Abstract
Background
Chromogenic in situ hybridization (CISH) is fast becoming a well established technique for easy and sensitive determination of HER2 gene status in breast cancer. However, for the chromogenic method to achieve status as a safe and reliable technique, the method needs to be validated against already known and validated FISH techniques.
Methods
Here it is reported from a comparative study where HER2 gene status obtained by HER2 CISH pharmDx™ Kit was compared to HER2 gene status obtained by the FDA approved HER2 FISH pharmDx™ Kit and the PathVysion HER-2 DNA probe Kit. The study included 365 formalin fixed and paraffin-embedded invasive breast cancer tissue specimens collected consecutively at a US reference laboratory.
Results
The data obtained revealed an overall HER2 status concordance of approximately 98% for comparisons of HER2 CISH pharmDx™ Kit to both HER2 FISH pharmDx™ Kit and PathVysion HER-2 DNA Probe Kit.
Conclusions
The concordance between results obtained using the recently FDA approved HER2 CISH pharmDx™ Kit with previously FDA approved FISH techniques for HER2 gene status determination indicate that the HER2 CISH pharmDx™ Kit is a reliable chromogenic alternative to fluorescence-based methods.
Similar content being viewed by others
Background
HER2 (Human Epidermal Growth Factor Receptor 2) is an important marker for invasive breast cancer. The assessment of the HER2 expression level is routinely done by examining protein expression and/or gene expression levels in formalin-fixed and paraffin-embedded (FFPE) histological sections. Overexpression of HER2 protein and/or HER2 (Human Epidermal Growth Factor Receptor 2 Gene) amplification is observed in approximately 22% of human breast cancers [1] and has been shown to be a marker of poor prognosis [2] and to predict benefit from treatment with the antibody based drug Herceptin (Genentech, San Francisco, CA, USA) [3].
Tissue based assessment of HER2 protein expression levels is commonly achieved using immunohistochemistry (IHC), whereas tissue based analysis of HER2 amplification is mostly done by in situ hybridization (ISH) techniques either fluorescence (FISH) [4] or chromogenic (CISH or SISH) [5]. In ISH the specific recognition of HER2 target sequences in the nuclei of tumor cells is done by fluorescence- or hapten-labeled sequence pairing probes. Implementation of CISH for determination of HER2 amplification in breast cancer has some advantages compared to FISH based detection [6–9]. Chromogenic signals do not fade over time and can therefore be archived and used for re-evaluation or retrospective studies. Furthermore, chromogenic visualization enables bright-field microscopy and easy access to tissue morphology to directly determine the appropriate tumor area for evaluation.
To implement CISH in the anatomical pathological laboratories for determination of HER2 status in breast cancer the technique needs to be safe and reliable [10]. One way to demonstrate this is to compare results obtained by the CISH method against results obtained from already known and validated HER2 FISH techniques. In this paper data is reported from the comparison of 365 breast cancer specimens using a new dual color HER2 CISH method (HER2 CISH pharmDx™ Kit, Dako Denmark A/S, Glostrup, Denmark) with two well established and FDA approved HER2 FISH techniques; HER2 FISH pharmDx™ Kit (Dako Denmark A/S, Glostrup, Denmark) and PathVysion HER-2 DNA Probe Kit (Abbot Laboratories, Illinois, USA).
