Abstract
Urban green spaces are known to be subjected to additional anthropogenic stress. Using native plants in monoculture, i.e., planting trees of the same species, may favour disease outbreaks and plant death. Non-native plants to be used in large cities for creating sustainable plantations are being searched for worldwide. Meanwhile, it is necessary to control plant pathogens in the variable conditions of the region and natural habitat. In Siberian cities, non-native European trees are used, and non-native Siberian plants are introduced in Belarus. This article reports long-term observations of foliar fungal pathogens attacking 21 woody plants (19 European, 2 Siberian) in Siberian and Belarusian cities. In both regions, 48 leaf fungal pathogens were detected, with powdery mildew fungi predominating in Belarus and leaf spotting fungi prevailing in Siberia. In both research regions, the greatest number of fungal species was found on Syringa vulgaris L. In Siberia, many pathogens were found on the non-native European plant Berberis vulgaris (9 species) and native plant Caragana arborescens Lam. (8 species). We have not detected the pathogens on European plants: Acer campestre L., Acer platanoides L., Euonymus europaeus L., Lonicera caprifolium L. in urban green areas in Siberia, while we have identified one to four foliar fungal pathogens on these plants in Belarus. To sum up: more pathogens were found on native plants in Siberia and Belarus; some leaf pathogen species (Sawadaea tulasnei (Fuckel) Homma, Erysiphe alphitoides (Griffon & Maubl.) U. Braun & S. Takam., Cladosporium syringae (Oudem.) Montem., Erysiphe syringae Schwein., Erysiphe palczewskii (Jacz.) U. Braun & S. Takam.) followed their host plants (Acer tataricum L., Syringa vulgaris L., Caragana arborescens Lam.) when introduced into new areas; and some local pathogens were also observed to spread to the non-native plants of closely related species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Woody plants are of particular importance for urban green spaces since they form the structure of gardens and parks, create a long-term shape of landscape objects, have a significant artistic impact, and considerably affect the microclimate and sanitary conditions of urban areas. The state of woody plants in urbanized areas largely determines the quality of the environment, the aesthetics of the urban landscape, and the physical and psycho-emotional health of urban residents. However, species sustainability and the extent of the impact of urban environmental factors on vegetation are known to influence the physiological state of plants, their ornamental features, longevity in gardens and parks (Sucharzewska, 2010; Timofeeva, 2014; Kumar, 2018; Desprez-Loustau et al., 2019; Faticov et al., 2020). The range of plants used in urban green areas tends to include native woody plants. More than 50% of native plants in plantings contribute to the development of more pathogens (Tomoshevich, 2019). Using plant monocultures favour disease outbreaks and the consequent mortality of trees in urban green areas (Heiniger and Rigling, 1994; Brasier and Buck, 2001; Brasier and Kirk, 2010; Buiteveld et al., 2015; Jurisoo et al., 2019; Selikhovkin et al., 2020).
Non-native trees are planted in urban greeneries in both Europe and Asia. The global practice of introducing non-native species, varieties, and forms of trees and shrubs in the landscaping of large cities has shown the need for multi-species plantations (various species of native and non-native woody plants) that are more resistant to pathogenic complexes (Koropachinskii et al., 2011). Non-native plants have been reported to be more resistant, at least at the first stages of introduction, due to differences in phases of ontogenesis of plants and local harmful species, the absence of syngenetic pathogens at the new site, and some other factors (Tomoshevich, 2009; Tomoshevich and Banaev, 2013).
However, research indicates the possibility for new variants of pathogen complexes to emerge and their infectious potential to increase ex situ (Lebeda et al., 2008; Takamatsu et al., 2016; Jakuschkin et al., 2016; Yin et al., 2020).
