Abstract
Objective
It is not known whether withdrawal of progesterone (P) action is a prerequisite for parturition in women or in nonhuman primates because concentrations of circulating progesterone or progesterone receptors (PR) in myometrium and decidua do not decrease before delivery. To examine this potentially important regulatory mechanism, we determined PR isoforms, PR localization, and mRNA in myometrium, decidua, and fetal membranes from rhesus monkeys during pregnancy and in spontaneous labor at term.
Methods
Gestational tissues were obtained midpregnancy (day 80–100), late pregnancy (day 130–145), and during spontaneous labor at term (day 161–167). Samples of rhesus monkey myometrium, decidua, chorion-decidua, and amnion were collected and analyzed for total nuclear and cytosolic PR by competitive binding assay. Progesterone receptor isoforms were identified and quantified by Western blot analysis, and PR mRNA was determined by a specific ribonuclease protection assay. Nuclear PR was localized by immunohistochemistry with monoclonal anti-PR (JZB39) after microwave stabilization.
Results
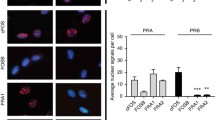
Myometrium and decidua showed no change in total PR during pregnancy and labor. Nuclear PR was not detected in fetal membranes by binding assay but was localized in amnion epithelial and mesenchymal cells and in chorion laeve cytotrophoblasts by immunohistochemistry. Staining for PR was substantially less by serial antibody dilution in fetal membranes than in decidua. Message for PR was confirmed in all tissues analyzed. A significant (P < .05) shift in the ratio of PR isoforms (from PR-B dominance at midpregnancy to PR-A dominance in labor) was observed in myometrium but not in decidua. Both PR-A and PR-B isoforms and PR nuclear staining were nearly undetectable in amnion obtained during labor.
Conclusion
A shift to PR-A dominance in myometrium at term together with a loss of PR in fetal membranes provides evidence for a functional progesterone withdrawal mechanism, which may facilitate the initiation of parturition in primates.
Similar content being viewed by others
References
Csapo AI. The “see-saw” theory of parturition. In: O’Connor M, Knight J, eds. The fetus and birth. New York: Elsevier, 1977:159–210.
Liggins GC, Thorburn GD. Initiation of parturition. In: Lamming GE, ed. Marshall’s physiology of reproduction. London: Chapman and Hall, 1994:863–1002.
Flint APP, Anderson ABM, Steele PA, Turnbull AC. The mechanisms by which foetal Cortisol controls the onset of parturition in the sheep. Biochem Soc Trans 1975;3:1189–94.
Lye SJ, Porter DG. Demonstration that progesterone “blocks” uterine activity in the ewe in vivo by a direct action on the myometrium. J Reprod Fertil 1978;52:87–94.
Tulchinsky D, Hobel CJ, Yeager E, Marshall JR. Plasma estrone, estradiol, estriol, progesterone, and 17-hydroxyprogesterone in human pregnancy. I. Normal pregnancy. Am J Obstet Gynecol 1972;112:1095–100.
Walsh SW, Stanczyk FZ, Novy MJ. Daily hormonal changes in the maternal, fetal and amniotic fluid compartments before parturition in a primate species. J Clin Endocrinol Metab 1984;58: 629–39.
Avrech OM, Golan A, Weinraub Z, Bukovsky I, Caspi E. Mifepristone (RU486) alone or in combination with a prostaglandin analogue for termination of early pregnancy: A review. Fertil Steril 1991;56:385–93.
Haluska GJ, Novy MJ. Hormonal modulation of uterine activity during primate parturition. Semin Reprod Endocrinol 1993;11: 261–71.
Selinger M, MacKenzie IZ, Gillmer MD, Phipps SL, Ferguson J. Progesterone inhibition in mid-trimester termination of pregnancy: Physiological and clinical effects. Br J Obstet Gynaecol 1987;94:1218–22.
