Abstract
Objective
To test the hypothesis that vascular relaxation kinetics are prolonged in pregnant rats treated chronically with Nω -nitro-L-arginine methyl ester (L-NAME).
Methods
Timed pregnant rats (on day 15 of a 22-day gestation) were implanted with infusion pumps containing vehicle (controls) or L-NAME (50 mg/d). L-NAME pumps were retained until day 22 (group 1), or removed on day 20 (group 2). All rats were killed at term. Aortic rings were mounted in organ chambers containing physiologic salt solution (PSS) for isometric tension recording, contracted with high-K+PSS (60 mM), and allowed to relax in normal-K+ PSS. Relaxation kinetics were quantified as time for 50% and 80% relaxation. After contraction with phenylephrine, responses to cumulative concentrations of methacholine were studied in the absence and presence of L-arginine (L-Arg) (10-3 M).
Results
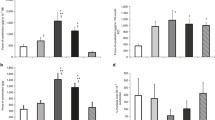
Responses to methacholine were inhibited completely in group 1 and partially in group 2 (P < .05). The inhibition in both groups was reversed by L-Arg. The rate of relaxation was significantly slower in groups 1 and 2 (P < .05) as compared with controls. Mechanical removal of the endothelium caused prolongation of relaxation in controls and group 2 (P < .05), but not in group 1. Preincubation of aortic rings from untreated controls with L-NAME (in vitro, 10-4 M) did not affect relaxation.
Conclusion
The endothelium modulates the rate of vascular relaxation by a factor other than nitric oxide. Nω-nitro-L-arginine methyl ester (L-NAME) prolongs vasorelaxation by endothelium-dependent and -independent mechanisms. Prolongation of vascular relaxation kinetics may be a mechanism to elevate blood pressure and peripheral vascular resistance in preeclampsia.
Similar content being viewed by others
References
Roberts JM. Pregnancy related hypertension. In: Creasy RK, Resnik R, eds. Maternal-fetal medicine: principals and practice. Philadelphia: WB Saunders, 1994;804–43.
Sibai BM. Hypertension in pregnancy. In: Gabbe SG, Niebyl JR, Simpson JL, eds. Obstetrics: normal and problem pregnancies. New York: Churchill Livingstone, 1996;935–96.
Poston L, McCarthy AL. Ritter JM. Control of vascular resistance in the maternal and feto-placental arterial beds. Pharmacol Ther 1995;65:215–39.
Magness RR, Gant NF. Control of vascular reactivity in pregnancy: the basis for therapeutic approaches to prevent pregnancy-induced hypertension. Semin Perinatol 1994;18:45–69.
McCarthy AL, Woolfson RG, Raju SK, Poston L. Abnormal endothelial cell function of resistance arteries from women with preeclampsia. Am J Obstet Gynecol 1993;168:1323–30.
Lyall F, Young A, Greer IA. Nitric oxide concentrations are increased in the fetoplacental circulation in preeclampsia. Am J Obstet Gynecol 1995;173:714–8.
Wang Y, Walsh SW, Parnell R, Han J. Placental production of nitric oxide and endothelin in normal and preeclamptic pregnancies. Hypertens Pregn 1994;13:171–8.
Vedernikov YP, Belfort MA, Saade GR, Moise KJ. Pre-eclamp-sia does not alter the response to endothelin-1 in human omental artery. J Cadiovasc Pharmacol 1995;26:S233–5.
Belfort MA, Saade GR, Suresh M, Kramer W, Vedernikov Y. Effects of selected vasoconstrictor agonists on isolated omental artery from premenopausal nonpregnant women and from normal and preeclamptic pregnant women. Am J Obstet Gynecol 1996;174:687–93.
Yallampalli C, Garfield RE. Inhibition of nitric oxide synthesis in rats during pregnancy produces signs similar to those of preeclampsia. Am J Obstet Gynecol 1993;169:1316–20.
Molnar M, Suto T, Toth T, Hertelendy F. Prolonged blockade of nitric oxide synthesis in gravid rats produces sustained hypertension, proteinuria, thrombocytopenia, and intrauterine growth retardation. Am J Obstet Gynecol 1994;170:1458–66.
Mayer B. Biochemistry and molecular pharmacology of nitric oxide synthases. In: Vincent SR, ed. Nitric oxide in the nervous system. New York: Academic Press, 1995;21–42.
Rees DD, Palmer RMJ, Schulz R, Hodson HF, Moncada S. Characterization of three inhibitors of endothelial nitric oxide synthase in vitro and in vivo. Br J Pharmacol 1990;101:746–52.