Studies conducted in Siberian cities found that among non-native species, the highest resistance was in plants of Far Eastern and North American origin (Tomoshevich, 2019; Tomoshevich and Banaev, 2013). European non-native plants in Siberia proved to have variable resistance to leaf pathogens. It is unclear whether the plant resistance to pathogens is due to the specific characteristics of the plant species or to the climate impact. In this regard, it is worth investigating and comparing the species composition of pathogens, taking into account their damage, on European plants in their homeland (Europe) and in the new climatic conditions, in Siberia. In European conditions, urban green areas of Belarusian cities feature the key factor for comparing the species composition of leaf pathogens, i.e., the similar principles in urban planting and the maximum contrasting climatic conditions. Of particular interest are European plants Quercus robur L. and Syringa vulgaris L., widely introduced into urban green spaces in Siberian cities in recent years, and Siberian plants Caragana arborescens Lam. and Rhamnus cathartica L., becoming increasingly common in Belarusian cities.
At the first stages, it is essential to identify the species composition of leaf pathogens of woody plants, the damage caused, and the frequency of occurrence. It is known that some pathogens that follow their host plant may fail to adapt to local climatic conditions or manifest themselves in some years under specific weather conditions and cause damage (Tomoshevich, 2019).
In this article, we summarize observations of leaf pathogens that attack European and Siberian woody plant species in urban green spaces in Siberia and Belarus (Europe), taking into account that European woody plants are non-native to Siberia and Siberian woody plants are non-native to Belarus (Europe). We also discuss how the species composition of pathogens on European non-native plants in Siberian cities will differ compared to the same plants in urban plantations in Belarus, where European plants are native. Moreover, we consider how non-native plants without close relatives in the local flora are affected by leaf pathogens and discuss the potential for new pathogens to be found that may pose a threat to native and non-native woody plants, both in Siberian and in Belarusian (European) conditions.
MATERIALS AND METHODS
Collecting Samples in Urban Green Areas
In 5 Siberian cities, 19 European plants were used in urban plantations, while only two Siberian woody plants were found in urban green spaces in Belarus; all these species were taken as model plants for research (Table 1).
Phytopathological surveys of 21 species of woody plants (19 European and 2 Siberian) were conducted annually in Siberian cities (Novosibirsk, Krasnoyarsk, Barnaul, Tomsk, Kemerovo) and Belarusian cities (Baranovichi, Braslav, Brest, Vitebsk, Gomel, Grodno, Dzerzhinsk, Minsk, Orsha, Polotsk, Soligorsk) (Fig. 1).
Inspection and sampling was continuously conducted for 19 years (2000–2018) throughout the summer periods in all cities. The following categories of urban green spaces were considered: parks, squares, gardens, highways, boulevards, territories adjacent to the house, intra-quarter territories, street plantings, etc. Trees and shrubs in all urban green areas (more than 100 plantation objects), including arboreta, were studied. In different parks, one woody plant species was found to be represented by a different number of specimens (from 3 to 50). At each plantation object, all plants of the same species were examined from four cardinal directions, large trees up to 2 m from the ground. The leaves with symptoms of the disease were incorporated into the herbarium. When examining woody plants, we made the records of disease damage. The plant damage area was evaluated according to a special scale: 1 point, 1–10% damage; 2 points, 11–25%; 3 points, 26–50%; and 4 points, >50% (Dudka et al., 1982).
Given the heterogeneous occurrence of woody plants in different categories of urban green spaces in Belarus and Siberia, we provide a general profile of the occurrence of plants in the study area in Table 1.
Microscopy and Identification of Pathogens
Sections of fruit bodies were made with a blade and examined under an Olympus SZ51 binocular microscope. Preparations of dry samples for further microscopy were prepared in a mixture of glycerol and alcohol, in a 3% KOH solution, and in distilled water. Erythrosine was used to increase the contrast of a number of preparations. Pathogens were identified using Zeiss Discovery V4, Olympus CX31, and Nikon Eclipse E200 microscopes. More than 1500 samples were examined during the study. When possible, the nomenclature corresponds to Index Fungorum, 2021 (www.indexfungorum.org). Siberian samples of foliar fungal pathogen species that were collected in this study are stored in the Central Siberian Botanical Garden of the Siberian Branch of the Russian Academy of Sciences (Herbarium of the Laboratory of Dendrology, NSC Collection). Belarusian samples are stored at V.F. Kuprevich Institute of Experimental Botany of the National Academy of Science of Belarus (Herbarium of the MSC-F mycology laboratory).
