies in molecular test results. Although we, and others [23], have hypothesized that pregabalin's mechanism of action is the disruption of CaVα2δ-1 trafficking, it remains unclear why expression of CACNA2D1 mRNA and CaVα2δ-1 protein is unaffected at the dorsal root ganglia. Together, these findings suggest that pregabalin's action is independent of translation of CaVα2δ-1 or transcription of CACNA2D1. As well, it would be anticipated that disrupted trafficking of CaVα2δ-1 would result in CaVα2δ-1 upregulation at the dorsal root ganglia, but this was not identified. However, pregabalin-mediated effects upon the presence of CaVα2δ-1 protein at the more proximal spinal nerve root and dorsal horn is indicative of pregabalin-induced suppression of the central trafficking of CaVα2δ-1 protein, as previously identified [23]. Rats received isoflurane anesthesia prior to intranasal delivery, but were tested for neuropathic pain at times that were many half lives after isoflurane discontinuation to avoid anesthetic effects upon behavioural testing. Finally, we designed this study to compare different modes of delivery of pregabalin upon neuropathic pain behaviours; however, our results cannot be used to interpret the site of action of pregabalin due to significant overlap in pregabalin's anatomical destinations with the intranasal and intrathecal delivery modes. However, we propose that a non-invasive form of pharmacologically targeting the dorsal horn and dorsal root ganglia, such as with intranasal delivery, may be a consideration for the management of human neuropathic pain.
Conclusions
The present data confirm the efficacy of pregabalin in the modulation of acute thermal and tactile hypersensitivity as features of neuropathic pain. The site of provision is critical, as exclusive delivery to the peripheral nerve has no impact; only delivery using intranasal or intrathecal delivery was associated with impact upon neuropathic pain behaviours and molecular outcomes. Furthermore, delivery using either intranasal or intrathecal methods led to diminished CaVα2δ-1 expression at the dorsal horn, suggesting that pregabalin inhibits central trafficking of the CaVα2δ-1 subunit from the dorsal root ganglia as previously shown by Bauer et al [23]. Future studies should examine the in vivo and in vitro trafficking of CaVα2δ-1 and the mechanisms by which pregabalin can influence the cell surface expression of CaVα2δ-1; another gabapentinoid, gabapentin, has been shown to prevent the recycling of the related protein CaVα2δ-2 from endosomes and the subsequent insertion in the plasma membrane without influencing internalization of CaVα2δ-2 [55]. The different impacts of pregabalin upon the central and peripheral axons of the dorsal root ganglia neurons are not yet understood. Finally, the intranasal delivery of agents such as pregabalin in order to directly target the nervous system may be a realistic method in managing neuropathic pain in humans, and may assist in the avoidance of systemic adverse effects.
Methods
Animals
All experiments were carried out using male Sprague Dawley rats (Charles River Laboratories), weighing 200-225 grams at initiation. Protocols were reviewed and approved by the University of Calgary Animal Care Committee using the Canadian Council of Animal Care guidelines. All attempts were made to minimize animal numbers and to maintain ethical standards. Experimental study groups were randomized and behavioural studies were performed by an experimenter who was unaware of treatment groups.
In all cases, rats were housed in plastic sawdust covered, pathogen-free cages with a normal light-dark cycle and free access to chow and water. Rats were anesthetized with pentobarbital (57 mg/kg) prior to all surgeries and terminal endpoints, and with inhaled isoflurane provided for all intranasal delivery points.
Sample size calculations
A sample size calculation for intervention groups was based upon an anticipated difference in neuropathic pain behavioral changes observed in treated diabetic rats to date, with an α of 0.05 and β of 0.5 providing a minimal sample size of n = 5 within each intervention group; this was increased to 6 rats due to anticipated minimal diabetes-related mortality over the three week study.
Radiolabeled Pregabalin Studies to Determine Localization of Delivery
We performed radiolabelled studies to determine the distribution of pregabalin reaching the central and peripheral nervous system after intranasal or intrathecal delivery. We examined the distribution of pregabalin at 1, 2, and 5 h after 72 hours of chronic delivery of intranasal, intrathecal, or near nerve delivery. A combination of 125I labeled pregabalin and unlabeled pregabalin was delivered during studies at the Alzheimer's Research Center at Regions Hospital in St. Paul, MN, USA. This procedure was approved by the Institutional Animal Care and Use Committee at Regions Hospital. Prior to experimentation, 30 diabetic rats (induced via streptozotocin injections, described below) were sedated for intranasal or intrathecal delivery using pentobarbital anesthesia (60 mg/kg). 125I-labelled pregabalin was provided to 10 rats via intrathecal delivery, to 10 rats via intranasal delivery, and to 10 rats via near nerve delivery (see below for procedure descriptions). Pregabalin (Pfizer Global, New York, New York) with an initial concentration 3.125 μg/μl was dissolved in PBS and custom labeled with 125I (GE Healthcare, Piscataway, New Jersey, USA). Synthesized radiolabelled pregabalin solution contained 266.7 μCi/μg. 125I-labeled pregabalin delivery (intranasal or intrathecal) was performed in a fume hood behind a lead impregnated shield, with anesthetized rats placed supine. A mixture of 125I pregabalin (19.2 μCr) and unlabeled pregabalin (30.0 μg) were administered intranasally or intrathecally. 125I pregabalin was intranasally administered over alternating nares as eight 6-μL drops with an Eppendorf pipetter every 2 minutes, for a total volume of 48 μL, provided twice daily for 3 days prior to a single delivery on day 4 as per a prior schedule [30]. For intrathecal delivery, 125I pregabalin was delivered using a placed intrathecal catheter (see below) attached to an Alzet pump (described below) with delivery of 2.04 mg/kg/d over 72 hours continually delivering a mixture of 125I pregabalin (19.2 μCr) and unlabeled pregabalin (30.0 μg) over each 24 hour period for 73, 74 and 75 hours prior to harvesting. For near nerve delivery, 125I pregabalin was delivered using a placed T-chamber around the proximal sciatic nerve (see below) attached to an Alzet pump (described below) with delivery of 2.04 mg/kg/d over 72 hours continually delivering a mixture of 125I pregabalin (19.2 μCr) and unlabeled pregabalin (30.0 μg) over each 24 hour period for 73, 74 and 75 hours prior to harvesting. Each desired dose contained a calculated radioactive dose of 40 μCi per day to best provide concentrations similar to the long-term duration experiments described below.
At each of 1, 2, and 5 hours after 72 hours of initiating either 125I pregabalin intranasal delivery, intrathecal or near-nerve delivery, cardiocentesis was performed for blood extraction. Euthanasia was performed via transcardial perfusion using 60 mL of saline, followed by 500 mL of 4% paraformaldehyde while the rat was maintained under anesthesia. To quantify 125I distribution, blood, urine, lymphatic (superficial perimandibular lymph nodes and cervical lymph nodes) and visceral organ structures (quadriceps muscle, kidney, liver, and lung), as well as portions of the central (olfactory bulb, anterior olfactory cortex, frontal cortex, caudate putamen, parietal cortex, temporal cortex, hippocampus, septum, thalamus, hypothalamus, midbrain, pons, medulla, cerebellum, cephalad cervical spinal cord, caudal spinal cord, mid-thoracic spinal cord, and lumbar spinal cord) and peripheral nervous systems (fifth lumbar dorsal root ganglia and proximal sciatic nerve) and associated tissues (ventral and dorsal cervical dura mater) were harvested. Gamma signal was recorded for each body region with autoradiographic imaging using a COBRA II Auto-Gamma Counter (Perkin-Elmer, Waltham, Mass., USA). Concentrations of 125I pregabalin were calculated based upon the gamma counting data, tissue weight, specific activity of the drug administered and standards measured. Results were studied for penetration into peripheral and central nervous system tissues.
To confirm patency and delivery with near-nerve chamber placement, infusion pumps containing India ink were implanted in four animals with pumps connected to the nerve regeneration chamber (see below). The presence of India ink was detected in all cases only at the catheter site for days 3, 7, and 14. Pump and catheter volume infusion rates were approximately 15 μl per day, or approximately 200 μl over 14 days for this India Ink experiment.