Methods
Specimens
The study included 365 FFPE invasive breast cancer tissue specimens with known fixation history (10% neutral buffered formalin, 18-24 h). The specimens were collected consecutively at a US reference laboratory and the first 304 specimens were included irrespective of HercepTest™ (Dako Denmark A/S, Glostrup, Denmark) IHC score and additional 61 specimens were included based on a IHC HER2 2+ score as determined by HercepTest™ Serial sections (5 μm) were cut from each specimen and stained with H&E, HercepTest™ for HER2 protein expresion, HER2 CISH pharmDx™ Kit (Dako Denmark A/S), HER2 FISH pharmDx™ Kit (Dako Denmark A/S) and PathVysion HER-2 DNA Probe Kit (Abbott Laboratories, Illinois, USA). Specimens were not individually identifiable and it was impossible to trace the identity of the patients. The study was performed in accordance with the current version of the World Medical Association Declaration of Helsinki and approval from an Institutional Review Board (Copernicus Group, Inc.) was granted prior to study start. Evaluation of specimens were performed by three different technologists for the three ISH tests and subsequently reviewed by the pathologists. One pathologist reviewed test results obtained with PathVysion HER-2 DNA Probe Kit and HER2 CISH pharmDx™ Kit with several months in between and another pathologist reviewed test results obtained with HER2 FISH pharmDx™ Kit. Knowledge of test results was not shared between technologists or between pathologists.
CISH testing
HER2 CISH staining was performed according to the manufactures instructions (Dako Denmark A/S) at the US reference laboratory. In short, specimens were subjected to heat-pre-treatment (microwave oven) and pepsin digestion at 37°C to prepare the tissue for probe hybridization. Denaturation for 5 min at 82°C and over-night hybridization at 45°C were performed simultaneously for the HER2/Texas Red labeled DNA probe and the CEN-17/FITC labeled PNA probe using a Hybridizer (Dako Denmark A/S). Specimens were subjected to stringent wash at 65°C for 10 min before transfer to a CISH wash buffer. The signals from the fluorescent probes were converted to chromogenic signals in an IHC staining reaction performed on an automated platform (Autostainer Plus, Dako Denmark A/S). The immunohistochemical staining included blocking of endogeneous peroxidase activity, incubation with horseradish peroxidase conjugated anti-FITC and alkaline phosphatase conjugated anti-Texas Red antibodies followed by development of chromogenic signals using red and blue chromogens. The slides were counterstained with hematoxylin and mounted in a permanent mounting medium.
HER2 CISH stained slides were interpreted using a bright field microscope with 40× and 60× objectives. The HER2/CEN-17 ratio was calculated based on the enumeration of 20 nuclei from the invasive tumor area. Based on ratio, the specimens were categorized into amplified (HER2/CEN-17 2.0) or non-amplified (HER2/CEN-17 < 2.0) categories. Specimens with a ratio between 1.8 and 2.2 (borderline cases) were subjected to additional enumeration of 20 nuclei and the ratio was then recalculated for the 40 nuclei to determine if amplification was present or not. Normal cells within the specimen served as an internal control for staining success. Normal cells should exhibit the ratio expected for normal diploid cells with a one to one relationship of red and blue signals.
FISH testing
HER2 FISH pharmDx™ was performed according to the manufacturer's instructions at Dako Denmark A/S and FISH using PathVysion HER-2 DNA Probe Kit was performed according to an internally validated procedure at the US reference laboratory.
HER2 status
In accordance with FDA approved guidelines for determination of HER2 status HER2/CEN-17 ratios obtained by CISH and FISH were translated to a HER2 gene status of amplified when the HER2/CEN-17 ratio was higher than or equal to 2.0 or non-amplified when the HER2/CEN-17 ratio was below 2.0.
IHC testing
All specimens were tested at the US reference laboratory using HercepTest™ as per the manufacturer's instructions to determine the IHC HER2 score.
Statistics
IBM SPSS Statistics were used for statistical work (SPSS Inc., Chicago, IL). Agreement calculations were reported with 95% confidence limits based on the binomial distribution using equal tailed Jeffreys prior intervals [11] as calculated by the PROPOR plug-in.
Results
HER2 immunohistochemistry scores of specimens included
A total of 365 breast cancer specimens were included in this investigation. An overview of the HER2 IHC scores obtained from HercepTest™ staining is provided in Table 1 for the entire population and for the specimens sampled consecutively. Among the 304 consecutively collected specimens 10.5% were HER2 3+, 18.8% were HER2 2+ and the remaining 70.8% were HER2 0 or 1+. In a recent meta-analysis the median percentage of specimens in the IHC 2+ and IHC 3+ category were found at 12.0% and 16.2% [12] which indicate a low percentage of IHC 3+ specimens in this investigation.