Interpretation and Analysis of Results
The following parameters were applied to assess the damage and occurrence of each pathogen on European and Siberian species of woody plants in green areas of Siberia and Belarus cities: A: pathogen is detected irregularly, i.e., not every year or not in every place where the plant is found; B: detected annually and in all places where the plant grows. A Mann–Whitney U-test was used to compare the series of data on the number of pathogens on leaves of woody plants.
RESULTS
Foliar fungal pathogens were found on 21 (19 European and 2 Siberian species) woody plant species inspected (Table 2). A total of 48 fungal species were detected, with 18 causing leaf spot; 17 species being powdery mildews, 7 species—saprotrophic fungal, 5 species—rusts, and one—fungal species, Taphrina acerina A.G. Eliasson, which caused leaf deformation.
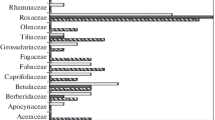
At the same, in green plantings of Siberian cities, no pathogens were found on five European woody plants: Acer campestre, A. platanoides, Euonymus europaeus, Lonicera caprifolium, L. nigra. In cities and parks of Belarus, from one to four pathogens were registered on these species (Fig. 2). Syringa vulgaris was found to be the most affected species in both regions of the study. In Siberia, Berberis vulgaris (European woody plants) were infected by nine pathogens, and Caragana arborescens (native Siberian woody plants) by eight pathogens. While, in Belarus, Berberis vulgaris were infected by six pathogens and Caragana arborescens by two pathogens.
Significant differences have been revealed in the number of pathogens on native and non-native plants. Non-native plants showed a lower number of pathogens, averaging 1.89 ± 0.57, whereas native plants showed 2.31 ± 0.43 (Mann–Whitney test Z = 2.01, p = 0.04).
In Siberian cities, the fungal-caused leaf spot, scab, and similar diseases are dominant (52%), while in Belarusian cities, powdery mildews were found to prevail (48%) (Fig. 3).
In Belarus, 35 pathogens were registered, with 15 not reported in Siberian cities (Table 2). In Siberia, a total of 29 species of fungi were detected, with 13 of them found only in Siberian cities. Twenty species of micromycetes were found on woody plants in both study regions, with 43% of them being powdery mildew fungi distributed on species: Acer tataricum, Berberis vulgaris, Chamaecytisus austriacus, Chamaecytisus ruthenicus, Quercus robur, Salix acutifolia, Salix daphnoides, Syringa josikaea, Syringa vulgaris, Caragana arborescens (Table 2). Four pathogens (Erysiphe lonicerae, Erysiphe penicillata, Podosphaera pannosa, Sawadaea bicornis) were also found in both study regions, but they do not occur on European woody plants in Siberia.
DISCUSSION
Leaf Pathogens Affecting Non-Native and Native Plants
We found that both in Siberia and Belarus, a greater number of pathogens were found on native plants. Siberian woody plant species in green spaces of Belarusian cities were found to be attacked by 1–2 pathogens, whereas in Siberia, 4–9 pathogens were registered on them. In Siberia, European woody plants were also infected with fewer fungal species (more often, 1‒2 fungus species) than in Belarusian cities. The most significant differences were identified for Acer platanoides that was not affected in Siberia but was infested by four pathogens in Belarus and for Caragana arborescens that was in contrast affected in Siberia by eight pathogens and in Belarus by two pathogens. This fact was confirmed by other researchers who found fewer plant pathogens on non-native plants than on native plants (Mitchell and Power, 2003; Kleunen and Fischer, 2009).
Analysis of the Species Composition of Pathogens Found on European Plants in Siberia and Belarus (Europe)
At the same, in green plantings of Siberian cities, no pathogens were found on five European woody plants: Acer campestre, A. platanoides, Euonymus europaeus, Lonicera caprifolium, L. nigra. In cities and parks of Belarus, from one to four pathogens were registered on these species (Table 2). Pathogens were not found only on one species Crataegus nigra in urban green areas in Belarus.
More surprisingly, four European woody plants (non-native) in green plantings in Siberia had 4‒9 pathogens: Berberis vulgaris, Chamaecytisus ruthenicus, Quercus robur, Syringa vulgaris (Table 2). It may be due to the wide distribution of these woody plants in plantations in Siberia (Table 1). Two of them—Berberis vulgaris, Chamaecytisus ruthenicus—were attacked by a greater number of leaf pathogens than in green plantings in Belarus (Fig. 2).