Models of Neuropathic Pain
Diabetes and Diabetic Peripheral Neuropathy
At the age of 1 month, rats intended to be diabetic were injected with streptozotocin diluted in sodium citratre (pH 4.7, Sigma, St. Louis, MO) intraperitoneally once daily for each of three consecutive days with doses of 60 mg/kg, 50 mg/kg, and then 40 mg/kg, while rats intended to be non-diabetic were injected with volume-matched carrier (sodium citrate pH 4.7) for three consecutive days. Streptozotocin ablates pancreatic β cells leading to an insulin deficient diabetic state. No insulin treatment was used at any time during the protocol. In all cases, whole blood glucose measurements were performed using puncture of the tail vein and a blood glucometer (OneTouch Ultra Meter, LifeScan Canada, Burnaby, BC, Canada). Hyperglycemia was verified 1 week after streptozotocin injections, with a fasting whole-blood glucose level of ≥ 16 mmol/l (normal 5-8 mmol/l) being our definition for experimental diabetes. Rats that did not meet these criteria for diagnosis of diabetes were excluded from further assessment. During diabetes, daily inspection of footpads occurred to rule out the presence of wounds or burns. No insulin or other diabetes therapies were provided during these investigations.
Spinal Nerve Ligation
Spinal nerve ligation was performed as described by Kim and Chung [74]. Briefly, the right L5/6 lumbar spinal nerves of male Sprague Dawley rats (200 g) were exposed in isoflurane/oxygen-anesthetised rats followed by tight ligation with 5.0 silk sutures between the DRG and proximal to the junctions forming the sciatic nerve. Sham operations were performed in the same way except that spinal nerves were not ligated. The left spinal nerves for all rats were left untouched.
Study Timelines, Animal Groups and Drug Administration
We conducted studies using the rat diabetic peripheral neuropathy model. A total of 118 male Sprague Dawley rats were induced diabetic while 26 male Sprague Dawley rats were studied as citrate-injected control littermates over the course of 3 weeks of diabetes without delivery of any other therapeutic agents other than pregabalin. Rates of conversion to diabetes were in excess of 95%. These rats were accustomized and studied with sensory behavioral testing every 3 days beginning prior to streptozotocin injections. Of the 118 rats receiving intraperitoneal streptozotocin injections, those with confirmed diabetes status were randomized to one of four varied dose groups within each of the intervention groups: 1) intranasal delivery with low dose (0.051 mg/kg/d) pregabalin (n = 9), medium dose (0.51 mg/kg/d) pregabalin (n = 8), or high dose (2.04 mg/kg/d) pregabalin (n = 8) or equal volumes of saline (n = 8); 2) intrathecal delivery with low dose (0.051 mg/kg/d) pregabalin (n = 9), medium dose (0.51 mg/kg/d) pregabalin (n = 8), or high dose (2.04 mg/kg/d) pregabalin (n = 8) or equal volumes of saline (n = 8); 3) near-nerve delivery with low dose (0.051 mg/kg/d) pregabalin (n = 9), medium dose (0.51 mg/kg/d) pregabalin (n = 8), or high dose (2.04 mg/kg/d) pregabalin (n = 8) or equal volumes of saline (n = 9). Those receiving intraperitoneal citrate injections (non-diabetic rats) were divided into intranasal (n = 7), intrathecal (n = 6) or near-nerve (n = 7) intervention groups. Following 7 days after diabetes confirmation occurred, intranasal, intrathecal, or near-nerve delivery began and occurred for a total of 14 continuous days. Neuropathic pain behaviors typically initiate within the first week after streptozotocin-induced diabetes confirmation [75], with initiation of interventions occurring 2 weeks after completion of streptozotocin injections.
We examined a model of traumatic neuropathy, the rodent spinal nerve ligation model. A total of 93 male Sprague Dawley rats received the spinal nerve ligation procedure while 23 male Sprague Dawley rats were studied as sham-operated control littermates. These rats were studied with sensory behavioral testing every 3 days beginning prior to spinal nerve ligation or sham procedure. The 93 rats receiving spinal nerve ligation were randomized to one of four varied dose groups within each of the intervention groups: 1) intranasal delivery with low dose (0.051 mg/kg/d) pregabalin (n = 7), medium dose (0.51 mg/kg/d) pregabalin (n = 6), or high dose (2.04 mg/kg/d) pregabalin (n = 6) or equal volumes of saline (n = 6); 2) intrathecal delivery with low dose (0.051 mg/kg/d) pregabalin (n = 7), medium dose (0.51 mg/kg/d) pregabalin (n = 6), or high dose (2.04 mg/kg/d) pregabalin (n = 6) or equal volumes of saline (n = 6); 3) near-nerve delivery with low dose (0.051 mg/kg/d) pregabalin (n = 7), medium dose (0.51 mg/kg/d) pregabalin (n = 6), or high dose (2.04 mg/kg/d) pregabalin (n = 6) or equal volumes of saline (n = 6). Those rats receiving sham procedures were divided into intranasal (n = 7), intrathecal (n = 7) or near-nerve (n = 6) intervention groups. Following 7 days after spinal nerve ligation occurred, intranasal, intrathecal, or near-nerve delivery began and occurred for a total of 14 continuous days. It has been demonstrated that spinal nerve ligation results in osnet of neuropathic pain behaviors in the first 1-7 days after spinal nerve injury [76].
Intranasal, Intrathecal and Near-Nerve Delivery Systems
Doses of pregabalin were selected based upon prior delivery of intrathecal gabapentin, a molecule similar to pregabalin but with less potency for the α2δ-1 subunit of voltage dependent calcium channels [52]. Intranasal pregabalin or saline delivery occurred twice daily during periods of therapy. Isoflurane-anesthetized rats were placed on their back with necks held in extension for intranasal administration, administered twice daily as ten 8 μl drops for a total volume of 80 μl for each nare, with delivery to alternating nares with 1-2 minutes between drops (i.e., ten drops per nare), for a total of 320 μl per day. Intrathecal delivery of pregabalin or saline occurred through Alzet mini osmotic pumps providing a continuous delivery of pregabalin/saline at a rate of 0.5 μl per hour. Near-nerve delivery of pregabalin or saline also occurred through Alzet mini osmotic pumps providing a continuous delivery of pregabalin/saline at a rate of 0.5 μl per hour. In each case where pregabalin was provided, there were three different total daily doses (low dose (0.2 μmol or 0.051 mg/kg), medium dose (2.0 μmol or 0.51 mg/kg), or high dose (8.0 μmol or 2.04 mg/kg)) of pregabalin delivered for each of the intervention systems.
For intrathecal delivery, we used the same surgical exposure as performed for spinal nerve ligation/sham, or created a new surgically exposed area over the right L5/6 spinal nerve region for diabetic/non-diabetic rats [10]. After incising the back skin at the lower lumbar region, a subcutaneous pouch was created. A silicone catheter (0.012 inches × 0.025 inches) was positioned into the lumbar intrathecal space in the created pouch between L6 and S1 vertebrae while connected to a two-week Alzet mini-osmotic infusion pump placed in the dorsal subcutaneous space of the rat. Alzet mini-osmotic infusion pumps are easily inserted into the subcutaneous space and require no external connections while providing a continuous, constant level of delivery to be provided over a two week period after placement. Surgical closure with 9-0 silk suture was performed around the subcutaneous pouch at the lumbar region. Surgeons were blinded as to the contents of the infusion pumps placed, which were randomized to either contain pregabalin (low (0.051 mg/kg/d), medium (0.51 mg/kg/d) or high dose (2.04 mg/kg/d)) or saline in a 3:1 ratio.
