Abstract
Fish introductions are known to be dangerous due to the unexpected spread of diseases and disorganization of the food web. In 1986, the resident sockeye salmon Oncorhynchus nerka from Kronotskoye Lake was introduced into an isolated lake in Kamchatka inhabited by the native lacustrine Dolly Varden charr Salvelinus malma. Thirty years later, we analyzed the stomach content, stable isotope ratio and lipid fractions in muscles, as well as the parasite fauna of fish to assess the trophic relationships between the two species. It has been found that the sockeye salmon has occupied the planktivorous niche; no diversification into trophic forms depending on the number of gill rakers, like in the ancestral population, was detected. The indigenous benthivorous S. malma is represented by three trophic groups. Mature dwarf individuals, as well as the juveniles, mainly consume insect larvae on the lake slope; large fish feed on amphipods in the same habitats; and 10% of the large Dolly Varden switches to facultative piscivory. Predators die as a result of Diphyllobothrium sp. hyper invasion, which infects fish when feeding on the sockeye salmon. The diversity of parasite fauna is 3/4 poorer in the introduced sockeye in comparison with the donor population; part of the parasite species is obtained by the sockeye from the Dolly Varden.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
In the 1970s–1980s, large-scale introducing of the sockeye salmon Oncorhynchus nerka Walb. from Kronotskoye Lake into the non-fisheries lakes was carried out in Kamchatka in order to increase the fish productivity of the water bodies (Pogodaev and Kurenkov, 2007). The naturalization of residential sockeye was successful in small lakes such as Klyuchevoye, Sevo, and Demidovskoye, as well as in Tolmachevo Reservoir, the largest reservoir on the peninsula (Bugaev and Kirichenko, 2008; Markevich, 2008). Interrogatory data also confirm the naturalization of the sockeye in Khangar Lake.
However, Lake Sevo turned out to be different from other water bodies with successful sockeye introduction because before the sockeye was introduced in 1986 and 1987, it had already been populated by a residential Dolly Varden charr Salvelinus malma Walb. (Pogodaev and Kurenkov, 2007). According to the data from 2007, the sockeye population of Lake Sevo increased significantly. The abundant food supply and the presence of predatory charrs were considered factors that determined successful naturalization (Pogodaev and Kurenkov, 2007). The influence of the sockeye salmon itself on the ecosystem of Lake Sevo was not considered. No study of the parasite fauna of the reservoir was carried before or after the experiment, either. However, the introduction is considered one of the most important reasons for the decline in biological diversity (Vitousek et al., 1997; Olden et al., 2004), since dangerous parasites are introduced into lakes and rivers along with introduced species, which promotes epizootic spread (Johnsen and Jensen, 2006; Boys et al., 2012; Lymbery et al., 2014), resource redistribution, and changes in the trophic structure of ecosystems (Baxter et al., 2004; Eby et al., 2006; Amundsen et al., 2013; Stolbunov et al., 2021). The fauna of small inland water bodies is considered especially vulnerable to the introduction of alien species (Gherardi, 2007; Vitule et al., 2009).
The history of salmonid fish introductions (Aprahamian et al., 2003; Crawford and Muir, 2008; Markevich and Esin, 2019) is full of cases ending in disaster for the recipient ecosystems and native species (Buoro et al., 2016). The introduction of Salmo spp. in the rivers of the Pacific coast of America led to a large-scale death of native Oncorhynchus spp. from vibriosis (Ginetz, 2002). Attempts to reintroduce Salmo salar L. into the Scandinavian rivers led to the death of local populations due to the parasite Gyrodactylus salaris epizooty (Bakkel et al., 1992). In other cases, introduced species displaced native salmon species from their food niches (Eloranta et al., 2015; Nasegava, 2020). The introduction of salmonids into isolated ecosystems with specific endemic fauna most often led to the extinction of the latter (Markevich et al., 2019).
Thus, the purpose of this study was to determine the trophic relationships between the native Dolly Varden and the introduced sockeye salmon in terms of their food composition, stable isotope and lipid ration, and parasite fauna.
MATERIALS AND METHODS
Lake Sevo is located in the spurs of the Valagin Range in central Kamchatka (54.479372′ N, 158.673444′ E) at an altitude of 620 m above sea level. It is a dammed lake, drainless; length of 3.5 km, average width 0.7 km, maximum depth 100 m (Esin and Markevich, 2017).