Frequencies of amplified and non-amplified specimens
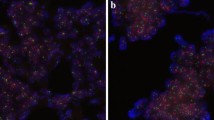
Frequencies of HER2 amplified and non-amplified test results found by HER2 CISH, HER2 FISH and PathVysion FISH are presented in Table 2 for all specimens. For the consecutively collected specimens only 10.8% of specimens with a successful test result were amplified by HER2 CISH, 11.4% were amplified by HER2 FISH and 11.0% were amplified by PathVysion FISH (data not shown). Figure 1 includes images illustrating a non-amplified and a cluster amplified breast cancer specimen stained by HER2 CISH pharmDx™ Kit. In both panels tumor cells having distinctive blue dots are observed corresponding to the reference CEN-17 probe signals (Figure 1). In the non-amplified specimen (right panel) single red dots corresponding to the HER2 signals are apparent. The amplified specimen (left panel) have cluster amplification in which red signals are overlapping, but some single red signals are also visible in some tumor cells.
HER2 status agreement
As indicated in Table 2, 13 specimens lack a HER2 CISH result and seven specimens lack a HER2 FISH result. Three of these specimens are overlapping and therefore, 348 specimens were eligible for comparison between HER2 CISH and HER2 FISH. Agreement calculations revealed an overall agreement of 98.3% (CI95: 96.5%; 99.3%) with positive agreement of 93.2% (CI95: 82.9%; 98.0%) and negative agreement of 99.0% (CI95: 97.4%; 99.7%) when comparing HER2 CISH and HER2 FISH (Table 3). The Kappa value was found at 0.92 [13]. McNemar's test for a systematic bias between HER2 CISH and HER2 FISH revealed a non-significant two-tailed p value of 1.00 showing that no bias was present [14]. Three of the six discordant cases for the comparison between HER2 CISH and HER2 FISH (Table 3) were HercepTest™ IHC 2+ equivocal cases, and the remaining three were 0, 1+ and 3+, respectively. The discordant cases had a HER2/CEN-17 ratio very close to or within the borderline area defined from 1.8 and 2.2 for at least one of the three methods performed. As indicated in Table 2, there are four specimens that lack a PathVysion FISH result and two of these specimens are overlapping with HER2 CISH resulting in agreement calculations based on a total of 350 specimens for this comparison. The overall HER2 status agreement between HER2 CISH and PathVysion FISH was 97.7% (CI95: 95.7%; 98.9%). Positive agreement was 90.9% (CI95: 79.8%; 96.9%) and negative agreement was 98.7% (CI95: 96.9%; 99.6%) (Table 4). The Kappa value was found at 0.90 [13]. McNemar's test for a systematic bias between HER2 CISH and PathVysion FISH revealed a non-significant two-tailed p value of 1.00 indicating the absence of bias. Five of the eight discordant cases for the comparison between HER2 CISH and PathVysion FISH (Table 4) were HercepTest™ IHC 2+ equivocal cases, and the remaining three were 3+. For the three IHC 3+ discordant cases cluster amplification of blue (CEN-17) signals was observed that covered red (HER2) signals or made them difficult to see. In these cases the blue signals in the normal cells surrounding the tumor cells were clear and distinct and cases could therefore pass the quality control. Therefore, in cases with cluster amplification of blue signals additional caution should be taken during interpretation and results from other test methods such as IHC or FISH should be included before a final HER2 status is given.
Success rates
Final success rates were determined after allowing for two staining runs. Of the 13 cases with a missing HER2 CISH test result (Table 2) the second staining run was not performed in four cases and, therefore, the success rate for HER2 CISH was 97.5% (352/361*100). The success rates for HER2 FISH and PathVysion FISH were 98.4 and 98.9%, respectively.