For near-nerve delivery, we used the same surgical exposure as performed for spinal nerve ligation/sham, or created a new surgically exposed area over the right spinal nerve region for diabetic/non-diabetic rats [10]. After incising the back skin at the lower lumbar region, a subcutaneous pouch was created. A two-week Alzet mini-osmotic infusion pump is placed in the dorsal subcutaneous pouch of the rodent. A nerve regeneration chamber is placed to surround the spinal nerve ligation site-the equipment used is composed of a 6.5 mm length (modified from 10 mm to compensate for the short distance of the spinal nerve) of silastic tubing (size: 1.98 mm inside diameter × 3.18 mm outside diameter; Dow Corning, Michigan). A small porthole cut into the side of the nerve chamber permits access with one end of a silicone catheter, with a small amount of cyanoacrylate cement (Instant Krazy Glue; Advanced Formula Gel; Elmer's Products Canada, Brampton, Ont.) placed at the outside of the end of the access tube before inserting it into the porthole. A second silastic tube (size: 0.76 mm inside diameter × 1.65 mm outside diameter) permits connection of the silicone catheter to the nerve regeneration chamber and Alzet pump on either end. Before their use, the entire apparatus used is autoclaved. Surgical closure with 9-0 silk suture was performed around the subcutaneous pouch and at the spinal nerve ligation exposure site. Surgeons were again blinded as to the contents of the infusion pumps placed, which were randomized to either contain pregabalin (low (0.051 mg/kg/d), medium (0.51 mg/kg/d) or high dose (2.04 mg/kg/d)) or saline in a 3:1 ratio.
During all intervention protocols, all animals were monitored post-operatively for signs of infection or other complications of surgery. For the purposes of data demonstration, Day 1 is considered to be the last of 7 days after either diabetes confirmation (for diabetic peripheral neuropathy studies) or 7 days after spinal nerve ligation occurred-this is one day before the start of interventions provided using intranasal, intrathecal, or near-nerve delivery of doses of pregabalin or saline. Note that after Day 15, no further intervention was provided prior to surgical harvesting on Day 18.
Sensory Behavioral Testing
In all cases, baseline testing was done beginning prior to streptozotocin injections or spinal nerve ligation procedure and ongoing testing continued until after pregabalin or saline delivery was completed, occurring every 3 days. During maintenance therapy, behavioural testing was performed at 2-4 hours after the morning dose of intranasal pregabalin/saline administration was delivered, during the time in which pregabalin concentrations were peaking in the nervous system. This latency period between dosing and behaviour testing was necessary to permit elimination of isoflurane anesthesia delivered during pregabalin/saline delivery and prevent confounding effects of anesthesia. A minimum of 1 hour was provided between forms of neuropathic pain behavior testing, and a minimum of 6 rats underwent behavioural testing at each time point.
Mechanical Sensitivity
Mechanical withdrawal thresholds were tested using a Dynamic Plantar Aesthesiometer (Ugo-Basile, Milan). In brief, rats were placed in clear acrylic boxes (22 × 16.5 × 14 cm) with a metal grid floor in a temperature controlled room (22°C) and were acclimatized for 15 minutes before testing. The stimulus was applied via a metal filament (0.5 mm) which applied a linearly increasing force ramp (2.5 g/s) to the middle of the plantar surface of the hind paw (within the sural nerve territory). A cut-off of 50 g was imposed to prevent any tissue damage. The force necessary to elicit a paw withdrawal was recorded. The paw withdrawal threshold was calculated as the average of three consecutive tests with at least 5 minutes between each test. Mechanical allodynia was defined as reduced threshold after induction of diabetes/spinal nerve ligation compared to a baseline paw withdrawal threshold.
Thermal Hyperalgesia
To quantitatively assess the thermal threshold of the hindpaw, rats were placed on the glass surface of a thermal testing apparatus with acclimatization for 15 minutes before testing. A mobile radiant heat source (Hargreaves apparatus) located under the glass was focused onto the middle of each of both individual hindpaws (within the sural nerve territory) for each rat for up to 60 seconds, with the latency (seconds) to withdrawal measured. Heating rate ramped from 30°C to 58°C over 60 seconds in consistent fashion on each occasion. The cutoff of 60 seconds was used to prevent potential tissue damage. Paws were inspected before and after thermal testing to ensure that no evidence of thermal damage was present. The mean withdrawal latency of both hindpaws from three consecutive trials was calculated as the thermal threshold. There were 5 minute intervals provided between each trial.
Locomotor Behavioral Testing
Potential changes in the locomotor function of the rats related to neuropathic pain states or therapeutic intervention were evaluated using Rotarod testing (Microprocessor Controlled Rota-Rod Treadmill for Rats, Model 57602, Ugo Basile, Italy). Acclimatization for walking on the revolving drum was performed over three training trials on the revolving drums at 10-15 rpm over three consecutive days. A maximum of 150 seconds for each Rotarod trial was used. The Rotarod performance time was measured at 1) 0.5, 1, 1.5, and 2 hours after intranasal pregabalin delivery following 72 hours of twice daily intranasal pregabalin/saline delivery; 2) 72 hours after initiation of intrathecal pregabalin/saline; and 3) 72 hours after near nerve pregabalin/saline delivery, each using varying doses of pregabalin (low (0.051 mg/kg/d), medium (0.51 mg/kg/d) or high dose (2.04 mg/kg/d)) or saline. The timepoint of after 72 hours was selected as occurring at least 5 half lives for pregabalin. Each test was performed at 5-10 minute intervals, and the average values obtained were compared. A total of 6 rats in each group were studied for locomotor activity, each of which had diabetes induced 7 days prior. A control rat cohort was studied 7 days after diabetes induction without any exposure to pregabalin or saline.
Surgical harvesting
Following 3 days after the last behavioral testing (4 days after completion of pregabalin/saline), rats from each cohort were sacrificed using pentobarbital intraperitoneal injections-a delay of four days was selected in order to assess the chronic effects of pregabalin delivery more than five half lives after pregabalin/saline termination, rather than detect any acute effects of pregabalin delivery. The following tissues were harvested from all rats: dorsal lumbar spinal cord from L2-S1 (left/right), bilateral lumbar DRG from L4-L6, bilateral sciatic nerves, bilateral sural nerves, and thalamus and primary sensory cortex (contralateral to injury in rats subjected to spinal nerve ligation). In diabetic/non-diabetic rats, left-sided tissues were placed in 2% Zamboni's fixative for later immunohistochemistry studies, while right-sided tissues were immediately fresh frozen at -80°C (Invitrogen, Burlington, ON) in liquid nitrogen and stored at -80°C for later protein/mRNA identification. In spinal nerve ligation/sham rats, right sided tissues ipsilateral to spinal nerve ligation were divided equally to be placed in 2% Zamboni's fixative or fresh frozen; left sided tissues were treated similarly.
Double Ligation Experiment
In order to examine the potential trafficking of CaVα2δ-1 anterograde from the dorsal root ganglia, an additional experiment was performed using two nerve ligations-spinal nerve ligation was performed along with ligature placed at the dorsal root. Although spinal nerve ligation forms a model of neuropathic pain, nerve ligation is also a well established method for determination of axonal protein trafficking, which when perturbed, leads to protein accumulation at the ligation site.
Twelve male Sprague Dawley rats had spinal nerve ligation performed (as above); another four rats had only dorsal root ligation performed (see below) another 4 rats had sham (used as controls) surgery performed (as above). Again, the right L5/6 lumbar spinal nerves of male Sprague Dawley rats (200 g) were exposed in isoflurane/oxygen-anesthetised rats followed by tight ligation with 5.0 silk sutures between the DRG and junctions forming the sciatic nerve. For the 4 rats receiving sham surgeries and for 8 of the 12 rats receiving spinal nerve ligation, ligation of the right L5/6 dorsal roots was performed as well using an additional laminectomy and dural splitting at the L4-6 intervertebral foramina in order to expose the dorsal roots. Dorsal root ligation was also performed in 4 rats not receiving spinal nerve ligation. Tight ligation with 5.0 silk sutures was performed for the dorsal root at the midpoint between the dorsal root ganglia and the dorsal horn of the spinal cord. Intrathecal delivery (see above) was performed for all 16 mice receiving procedures. Surgeons were blinded as to the contents of the infusion pumps placed, which were randomized to either contain pregabalin (high dose (8.0 μmol or 2.04 mg/kg)) or saline in a 1:1 ratio for those rats receiving both spinal and dorsal root nerve ligation procedures. For the 8 rats receiving one of spinal nerve ligation or dorsal root ligation, only intrathecal saline delivery was provided. Closure of the dura used 9-0 silk sutures, with surgical closure of the back of each rat performed otherwise as described above. Pregabalin or saline delivery began immediately post-surgery, considered day 0. The four rats with only a sham procedure performed were used as controls and did not receive any intrathecal delivery.