Li J-S, Schiffrin EL. Resistance artery structure and neuroeffector mechanisms in hypertension induced by inhibition of nitric oxide synthase. Am J Hypertens 1994;7:996–1004.
Knppeit-Drews P, Welker S, Drews G. Effects of the nitric oxide synthase inhibitor N omega nitro-L-arginine methyl ester on electrical activity and ion channels of mouse pancreatic B cells. Biochem Biophys Res Commun 1996;224:199–205.
Yang Y, Macdonald GJ, Duggan KA. Changes in angiotensin II metabolism contribute to the increased pressor response to angiotensin after chronic treatment with L-NAME in the spontaneously hypertensive rat. Clin Exp Pharmacol Physiol 1996;23: 611–3.
Takemoto M, Egashira K, Usui M, et al. Important role of tissue angiotensin-converting enzyme activity in the pathogenesis of coronary and myocardial structural changes induced by long-term blockade of nitric oxide synthesis in rats. J Clin Invest 1997;99:278–87.
Kadokami T, Egashira K, Kuwata K, et al. Altered serotonin receptor subtypes mediate coronary microvascular hyperreactivity in pigs with chronic inhibition of nitric oxide synthesis. Circulation 1996;94:182–9.
Okamura T, Ayajiki K, Toda N. Neural mechanism of pressor action of nitric oxide synthase inhibitor in anaesthetized monkeys. Hypertension 1996;28:341–6.
Cohen ML, Berkowitz BA. Decreased vascular relaxation in hypertension. J Pharmacol Exp Ther 1976;196:396–406.
Sunano S, Shimada T, Moriyama K, Shimamura K. Relaxation of mesenteric artery of stroke prone spontaneously hypertensive rats by calcium removal. Clin Exp Physiol Pharmacol 1990;17: 413–25.
Jam V, Vedernikov YP, Saade GR, Chwalisz K, Garfield RE. Relaxation kinetics of rat aorta during pregnancy. J Soc Gynecol Invest 1998;5:81–6.
Griffith TM. Temporal chaos in the microcirculation. Cardiovas Res 1996;31:342–58.
Achakri H, Stergiopulos N, Hoogerwerf N, Hayoz D, Brunner HR, Meister JJ. Intraluminal pressure modulates the magnitude and the frequency of induced vasomotion in rat arteries. J Vase Res 1995;32:237–46.
Vanhoutte PM. Endothelial dysfunction in hypertension. J Hypertens 1996;14:S83–93.
Weiner CP, Liu KZ, Thompson L. Effect of pregnancy on endothelium and smooth muscle: their role in reduced adrenergic sensitivity. Am J Physiol 1991;261:H1275–83.
Pfeiffer S, Leopold E, Schmidt K, Brunner F, Mayer B. Inhibition of nitric oxide synthesis by NG-nitro-L-arginine methyl ester (L-NAME): requirement for bioactivation to free acid, NG-nitro-L-arginine. Br J Pharmacol 1996;118:1433–40.
Vanhoutte PM. ed. Endothelium-derived hyperpolarizing factor. Amsterdam: Harwood Academic Publishers, 1996.
Waugh WH. Role of calcium in contractile excitation of vascular smooth by epinephrine and potassium. Circ Res 1962;11:927– 40.
Kamrn KE, Stull JT. The function of myosin and myosin light chain kinase phosphorylation in smooth muscle. Annu Rev Pharmacol Toxicol 1985;25:593–620.
Verbist J, Droogmans G, Wuytack F, Casteels R. Ca distribution and its regulation in smooth muscle cells. In: Margo A, Oswald W, Vanhoutte PM, eds. Central and peripheral mechanisms of cardiovascular regulation. New York: Plenum, 1986;33–8.
Kolpakov V, Gordon D, Kulik TJ. Nitric oxide-generating compounds inhibit total protein and collagen synthesis in cultured vascular smooth muscle cells. Circ Res 1995;76:305–9.
Dunn WR. Gardiner SM. Ńo evidence for vascular remodelling during hypertension induced by chronic inhibition of nitric oxide synthase in Brattleboro rats. J Hypertens 1995;14:849–57.
Roberts JM, Redman CW. Pre-eclampsia: more than pregnancy-induced hypertension. Lancet 1993;341:1447–51.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
jain, V., Vedernikov, Y.P., Saade, G.R. et al. Relaxation Kinetics of the Aorta in Nω-nitro-L-arginine Methyl Ester-Treated Pregnant Rats. Reprod. Sci. 6, 11–16 (1999). https://doi.org/10.1177/107155769900600104
Published:
Issue Date:
DOI: https://doi.org/10.1177/107155769900600104