Haluska GJ, Cook MJ, Novy MJ. Inhibition and augmentation of progesterone production during pregnancy: Effects on parturition in rhesus monkeys. Am J Obstet Gynecol 1997;176:682–91.
Novy MJ, Haluska GJ. Endocrine and paracrine control of parturition in rhesus monkeys. In: McNellis D, Challis JRG, MacDonald PC, Nathanielsz PW, Roberts JM, eds. The onset of labor: Cellular and integrative mechanisms. New York: Perinatology Press, 1988:321–37.
Challis JRG, Matthews SG, Gibb W, Lye SJ. Endocrine and paracrine regulation of birth at term and preterm. Endocr Rev 2000;21:514–50.
Vadillo-Ortega F, Gonzalez-Avila G, Furth E, et al. 92-kd type IV collagenase (matrix metalloproteinase-9) activity in human amniochorion increases with labor. Am J Pathol 1995;146:148–56.
Kelly RW. Pregnancy maintenance and parturition: The role of prostaglandin in manipulating the immune and inflammatory response. Endocr Rev 1994;15:684–706.
MacDonald PC, Koga S, Casey ML. Decidual activation in parturition: Examination of amniotic fluid for mediators of the inflammatory response. Ann N Y Acad Sci 1991;622:315–30.
Bethea CL, Gravett MG, Sadowsky DW, Haluska GJ, Axthelm MK, Novy MJ. Amniotic fluid prolactin is decreased by experimental intrauterine infection or IL-1β infusion but not via prostaglandins in pregnant rhesus macaques. Biol Reprod 1998; 58:1385–93.
Mitchell BF, Wong S. Changes in 17β,20α-hydroxysteroid dehydrogenase activity supporting an increase in the estrogen/ progesterone ratio of human fetal membranes at parturition. Am J Obstet Gynecol 1993;168:1377–85.
Henderson D, Wilson T. Reduced binding of progesterone receptor to its nuclear response element after human labor onset. Am J Obstet Gynecol 2001;185:579–85.
Vegeto E, Shahbaz MM, Wen DX, Goldman ME, O’Malley BW, McDonnell DP. Human progesterone receptor A form is a cell- and promoter-specific repressor of human progesterone receptor B function. Mol Endocrinol 1993;7:1244–55.
Duffy DM, WeUs TR, Haluska GJ, Stouffer RL. The ratio of progesterone receptor isoforms changes in the monkey corpus luteum during the luteal phase of the menstrual cycle. Biol Reprod 1997;57:693–9.
Chibbar R, Wong S, Miller FD, Mitchell BF. Estrogen stimulates oxytocin gene expression in human choriodecidua. J Clin Endocrinol Metab 1995;80:567–72.
Hild-Petito S, Verhage HG, Fazleabas AT. Immunocytochemical localization of estrogen and progestin receptors in the baboon (Papio anubis) uterus during implantation and pregnancy. Endocrinology 1992;130:2343–53.
Padayachi T, Pegoraro RJ, Rom L, Joubert SM. Enzyme immunoassay of estrogen and progesterone receptors in uterine and intrauterine tissue during human pregnancy and labor. J Steroid Biochem Mol Biol 1990;37:509–11.
Rezapour M, Backstrom T, Lindblom B, Ulmsten U. Sex steroid receptors and human parturition. Obstet Gynecol 1997;89:918–24.
Slayden OD, Koji T, Brenner RM. Microwave stabilization enhances immunocytochemical detection of estrogen receptor in frozen sections of macaque oviduct. Endocrinology 1995; 136: 4012–21.
Slayden OD, Hirst JJ, Brenner RM. Estrogen action in the reproductive tract of rhesus monkeys during antiprogestin treatment. Endocrinology 1993;132:1845–56.
Green GL, Harris K, Bova R, Kinders R, Moore B, Nolan C. Purification of T47D human progesterone receptor and immunochemical characterization with monoclonal antibodies. Mol Endocrinol 1988;2:714–26.
