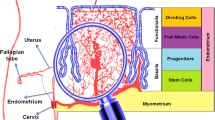
Abstract
The mesenchymal stem cells (MSCs) have awakened interest in regenerative medicine due to its high capability to proliferate and differentiate in multiple specialized lineages under defined conditions. The reproductive system is considered a valuable source of MSCs, which needs further investigations. Many factors have been reported as critical for these cell lineage specification and determination. In this review, we discuss the main effects of extracellular matrix or tissue environment and growth factors in the cell lineage commitment, including the reproductive stem cells. The MSCs responses to culture medium stimuli or to soluble factors probably occur through several intracellular activation pathways. However, the molecular mechanisms in which the cells respond to these mechanical or chemical perturbations remain elusive. Recent findings suggest a synergic effect of microenvironment and soluble cell culture factors affecting cell differentiation. For future applications in cell therapy, protocols of reproductive MSCs differentiation must be established.
Similar content being viewed by others
References
World Health Organization. Make every mother and child count. World health report. 2005; 1211 Geneva 27, Switzerland.
Masuda H, Matsuzaki Y, Hiratsu E, et al. Stem cell-like properties of the endometrial side population: implication in endometrial regeneration. PloS one. 2010;5(4):e10387.
Taylor HS, Osteen KG, Bruner-Tran KL, et al. Novel therapies targeting endometriosis. Reprod Sci. 2011;18(9):814–823.
Du H, Taylor SH. Stem cells and reproduction. Curr Opin Obstet Gynecol. 2010;22(3):235–241.
Hayashi Y, Saitou M, Yamanaka S. Germline development from human pluripotent stem cells toward disease modeling of infertility. Fertil Steril. 2012;97(6):1250–1259.
Hutt KJ, Albertini DF. Clinical applications and limitations of current ovarian stem cell research: A review. J Exp Clin Assist Reprod. 2006;3(6). doi: 10.1186/1743-1050-3-6.
Bukovsky A, Caudle MR, Carson RJ, et al. Immune physiology in tissue regeneration and aging, tumor growth and regenerative medicine. Aging. 2009;1(2):157–181.
Taylor HS. Endometrial cells derived from donor stem cells in bone marrow transplant recipients. JAMA. 2004;292(1):81–85.
Wolff EF, Wolff AB, Hongling DU, Taylor HS. Demonstration of multipotent stem cells in the adult human endometrium by in vitro chondrogenesis. Reprod Sci. 2007;14(6):524–533.
Du H, Taylor SH. Stem cells and female reproduction. Reprod Sci. 2009;16(2):126–139.
Martin DR, Cox NR, Hathcock TL, Niemeyer GP, Baker HJ. Isolation and characterization of multipotential mesenchymal stem cells from feline bone marrow. Exp Hematol. 2002;30(8): 879–886.
Herzog EL, Chai L, Krause DS. Plasticity of marrow-derived stem cells. Blood. 2003;102(10):3483–3493.
Mohr S, Portmann-lanz CB, Schoeberlein A, Sager R, Surbek DV. Generation of an osteogenic graft from human placenta and placenta-derived mesenchymal stem cells. Reprod Sci. 2010; 17(11):1006–1015.
Abreu SC, Antunes MA, Pelosi P, Morales MM, Rocco PRM. Mechanisms of cellular therapy in respiratory diseases. Intens Care Med. 2011;37(9):1421–1431.
Kuijk EW, Lopes SMCS, Geijsen N, Macklon N, Roelen BAJ. The different shades of mammalian pluripotent stem cells. Hum Reprod Update. 2011;17(2):254–271.
Lin GL, Hankenson KD. Integration of BMP, Wnt, and notch signaling pathways in osteoblast differentiation. J Cell Biochem. 2011;112(12):3491–3501.
Semb H. Human embryonic stem cells: origin, properties and applications. APMIS. 2005;113(11–12):743–750.
Gattegno-Ho D, Argyle SA, Argyle DJ. Stem cells and veterinary medicine: tools to understand diseases and enable tissue regeneration and drug discovery. Vet J (London, England: 1997). 2012; 191(1):19–27.
Hayashi Y, Tsuji S, Tsujii M, et al. Topical transplantation of mesenchymal stem cells accelerates gastric ulcer healing in rats. Am J Physiol Gastrointest Liver Physiol. 2008;294(3): G778–G786.
Bianchi G, Muraglia A, Daga A, Corte G, Cancedda R, Quarto R. Microenvironment and stem properties of bone marrow-derived mesenchymal cells. Wound Repair Regen. 2001;9(6):460–466.
