Abstract
Background
The Wnt signaling pathway is a conserved pathway and plays a crucial role in regulating trophoblast functions. Abnormal expression of the Wnt pathway may result in the dysfunction of the trophoblast that can contribute to the pathogenesis of preeclampsia (PE). However, published data regarding the association between Wnt pathway and PE in human pregnancy is rare.
Objective
The aims of this study were to investigate the expression pattern of Wnt2 and secreted frizzled-related protein 4 (sFRP4) in the third trimester human placenta and to evaluate the relationship between changes in placental Wnt2 and sFRP4 expression and severe PE.
Methods
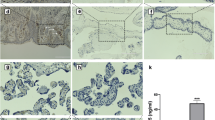
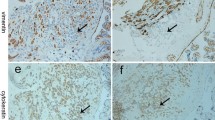
The expression of Wnt2 and sFRP4 in normal and severe PE placentas was examined using immunohistochemistry (IHC), real-time polymerase chain reaction, and Western blot.
Results
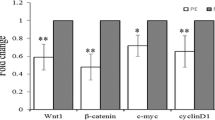
Compared to the controls, the relative expression of Wnt2 messenger RNA was remarkably downregulated in the PE placentas, while there was no significant difference in sFRP4 between the 2 groups. The IHC indicated that Wnt2 and sFRP4 were expressed predominantly in the villous syncytiotrophoblast and the extravillous trophoblast, whereas Wnt2 in the control group showed higher staining intensity than in the PE group, and sFRP4 in the PE group had a higher staining intensity than in the control group. Furthermore, the results of the Western blots were consistent with the IHC.
Conclusions
The Wnt signaling pathway was detected in human third trimester placentas, and the decreased placental expression of Wnt2 and increased placental expression of sFRP4 may be associated with the pathogenesis of severe PE.
Similar content being viewed by others
References
Beaufils M. Pregnancy hypertension. Nephrol Ther. 2010;6(3):200–214.
Wang A, Rana S, Karumanchi SA. Preeclampsia: the role of angiogenic factors in its pathogenesis. Physiology (Bethesda). 2009;24(3):147–158.
Roberts JM, Escudero C. The placenta in preeclampsia. Pregnancy Hypertens. 2012;2(2):72–83.
Pennington KA, Schlitt JM, Jackson DL, Schulz LC, Schust DJ. Preeclampsia: multiple approaches for a multifactorial disease. Dis Model Mech. 2012;5(1):9–18.
Sonderegger S, Pollheimer J, Knofler M. Wnt signalling in implantation, decidualisation and placental differentiation– review. Placenta. 2010;31(10):839–847.
Fitzgerald JS, Germeyer A, Huppertz B, et al. Governing the invasive trophoblast: current aspects on intra- and extracellular regulation. Am J Reprod Immunol. 2010;63(6):492–505.
Sonderegger S, Haslinger P, Sabri A, et al. Wingless (Wnt)-3A induces trophoblast migration and matrix metalloproteinase-2 secretion through canonical Wnt signaling and protein kinase B/ AKT activation. Endocrinology. 2010;151(1):211–220.
Sonderegger S, Husslein H, Leisser C, Knofler M. Complex expression pattern of Wnt ligands and frizzled receptors in human placenta and its trophoblast subtypes. Placenta. 2007;28(suppl A):S97–S102.
Miller JR. The Wnts. Genome Biol. 2002;3(1):REVIEWS3001.
Turashvili G, Bouchal J, Burkadze G, Kolar Z. Wnt signaling pathway in mammary gland development and carcinogenesis. Pathobiology. 2006;73(5):213–223.
Katoh M. WNT2 and human gastrointestinal cancer (review). Int J Mol Med. 2003;12(5):811–816.
Fu L, Zhang C, Zhang LY, et al. Wnt2 secreted by tumour fibroblasts promotes tumour progression in oesophageal cancer by activation of the Wnt/beta-catenin signalling pathway. Gut. 2011;60(12):1635–1643.
Mazieres J, You L, He B, et al. Wnt2 as a new therapeutic target in malignant pleural mesothelioma. Int J Cancer. 2005; 117(2):326–332.
Park JK, Song JH, He TC, Nam SW, Lee JY, Park WS. Overexpression of Wnt-2 in colorectal cancers. Neoplasma. 2009;56(2):119–123.
Wang GX, Zhang ZY, Pu PY, et al. Expression of core components of Wnt2 signaling pathway in gliomas. Zhonghua Bing Li Xue Za Zhi. 2009;38(7):481–482.
