Abstract
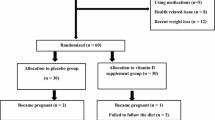
Dietary fish oil restores ovarian function in subfertile rats, which is thought to be associated with decreased transcription of follicle-stimulating hormone (FSH) β-subunit. We have previously demonstrated a reduction in early follicular serum FSH levels in normal weight but not obese women after treatment with omega-3 polyunsaturated fatty acids (PUFA). Herein, we report the effect of supplementation with omega-3 PUFA on urinary reproductive hormones across the whole menstrual cycle. This interventional study included 17 eumenorrheic women, aged 24–41 years. One month of daily morning urine was collected before and after 1 month of omega-3 PUFA supplementation with 4 g of eicosapentaenoic acid and docosahexaenoic acid daily. Measurements included urinary FSH, luteinizing hormone (LH) and estrogen and progesterone metabolites, plasma fatty acid composition, and markers of endoplasmic reticulum stress. Compliance with dietary supplementation was verified by significantly reduced ratios of omega-6 to omega-3 PUFA for all subjects after treatment (P <.01). After 1 month of omega-3 PUFA supplementation, urinary FSH was significantly decreased in normal weight, but not obese women, in both follicular and luteal phases (-28.4% and -12.6%, respectively, both P=.04). No significant changes were seen in LH or sex steroids for either weight group. The selective and specific decrease in FSH suggests that omega-3 PUFA supplementation merits further investigation in normal weight women with decreased fertility and/or diminished ovarian reserve.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Clarke TC, Black LI, Stussman BJ, Barnes PM, Nahin RL. Trends in the use of complementary health approaches among adults: United States, 2002-2012. Natl Health Stat Report. 2015;79:1–16.
Palmsten K, Flores KF, Chambers CD, Weiss LA, Sundaram R, Buck Louis GM. Most frequently reported prescription medications and supplements in couples planning pregnancy: the LIFE study. Reprod Sci (Thousand Oaks, Calif). 2018;25(1):94–101.
Nehra D, Le HD, Fallon EM, et al. Prolonging the female reproductive lifespan and improving egg quality with dietary omega-3 fatty acids. Aging Cell. 2012;11(6):1046–1054.
Mumford SL, Chavarro JE, Zhang C, et al. Dietary fat intake and reproductive hormone concentrations and ovulation in regularly menstruating women. Am J Clin Nutr. 2016;103(3):868–877.
Martin JC, Zhou SJ, Flynn AC, Malek L, Greco R, Moran L. The assessment of diet quality and its effects on health outcomes prepregnancy and during pregnancy. Semin Reprod Med. 2016;34(2):83–92.
Hammiche F, Vujkovic M, Wijburg W, et al. Increased preconception omega-3 polyunsaturated fatty acid intake improves embryo morphology. Fertil Steril. 2011;95(5):1820–1823.
Moran LJ, Tsagareli V, Noakes M, Norman R. Altered preconception fatty acid intake is associated with improved pregnancy rates in overweight and obese women undertaking in vitro fertilisation. Nutrients. 2016;8(1):pii: E10.
Wonnacott KE, Kwong WY, Hughes J, et al. Dietary omega-3 and -6 polyunsaturated fatty acids affect the composition and development of sheep granulosa cells, oocytes and embryos. Reproduction (Cambridge, England). 2010;139(1):57–69.
Wise LA, Wesselink AK, Tucker KL, et al. Dietary fat intake and fecundability in two preconception cohort studies. Am J Epidemiol. 2017;187(1):60–74.
Polotsky AJ, Hailpern SM, Skurnick JH, Lo JC, Sternfeld B, Santoro N. Association of adolescent obesity and lifetime nulliparity- the Study of Women’s Health Across the Nation (SWAN). Fertil Steril. 2010;93(6):2004–2011.
Brunner Huber LR, Hogue CJ. The association between body weight, unintended pregnancy resulting in a livebirth, and contraception at the time of conception. Matern Child Health J. 2005;9(4):413–420.
Lan L, Harrison CL, Misso M, et al. Systematic review and metaanalysis of the impact of preconception lifestyle interventions on fertility, obstetric, fetal, anthropometric and metabolic outcomes in men and women. Hum Reprod (Oxford, England). 2017;32(9):1925–1940.
Edelman AB, Cherala G, Munar MY, McInnis M, Stanczyk FZ, Jensen JT. Correcting oral contraceptive pharmacokinetic alterations due to obesity: a randomized controlled trial. Contraception. 2014;90(5):550–556.
Kawwass JF, Kulkarni AD, Hipp HS, Crawford S, Kissin DM, Jamieson DJ. Extremities of body mass index and their association with pregnancy outcomes in women undergoing in vitro fertilization in the United States. Fertil Steril. 2016;106(7):1742–1750.
Al-Safi ZA, Liu H, Carlson NE, et al. Omega-3 fatty acid supplementation lowers serum FSH in normal weight but not obese women. J Clin Endocrinol Metab. 2016;101(1):324–333.
Moriyama R, Yamazaki T, Kato T, Kato Y. Long-chain unsaturated fatty acids reduce the transcriptional activity of the rat follicle-stimulating hormone beta-subunit gene. J Reprod Dev. 2016;62(2):195–199.