The study material was collected in August 2015. Fish were caught with gillnets (mesh pitch 20–35 mm) throughout the lake littoral; two large dying Dolly Varden charrs were sampled from the surface of the water. In total, 65 Dolly Varden specimens and 52 specimens of the sockeye salmon were studied according to the standard protocol (45% of fish catches were the Dolly Varden). For each freshly caught fish, FL (cm), Q (g), and maturity stage (MS, points) were determined (Murza and Khristoforov, 1991). In the sockeye salmon, the number of gill rakers on the first gill arch was also counted.
The fish feeding strategy was analyzed from stomach contents (Amundsen et al., 1996). We calculated the frequency of occurrence of various food object groups, i.e., the ratio of the number of fish with a given type of food to the number of fish in the sample (%), as well as the average number of food objects of each group per one fish in the sample (ind.). The commonality of food niches was assessed using the Morisita index modified by Horn (Horn, 1966); the proportion of groups in the diet was determined by the number of victims.
Since a rapid shift to feeding on mass prey is possible for charrs in summer (Tveiten et al., 1996; Jorgensen and Johnsen, 2014), a retrospective assessment of trophic niches was additionally carried out. For this, fish at the 3–4 MS were compared by the ratio of stable isotopes of 14N/15N nitrogen and 12C/13C carbon in muscle tissue (Peterson and Fry, 1987), by the lipid composition of muscles (Bogdan et al., 2001), and by the composition of parasite fauna (Knudsen et al., 2004).
For isotope analysis, samples of muscle tissue were cut out from under the posterior edge of the dorsal fin and dried to constant weight. Mass spectrometric analysis of the samples was carried out at the Instrumental Methods in Ecology Shared Use Center at the Severtsov Institute for Ecology and Evolution of the Russian Academy of Sciences on a Thermo Finnigan Delta V Plus device equipped with a Flash 1112 analyzer. The isotopic composition is presented in pro mille of a deviation (‰) from the standards (atmospheric nitrogen and belemnite sample). Since the δ13C values varied within 10‰, normalization by lipid composition was not performed (Post et al., 2007).
The content of the main lipid fractions in the muscles was assessed spectrophotometrically using a Hatch DR2400 device. Extraction (Folch et al., 1957) was performed from tissue fragments weighing 0.4 ± 0.001 g, followed by washing in 1% KCl solution. The gross lipid concentration was determined by reaction with phosphoric vanillin in 10% H2SO4; optical density was determined at a wavelength of 520 nm (Knicht et al., 1972). The content of triacylglycerides and phospholipids was determined by enzyme-photometrical approach at a wavelength of 505 nm using commercial Spinreact kits in accordance with the manufacturer’s instructions. The values were expressed as % by weight of the tissue samples.
A parasitological analysis of fish was carried out using an incomplete parasitological dissection (Bykhovskaya-Pavlovskaya, 1985). The fish were examined for the presence of parasites on the body surface, in the oral and gill cavities, in the eyes (in the internal environment and lenses), in the body cavity, on the surface of the heart, in the swim bladder, in the ureters, and on the walls and in the cavity of the stomach and intestines. The fish were not examined for the presence of protozoa, myxosporidium (except for Henneguya zschokkei (Gurley, 1894)), or monogeneans. Most of the parasites were identified up to the species level (Opredelitel’…, 1985, 1987). Traditional indicators of infestation were used: extensiveness of invasion is the proportion of infected fish in the sample, %; the abundance index is the average number of parasites per fish in the sample, ind.
To determine the statistical significance of differences between groups of fish in their feeding strategies, isotopic and lipid composition, and parasite infestation, the nonparametric Mann–Whitney test was used after the presence of differences was confirmed by the Kruskal–Wallis test (Conover, 1999). The significance of size trends in changes in the proportion of food objects in the stomachs, the ratio of isotopes and lipids in muscles, and infestation with parasites was determined by the method of least squares (Plokhinskii, 1970). Mathematical data processing was carried out using the StatSoft Statistica v.13 software.
RESULTS
Biological characteristics of fish. In our catches, the Dolly Varden was represented by juveniles with gonads of the second MS (17 specimens, FL 10–20 cm, Q 10–70 g), adults of both sexes at MS 4+ (11 specimens), so-called “dwarfs” with FL 14–20 (on average 17.2 ± 4.28) cm and Q 32–60 (43.7 ± 3.01) g, and large adults of both sexes on the third–fourth MS (37 specimens) with FL 20–42 (27.3 ± 8.90) cm and Q 68–580 (208.9 ± 19.94) g.