After 7 days, the following tissues were harvested from all rats: dorsal lumbar spinal cord from L2-S1 (left/right), dorsal root proximal to dorsal root ligature, dorsal root distal to dorsal root ligature, spinal nerve proximal to spinal nerve ligation, and spinal nerve distal to dorsal root ligature. The tissues from one rat from each group were used for immunohistochemistry studies to identify CaVα2δ-1 (see below), while the tissues from the other 3 rats in each group were placed in liquid nitrogen and stored at -80°C for later protein (CaVα2δ-1 and β-actin) identification. Due to the small amount of tissues obtained, pooling of tissues from the rats within each cohort was required for Western blot analysis (see below).
Immunohistochemistry
After spinal cord/DRG/peripheral nerve specimens were fixed in 2% Zamboni's fixative overnight at 4°C, they were washed in PBS, kept overnight in 25% sucrose PBS solution, and then embedded in optimal cutting temperature embedding solution (Tissue Tek, Sakura Finetek, USA), before storage at -80°C until sectioning. Cryostat transverse and longitudinal nerve sections (10 μm) were placed onto poly-l-lysine-and acetone-coated slides (SuperFrost Plus, Fisher Scientific, USA). Antigen retrieval was performed with slides placed in sodium citrate in an 80°C water bath, a PBS wash for 5 min, blocking with 10% goat serum for 1 h, and further PBS washing. In all cases, slides were incubated with primary antibody overnight at 4°C. After PBS washing, secondary fluorescent antibody was applied with incubation for 1 h at room temperature, followed by PBS washing and slide mounting. All immunohistochemistry was visualized using a Zeiss Imager Z1 (Zeiss, UK) fluorescence microscope. Calculation of the number of immunofluorescent profiles as well as the relative luminosity was performed using Adobe Photoshop (Adobe Photoshop 9.0, Adobe, San Jose, CA, 2005).
Primary antibodies used were to identify CaVα2δ-1 (1:200, produced in rabbit, Lifespan Biosciences, Seattle, WA), glial fibrillary acidic protein (GFAP) for Schwann cell identification (1:200, produced in mouse, Sigma Aldrich Canada, Oakville, ON), neurofilaments (NF) 200 for axon and neuron identification (1:100, produced in mouse, Santa Cruz, Santa Cruz, CA), goat anti-ionized calcium-binding adaptor molecule 1 (Iba-1) (1:1000, produced in goat, Abcam, Cambridge, MA) and microtubule associated protein-2 (MAP-2) (1:500, produced in rabbit, Sigma Aldrich Canada). Secondary antibodies used were either anti-rabbit IgG fluorescein isothiocyanate (FITC) labelled (1:100; Zymed, San Francisco, CA), donkey anti-goat IgG CY3 labeled (1:200, Fitzgerald Industries, Concord, MA), or goat anti-mouse IgG CY3 labeled (1:200, Sigma Aldrich Canada, Oakville, ON). Slides were cover-slipped with Vectashield mounting medium containing DAPI for nuclear identification (Vector Laboratories, CA, USA). Iba-1, MAP-2 and GFAP-positive cells were determined by counting the number of profiles (cell bodies) as described previously [10].
Samples of spinal cord/DRG/spinal nerve prepared for immunohistochemistry were examined for expression of CaVα2δ-1 along with identification of cell types (GFAP, MAP-2, NF200). For identification of microglia, isolated staining with Iba-1 was performed. Calculation of the number of immunofluorescent fibers as well as the luminosity of individual nerve fibers was performed for CaVα2δ-1. From each of the L4-6 DRGs, neurons were counted in six sections through the midportion of the DRG: total neuron numbers per transverse section, total numbers of neurons immunolabeled, and numbers of neurons with intense expression of channel of interest, as defined using two previously defined cutoff values with Adobe Photoshop [10]. Dorsal root ganglia neurons were counted as those with nuclear profiles that were visible in one section, but not in the subsequent section. Sensory neurons were differentiated from satellite and Schwann cells based on size and appearance as well as positive immunoprofiling for NF200. Total neuronal number was then calculated based upon the summation of neurons with newly identified nuclei identified through all sections of an individual dorsal root ganglion. All counting was performed with the microscopist masked to the identity of the experimental group. All of these measures were easily distinguished among immunolabeled neurons by a single examiner blinded to the identity of the groups using a calculated luminosity measurement of fluorescence for individual fibers examined under 400 × magnifications. Luminosity was classified as none-low (luminosity value of 0-150), moderate (150-250) or high (> 250) using Adobe Photoshop software (scale of 0-255 with arbitrary units).
Transverse sections through the lumbar spinal cord were also immunolabeled for channel expression with specific attention directed toward the dorsal horn. The relative fluorescence intensity was measured for each side ipsilateral and contralateral to spinal nerve ligation injury, or bilaterally for diabetic rats, at a pre-determined exposure time with a digital camera (Zeiss Axioscope, Zeiss) which provided an image of the entire spinal cord. Luminosity of each dorsal quadrant of the spinal cord was calculated using Adobe Photoshop [10].
In dorsal spinal cord regions within the thoracic and lumbar regions, the total numbers of microglia per transverse section were identified using Iba-1 immunohistochemistry. The lateral, central and medial dorsal horn regions, representing laminae 1-3, were examined. For cellular densities, a box measuring 104 μm2 was placed onto areas of dorsal horn for regions between T2 and S1. A quantitative estimate (proportional area) of changes in the activation state of microglial cells was performed [77–79] in the dorsal spinal cord based on atlas boundaries and after subtraction of background signal. A resting microglia was classified as having a small, compact soma with long, thin, ramified processes. Activated microglia, in contrast, exhibit marked cellular hypertrophy, and retracted processes with process length less than soma diameter. A total of 25 randomly chosen areas of dorsal spinal cord, from a minimum of 4 animals per cohort group, were examined for activated microglia quantification. All measurements were performed by a single examiner blinded to the group identity.
Western Immunoblotting
In order to determine protein expression for membrane bound channels, we performed Western blot with a specialized protocol to protect membrane structure. Tissue portions from the dorsal spinal cord, DRG, thalamus and primary sensory cortex were placed in chilled phosphate buffer solution (PBS), followed by centrifugation at 400 rpm. Tissue was ground down with a pestle in ice-cold lysis buffer (HEPES 15 mM at pH 7.9, 1.5 mM MgCl2, 10 mM KCl, 1 mM 2Na-EDTA, 5% glycerol, 0.5% Nonidet 40 (NP-40), 1 mM Phenylmethylsulfonyl Fluoride, Roche Mini-Complete Protease Inhibitors, and double distilled H20) at a ratio of 1 g of tissue: 10 ml of lysis buffer. Centrifugation of samples at 5,000 rpm followed for 15 minutes at 4°C. Supernatant was kept in tubes, and the pellet was homogenized again with half amounts of lysis buffer, followed by repeat centrifugation at 5,000 rpm for 15 minutes at 4°C. Both supernatants were transferred to ultracentrifuge rubes for repeat centrifuge at 26,000 rpm for 15 minutes at 4°C. Pellets were taken and resuspended in 25-50 μl of resuspension buffer (75 mM Tris at pH 7.4, 12.5 mM MgCl2, 5 mM 2Na-EDTA, 1.5% SDS, 0.1% Triton X-100, and double distilled H20) for protein quantification. These protein samples were then separated by SDS-PAGE techniques under conditions previously described [34]. Blots were probed for CaVα2δ-1 (1:4000, produced in rabbit, Lifespan Biosciences, Seattle, WA). For a loading control, anti-β-actin (1:100, Biogenesis Ltd. Poole, UK) was analyzed as well. Signal detection was performed by exposing of the blot to enhanced chemi-luminescent reagents (Amersham, GE Healthcare, USA) and captured on Kodac X-OMAT K film. In each case, 4-6 blots were performed for each protein of interest from different rats in each cohort. Blots were analyzed with Adobe Photoshop (Adobe Photoshop 9.0, Adobe, San Jose, CA, 2005) for semi-quantification of blotting density [34].