Exploratory analysis of copy numbers and ratio
Exploratory analysis of HER2/CEN-17 ratios was performed by plotting ratios obtained by HER2 CISH as a function of ratios obtained by HER2 FISH (Figure 2) or PathVysion FISH (Figure 3). The plot of the ratio data revealed a good correlation between the HER2 CISH ratio and the ratios obtained by the two HER2 FISH assays. Since the data showed heteroscedasticity with apparent elevated variances at higher ratios linear regression was not carried out. The plots of HER2/CEN-17 ratios in Figure 2 and Figure 3 also reveal that lower HER2 CISH ratios are observed at ratios above 3.0 compared to ratios obtained by FISH. During the exploratory analysis mean HER2/CEN-17 ratios and mean HER2 and CEN-17 signal copy numbers (normalized to 20 nuclei) were calculated for all three assays and tabulated for comparison for all the valid specimens (Table 5). In order to evaluate HER2 CISH assay performance closer to the cut-off this data were also presented for all valid specimens having a CISH HER2/CEN-17 ratio below 3.0 (Table 6). Generally the standard deviations on copy numbers and HER2/CEN-17 ratios for HER2 CISH seemed lower compared to HER2 FISH and PathVysion FISH (Table 5 and Table 6). The comparison also revealed that significantly higher HER2/CEN-17 ratios and higher CEN-17 signal copy numbers were observed for HER2 CISH compared to HER2 FISH and PathVysion FISH, whereas no significant difference were observed between HER2 signals (Table 5). Interestingly, the examination of cases with a HER2/CEN-17 ratio below 3.0 revealed that significant higher HER2 and CEN-17 signal copy numbers were obtained with HER2 CISH whereas HER2/CEN-17 ratios were identical (Table 6).
Evaluation time
During the study, the evaluation time for each specimen was recorded for HER2 CISH and PathVysion FISH assays allowing for a comparison of evaluation times. The mean evaluation time for HER2 CISH was 3:12 (min:sec) compared to 4:02 for PathVysion FISH (data not shown). This difference was statistically significant using a two-tailed, paired t-test (n = 350, p < 0.001).
Discussion
Previously published studies have reported high concordance between HER2 FISH and HER2 CISH performed in breast cancer specimens [7, 15–19], however, larger studies that validate the use of HER2 CISH have not yet been presented. In the current investigation 365 primary breast cancer specimens were included and an overall HER2 status agreement close to 98% is reported when comparing test results obtained by HER2 CISH pharmDx™ Kit to results obtained by HER2 FISH pharmDx™ and PathVysion HER-2 FISH DNA Probe Kit. The study population was enriched for HercepTest™ IHC 2+ specimens because most testing modalities pass on such equivocal specimens to a genetic test. For overall agreement the lower 95% confidence interval limits were at or above 96% in the two comparisons, further stressing the reliability of the HER2 CISH pharmDx™ in this comparison to the two FISH analysis methods.
The number of HER2 amplified or HER2 positive consecutively collected specimens reported in this study by the three ISH assays (10.8%, 11.4%, 11.0%) and by HercepTest™ IHC (10.5%) seems to be lower than expected based on previously published data [1] in which the overall HER2 positivity rate was found at 22% with observations ranging from 9%-74%. However, Ross et al. (2009) [1] indicate that the true HER2 positivity rate probably is in the range 15-20%, with national reference labs and community hospitals reporting lower rates and tertiary hospitals and cancer centers reporting slightly higher rates.
In the current data analysis a significantly higher average CEN-17 signal counts in HER2 CISH compared to HER2 FISH and PathVysion FISH with no significant difference in HER2 signal counts between the three assays were observed. This resulted in significantly lower HER2/CEN-17 ratios observed for HER2 CISH. There was no clinical diagnostic impact of this change as the concordance agreement calculations revealed very fine agreement between HER2 CISH and the two FISH assays. In support of the good agreement the analysis of test results for specimens having a HER2/CEN-17 CISH ratio below 3.0 revealed that identical ratios were obtained for HER2 CISH, HER2 FISH and PathVysion FISH. Surprisingly, average copy numbers for both HER2 and CEN-17 were higher for the HER2 CISH method, whereas, HER2 CISH standard deviations seemed to be lower for the average HER2/CEN-17 ratio, HER2 and CEN-17 copy numbers compared to PathVysion FISH. This could be interpreted as the HER2 CISH assay being the most sensitive assay.