Ascochyta borjomi and Phyllosticta caraganae, fungi widespread on the Siberian native woody plant Caragana arborescens, were found on Chamaecytisus ruthenicus (non-native woody plant).
Of particular interest is the species Pleiochaeta setosa, causing leaf spot and root rot on various herbaceous lupin species (Lupinus sp.). Pleiochaeta setosa, a dangerous pathogen infesting lupin worldwide (Paulitz, 1992; Yang and Sweetingham, 2002; Garibaldi et al., 2012; Gur and Om, 2015; Luckett et al., 2009), was also registered in Belarus (Mikobiota Belorussko-Valdajskogo poozer’ya, 2013). Nevertheless, the pathogen was found in Siberian greenspaces on the woody plant Chamaecytisus ruthenicus in the medium degree of damage (Fig. 4). Earlier, Pleiochaeta setosa was detected on woody plants of the legume family: on Genista tinctoria in Oregon (USA) (Sahakian, 1996) and Chamaecytisus supinus (L.) Link. in Poland (Mułenko et al., 2008).
Analysis of the Species Composition of Pathogens Found on Siberian Plants in Siberia and Belarus (Europe)
In green spaces of Belarusian cities, Siberian species of woody plants Caragana arborescens and Rhamnus cathartica were attacked mainly by powdery mildew fungi: Erysiphe palczewskii, Erysiphe robiniae, Erysiphe friesii. In Siberia, they were also found to have rust and leaf spots (Table 2).
In the 1960s and 1970s, the leaf pathogen Erysiphe palczewskii was endemic to the Far East (Koval’ and Nelen, 1970; Nelen, 1963, 1972). Extensive use of the Siberian species Caragana arborescens as an ornamental plant in the landscaping of cities in the Far East allowed Е. palczewskii to displace Trichocladia caragana. At the beginning of the 1980s, the occurrence of the fungus in the European part of the former USSR and the epiphytotic character of the disease development were reported (Gelyuta, 1981; Gelyuta and Gorlenko, 1984). Recently, many researchers have been observing this fungus further advancing from the East to the West (Huhtinen et al., 2001; Vajna, 2006; Lebeda et al., 2008; Mieslerová et al., 2020). Most likely, Erysiphe palczewskii entered Belarusian cities together with the Siberian plant Caragana arborescens.
Leaf Pathogens of Non-native Plants with no Close Relatives in the Local Flora
Some pathogens have been found to affect both native and closely related European woody plants in Siberia. For example, Coryneum foliicola is widely found in Siberia on a native plant Crategus sanguinea Pall. and has been found on Crataegus nigra (European woody plant). Similarly, Passalora rosae actively attacks native species Rosa acicularis Lindl., R. majalis Herrn, R. spinosissima L. and has been found on European species Rosa glauca (Fig. 5). These pathogens have a vast distribution area but were not registered on native European woody plants in Belarusian cities.
Meanwhile, some species of leaf pathogens (Sawadaea tulasnei, Erysiphe alphitoides, Cladosporium syringae, Erysiphe syringae) were possibly brought to Siberia with their plant hosts (Acer tataricum, Quercus robur, Syringa vulgaris (Fig. 6)), since there are no closely related species in this Region.
Symptoms of powdery mildew infection on plant-hosts: (a) Acer tataricum in Kemerovo; (b) Acer tataricum in Novosibirsk; (c) chasmothecia Sawadaea tulasnei; symptoms of powdery mildew infection on plant-hosts: (d) Quercus robur in Novosibirsk; (e) Quercus robur in Krasnoyarsk; (f) chasmothecia Erysiphe alphitoides; (g) symptoms of powdery mildew infection on Syringa vulgaris in Barnaul; (h) chasmothecia Erysiphe syringae.