Quantitative Reverse-Transcriptase Polymerase Chain Reactions
Total RNA was extracted from peripheral nerve and spinal cord regions using Trizol reagent (Invitrogen). Total RNA (1 μg) was processed directly to cDNA synthesis using the Superscript II Reverse Transcriptase® system (Invitrogen). CACNA2D1 primers were: Forward 5'-GAAAGGCTTTAGCTTCGCGTTT-3', Reverse 5'-TCTCTCTTCTCCTCCATCCGTG-3' [GenBank: NM_001110847.1]. For a housekeeping gene, ribosomal protein (large P0) (RPLP0) was used, with primers: Forward 5'-TACCTGCTCAGAACACCGGTCT-3', Reverse 5'-GCACATCGCTCAGGATTTCAA-3' [GenBank: NM_022402.2]. qRT-PCR was done using SYBR Green dye. All reactions were performed in duplicate in an ABI PRISM 7000 Sequence Detection System. Data were calculated by the 2-ΔΔCT method and are presented as the fold induction of mRNA for the specific target in diabetic tissues normalized to RPLP0 and compared to control tissues (defined as 1.0-fold). The 2-ΔΔCT method is a standard technique to permit analysis of quantitiative real-time polymerase chain reaction results; this is an approximation technique which assumes similar efficiencies of reactions for both standard and target genes, permitting comparisons of differences in messenger RNA quantities following polymerase chain reactions after baseline measurements.
Data analysis
The presence of multiple doses of pregabalin within each intervention led to the use of two-way repeated unmatched ANOVA measurements followed by Tukey's test were performed for analysis of behavioural studies. We chose to analyze behavioral data based upon comparison of either low (0.051 mg/kg/d), medium (0.51 mg/kg/d) or high dose (2.04 mg/kg/d) pregabalin intervention with either the sham group (for spinal nerve ligation) or non-diabetic cohort group as appropriate. For immunohistochemistry, we chose to analyze the tissues from rodents receiving the highest doses for each intervention with those tissues from rats receiving placebo using mean values-therefore, one-way unmatched ANOVA followed by Tukey's test was used for immunohistochemistry analysis as well. Data collected in the groups were expressed as mean ± standard error in all cases. For immunohistochemistry comparisons demonstrated as low/medium/high intensity, the individual values were compared using unmatched ANOVA testing. Bonferroni corrections were applied as appropriate in all cases.
Authors' information
CT is the Director of the Neuropathic Pain Clinic, University of Calgary and is a Clinician-Scientist. WHF is the Director of the Alzheimer's Research Centre at the University of Minnesota and is the patent owner for delivery of intranasal compounds for neurodegenerative conditions.
Abbreviations
- ANOVA:
-
Analysis of variance
- CaVα2δ-1:
-
The Alpha-2 Delta-1 protein subunit of the calcium channel
- CACNA2D1:
-
The gene and mRNA for the Alpha-2 Delta-1 subunit of the calcium channel
- DRG:
-
Dorsal root ganglia
- EDTA:
-
Ethylenediaminetetraacetic acid
- FITC:
-
Fluorescein isothiocyanate
- GFAP:
-
Glial fibrillary acidic protein neurofilaments
- HEPES:
-
4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid
- HRP:
-
Horseradish peroxidase
- Iba-1:
-
Ionized calcium binding adaptor molecule 1
- MAP-2:
-
Microtubule associated protein-2
- NF200:
-
Neurofilament 200
- PBS:
-
Phosphate Buffered Solution
- PMSF:
-
Phenylmethanesulphonylfluoride
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis.
References
Dickenson AH, Matthews EA, Suzuki R: Neurobiology of neuropathic pain: mode of action of anticonvulsants. Eur J Pain 2002,6(Suppl A):51–60.
Toth C, Lander J, Wiebe S: The prevalence and impact of chronic pain with neuropathic pain symptoms in the general population. Pain Med 2009, 10: 918–929. 10.1111/j.1526-4637.2009.00655.x
Bouhassira D, Lanteri-Minet M, Attal N, Laurent B, Touboul C: Prevalence of chronic pain with neuropathic characteristics in the general population. Pain 2008, 136: 380–387. 10.1016/j.pain.2007.08.013
Bennett MI, Bouhassira D: Epidemiology of neuropathic pain: can we use the screening tools? Pain 2007, 132: 12–13. 10.1016/j.pain.2007.09.003
Davies A, Hendrich J, Van Minh AT, Wratten J, Douglas L, Dolphin AC: Functional biology of the alpha(2)delta subunits of voltage-gated calcium channels. Trends Pharmacol Sci 2007, 28: 220–228. 10.1016/j.tips.2007.03.005
Newton RA, Bingham S, Case PC, Sanger GJ, Lawson SN: Dorsal root ganglion neurons show increased expression of the calcium channel alpha2delta-1 subunit following partial sciatic nerve injury. Brain Research Mollecular Brain Research 2001, 95: 1–8. 10.1016/S0169-328X(01)00188-7
Wang H, Sun H, Della Penna K, Benz RJ, Xu J, Gerhold DL, Holder DJ, Koblan KS: Chronic neuropathic pain is accompanied by global changes in gene expression and shares pathobiology with neurodegenerative diseases. Neuroscience 2002, 114: 529–546. 10.1016/S0306-4522(02)00341-X
Li CY, Song YH, Higuera ES, Luo ZD: Spinal dorsal horn calcium channel alpha2delta-1 subunit upregulation contributes to peripheral nerve injury-induced tactile allodynia. J Neurosci 2004, 24: 8494–8499. 10.1523/JNEUROSCI.2982-04.2004
Li CY, Zhang XL, Matthews EA, Li KW, Kurwa A, Boroujerdi A, Gross J, Gold MS, Dickenson AH, Feng G, Luo ZD: Calcium channel alpha2delta1 subunit mediates spinal hyperexcitability in pain modulation. Pain 2006, 125: 20–34. 10.1016/j.pain.2006.04.022
Toth C, Brussee V, Martinez JA, McDonald D, Cunningham FA, Zochodne DW: Rescue and regeneration of injured peripheral nerve axons by intrathecal insulin. Neuroscience 2006, 139: 429–449. 10.1016/j.neuroscience.2005.11.065
Cole RL, Lechner SM, Williams ME, Prodanovich P, Bleicher L, Varney MA, Gu G: Differential distribution of voltage-gated calcium channel alpha-2 delta (alpha2delta) subunit mRNA-containing cells in the rat central nervous system and the dorsal root ganglia. JComp Neurol 2005, 491: 246–269. 10.1002/cne.20693
Woolf CJ: Excitability changes in central neurons following peripheral damage. In Hyperalgesia and Allodynia Edited by: Willis Jr WD. 1991, 221–243.
Wen XJ, Li ZJ, Chen ZX, Fang ZY, Yang CX, Li H, Zeng YM: Intrathecal administration of Cav3.2 and Cav3.3 antisense oligonucleotide reverses tactile allodynia and thermal hyperalgesia in rats following chronic compression of dorsal root of ganglion. Acta Pharmacol Sin 2006, 27: 1547–1552. 10.1111/j.1745-7254.2006.00461.x
Khosravani H, Altier C, Simms B, Hamming KS, Snutch TP, Mezeyova J, McRory JE, Zamponi GW: Gating effects of mutations in the Cav3.2 T-type calcium channel associated with childhood absence epilepsy. J Biol Chem 2004, 279: 9681–9684.