Meirelles LS, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119(Pt 11):2204–2213.
Gandolfi F, Brevini TAL, Cillo F, Antonini S. Cellular and molecular mechanisms regulating oocyte quality and the relevance for farm animal reproductive efficiency. Rev Sci Tech. 2005;24(1):413–423.
Hu FF, Jing XU, Cui YG, et al. Isolation and characterization of side population cells in the postpartum murine endometrium. Reprod Sci. 2010;17(7):629–642.
Deans RJ, Moseley AB. Mesenchymal stem cells: Biology and potential clinical uses. Exp Hematol. 2000;28(8):875–884.
Vããnãnen HK. Mesenchymal stem cells. Ann Med. 2005;37(7): 469–479.
Bittencourt RAC, Pereira HR, Felisbino SL, Murador P, Oliveira APE, Deffune E. Isolation of bone marrow mesenchymal stem cells. Acta Ortop Bras. 2011;14(1):22–24.
Kanitkar M, Tailor HD, Khan WS. The use of growth factors and mesenchymal stem cells in orthopaedics. Open Orthop J. 2011; 5(2):271–275.
Bae KS, Park JB, Kim HS, Kim DS, Park DJ, Kang SJ. Neuronlike Differentiation of bone marrow-derived mesenchymal stem cells. Yonsei Med J. 2011;52(3):401–412.
Orlic D, Kajstura J, Chimenti S, et al. Mobilized bone marrow cells repair the infracted heart, improving function and survival. Proc Natl Acad Sci U S A. 2001;98(18):10344–10349.
Reyes M, Dudek A, Jahagirdar B, Koodie L, Marker PH, Verfaillie CM. Origin of endothelial progenitors in human postnatal bone marrow. J Clin Invest. 2002;109(3):337–346.
Hayashi Y, Tsuji S, Tsujii M, et al. Topical Implantation of Mesenchymal Stem Cells Has Beneficial Effects on Healing of Experimental Colitis in Rats. J Pharmacol Exp Ther. 2008; 326(2):523–531.
Lange C, Bassler P, Lioznov MV, et al. Liver-specific gene expression in mesenchymal stem cells is induced by liver cells. World J Gastroenterol. 2005;11(29):4497–4504.
Sato Y, Araki H, Kato J, et al. Human mesenchymal stem cells xenografted directly to rat liver are differentiated into human hepatocytes without fusion. Blood. 2005;106(2):756–763.
Morelli SS, Yi P, Goldsmith LT. Endometrial stem cells and reproduction [published online January 12, 2012]. Obstet Gynecol Int. 2012.
Sanchez-Ramos J, Song S, Cardozo-Pelaez, et al. Adult bone marrow stromal cells differentiate into neural cells in vitro. Exp Neurol. 2000;164(2):247–256.
Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418(6893):41–49.
Bianco P, Riminucci M, Gronthos S, Robey PG. Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells. 2001;19(3):180–192.
Woodbury D, Schwarz EJ, Prockop DJ, Black IB. Adult rat and human bone marrow stromal cells differentiate into neurons. J Neurosci Res. 2000;61(4):364–370.
Houghton J, Stoicov C, Nomura S, et al. Gastric cancer originating from bone marrow-derived cells. Science. 2004;306(5701): 1568–1571.
Barry FP, Murphy JM. Mesenchymal stem cells: clinical applications and biological characterization. Int J Biochem Cell B. 2004; 36(4):568–584.
McBeath R, Pirone DM, Nelson CM, Bhadriraju K, Chen CS. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev Cell. 2004;6(4):483–495.
Grassel S, Ahmed N. Influence of cellular microenvironment and paracrine signals on chondrogenic differentiation. Front Biosci. 2007;12:4946–4956. doi: 10.2741/2440.
Bianco P, Robey PG. Stem cells in tissue engineering. Nature. 2001;414(6859):118–121.
Schneider PRA, Buhrmann C, Mobasheri A, Matis U, Shakibaei M. Three-dimensional high-density co-culture with primary tenocytes induces tenogenic differentiation in mesenchymal stem cells. J Orthopaed Res. 2011;29(9):1351–1360.
Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4):677–689.
Even-Ram S, Artym V, Yamada KM. Matrix control of stem cell fate. Cell. 2006;126(4):645–647.
Hwang NS, Varghese S, Li H, Elisseeff J. Regulation of osteogenic and chondrogenic differentiation of mesenchymal stem cells in PEG-ECM hydrogels. Cell Tissue Res. 2011;344(3): 499–509.