Also, non-native European plants Acer campestre, A. platanoides, Euonymus europaeus, Alnus glutinosa, A. incana have no closely related representatives in Siberia, but they have not brought pathogens from the European region. The fungus Mycopappus alni was registered on Alnus glutinosa and A. incana plants in Novosibirsk, while it remains to be determined how it appeared in Siberia. M. alni was first found in Canada and the United States on Alnus rubra and A. sinuata in the late 1980s (Redhead and White, 1985). Later, M. alni was detected in the Far East on leaves of Alnus hirsuta, as well as on hawthorn and chokeberry leaves in Korea and Turkey (Braun et al., 2000; Lee et al., 2013; Park et al., 2013). We detected this fungus for the first time in Russia in 2005 in the arboretum of the Siberian Branch of the Russian Academy of Sciences on Alnus glutinosa and A. incana (Tomoshevich, 2008). The fungus is not found every year, but under favorable weather conditions, it causes serious damage (leaf damage up to 80–100%), resulting in curling and falling of leaves on lower branches by mid-August. Earlier, we noted that the fungus Mycopappus alni was likely to appear in Europe (Tomoshevich et al., 2013).
Pathogen Impact on Woody Plants in Siberia and Belarus
It should be noted that in green areas of Belarus, pathogens, excluding Ascochyta syringae and Cladosporium macrocarpum, are found every year and at all locations (Flora of Belarus, 2015). However, in Siberia, only seven pathogens (Erysiphe adunca, Erysiphe alphitoides, Erysiphe berberidis, Erysiphe palczewskii, Erysiphe syringae, Sawadaea tulasnei, Septoria syringae) are found every year and at all locations where the plants grow (values A and B, Table 3).
In Belarus (Europe), seven pathogens (Erysiphe adunca, Erysiphe alphitoides, Erysiphe berberidis, Erysiphe palczewskii, Erysiphe syringae, Sawadaea tulasnei, Puccinia graminis) cause 26–50% or more damage, whereas in Siberia, only three pathogens (Septoria syringae, Sawadaea tulasnei, Erysiphe trifoliorum) are more aggressive (values 3–4, Table 3). This fact proves native species to be affected to a greater extent in the region of origin.
Leaf pathogens Alternaria alternata, Cladosporium herbarum, Erysiphe alphitoides, Erysiphe palczewskii, Sphaerulina berberidis were found to develop during in the same months in Siberia and Belarus (Table 3). However, most fungi occur in green areas of Siberia a month later or have a longer period of development than in the Belarusian cities. The exception is Puccinia graminis recorded in Siberia in May–June. The later occurrence of leaf pathogens in Siberia is related to the delayed phenophases of European woody plants. For example, in the European part of Russia, the first oak shoots open in mid-April (Shimanyuk, 1964). In Siberia, the opening of buds and the growth of Quercus robur shoots were recorded more than a month later (late May-early June) (Vstovskaya and Koropachinskiy, 2005).
CONCLUSIONS
The research has shown that using native and non-native woody plants in parks and gardens enables creating more sustainable green spaces. However, continuous monitoring of European woody plants in Siberia and Siberian plants in Belarus should be carried out since the transmission of local pathogens to introduced plants and increased aggressiveness of pathogens that were brought to the region by their host plants are observed. It is particularly the case for powdery mildew fungi since these biotrophic pathogens can persist in twigs and seeds, such as oak acorns.
When adapting to environmental conditions, the non-native woody plants change their development rhythm and decrease the resistance, affecting the degree of pathogenic fungi development. For example, in the sharply continental conditions of Siberia, the delayed appearance of disease symptoms is observed due to the shift in the phenophases of plants, but the aggressiveness of pathogens increases compared to the regions of Belarus.
The new findings on the development of Mycopappus alni and Pleiochaeta setosa for Siberia suggest that they are likely to appear on woody plants in Europe and have a possible invasive status.
The first findings of pathogen analysis on non-native and native plants in Siberia and Belarus (Europe) show that using non-native plants in urban plantations can cause new associations between plants and pathogens and even more severe plant lesions. Future molecular studies are needed to understand the distribution of the fungus more accurately, especially in urban areas. In controversial cases, pathogen identification and replenishment of GenBank with parallel analyses of samples of Siberian and European fungus samples will be required.