The sockeye salmon with FL 14–29 (19.9 ± 3.36) cm and Q 20–110 (53.3 ± 2.40) g had gonads on the fourth MS. In the ancestral (donor) resident population of Kronotskoye Lake, there are two groups in the sockeye salmon population: the one of benthivorous sockeye with few gill rakers (<39) and the other of planktivorous sockeye with dense gill rakers (≥39), which differ in body size (Kurenkov, 1977). In Lake Sevo (as well as in other Kamchatka lakes), the planktivorous densely-rakered sockeye salmon was introduced. In 2015, a sample from Lake Sevo included fish with few (<39) and many (≥39) gill rakers (11 ind. and 41 ind., respectively). These individuals did not differ in body size (t‑test p = 0.281).
Differences in fish nutrition. Eight groups of food objects were distinguished in the stomachs of the studied fish (Table 1). Juveniles and dwarfs of the Dolly Varden predominately fed on chironomid larvae and did not differ in the number of victims of all groups in the stomachs (U‑tests p > 0.05). According to the feeding strategy, large Dolly Varden was divided into benthivorous (58 ind.) and piscivorous (7 ind.). In the stomachs of the first group, besides gammarids (amphipods), chironomid larvae and mollusks were also noted. Fish and benthos were present in the stomachs of the predatory Dolly Varden. Predators were larger than “large” benthophages: FL 31–42 (on average 35.1) cm and Q 215–580 (386.8) g, versus FL 20–39 (26.1) cm and Q 68–480 (181.3) g (t-tests p = 0.009 and p = 0.004 for FL and Q, respectively).
The Sockeye salmon with different number of gill rakers (more or less than 39) did not differ in regards to stomach contents (U-tests, p > 0.05 for all groups of objects). In August, it fed on planktonic crustaceans, gammarids, and insect imagoes. The Sockeye salmon significantly differed from the dwarf, large benthivorous, or predatory Dolly Varden by the high content of planktonic crustaceans in the stomachs (p < 0.001); it differed from the dwarf and benthivorous Dolly Varden by a smaller number of gammarids and mollusks (p < 0.001); insect imagoes (p = 0.010); and chironomid larvae, mayflies, and caddisflies (p < 0.001). The Sockeye salmon had a higher stomach fullness compared to the Dolly Varden (р = 0.006).
The food niches of the Dolly Varden and the sockeye salmon did not overlap. The Morisita index in pairs of sockeye salmon–dwarf Dolly Varden, sockeye salmon–large benthivorous Dolly Varden, and sockeye salmon–predatory Dolly Varden was less than 0.1; in the pair of piscivorous–benthivorous Dolly Varden it was 0.5. The biological significance of the Morisita index was 0.6 (Horn, 1966).
The increase in the size of the benthivorous Dolly Varden coincides with a decrease in the proportion of insect larvae in diet and an increase in the proportion of gammarids. The value of the linear approximation R of the relation FL–the number of chironomid larvae in the stomach reached 0.66 (p = 0.037); for FL–gammarids it was 0.57 (p = 0.043). The Sockeye salmon did not show any change in food composition associated with an increase of FL, R < 0.30 (p > 0.05).
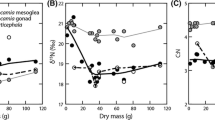
Ratio of stable isotopes in the muscle tissue of fish. The Adult benthivorous Dolly Varden, piscivorous Dolly Varden, and sockeye salmon formed nonoverlapping areas in the δ13С–δ15N space (Fig. 1). The benthivorous Dolly Varden differed from predators of this species, as well as from the sockeye salmon, by a higher δ13С (H2;48 = 36.22, p < 0.001; U-tests p = 0.041 and p < 0.001, respectively). Dwars differed from adult large benthivorous fish by a reduced δ15N (р = 0.045). The predatory Dolly Varden has higher δ15N in comparison to the benthivorous Dolly Varden and the sockeye salmon (H2;48 = 16.67, р = 0.002; U-tests р < 0.001). The Sockeye salmon did not differ significantly in nitrogen signature from benthivorous Dolly Varden (р = 0.322) or in carbon signature from the predatory Dolly Varden (р = 0.287).
For the benthivorous and predatory Dolly Varden, there was a positive correlation between an increase in δ15N and body size (R = 0.72, р = 0.035).