Ravindran S, Gao Q, Kotecha M, Magin RL, Karol S, Bedran-Russo A, George A. Biomimetic ECM Incorporated Scaffold Induces Osteogenic Gene Expression in Human Marrow Stromal Cells. Tissue Eng. Part A. 2012;18(3–4):295–309.doi: 10.1089/ten.TEA.0136, 1–40.
Huang S, Ingber DE. Cell tension, matrix mechanics, and câncer development. Cancer cell. Previews. 2005;8(3):175–176.
Bershadsky AD, Balaban NQ, Geiger B. Adhesion-dependent cell mechanosensitivity. Annu Rev Cell Dev Biol. 2003;19:677–695. doi: 10.1146/annurev.cellbio.19.111301.153011.
Discher DE, Janmey P, Wang Y. Tissue cells feel and respond to the stiffness of their substrate. Science. 2005;310(5751): 1139–1143.
Sawada Y, Tamada M, Thaler BJD, et al. Force sensing by mechanical extension of the src family kinase substrate p130Cas. Cell. 2006;127(5):1015–1026.
Griffith LG, Swartz MA. Capturing complex 3D tissue physiology in vitro. Nat Rev Mol Cell Biol. 2006;7(3):211–224.
Giannone G, Sheetz MP. Substrate rigidity and force define form through tyrosine phosphatase and kinase pathways. Trends Cell Biol. 2006;16(4):213–223.
Ingber DE. Cellular mechanotransduction: putting all the pieces together again. FASEB J. 2006;20(7):811–827.
Davis HE, Case EM, Miller SL, Genetos DC, Leach JK. Osteogenic response to BMP-2 of hMSCs grown on apatite-coated scaffolds. Biotechnol Bioeng. 2011;108(11):2727–2735.
Vogel V, Sheetz M. Local force and geometry sensing regulate cell functions. Nat Rev Mol Cell Biol. 2006;7(4):265–275.
Raghavan S, Shen CJ, Desai RA, Sniadecki NJ, Nelson CM, Chen CS. Decoupling diffusional from dimensional control of signaling in 3D culture reveals a role for myosin in tubulogenesis. J Cell Sci. 2010;123(17):2877–2883.
Curran JM, Chen R, Hunt JA. The guidance of human mesenchymal stem cell differentiation in vitro by controlled modifications to the cell substrate. Biomaterials. 2006;27(27):4783–4793.
Jurgens WJFM, Lu Z, Zandieh-Doulabi B, Kuik DJ, Ritt MJPF, Helder MN. Hyperosmolarity and hypoxia induce chondrogenesis of adipose-derived stem cells in a collagen type 2 hydrogel. J Tissue Eng Regen Med. 2011;6(7):570–578.
Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861–872.
Schmitt B, Ringe J, Haupl T, et al. BMP2 initiates chondrogenic lineage development of adult human mesenchymal stem cells in high-density culture. Differentiation. 2003;71:567–577.
Davidson D, Blanc A, Filion D, et al. Fibroblast growth factor (FGF) 18 signals through FGF receptor 3 to promote chondrogenesis. J Biol Chem. 2005;280(21):20509–20515.
Hwang NS, Varghese S, Puleo C, Zhang Z, Elisseeff J. Morphogenetic signals from chondrocytes promote chondrogenic and osteogenic differentiation of mesenchymal stem cells. J Cell Physiol. 2007;212(2):281–284.
Hanada K, Dennis JE, Caplan AJ. Stimulatory effects of basic fibroblast growth factor and bone morphogenetic protein-2 on osteogenic differentiation of rat bone marrow derived mesenchymal stem cells. J Bone Miner Res. 1997;12(10):1606–1614.
Zhou S, Turgeman G, Harris SE, et al. Estrogens activate bone morphogenetic protein-2 gene transcription in mouse mesenchymal stem cells. Mol Endocrinol. 2003;17(1):56–66.
Fierro FA, Kalomoiris S, Sondergaard CS, Nolta JA. Effects on proliferation and differentiation of multipotent bone marrow stromal cells engineered to express growth factors for combined cell and gene therapy. Stem Cells. 2011;29(11):1727–1737.
Gorter DJJ, van Dinther M, Korchynskyi O, ten Dijke P. Biphasic effects of transforming growth factor b on bone morphogenetic protein–induced osteoblast differentiation. J Bone Miner Res. 2011;26(6):1178–1187.