REFERENCES
Belomesyantseva, D.B., Shabashova, T.G., and Parfenov, V.I., Flora of Belarus. Fungi, vol. 2: Anamorphic fungi, Book 1: Dark-colored hyphomycetes (Flora Belarusi. Griby, Tom 2: Anamorfnye Griby, Kniga 1: Temnookrashennye Gifomitsety), Minsk: Belarus. Navuka, 2015.
Brasier, C.M. and Buck, K.W., Rapid evolutionary changesin a globally invading fungal pathogen (Dutch elm disease), Biol. Invasions, 2001, vol. 3, pp. 223–233.
Brasier, C.M. and Kirk, S.A., Rapid emergence of hybridsbetween the two subspecies of Ophiostoma novo-ulmiwith ahigh level of pathogenic fitness, Plant Pathol., 2010, vol. 59, pp. 186–199. https://doi.org/10.1111/j.1365-3059.2009.02157.x
Braun, U., Mel’nik, V., Huseyinov, E., and Selcuk, F., Mycopappus alni on species of Betula and Pyrus from Turkey, Mycol. Phytopathol., 2000, vol. 34, no. 6, pp. 1–2.
Buiteveld, J., Werf, B., and Hiemstra, J.A., Comparison of commercial elm cultivars and promising unreleased Dutch clones for resistance to Ophiostoma novo-ulmi, iForest - Biogeosci. For., 2015, vol. 8, no. 2, pp. 158–164. https://doi.org/10.3832/ifor1209-008
Desprez-Loustau, M.-L., Frederic Hamelin, M., and Marçais, B., The ecological and evolutionary trajectory of oak powdery mildew in Europe, in Wildlife Disease Ecology: Linking Theory to Data and Application (Ecological Reviews), Wilson, K., Fenton, A., and Tompkins, D., Eds., Cambridge: Cambridge Univ. Press, 2019, pp. 429–457. https://doi.org/10.1017/9781316479964.015
Dudka, I.A., Vasser, S.P., and Ellanskaya, I.A., Metody eksperimental’noi mikologii (Methods of Experimental Mycology), Kiev: Naukova Dumka, 1982.
Faticov, M., Ekholm, A., Roslin, T., Ayco, J., and Tack, M., Climate and host genotype jointly shape tree phenology, disease levels and insect attacks, Oikos, 2020, vol. 129, pp. 391–401. https://doi.org/10.1111/oik.06707
Garibaldi, A., Bertetti, D., Poli, A., and Gullino, M.L., First report of leaf spot of garden lupin (Lupinus polyphyllus) caused by Pleiochaeta setosa in Italy, Plant Dis., 2012, vol. 96, p. 909.
Gelyuta, V.P., New species of genus Microsphaera Lev., Ukr. Bot. J., 1981, vol. 38, no. 6, pp. 50–52.
Gelyuta, V.P. and Gorlenko, V.P., Microsphaera palczewskii Jacz. in USSR, Mycol. Phytopathol., 1984, vol. 18, no. 3, pp. 177–182.
Girilovich, I.S., Powdery Mildews (Order Erysiphales) in Belarus, Minsk: BSU, 2018.
Gur, L. and Om, F., First report of leaf spot on blue lupin (Lupinus pilosus) in Israel caused by Pleiochaeta setosa, Plant Dis., 2015, vol. 100, p. 2. https://doi.org/10.1094/PDIS-05-15-0558-PDN
Heiniger, U. and Rigling, D., Biological control of chestnut blight in Europe, Annu. Rev. Phytopathol., 1994, vol. 32, pp. 581–599.
Huhtinen, S., Alanko, P., and Makinen, Y., The invasion history of Microsphaera palczewskii (Erysiphales) in Finland, Karstenia, 2001, vol. 41, pp. 31–36.
Jakuschkin, B., Fievet, V., Schwaller, L., Fort, Th., Robin, C., and Vacher, C., Deciphering the pathobiome: intra- and interkingdom interactions involving the pathogen Erysiphe alphitoides, Microb. Ecol., 2016, vol. 72, pp. 870–880. https://doi.org/10.1007/s00248-016-0777-x
Jurisoo, L., Adamson, K., Padari, A., and Drenkhan, R., Health of elms and Dutch elm disease in Estonia, Eur. J. Plant Pathol., 2019, vol. 154, p. 7. https://doi.org/10.1007/s10658-019-01707-0
Kleunen, M. and Fischer, M., Release from foliar and floral fungal pathogen species does not explain the geographic spread of naturalized North American plants in Europe, J. Ecol., 2009, vol. 97, pp. 385–392.