Watanabe O, Imamura H, Shimizu T, et al. Expression of twist and wnt inhuman breast cancer.Anticancer Res. 2004;24(6):3851–3856.
Kuster K, Koschel A, Rohwer N, Fischer A, Wiedenmann B, Anders M. Downregulation of the coxsackie and adenovirus receptor in cancer cells by hypoxia depends on HIF-1alpha. Cancer Gene Ther. 2010;17(2):141–146.
You L, He B, Xu Z, et al. An anti-Wnt-2 monoclonal antibody induces apoptosis in malignant melanoma cells and inhibits tumor growth. Cancer Res. 2004;64(15):5385–5389.
Gong YD, Dong MS, Lee SB, Kim N, Bae MS, Kang NS. A novel 3-arylethynyl-substituted pyrido[2,3,-b]pyrazine derivatives and pharmacophore model as Wnt2/beta-catenin pathway inhibitors in non-small-cell lung cancer cell lines. Bioorg Med Chem. 2011;19(18):5639–5647.
Klein D, Demory A, Peyre F, et al. Wnt2 acts as an angiogenic growth factor for non-sinusoidal endothelial cells and inhibits expression of stanniocalcin-1. Angiogenesis. 2009; 12(3):251–265.
Monkley SJ, Delaney SJ, Pennisi DJ, Christiansen JH, Wainwright BJ. Targeted disruption of the Wnt2 gene results in placentation defects. Development. 1996;122(11):3343–3353.
Bafico A, Gazit A, Pramila T, Finch PW, Yaniv A, Aaronson SA. Interaction of frizzled related protein (FRP) with Wnt ligands and the frizzled receptor suggests alternative mechanisms for FRP inhibition of Wnt signaling. J Biol Chem. 1999;274(23):16180–16187.
Carmon KS, Loose DS. Secreted frizzled-related protein 4 regulates two Wnt7a signaling pathways and inhibits proliferation in endometrial cancer cells. Mol Cancer Res. 2008;6(6):1017–1028.
Jacob F, Ukegjini K, Nixdorf S, et al. Loss of secreted frizzled-related protein 4 correlates with an aggressive phenotype and predicts poor outcome in ovarian cancer patients. PLoS One. 2012; 7(2):e31885.
Castro M, Grau L, Puerta P, et al. Multiplexed methylation profiles of tumor suppressor genes and clinical outcome in lung cancer. J Transl Med. Sep 2010;8:86–96.
Hewitt DP, Mark PJ, Dharmarajan AM, Waddell BJ. Placental expression of secreted frizzled related protein-4 in the rat and the impact of glucocorticoid-induced fetal and placental growth restriction. Biol Reprod. 2006;75(1):75–81.
Muley A, Majumder S, Kolluru GK, et al. Secreted frizzled-related protein 4: an angiogenesis inhibitor. Am J Pathol. 2010; 176(3):1505–1516.
Tsai IH, Chen CP, Sun FJ, Wu CH, Yeh SL. Associations of the pre-pregnancy body mass index and gestational weight gain with pregnancy outcomes in Taiwanese women. Asia Pac J Clin Nutr. 2012;21(1):82–87.
Marshall NE, Guild C, Cheng YW, Caughey AB, Halloran DR. Maternal superobesity and perinatal outcomes. Am J Obstet Gynecol. 2012;206(5):417 e411-e416.
Smolich BD, McMahon JA, McMahon AP, Papkoff J. Wnt family proteins are secreted and associated with the cell surface. Mol Biol Cell. 1993;4(12):1267–1275.
Kemp C, Willems E, Abdo S, Lambiv L, Leyns L. Expression of all Wnt genes and their secreted antagonists during mouse blastocyst and postimplantation development. Dev Dyn. 2005;233(3):1064–1075.
White L, Suganthini G, Friis R, Dharmarajan A, Charles A. Expression of secreted frizzled-related protein 4 in the primate placenta. Reprod Biomed Online. 2009;18(1):104–110.
Fujita M, Ogawa S, Fukuoka H, et al. Differential expression of secreted frizzled-related protein 4 in decidual cells during pregnancy. J Mol Endocrinol. 2002;28(3):213–223.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Z., Zhang, L., Zhang, L. et al. Association of Wnt2 and sFRP4 Expression in the Third Trimester Placenta in Women With Severe Preeclampsia. Reprod. Sci. 20, 981–989 (2013). https://doi.org/10.1177/1933719112472740
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719112472740