Lipid composition of fish muscle tissue. The sockeye salmon differed from the adult Dolly Varden in a reduced fat content of the muscle tissue (H2;48 = 26.05, p < 0.001; U-tests, p < 0.001); no significant differences in the total lipid concentration were obtained for the various trophic groups of the Dolly Varden (p = 0.174) (Fig. 2a). According to the content of triacylglycerides, dwarfs differed from large benthivorous fish (p = 0.039) and predators (p = 0.003), while large benthophages and predators did not differ significantly (p > 0.05) (Fig. 2b). The sockeye salmon differed by a reduced phospholipid content from the dwarf large benthic, and predatory Dolly Varden (p = 0.021, 0.004, 0.001, respectively), while in both groups of benthivorous Dolly Varden this lipid fraction was lower than in predators (p = 0.049) (Fig. 2c).
Lipid composition in the muscle tissue of the Dolly Varden and the planktivorous sockeye salmon from Lake Sevo: (a) gross concentration of lipids, (b) triacylglycerides, and (c) phospholipids. Mean values (transverse lines), standard errors (rectangles), and limits of variation (vertical lines) are given. Designations of fish groups are same as in Fig. 1.
The growth of the large benthivorous Dolly Varden was associated with a trend towards an increase in the fat content of its muscle tissue (R = 48, p = 0.05).
Composition of fish parasites. Nine species of parasites were found in the examined fish of both species (Table 2). Salmincola edwardsii Olsson, 1869 from the gill cavity and Paracanthobdella livanowi (Epstein, 1966) from the body surface have direct development. The rest of the parasites marked various food preferences of fish, as they were associated with the consumption of different groups of invertebrates. On the surface of the fish body, cysts of the myxosporidium Henneguya zschokkei (Gurley, 1894), whose main hosts are benthic annelids (Okamura et al., 2015), were noted. In the fish eyes, there were metacercarids Diplostomum sp., which infect fish upon contact with lung mollusks (Sudarikov et al., 2002). Crepidostomum farionis Muller, 1874 was determined in the intestines of fish. Its intermediate host is mayfly larvae (Crawford, 1943). Cystidicola farionis Fisher, 1798 was noted in the swim bladder and Echinorhynchus salmonis Muller, 1780, in the intestine. Both of these parasites are associated with amphipods as intermediate hosts (Black and Lankester, 1980; Measures and Bosse, 1993). Plerocercoids of Diphyllobothrium sp., whose intermediate hosts are planktonic crustaceans, were present in the walls of the stomach and on the surface of the internal organs of fish (Delyamure et al., 1985). Proteocephalus longicollis Zeder, 1800 was found in the intestines of fish; its intermediate hosts are cyclops and diaptomuses (Willemse, 1969).
The Benthivorous Dolly Varden, piscivorous Dolly Varden, and sockeye salmon differed in parasite infestation. The first differed from the predatory Dolly Varden and the sockeye salmon by the low abundance of Diphyllobothrium sp. (U-tests, p < 0.001). At the same time, dwarfs differed from large benthivorous fish by a greater abundance of Proteocephalus longicollis (p = 0.008) and a lower invasion of Echinorhynchus salmonis and Cystidicola farionis (p < 0.001). The predatory Dolly Varden differed by an extremely high infestation with Diphyllobothrium sp. (up to 524 ind./fish). The Sockeye salmon was characterized by significantly low invasion of Diplostomum sp., Cystidicola farionis, and Echinorhynchus salmonis (p < 0.001), as well as the absence of Salmincola edwardsii.
The growth of the Dolly Varden of all trophic groups was characterized by an increase in the number of Echinorhynchus salmonis (R = 0.87, p = 0.001) and Cystidicola farionis (R = 0.77, p = 0.003) against the background of a decrease in the number of Proteocephalus longicollis (R = 0.65, p = 0.013).
DISCUSSION
Our study showed that Dolly Varden from Lake Sevo is polymorphic in its ecology. Juveniles mainly feed on insect larvae. With age, the food spectrum expands: gammarids and mollusks are included in the diet. Adults >20 cm feed almost exclusively on gammarids. At the same time, some fish (~10%) matured in the size range of juveniles (males at FL ≥ 13 cm, females at FL ≥ 15 cm) continue feeding on insect larvae without switching to gammarids. Such fish differ from gammarid-consumers in their low nitrogen signature, since amphipods themselves are benthos consumers and enrich large Dolly Varden with heavy isotopes (MacNeil et al., 1997). Despite the obviously higher calorie content of gammarus compared to chironomid larvae, dwarfs store lipids more efficiently than large benthivorous fish.