Janderova L, McNeil M, Murrell AN, Mynatt RL, Smith SR. Human mesenchymal stem cells as an in vitro model for human adipogenesis. Obes Res. 2003;11(1):65–74.
Neubauer M, Fischbach C, Bauer-Kreisel P, et al. Basic fibroblast growth factor enhances PPARγ ligand-induced adipogenesis of mesenchymal stem cells. FEBS Lett. 2004;577(1–2):277–283.
Kakudo N, Shimotsuma A, Kusumoto K. Fibroblast growth factor-2 stimulates adipogenic differentiation of human adipose-derived stem cells. Biochem Bioph Res Co. 2011; 359(2):239–244.
van den Bos C, Mosca J, Winkles J, Kerrigan L, Burgess WH, Marshak DR. Human mesenchymal stem cells respond to fibroblast growth factors. Human Cell. 1997;10(1):45–50.
MacDonald KK, Cheung CY, Anseth KS. Cellular delivery of TGFβ1 promotes osteoinductive signalling for bone regeneration. J Tissue Eng Regen Med. 2007;1(4):314–317.
Del Carlo RJ, Monteiro BS, Argôlo Neto NM. Células-tronco e fatores de crescimento na reparação tecidual. Ciênc vet Tróp. 2008;11(1):167–169.
Kuznetsov SA, Friedenstein AJ, Robey PG. Factors required for bone marrow stromal fibroblast colony formation in vitro. Brit J Haematol. 1997;97(3):561–570.
Schwab KE, Chan RW, Gargett CE. Putative stem cell activity of human endometrial epithelial and stromal cells during the menstrual cycle. Fertil Steril. 2005;84(suppl 2):1124–1130.
Chan RW, Schwab KE, Gargett CE. Clonogenicity of human endometrial epithelial and stromal cells. Biol Reprod. 2004; 70(6):1738–1750.
Kulak JJ, Ferriani AR, Komm BS, Taylor HS. Tissue selective estrogen complexes (TSECs) differentially modulate markers of proliferation and differentiation in endometrial cells. Reprod Sci. 2013;20(2):129–137.
Cuevas P, Burgos J, Baird A. Basic fibroblast growth factor (FGF) promotes cartilage repair in vivo. Biochem Bioph Res Co. 1988; 156(2):611–618.
Mackay AM, Beck SC, Murphy JM, Barry FP, Chichester CO, Pittenger MF. Chondrogenic differentiation of cultured human mesenchymal stem cells from marrow. Tissue Eng. 1998;4(4): 415–428.
Chen D, Ji X, Harris MA, et al. Differential roles for bone marrow morphogenetic protein (BMP) receptor type IB and IA in differentiation and specification of mesenchymal precursor cells to osteoblast and adipocyte lineages. J Cell Biol. 1998;142(1): 295–305.
Yonezawa T, Lee JW, Hibino A. Harmine promotes osteoblast differentiation through bone morphogenetic protein signaling. Biochem Bioph Res Co. 2011;409(2):260–265.
Grgurevic L, Macek B, Mercep M, et al. Bone morphogenetic protein (BMP)1-3 enhances bone repair. Biochem Bioph Res Co. 2011;408(1):25–31.
Wen Q, Zhou L, Zhou C, Zhou M, Luo W, Ma L. Change in hepatocyte growth factor concentration promote mesenchymal stem cell-mediated osteogenic regeneration. J Cell Mol Med. 2012; 16(6):1260–1273.
Wang Y, Zhang D, Ashraf M, et al. Combining neuropeptide Y and mesenchymal stem cells reverses remodeling after myocardial infarction. Am J Physiol-Heart C. 2010;298(1):275–286.
Delcroix GJR, Curtis KM, Schiller PC, Montero-Menei CN. EGF and bFGF pre-treatment enhances neural specification and the response to neuronal commitment of MIAMI cells. Differentiation. 2010;80(4–5):213–227.
Pulavendran S, Rose C, Mandal AB. Hepatocyte growth factor incorporated chitosan nanoparticles augment the differentiation of stem cell into hepatocytes for the recovery of liver cirrhosis in mice. J Nanobiotechnology. 2011;9(15):1–11.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vidane, A.S., Zomer, H.D., Oliveira, B.M.M. et al. Reproductive Stem Cell Differentiation: Extracellular Matrix, Tissue Microenvironment, and Growth Factors Direct the Mesenchymal Stem Cell Lineage Commitment. Reprod. Sci. 20, 1137–1143 (2013). https://doi.org/10.1177/1933719113477484
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719113477484