Chandrasekher YA, Melner MH, Nagalla SR, Stouffer RL. Progesterone receptor, but not estradiol receptor, messenger ribonucleic acid is expressed in luteinizing granulosa cells and the corpus luteum in rhesus monkeys. Endocrinology 1994;135:307–14.
Giannopoulos G, Tulchinsky D. Cytoplasmic and nuclear progestin receptors in human myometrium during the menstrual cycle and in pregnancy at term. J Clin Endocrinol Metab 1979; 49:100–6.
Khan-Dawood FS, Dawood MD. Estrogen and progesterone receptor and hormone levels in human myometrium and placenta in term pregnancy. Am J Obstet Gynecol 1984;150:501–5.
Stanczyk FZ, Hess DL, Namkung PC, Senner JW, Petra PH, Novy MJ. Alterations in sex steroid-binding protein (SBP), corticosteroid-binding globulin (CBG), and steroid hormone concentrations during pregnancy in rhesus macaques. Biol Reprod 1986;35:126–32.
Bernard A, Duffek L, Torok I, Kosasa S. Progesterone and oestradiol levels and cytoplasmic receptor concentrations in the human myometrium at term, before labour and during labour. Acta Physiol Hungary 1988;71:507–10.
McCormick PD, Razel AJ, Spelsberg TC. Absence of high-affinity binding of progesterone (R5020) in human placenta and fetal membranes. Placenta 1981;3:123–32.
Younes MA, Besch NF, Besch PK. Estradiol and progesterone binding in human term placental cytosol. Am J Obstet Gynecol 1981;141:170–4.
Rivera J, Cano A. Oestrogen and progesterone receptors in human term placenta: Measurement by binding assays and immunological methods. Placenta 1989;10:579–99.
Karalis K, Goodwin G, Majzoub JA. Cortisol blockade of progesterone: A possible molecular mechanism involved in the initiation of human labor. Nat Med 1996;2:556–60.
Rossmanith WG, Wolfahrt S, Ecker A, Eberhardt E. The demonstration of progesterone, but not of estrogen, receptors in the developing human placenta. Horm Metab Res 1997;29:604–10.
Shanker YG, Rao AJ. Progesterone receptor expression in the human placenta. Mol Hum Reprod 1998;5:481–6.
Vadillo-Ortega F, Sadowsky DW, Haluska GJ, et al. Identification of matrix metalloproteinase-9 in amniotic fluid and amniochorion in spontaneous labor and after experimental intrauterine infection or interleukin-lβ infusion in pregnant rhesus monkeys. Am J Obstet Gynecol 2002;186:128–38.
Wilson T, Liggins GC, Aimer GP, Watkins EJ. The effects of progesterone on the release of arachidonic acid from human endometrial cells stimulated by histamine. Prostaglandins 1986; 31:343–60.
Ishihara O, Matsuoka K, Kinoshita K, Sullivan MH, Elder MG. Interleukin-1 beta-stimulated PGE2 production from early first trimester human decidual cells is inhibited by dexamethasone and progesterone. Prostaglandins 1995;49:15–26.
Kalkhoven E, Wissink S, van der Saag PT, van der Burg B. Negative interaction between the ReIA (p65) subunit of NF-KB and the progesterone receptor. J Biol Chem 1996;271:6217–24.
Kosaka T, Miyara A, Ihara H, et al. Characterization of the human gene (PTGS-2) encoding prostaglandin endoperoxide synthease 2. Eur J Cell Biol 1994;221:889–97.
Kelly RW, King AE, Critchley HOD. Cytokine control in human endometrium. Reproduction 2001;121:3–19.
Allport VC, Pieber D, Slater DM, Newton R, White JO, Bennett PR. Human labour is associated with nuclear factor-kappaB activity which mediates cyclo-oxygenase-2 expression and is involved with functional progesterone withdrawal. Mol Hum Reprod 2001;7:581–6.
Greenland KJ, Jantke I, Jenatschke S, Bracken KE, Vinson C, Gellersen B. The human NAD+-dependent 15-hydroxyprostaglandin dehydrogenase gene promoter is controlled by Ets and activating protein-1 transcription factors and progesterone. Endocrinology 2000;141:581–97.