Zhuang ZY, Wen YR, Zhang DR, Borsello T, Bonny C, Strichartz GR, Decosterd I, Ji RR: A peptide c-Jun N-terminal kinase (JNK) inhibitor blocks mechanical allodynia after spinal nerve ligation: respective roles of JNK activation in primary sensory neurons and spinal astrocytes for neuropathic pain development and maintenance. J Neurosci 2006, 26: 3551–3560. 10.1523/JNEUROSCI.5290-05.2006
Scholz J, Woolf CJ: The neuropathic pain triad: neurons, immune cells and glia. Nat Neurosci 2007, 10: 1361–1368. 10.1038/nn1992
Watkins LR, Maier SF: Glia: a novel drug discovery target for clinical pain. Nat Rev Drug Discov 2003, 2: 973–985. 10.1038/nrd1251
Tsuda M, Ueno H, Kataoka A, Tozaki-Saitoh H, Inoue K: Activation of dorsal horn microglia contributes to diabetes-induced tactile allodynia via extracellular signal-regulated protein kinase signaling. Glia 2008, 56: 378–386. 10.1002/glia.20623
Gee NS, Brown JP, Dissanayake VU, Offord J, Thurlow R, Woodruff GN: The novel anticonvulsant drug, gabapentin (Neurontin), binds to the alpha2delta subunit of a calcium channel. JBiol Chem 1996, 271: 5768–5776. 10.1074/jbc.271.10.5768
Field MJ, Oles RJ, Singh L: Pregabalin may represent a novel class of anxiolytic agents with a broad spectrum of activity. Br J Pharmacol 2001, 132: 1–4. 10.1038/sj.bjp.0703794
Brown JP, Dissanayake VU, Briggs AR, Milic MR, Gee NS: Isolation of the [3H]gabapentin-binding protein/alpha 2 delta Ca2+ channel subunit from porcine brain: development of a radioligand binding assay for alpha 2 delta subunits using [3H]leucine. Anal Biochem 1998, 255: 236–243. 10.1006/abio.1997.2447
Field MJ, Cox PJ, Stott E, Melrose H, Offord J, Su TZ, Bramwell S, Corradini L, England S, Winks J, Kinloch RA, Hendrich J, Dolphin AC, Webb T, Williams D: Identification of the alpha2-delta-1 subunit of voltage-dependent calcium channels as a molecular target for pain mediating the analgesic actions of pregabalin. Proc Natl Acad Sci USA 2006, 103: 17537–17542. 10.1073/pnas.0409066103
Bauer CS, Nieto-Rostro M, Rahman W, Tran-Van-Minh A, Ferron L, Douglas L, Kadurin I, Sri Ranjan Y, Fernandez-Alacid L, Millar NS, Dickenson AH, Lujan R, Dolphin AC: The increased trafficking of the calcium channel subunit alpha2delta-1 to presynaptic terminals in neuropathic pain is inhibited by the alpha2delta ligand pregabalin. J Neurosci 2009, 29: 4076–4088. 10.1523/JNEUROSCI.0356-09.2009
Toth C, Brussee V, Zochodne DW: Remote neurotrophic support of epidermal nerve fibres in experimental diabetes. Diabetologia 2006, 49: 1081–1088. 10.1007/s00125-006-0169-8
Reger MA, Craft S: Intranasal insulin administration: a method for dissociating central and peripheral effects of insulin. Drugs Today (Barc) 2006, 42: 729–739. 10.1358/dot.2006.42.11.1007675
Frey W: Neurologic Agents for Nasal Administration to the Brain. WIP Organization, editor Book Title, Vol. Volume. City: Publisher, Year. p.^pp. Pages
Hanson LR, Frey WH: Intranasal delivery bypasses the blood-brain barrier to target therapeutic agents to the central nervous system and treat neurodegenerative disease. BMC Neurosci 2008,9(Suppl 3):S5. 10.1186/1471-2202-9-S3-S5
Dhuria SV, Hanson LR, Frey WH: Intranasal delivery to the central nervous system: Mechanisms and experimental considerations. J Pharm Sci 2009.
Valiveti S, Agu RU, Hammell DC, Paudel KS, Earles DC, Wermeling DP, Stinchcomb AL: Intranasal absorption of Delta(9)-tetrahydrocannabinol and WIN55,212–2 mesylate in rats. Eur J Pharm Biopharm 2007, 65: 247–252. 10.1016/j.ejpb.2006.08.009
Thorne RG, Pronk GJ, Padmanabhan V, Frey WHn: Delivery of insulin-like growth factor-I to the rat brain and spinal cord along olfactory and trigeminal pathways following intranasal administration. Neuroscience 2004, 127: 481–496. 10.1016/j.neuroscience.2004.05.029
Reger MA, Watson GS, Green PS, Wilkinson CW, Baker LD, Cholerton B, Fishel MA, Plymate SR, Breitner JC, DeGroodt W, Mehta P, Craft S: Intranasal insulin improves cognition and modulates beta-amyloid in early AD. Neurology 2008, 70: 440–448. 10.1212/01.WNL.0000265401.62434.36
Francis GJ, Martinez JA, Liu WQ, Xu K, Ayer A, Fine J, Tuor UI, Glazner G, Hanson LR, Frey WH, Toth C: Intranasal insulin prevents cognitive decline, cerebral atrophy and white matter changes in murine type I diabetic encephalopathy. Brain 2008, 131: 3311–3334. 10.1093/brain/awn288
Alcala-Barraza SR, Lee MS, Hanson LR, McDonald AA, Frey WH, McLoon LK: Intranasal delivery of neurotrophic factors BDNF, CNTF, EPO, and NT-4 to the CNS. J Drug Target 2009.
Francis G, Martinez J, Liu W, Nguyen T, Ayer A, Fine J, Zochodne D, Hanson LR, Frey WH, Toth C: Intranasal Insulin Ameliorates Experimental Diabetic Neuropathy. Diabetes 2009, 58: 934–945. 10.2337/db08-1287
Huge V, Lauchart M, Magerl W, Schelling G, Beyer A, Thieme D, Azad SC: Effects of low-dose intranasal (S)-ketamine in patients with neuropathic pain. Eur J Pain 2009.
Toth CC, Jedrzejewski NM, Ellis CL, Frey WH: Cannabinoid-mediated modulation of neuropathic pain and microglial accumulation in a model of murine type I diabetic peripheral neuropathic pain. Mol Pain 2010, 6: 16. 10.1186/1744-8069-6-16
Scranton RA, Fletcher L, Sprague S, Jimenez DF, Digicaylioglu M: The rostral migratory stream plays a key role in intranasal delivery of drugs into the CNS. PLoS ONE 2011, 6: e18711. 10.1371/journal.pone.0018711
Gajraj NM: Pregabalin: its pharmacology and use in pain management. Anesth Analg 2007, 105: 1805–1815. 10.1213/01.ane.0000287643.13410.5e
Wilson-Gerwing TD, Verge VM: Neurotrophin-3 attenuates galanin expression in the chronic constriction injury model of neuropathic pain. Neuroscience 2006, 141: 2075–2085. 10.1016/j.neuroscience.2006.05.056
Clark AK, Yip PK, Grist J, Gentry C, Staniland AA, Marchand F, Dehvari M, Wotherspoon G, Winter J, Ullah J, Bevan S, Malcangio M: Inhibition of spinal microglial cathepsin S for the reversal of neuropathic pain. Proc Natl Acad Sci USA 2007, 104: 10655–10660. 10.1073/pnas.0610811104
Toth C, Martinez JA, Liu WQ, Diggle J, Guo GF, Ramji N, Mi R, Hoke A, Zochodne DW: Local erythropoietin signaling enhances regeneration in peripheral axons. Neuroscience 2008, 154: 767–783. 10.1016/j.neuroscience.2008.03.052
Marcol W, Kotulska K, Larysz-Brysz M, Kowalik JL: BDNF contributes to animal model neuropathic pain after peripheral nerve transection. Neurosurg Rev 2007, 30: 235–243. discussion 243 10.1007/s10143-007-0085-5
Hofmann F, Lacinova L, Klugbauer N: Voltage-dependent calcium channels: from structure to function. Rev Physiol Biochem Pharmacol 1999, 139: 33–87. 10.1007/BFb0033648
Catterall WA: Structure and regulation of voltage-gated Ca2+ channels. Annu Rev Cell Dev Biol 2000, 16: 521–555. 10.1146/annurev.cellbio.16.1.521
Klugbauer N, Marais E, Hofmann F: Calcium channel alpha2delta subunits: differential expression, function, and drug binding. J Bioenerg Biomembr 2003, 35: 639–647.