Also, there was a tendency towards differences between HER2/CEN-17 ratios in highly amplified specimens between HER2 CISH and FISH assays (see Figure 2 and 3). This is probably due to the variation in cluster estimation and is not a problem in relation to achieving the correct diagnosis.
As a parallel study, the slide evaluation time was compared between HER2 CISH and PathVysion FISH and as could be expected the average evaluation time for a CISH staining was significantly shorter. This is most likely due to the easy access to the morphological information and hence a faster selection of areas for enumeration. In HER2 CISH a higher number of specimens were reported failed in comparison to staining with PathVysion FISH. This is likely to be due to CISH being a new technique implemented in the US reference laboratory and PathVysion FISH being a well established test method at this site. With respect to HER2 CISH the normal cells in the tissue surrounding the tumor area can be used as internal control securing good staining quality and preventing wrong diagnosis being based on a failed slide.
Conclusion
From this study based on HER2 CISH and FISH data from 365 different primary breast cancer specimens it is confirmed that the FDA approved HER2 CISH pharmDx™ Kit is a reliable chromogenic alternative to today's FDA approved FISH techniques for HER2 gene status determination in FFPE breast carcinoma specimens.
References
Ross JS, Slodkowska EA, Symmans WF, Pusztai L, Ravdin PM, Hortobagyi GN: The HER-2 receptor and breast cancer: ten years of targeted anti-HER-2 therapy and personalized medicine. Oncologist. 2009, 14 (4): 320-368. 10.1634/theoncologist.2008-0230.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL: Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987, 235 (4785): 177-182. 10.1126/science.3798106.
Ross JS, Fletcher JA: The HER-2/neu oncogene in breast cancer: prognostic factor, predictive factor, and target for therapy. Stem Cells. 1998, 16 (6): 413-428. 10.1002/stem.160413.
Kallioniemi OP, Kallioniemi A, Kurisu W, Thor A, Chen LC, Smith HS, Waldman FM, Pinkel D, Gray JW: ERBB2 amplification in breast cancer analyzed by fluorescence in situ hybridization. Proc Natl Acad Sci USA. 1992, 89 (12): 5321-5325. 10.1073/pnas.89.12.5321.
Tanner M, Gancberg D, Di LA, Larsimont D, Rouas G, Piccart MJ, Isola J: Chromogenic in situ hybridization: a practical alternative for fluorescence in situ hybridization to detect HER-2/neu oncogene amplification in archival breast cancer samples. Am J Pathol. 2000, 157 (5): 1467-1472. 10.1016/S0002-9440(10)64785-2.
Bhargava R, Lal P, Chen B: Chromogenic in situ hybridization for the detection of HER-2/neu gene amplification in breast cancer with an emphasis on tumors with borderline and low-level amplification: does it measure up to fluorescence in situ hybridization?. Am J Clin Pathol. 2005, 123 (2): 237-243. 10.1309/C4PEBGB9LN830TVL.
Arnould L, Denoux Y, MacGrogan G, Penault-Llorca F, Fiche M, Treilleux I, Mathieu MC, Vincent-Salomon A, Vilain MO, Couturier J: Agreement between chromogenic in situ hybridisation (CISH) and FISH in the determination of HER2 status in breast cancer. Br J Cancer. 2003, 88 (10): 1587-1591. 10.1038/sj.bjc.6600943.
Lambros MB, Natrajan R, Reis-Filho JS: Chromogenic and fluorescent in situ hybridization in breast cancer. Hum Pathol. 2007, 38 (8): 1105-1122. 10.1016/j.humpath.2007.04.011.