Mitchell BF, Challis JRG. Estrogen and progesterone metabolism in human fetal membranes. In: Mitchell BF, ed. The physiology and biochemistry of human fetal membranes. Ithaca, NY: Perinatology Press, 1988:5–28.
Sun M, Ramirez M, Challis JRG, Gibb W. Immunohistochemical localization of the glucocorticoid receptor in human fetal membranes and decidua throughout gestation. J Endocrinol 1996;149:243–8.
Grazzini E, Guillon G, Mouillac B, Zingg HH. Inhibition of oxytocin receptor function by direct binding of progesterone. Nature 1998;392:509–12.
Challis JRG, Patel FA, Pomini F. Prostaglandin dehydrogenase and the initiation of labor. J Perinatal Med 1999;27:26–34.
Ogle TF, Dai D, George P, Mahesh VB. Regulation of the progesterone receptor and estrogen receptor in decidua basalis by progesterone and estradiol during pregnancy. Biol Reprod 1998; 58:1188–98.
Kastner P, Krust A, Turcotte B. Two distinct estrogen regulated promoters generate transcripts encoding the two functionally different human progesterone receptor forms A and B. EMBO J 1990;9:1603–14.
Sartorius CA, Melville MY, Hovland AR, Tung L, Takimoto GS, Horwitz KB. A third transactivation function (AF3) of human progesterone receptors located in the unique N-terminal segment of the B-isoform. Mol Endocrinol 1994;8:1347–60.
Conneely OM, Lydon JP, DeMayo F, O’Malley BW. Reproductive functions of the progesterone receptor. J Soc Gynecol Investig 2000;7:25–32.
Pieber D, Allport VC, Hills F, Johnson M, Bennett PR. Interactions between progesterone receptor isoforms in myometrial cells in human labour. Mol Hum Reprod 2001;7:875–9.
Gao J, Mazella J, Tang M, Tseng L. Ligand-activated progesterone receptor isoform hPR-A is a stronger transactivator than hPR-B for the expression of IGFBP-1 (insulin-like growth factor binding protein-1) in human endometrial stromal cells. Mol Endocrinol 2000;14:1954–61.
Jones CA, Finlay-Jones JJ, Hart PH. Type-1 and type-2 cytokines in human late-gestation decidual tissue. Biol Reprod 1997; 57:303–11.
Haluska GJ, Kaler CA, Cook MJ, Novy MJ. Prostaglandin production during spontaneous labor and after treatment with RU486 in pregnant rhesus macaques. Biol Reprod 1994;51: 760–5.
Mijovic JE, Zakar T, Angelova J, Olson DM. Prostaglandin endoperoxide H synthase mRNA expression in the human amnion and decidua during pregnancy and in the amnion at preterm labour. Mol Hum Reprod 1999;5:182–7.
Casey ML, MacDonald PC. Human parturition: Distinction between the initiation of parturition and the onset of labor. Semin Reprod Endocrinol 1993;11:272–84.
Weiss G. Endocrinology of parturition. J Clin Endocrinol Metab 2000;85:4421–5.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by NIH grants HD06159 (to MJN), HD19182 (to RMB), RR00163, and HD18185.
We gratefully acknowledge the expert assistance of Noreen Currier, Kunie Mah, and Michael Cook in carrying out this work. We thank Dr. Geoffrey Greene, University of Chicago, for the JZB39 antibody.
Rights and permissions
About this article
Cite this article
Haluska, G.J., Wells, T.R., Hirst, J.J. et al. Progesterone Receptor Localization and Isoforms in Myometrium, Decidua, and Fetal Membranes From Rhesus Macaques: Evidence for Function Progresterone Withdrawal at Parturtion. Reprod. Sci. 9, 125–136 (2002). https://doi.org/10.1177/107155760200900303
Published:
Issue Date:
DOI: https://doi.org/10.1177/107155760200900303