Jay SD, Sharp AH, Kahl SD, Vedvick TS, Harpold MM, Campbell KP: Structural characterization of the dihydropyridine-sensitive calcium channel alpha 2-subunit and the associated delta peptides. J Biol Chem 1991, 266: 3287–3293.
Boroujerdi A, Kim HK, Lyu YS, Kim DS, Figueroa KW, Chung JM, Luo ZD: Injury discharges regulate calcium channel alpha-2-delta-1 subunit upregulation in the dorsal horn that contributes to initiation of neuropathic pain. Pain 2008, 139: 358–366. 10.1016/j.pain.2008.05.004
Sun H, Ren K, Zhong CM, Ossipov MH, Malan TP, Lai J, Porreca F: Nerve injury-induced tactile allodynia is mediated via ascending spinal dorsal column projections. Pain 2001, 90: 105–111. 10.1016/S0304-3959(00)00392-4
Pitcher GM, Henry JL: Nociceptive response to innocuous mechanical stimulation is mediated via myelinated afferents and NK-1 receptor activation in a rat model of neuropathic pain. Exp Neurol 2004, 186: 173–197. 10.1016/j.expneurol.2003.10.019
Snutch TP: Targeting chronic and neuropathic pain: the N-type calcium channel comes of age. NeuroRx 2005, 2: 662–670. 10.1602/neurorx.2.4.662
Patel MK, Gonzalez MI, Bramwell S, Pinnock RD, Lee K: Gabapentin inhibits excitatory synaptic transmission in the hyperalgesic spinal cord. Br J Pharmacol 2000, 130: 1731–1734. 10.1038/sj.bjp.0703530
Coderre TJ, Kumar N, Lefebvre CD, Yu JS: Evidence that gabapentin reduces neuropathic pain by inhibiting the spinal release of glutamate. J Neurochem 2005, 94: 1131–1139. 10.1111/j.1471-4159.2005.03263.x
Moore KA, Baba H, Woolf CJ: Gabapentin-- actions on adult superficial dorsal horn neurons. Neuropharmacology 2002, 43: 1077–1081. 10.1016/S0028-3908(02)00226-5
Bayer K, Ahmadi S, Zeilhofer HU: Gabapentin may inhibit synaptic transmission in the mouse spinal cord dorsal horn through a preferential block of P/Q-type Ca2+ channels. Neuropharmacology 2004, 46: 743–749. 10.1016/j.neuropharm.2003.11.010
Tran-Van-Minh A, Dolphin AC: The alpha2delta ligand gabapentin inhibits the Rab11-dependent recycling of the calcium channel subunit alpha2delta-2. J Neurosci 2010, 30: 12856–12867. 10.1523/JNEUROSCI.2700-10.2010
Davies A, Douglas L, Hendrich J, Wratten J, Tran Van Minh A, Foucault I, Koch D, Pratt WS, Saibil HR, Dolphin AC: The calcium channel alpha2delta-2 subunit partitions with CaV2.1 into lipid rafts in cerebellum: implications for localization and function. J Neurosci 2006, 26: 8748–8757. 10.1523/JNEUROSCI.2764-06.2006
Maneuf YP, McKnight AT: Block by gabapentin of the facilitation of glutamate release from rat trigeminal nucleus following activation of protein kinase C or adenylyl cyclase. Br J Pharmacol 2001, 134: 237–240. 10.1038/sj.bjp.0704227
Zhang Y, Helm JS, Senatore A, Spafford JD, Kaczmarek LK, Jonas EA: PKC-induced intracellular trafficking of Ca(V)2 precedes its rapid recruitment to the plasma membrane. J Neurosci 2008, 28: 2601–2612. 10.1523/JNEUROSCI.4314-07
Canti C, Nieto-Rostro M, Foucault I, Heblich F, Wratten J, Richards MW, Hendrich J, Douglas L, Page KM, Davies A, Dolphin AC: The metal-ion-dependent adhesion site in the Von Willebrand factor-A domain of alpha2delta subunits is key to trafficking voltage-gated Ca2+ channels. Proc Natl Acad Sci USA 2005, 102: 11230–11235. 10.1073/pnas.0504183102
Hendrich J, Van Minh AT, Heblich F, Nieto-Rostro M, Watschinger K, Striessnig J, Wratten J, Davies A, Dolphin AC: Pharmacological disruption of calcium channel trafficking by the alpha2delta ligand gabapentin. Proc Natl Acad Sci USA 2008, 105: 3628–3633. 10.1073/pnas.0708930105
Heblich F, Tran Van Minh A, Hendrich J, Watschinger K, Dolphin AC: Time course and specificity of the pharmacological disruption of the trafficking of voltage-gated calcium channels by gabapentin. Channels (Austin) 2008, 2: 4–9. 10.4161/chan.2.1.6045
Boucher TJ, Okuse K, Bennett DL, Munson JB, Wood JN, McMahon SB: Potent analgesic effects of GDNF in neuropathic pain states. Science 2000, 290: 124–127. 10.1126/science.290.5489.124
Wu G, Ringkamp M, Hartke TV, Murinson BB, Campbell JN, Griffin JW, Meyer RA: Early onset of spontaneous activity in uninjured C-fiber nociceptors after injury to neighboring nerve fibers. Journal of Neuroscience 2001, 21: RC140.
Djouhri L, Koutsikou S, Fang X, McMullan S, Lawson SN: Spontaneous pain, both neuropathic and inflammatory, is related to frequency of spontaneous firing in intact C-fiber nociceptors. J Neurosci 2006, 26: 1281–1292. 10.1523/JNEUROSCI.3388-05.2006
You HJ, Lei J, Arendt-Nielsen L: Selective inhibitory effects of pregabalin on peripheral C but not A-delta fibers mediated nociception in intact and spinalized rats. Neuroscience 2009, 164: 1845–1853. 10.1016/j.neuroscience.2009.09.047
Suzuki R, Rahman W, Rygh LJ, Webber M, Hunt SP, Dickenson AH: Spinal-supraspinal serotonergic circuits regulating neuropathic pain and its treatment with gabapentin. Pain 2005, 117: 292–303. 10.1016/j.pain.2005.06.015
Tanabe M, Takasu K, Takeuchi Y, Ono H: Pain relief by gabapentin and pregabalin via supraspinal mechanisms after peripheral nerve injury. J Neurosci Res 2008, 86: 3258–3264. 10.1002/jnr.21786
Takasu K, Kinoshita Y, Ono H, Tanabe M: Protein kinase A-dependence of the supraspinally mediated analgesic effects of gabapentin on thermal and mechanical hypersensitivity. J Pharmacol Sci 2009, 110: 223–226. 10.1254/jphs.09091SC
Tawfik VL, Nutile-McMenemy N, Lacroix-Fralish ML, Deleo JA: Efficacy of propentofylline, a glial modulating agent, on existing mechanical allodynia following peripheral nerve injury. Brain Behav Immun 2007, 21: 238–246. 10.1016/j.bbi.2006.07.001
Svensson CI, Marsala M, Westerlund A, Calcutt NA, Campana WM, Freshwater JD, Catalano R, Feng Y, Protter AA, Scott B, Yaksh TL: Activation of p38 mitogen-activated protein kinase in spinal microglia is a critical link in inflammation-induced spinal pain processing. J Neurochem 2003, 86: 1534–1544. 10.1046/j.1471-4159.2003.01969.x
Wodarski R, Clark AK, Grist J, Marchand F, Malcangio M: Gabapentin reverses microglial activation in the spinal cord of streptozotocin-induced diabetic rats. Eur J Pain 2009, 13: 807–811. 10.1016/j.ejpain.2008.09.010
Gustafsson H, Sandin J: Oral pregabalin reverses cold allodynia in two distinct models of peripheral neuropathic pain. Eur J Pharmacol 2009, 605: 103–108. 10.1016/j.ejphar.2009.01.014
Ling B, Coudore F, Decalonne L, Eschalier A, Authier N: Comparative antiallodynic activity of morphine, pregabalin and lidocaine in a rat model of neuropathic pain produced by one oxaliplatin injection. Neuropharmacology 2008, 55: 724–728. 10.1016/j.neuropharm.2008.06.007
Kim SH, Chung JM: An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 1992, 50: 355–363. 10.1016/0304-3959(92)90041-9
Messinger RB, Naik AK, Jagodic MM, Nelson MT, Lee WY, Choe WJ, Orestes P, Latham JR, Todorovic SM, Jevtovic-Todorovic V: In vivo silencing of the Ca(V)3.2 T-type calcium channels in sensory neurons alleviates hyperalgesia in rats with streptozocin-induced diabetic neuropathy. Pain 2009, 145: 184–195. 10.1016/j.pain.2009.06.012
Calvo M, Zhu N, Tsantoulas C, Ma Z, Grist J, Loeb JA, Bennett DL: Neuregulin-ErbB signaling promotes microglial proliferation and chemotaxis contributing to microgliosis and pain after peripheral nerve injury. J Neurosci 30: 5437–5450.