Ross JS, Fletcher JA, Bloom KJ, Linette GP, Stec J, Symmans WF, Pusztai L, Hortobagyi GN: Targeted therapy in breast cancer: the HER-2/neu gene and protein. Molecular Cell Proteomics. 2004, 3 (4): 379-398. 10.1074/mcp.R400001-MCP200.
Arena V, Pennacchia I, Vecchio FM, Carbone A: "CISH the FISH" for HER2: our laboratory experience. Am J Clin Pathol. 2010, 134 (2): 347-348. 10.1309/AJCPVH1IGEG8CPXB.
Brown LD, Cai T, DasGupta A: Interval Estimation for a Binomial Proportion. Stat Sci. 2001, 16 (2): 101-133.
Dendukuri N, Khetani K, McIsaac M, Brophy J: Testing for HER2-positive breast cancer: a systematic review and cost-effectiveness analysis. CMAJ. 2007, 176 (10): 1429-1434. 10.1503/cmaj.061011.
Cohen J: A Coefficient of Agreement for Nominal Scales. Educ Psychol Meas. 1960, 20 (1): 37-46. 10.1177/001316446002000104.
Dwyer AJ: Matchmaking and McNemar in the comparison of diagnostic modalities. Radiology. 1991, 178 (2): 328-330.
García-Caballero T, Grabau D, Green AR, Gregory J, Schad A, Kohlwes E, Ellis IO, Watts S, Mollerup J: Determination of HER2 amplification in primary breast cancer using dual-color chromogenic in situ hybridization is comparable to fluorescence in situ hybridization: a European multicenter study involving 168 specimens. Histopathology. 2010, 56: 472-480. 10.1111/j.1365-2559.2010.03503.x.
Pedersen M, Rasmussen BB: The correlation between dual-color chromogenic in situ hybridization and fluorescence in situ hybridization in assessing HER2 gene amplification in breast cancer. Diagn Mol Pathol. 2009, 18 (2): 96-102. 10.1097/PDM.0b013e31817f5227.
Hoff K, Jorgensen JT, Muller S, Rongaard E, Rasmussen O, Schonau A: Visualization of FISH Probes by dual-color chromogenic in situ hybridization. Am J Clin Pathol. 2009, 133 (2): 205-211.
Kato N, Itoh H, Serizawa A, Hatanaka Y, Umemura S, Osamura RY: Evaluation of HER2 gene amplification in invasive breast cancer using a dual-color chromogenic in situ hybridization (dual CISH). Pathol Int. 2010, 60 (7): 510-515. 10.1111/j.1440-1827.2010.02553.x.
Dandachi N, Dietze O, Hauser-Kronberger C: Chromogenic in situ hybridization: a novel approach to a practical and sensitive method for the detection of HER2 oncogene in archival human breast carcinoma. Lab Invest. 2002, 82 (8): 1007-1014.
Pre-publication history
The pre-publication history for this paper can be accessed here:http://www.biomedcentral.com/1472-6890/12/3/prepub
Acknowledgements
We thank USLABS in Irvine for a successful and professional collaboration. Jan Trøst Jørgensen is acknowledged for excellent scientific help.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
JM, UH, SM and AS are employees at Dako Denmark A/S.
Authors' contributions
The clinical trial was planned by UH, AS and JM. Study protocols were written by JM with help from UH and AS. Training of the US reference laboratory was performed by UH and SM. Data analysis was performed by JM and JM wrote the manuscript with help from UH, SM and AS. All authors read and approved the final manuscript
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Mollerup, J., Henriksen, U., Müller, S. et al. Dual color chromogenic in situ hybridization for determination of HER2 status in breast cancer: a large comparative study to current state of the art fluorescence in situ hybridization. BMC Clin Pathol 12, 3 (2012). https://doi.org/10.1186/1472-6890-12-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1472-6890-12-3