The composition of Dolly Varden parasites indicates that it feeds on benthos near the lake bottom. Five of the seven parasite species infect fish either by eating benthic organisms (Crepidostomum farionis, Cystidicola farionis, and Echinorhynchus salmonis) or by contact with them (Diplostomum spp., Henneguya zschokkei). Only Proteocephalus longicollis and Diphyllobothrium sp. infect Dolly Varden when feeding on plankton. The large benthivorous Dolly Varden is heavily infested with Cystidicola farionis and Echinorhynchus salmonis, which is associated with its feeding on gammarids.
Among the older and already mature fish ~10% become piscivorous and accelerate in growth. This phenomenon is typical for charrs in isolated lakes of Kamchatka (Esin and Markevich, 2019) and throughout the range of the genus (Reist et al., 2013; Esin and Markevich, 2017). Feeding on their own juveniles and sockeye salmon, the predatory Dolly Varden is reinvaded by Cystidicola farionis, Echinorhynchus salmonis, and Diphyllobothrium sp., and also acquires higher δ15N values (Minagawa and Wada, 1984). The specific lipid composition of predators indicates a good food supply and an increased level of metabolism compared to benthivorous individuals (Houlihan et al., 1993).
Sockeye salmon in Lake Sevo is uniform in nutrition; fish with different numbers of gill rakers (<39 and ≥39) mainly feed on planktonic crustaceans (average 168 ind./fish). The composition of parasites confirms the preferential feeding of sockeye salmon on plankton. Compared to the benthivorous Dolly Varden, sockeye salmon is invaded more by Diphyllobothrium sp., which infects fish when feeding on cyclops, and less by Diplostomum sp., Cystidicola farionis, and Echinorhynchus salmonis, which are transmitted to fish through mollusks and amphipods.
Thus, 30 years after the introduction of the sockeye salmon (about eight generations) in the ecosystem of Lake Sevo, there is stable food segregation between the native Dolly Varden and the introduced species. In contrast to other salmon lakes, such as Inari Lake in Finland, the native species was not displaced from its trophic niche (Eloranta et al., 2015). The food niches of the Dolly Varden and sockeye salmon, according to the results of the Morisita index, do not overlap. Species are separated by isotopic signature, showing segregation of trophic niches over several months, i.e., throughout the spring–summer feeding period (Peterson and Fry, 1987) and, judging by the difference in infestation of Cystidicola farionis and Echinorhynchus salmonis, for at least the last two years (Black and Lankester, 1980; Mikhailova, 2015). The reduced δ13C of the sockeye salmon is associated with its involvement in the secondary cycle of organic matter in the hypolimnion (Wada, 2009). Despite more actively feeding in August, sockeye salmon is characterized by a lower fat content, which may indicate worse feeding conditions when compared with benthivorous Dolly Varden (Bogdan et al., 2001; Houlihan et al., 1993).
Sockeye salmon of Lake Sevo differs from its parental multiraker population from Lake Kronotskoye: in Sevo, individuals with a number of gill rakers <39 appeared, which is typical for specimens with few rakers from Kronotskoye Lake (Markevich, 2008). At the same time, no differences in nutrition were found between the specimens with many and few rakers in Lake Sevo, as well as differences in their infestation with parasites, biological parameters, and stages of gonadal maturity. Thus, in Sevo, sockeye salmon did not follow the scenario of the formation of independent ecological forms.
In addition to Lake Sevo, we have studied the diet and parasite fauna of the sockeye salmon introduced into the Tolmachevo Reservoir (Busarova and Esin, 2017). This population also differs from the parental one (Table 3). Like sockeye salmon from Sevo, the fish has a relatively smaller size and body weight and does not differentiate in terms of nutrition, despite the fact that a third of individuals have a number of gill rakers <39. At the same time, in the Tolmachevsky Reservoir, sockeye salmon occupied the niche of a benthivorous fish. In lake Sevo this did not happen, because the native Dolly Varden, which is characterized by feeding from the bottom (Esin and Markevich, 2017), has competitive advantages over the sockeye salmon when feeding on benthophages.