Khedr NF. Fish oil and wheat-germ oil supplementation restores ovarian function in streptozotocin-diabetic rats. Reprod Fertil Dev. 2017;29(9):1689–1698.
Meyer PM, Zeger SL, Harlow SD, et al. Characterizing daily urinary hormone profiles for women at midlife using functional data analysis. Am J Epidemiol. 2007;165(8):936–945.
Wu LL, Russell DL, Wong SL, et al. Mitochondrial dysfunction in oocytes of obese mothers: transmission to offspring and reversal by pharmacological endoplasmic reticulum stress inhibitors. Development (Cambridge, England). 2015;142(4):681–691.
Fu Z, Lofqvist CA, Shao Z, et al. Dietary omega-3 polyunsaturated fatty acids decrease retinal neovascularization by adiposeendoplasmic reticulum stress reduction to increase adiponectin. Am J Clin Nutr. 2015;101(4):879–888.
Aksoy MO, Kim V, Cornwell WD, et al. Secretion of the endoplasmic reticulum stress protein, GRP78, into the BALF is increased in cigarette smokers. Respir Res. 2017;18(1):78.
Guzel E, Arlier S, Guzeloglu-Kayisli O, et al. Endoplasmic reticulum stress and homeostasis in reproductive physiology and pathology. Int J Mol Sci. 2017;18(4):pii: E792.
Billman G. Effect of dietary omega-3 polyunsaturated fatty acids on heart rate and heart rate variability in animals susceptible or resistant to ventricular fibrillation. Front Physiol. 2012;3(71):1–10.
Kassam A, Overstreet JW, Snow-Harter C, De Souza MJ, Gold EB, Lasley BL. Identification of anovulation and transient luteal function using a urinary pregnanediol-3-glucuronide ratio algorithm. Environ Health Perspect. 1996;104(4):408–413.
Jungheim ES, Macones GA, Odem RR, Patterson BW, Moley KH. Elevated serum alpha-linolenic acid levels are associated with decreased chance of pregnancy after in vitro fertilization. Fertil Steril. 2011;96(4):880–883.
Kermack AJ, Calder PC, Houghton FD, Godfrey KM, Macklon NS. A randomised controlled trial of a preconceptional dietary intervention in women undergoing IVF treatment (PREPARE trial). BMC Womens Health. 2014;14:130.
Messerlian C, Williams PL, Ford JB, et al. The Environment and Reproductive Health (EARTH) study: a prospective preconception cohort. Hum Reprod Open. 2018;2018(2):pii: hoy001.
Cnop M, Foufelle F, Velloso LA. Endoplasmic reticulum stress, obesity and diabetes. Trends Mol Med. 2012;18(1):59–68.
Huang N, Yu Y, Qiao J. Dual role for the unfolded protein response in the ovary: adaption and apoptosis. Protein Cell. 2017;8(1):14–24.
Li S, Mbong EF, John DT, Terasaka T, Li D, Lawson MA. Induction of stress signaling in vitro and suppression of gonadotropin secretion by free fatty acids in female mouse gonadotropes. Endocrinology. 2018;159(2):1074–1087.
Babayev E, Lalioti MD, Favero F, Seli E. Cross-talk between FSH and endoplasmic reticulum stress: a mutually suppressive relationship. Reprod Sci (Thousand Oaks, Calif). 2016;23(3):352–364.
Begum G, Yan HQ, Li L, Singh A, Dixon CE, Sun D. Docosahexaenoic acid reduces ER stress and abnormal protein accumulation and improves neuronal function following traumatic brain injury. J Neurosci. 2014;34(10):3743–3755.
Okada L, Oliveira CP, Stefano JT, et al. Omega-3 PUFA modulate lipogenesis, ER stress, and mitochondrial dysfunction markers in NASH - proteomic and lipidomic insight. Clin Nutr. 2017;37(5):1474–1484.
Kiranmayee D, Praveena T, Himabindu Y, Sriharibabu M, Kavya K, Mahalakshmi M. The effect of moderate physical activity on ovarian reserve markers in reproductive age women below and above 30 years. J Hum Reprod Sci. 2017;10(1):44–48.
Reame NE, Kelche RP, Beitins IZ, Yu MY, Zawacki CM, Padmanabhan V. Age effects of follicle-stimulating hormone and pulsatile luteinizing hormone secretion across the menstrual cycle of premenopausal women. J Clin Endocrinol Metab. 1996;81(4):1512–1518.
van Rooij IA, Broekmans FJ, Scheffer GJ, et al. Serum antimullerian hormone levels best reflect the reproductive decline with age in normal women with proven fertility: a longitudinal study. Fertil Steril. 2005;83(4):979–987.
Acknowledgments
The authors acknowledge the assistance of Bill Lasley, PhD, and the Endocrine Core at the California National Primate Research Center, for the urinary steroid assays. The authors also acknowledge the University of Virginia, Center for Research in Reproduction Ligand Assay and Analysis Core for assistance with hormonal assays and thank Jack Dunker for assistance with formatting the figures and tables.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Bauer, J.L., Kuhn, K., Bradford, A.P. et al. Reduction in FSH Throughout the Menstrual Cycle After Omega-3 Fatty Acid Supplementation in Young Normal Weight but not Obese Women. Reproductive Sciences 26, 1025–1033 (2019). https://doi.org/10.1177/1933719119828099
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719119828099