Popovich PG, Wei P, Stokes BT: Cellular inflammatory response after spinal cord injury in Sprague-Dawley and Lewis rats. J Comp Neurol 1997, 377: 443–464. 10.1002/(SICI)1096-9861(19970120)377:3<443::AID-CNE10>3.0.CO;2-S
Hains BC, Waxman SG: Activated microglia contribute to the maintenance of chronic pain after spinal cord injury. J Neurosci 2006, 26: 4308–4317. 10.1523/JNEUROSCI.0003-06.2006
Zhao P, Waxman SG, Hains BC: Extracellular signal-regulated kinase-regulated microglia-neuron signaling by prostaglandin E2 contributes to pain after spinal cord injury. J Neurosci 2007, 27: 2357–2368. 10.1523/JNEUROSCI.0138-07.2007
Acknowledgements
This project was supported by an unrestricted grant from Pfizer Global. Pregabalin used in this work was generously donated by Pfizer Global.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
CT receives salary support from the Alberta Heritage Foundation for Medical Research, and also receives research operating funds from the Juvenile Diabetes Research Foundation, The University of Calgary, The University of Calgary Department of Clinical Neurosciences, Pfizer Global, Valeant Canada, and Talecris Canada. CT has received honoraria for medical education seminars and consultancy from Pfizer Global, Valeant Canada, and Talecris Canada.
Authors' contributions
CT conceived of the study, participated in its design and coordination and drafted the manuscript. CT also performed the surgeries and molecular studies related to the project. JM and MK carried out the delivery of agents and performed behavioral testing in all cases, and performed surgeries for harvesting of tissues. LH and WF participated in study design, assisted in performance of radiolabelled studies, and helped to edit the manuscript. All authors read and approved the final manuscript.
An erratum to this article can be found online at 10.1186/1744-8069-10-20.
A retraction note to this article can be found online at http://dx.doi.org/10.1186/1744-8069-10-20.
Electronic supplementary material
12990_2011_460_MOESM1_ESM.TIFF
Additional file 1: Supplementary Figure 1- Pregabalin Detection. Concentrations of pregabalin within specific regions of the brain and spinal cord in the hours after delivery of either intranasal or intrathecal pregabalin were measured beginning at 72 hours after initiation of either therapy. Intranasal (A) pregabalin delivery led to peak tissue concentrations of pregabalin at about the 74 hour timepoint, 2 hours after most recent intranasal delivery before falling at the 77 hour timepoint; intranasal delivery led to highest concentrations in the cerebral structures. Intrathecal pregabalin delivery (B) led to concentrations that were essentially stable over time and greatest in the lumbar cord region. Doses provided through either intranasal or intrathecal delivery were comparable to medium doses (0.51 mg/kg/d) of pregabalin in later subexperiments. [n = 3-4 rats in each rat cohort for each time point]. (TIFF 667 KB)
12990_2011_460_MOESM2_ESM.TIFF
Additional file 2: Supplementary Figure 2-CaVα 2 δ-1 nerve immunohistochemistry. Co-localized immunohistochemistry pictures for CaVα2δ-1 demonstrated for sciatic peripheral nerve of control (non-diabetic) rat (A) or diabetic rat (B), and spinal nerve of control (no injury) rat (C) or spinal nerve nerve distal to spinal nerve ligation (D). Axonal fibers are demonstrated with neurofilament 200 (NF200) (red), Schwann cells with glial fibrillary acidic protein (GFAP) (green), and CaVα2δ-1 presence is also demonstrated (blue). Axons expressing CaVα2δ-1 appear purple-blue after co-localization of images. A bar graph demonstrates a greater number of CaVα2δ-1 positive sciatic fibers in diabetic nerve with diabetic peripheral neuropathy (DPN) (E) and in spinal nerve at or distal to spinal nerve ligation (SNL) when compared to control rat nerve (F). There was no impact of pregabalin interventions upon expression of CaVα2δ-1 within the peripheral nerve itself distal to the site of traumatic injury when compared to saline delivery (values are mean ± S.E.M., unmatched ANOVA testing between intervention groups, * indicates a significant difference between the control rat group as compared to each of the intervention groups receiving either pregabalin or saline (p < 0.0125 after Bonferroni corrections). Bar = 10 μm. (TIFF 7 MB)
12990_2011_460_MOESM3_ESM.TIFF
Additional file 3: Supplementary Figure 3-CaVα 2 δ-1 dorsal root ganglia immunohistochemistry. Co-localized immunohistochemistry pictures demonstrating DRG neurons of control (non-diabetic) rat (A), diabetic rat (B), control (no injury) rat (C), and DRG ipsilateral to spinal nerve ligation (D). Neurons are demonstrated with neurofilament 200 (NF200) (red), while CaVα2δ-1 presence is also demonstrated (green). Neurons expressing CaVα2δ-1 appear yellow-green after co-localization of images. A bar graph demonstrates a greater number of medium-high CaVα2δ-1 expressing DRG neurons ipsilateral to spinal nerve ligation (SNL) (E) and in diabetic DRG neurons with diabetic peripheral neuropathy (DPN) (F). There was no impact of pregabalin interventions upon expression of CaVα2δ-1 within the DRG neurons themselves when compared with saline interventions (values are mean ± S.E.M., unmatched ANOVA testing between intervention groups, * indicates a significant difference between the control rat group as compared to each of the intervention groups receiving pregabalin or saline (p < 0.0125 after Bonferroni corrections). Bar = 10 μm. (TIFF 618 KB)
12990_2011_460_MOESM4_ESM.TIFF
Additional file 4: Supplementary Figure 4-Central nervous system CaVα 2 δ-1 protein detection. Western blotting identified no increase in levels of CaVα2δ-1 protein in either the thalamus or sensory cortex for rats exposed to either diabetic peripheral neuropathy (DPN) or spinal nerve ligation (SNL) when compared to control rats. Levels of CaVα2δ-1 protein in the thalamus and sensory cortex were also unchanged with either intrathecal or intranasal delivery of high dose (2.04 mg/kg/d) pregabalin or with saline delivery. β-actin was used as a loading control as displayed. (TIFF 191 KB)
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Martinez, J.A., Kasamatsu, M., Rosales-Hernandez, A. et al. Retracted: Comparison of central versus peripheral delivery of pregabalin in neuropathic pain states. Mol Pain 8, 3 (2012). https://doi.org/10.1186/1744-8069-8-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1744-8069-8-3