Koropachinskii, I.Yu., Vstovskaya, T.N., and Tomoshevich, M.A., Immediate tasks of introduction of woody plants in Asian Russia, Contemp. Probl. Ecol., 2011, vol. 4, no. 2, pp. 107–125. https://doi.org/10.1134/S1995425511020019
Koval’, E.Z. and Nelen, E.S., Powdery mildew of Siberian peashrub in the Far East, Byull. Gl. Bot. Sada, 1970, vol. 76, pp. 92–95.
Kumar, V., Effect of epidemiological factors on percent disease index of rose powdery mildew caused by Podosphaera pannosa (Wallr.) de Bary, J. Crop Weed, 2018, vol.14, no. 2, pp. 137–142.
Lebeda, A., Mieslerova, B., and Sedla, M., First report of Erysiphe palczewskii on Caragana arborescens in the Czech Republic, Plant Pathol., 2008, vol. 57, p. 779.
Lee, S.C., Han, K.S., Park, J.H., and Shin, H.D., First report of frosty mildew caused by Mycopappus alni on Asian pear in Korea, Plant Dis., 2013, vol. 97, no. 1, pp. 147–147. https://doi.org/10.1094/PDIS-08-12-0730-PDN
Luckett, D.J., Cowley, R.B., Richards, M.F., and Roberts, D.M., Breeding Lupinus albus for resistance to the root pathogen Pleiochaeta setosa, Eur. J. Plant Pathol., 2009, vol. 125, pp. 131–141.
Mieslerova, B., Sedlarova, M., Michutova, M., Petrekova, V., Cook, R., and Lebeda, A., Powdery mildews on trees and shrubs in botanical gardens, parks and urban green areas in the Czech Republic, Forests, 2020, vol. 11, p. 967. https://doi.org/10.3390/f11090967
Mikobiota Belorussko-Valdajskogo poozer’ya. (Mycobiota of the Belarusian-Valdai Lakeland), Kovalenko, A.E., Ed., Moscow: KMK, 2013.
Mitchell, C.E. and Power, A.G., Release of invasive plants from fungal and viral pathogens, Nature, 2003, vol. 421, pp. 625–627.
Mułenko, W., Majewski, T., and Ruszkiewicz-Michalska, M.A., Preliminary Checklist of Micromycetes in Poland, Krakow: W. Szafer. Inst. Bot., Polish Acad. Sci., 2008. 2008ISBN 978-83-89648-75-4
Nelen, E.S., Pathogenic mycoflora of green plantations in the cities of Amur oblast, Soobshch. Dal’nevost. Fil. Akad. Nauk SSSR, 1963, vol. 17, pp. 69–72.
Nelen, E.S., Diseases of decorative trees and scrubs in Amur region, Byull. Gl. Bot. Sada, 1972, vol. 84, pp. 102–106.
Park, J.H., Cho, S-E., Lee, S.H., and Shin, H-D., First Report of Frosty Mildew on Salix koreensis Caused by Mycopappus alni in Korea, J. Phytopathol., 2013, vol. 161, pp. 11–12. https://doi.org/10.1111/jph.12133
Paulitz, T.C., First report of brown spot of lupines caused by Pleiochaeta setosa in Canada, Plant Dis., 1992, vol. 76, no 11, p. 1185. https://doi.org/10.1094/PD-76-1185C
Redhead, S.A. and White, G.P., Mycopappus, a new genus of leaf pathogens, and two parasitic Anguillospora species, Can. J. Bot., 1985, vol. 63, pp. 1429–1435.
Sahakian, V., First Report of Pleiochaeta setosa Leaf Spot on Genista tinctoria in Oregon, Plant Dis., 1996, vol. 80, p. 710. https://doi.org/10.1094/PD-80-0710C
Selikhovkin, A.V., Drenkhan, R., Mandelshtam, M., and Musolin, D., Invasions of insect pests and fungal pathogens of woody plants into the northwestern part of European Russia, Earth Sci., 2020, vol. 65, no. 2, pp. 263–283. https://doi.org/10.21638/spbu07.2020.203
Shimanyuk, A.P., Biologiya drevesnyh i kustarnikovyh porod SSSR (Biology of Wood and Scrubby Plants in USSR), Moscow: Prosveshchenie, 1964.
Sucharzewska, E., Key survival strategies of the Sawadaea tulasnei Parasite on its Acer platanoides host under conditions of varied anthropopression, Pol. J. Environ. Stud., 2010, vol. 19, no 5, pp. 1013–1017.
Takamatsu, S., Shiroya, Y., and Seko, Y., Geographical and spatial distributions of two Erysiphe species occurring on lilacs (Syringa spp.), Mycoscience, 2016, vol. 57, pp. 349–355.
Timofeeva, V.A., Bolezni i vrediteli dekorativnykh rastenii v nasazhdeniyakh Belarusi (Diseases and Pests of Ornamental Plants in the Plantations of Belarus), Minsk: Belarus. Navuka, 2014.
Tomoshevich, M.A., First record of Mycopappus alni from Russia, Mycol. Phytopathol., 2008, vol. 42, no 5, pp. 498–499.
Tomoshevich, M.A., Pathogenic mycobiota on trees in Novosibirsk plantations, Contemp. Probl. Ecol., 2009, vol. 2, no. 4, pp. 382–387.
Tomoshevich, M.A. and Banaev, E.V., Concerning regularities in the structure of pathogenic micromycetes on leaves of woody plants in urban ecosystems of Siberia, Contemp. Probl. Ecol., 2013, vol. 6, no. 4, pp. 396–401.
Tomoshevich, M., Kirichenko, N., Holmes, K., and K-enis, M., Foliar fungal pathogens of European woody plants in Siberia: an early warning of potential threats?, For. Pathol., 2013, vol. 43, no. 5, pp. 345–359. https://doi.org/10.1111/efp.12036
Tomoshevich, M.A., Interrelations between Alien and native foliar fungal pathogens and woody plants in Siberia, Contemp. Probl. Ecol., 2019, vol. 12, no. 6, pp. 642–657. https://doi.org/10.1134/S1995425519060143
Vajna, L., First report of powdery mildew on Caragana arborescens in Hungary caused by Erysiphe palczewskii, Plant Pathol., 2006, vol. 55, p. 814.
Vstovskaya, T.N. and Koropachinskiy, I.Yu., Drevesnye rasteniya Central’nogo sibirskogo botanicheskogo sada (Woody Plants of the Central Siberian Botanical Garden), Novosibirsk: Geo, 2005.
Yang, H.A. and Sweetingham, M.W., Variation in morphology and pathogenicity of Pleiochaeta setosa isolates from Lupinus spp. and other legumes, Aust. Plant Pathol., 2002, vol. 31, pp. 273–280.
Yin, C., Zhang, Sh., Liu, L., and Zhang, Y., First report of powdery mildew caused by Erysiphe berberidis on Berberis fortunei in China, Plant Dis., 2020, vol. 105, no. 1, p. 222. https://doi.org/10.1094/PDIS-06-20-1300-PDN
Funding
This research was funded by the National Academy of Sciences of Belarus, State program of scientific research “Nature management and ecology” Projects no. 20190899, 20190898 and was funded by the Ministry of Science and Higher Education of the Russian Federation program of scientific research “Theoretical and applied aspects of studying gene pools of natural plant populations and conservation of plant diversity “outside the typical environment” (ex situ)” (АААА-А21-121011290027-6). In preparing the publication, materials of the bioresource scientific collection of the CSBG SB RAS “Collections of living plants indoors and outdoors” USU_440534 (Novosibirsk, Russia) were used.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Tomoshevich, M.A., Belomesyatseva, D., Banaev, E.V. et al. Comparative Analysis of Foliar Diseases of Some Native and Non-Native Tree Species in Belarus and Siberia. Contemp. Probl. Ecol. 16, 217–229 (2023). https://doi.org/10.1134/S1995425523020166
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995425523020166