Both daughter populations of the sockeye salmon are characterized by a depleted composition of the parasite fauna. In Lake Kronotskoye, we noted 17 species of parasites in the sockeye salmon (Busarova et al., 2016), which, together with sockeye salmon spawners, should have entered the recipient lakes. However, by 2015, not all types of parasites were preserved in the daughter populations. Therefore, the sockeye salmon of Lake Sevo (taking into account the incomplete parasitological dissection) housed eight species of parasites, of which only five species are common for the sockeye salmon of Lake Kronotskoye (Crepidostomum farionis, Diplostomum sp., Diphyllobothrium sp., Proteocephalus longicollis, Cystidicola farionis), and three species (Henneguya zschokkei, Echinorhynchus salmonis, Paracanthobdella livanowi) were absent in the parental population, i.e. were purchased from the native Dolly Varden. In the sockeye salmon of the Tolmachevo Reservoir, we noted only two species of parasites; the sockeye salmon has lost over 90% of the diversity of its parasite fauna (Busarova and Esin, 2017). At the same time, both Sevo and the Tolmachevskoye Reservoir are inhabited by invertebrates that could serve as links in parasitic cycles.
Among the consequences of the introduction of the sockeye salmon in Lake Sevo, the hyperinvasion of predatory Dolly Varden by Diphyllobothrium sp. should also be noted. It is known that the high infestation of Diphyllobothrium sp. leads to multiple functional disorders in fish and their death (Pronin et al., 1991; Knudsen and Klemetsen, 1994). The dead Dolly Varden that we found in Lake Sevo was extremely heavily infested with this parasite (200–524 ind./fish). The predatory Dolly Varden accumulates representatives of the genus Diphyllobothrium when feeding on the sockeye salmon; on average, one sockeye salmon in the sample contained 8.1 ind. of Diphyllobothrium sp. Juvenile Dolly Varden, which also serves as food for predators, is minimally infected with this parasite. Larvae of Diphyllobothrium sp. in predators were located not only in the walls of the stomach, but also on the intestines, the walls of the abdominal cavity, and on the liver and in its tissue. Other individuals of the predatory Dolly Varden also had a high invasion of Diphyllobothrium sp. (invasion intensity 19–56 ind./fish).
CONCLUSIONS
According to our data, the introduction of the resident planktivorous sockeye salmon into isolated Lake Sevo, already inhabited by the Dolly Varden, did not lead to a change in the feeding strategies of these species. Dolly Varden continued to use the benthic food web. This native species is not uniform in nutrition: small individuals mainly consume chironomid larvae; the large-sized individuals switch to feeding on gammarids, while minor number of the fish at an older age begin to search for fish prey, hunting their own juveniles and the sockeye salmon. The elimination of the predatory individuals as a result of the accumulation of Diphyllobothrium sp. obtained from the sockeye salmon was noted. The sockeye salmon of Lake Sevo differs from the ancestral population from Lake Kronotskoye by a smaller size. The diversity of its parasitic fauna has decreased in comparison with the ancestral population.
REFERENCES
Amundsen, P.-A., Gabler, H.-M., and Staldvik, F.J., A new approach to graphical analyses of feeding strategy from stomach contents data—modification of the Costello (1990) method, J. Biol., 1996, vol. 48, no. 4, p. 607. https://doi.org/10.1111/1095-8649.1996.tb01455.x
Amundsen, P.-A., Lafferty, K.D., Knudsen, R., et al., New parasites and predators follow the introduction of two fish species to a subarctic lake: implications for food-web structure and functioning, Oecologia, 2013, vol. 171, no. 4, p. 993. https://doi.org/10.1007/s00442-012-2461-2
Aprahamian, M.W., Martin-Smith, K.M., and Mcginnity, P., Restocking of salmonids—opportunities and limitations, Fish. Res., 2003, vol. 62, no. 2, p. 211. https://doi.org/10.1016/S0165-7836(02)00163-7
Bakkel, T.A., Harris, P.D., Jansenl, P.A., and Hansen, L.P., Host specificity and dispersal strategy in Gyrodactylid monogeneans, with particular reference to Gyrodactylus salaris (Platyhelminthes, Monogenea), Dis. Aquat. Org., 1992, vol. 13, p. 63.
Baxter, C.V., Fausch, K D., Murakami, M., and Chapman, P.L., Fish invasion restructures stream and forest food webs by interrupting reciprocal prey subsidies, Ecology, 2004, vol. 85, no. 10, p. 2656. https://doi.org/10.1890/04-138
Black, G.A. and Lankester, M.W., Migration and development of swim-bladder nematodes, Cystidicola spp. (Habronematoidea), in their definitive hosts, Can. J. Zool., 1980, vol. 58, no. 11, p. 1997. https://doi.org/10.1139/z80-275
Bogdan, V.V., Sidorov, V.S., and Zekina, L.M., Role of fish lipids in adaptation to the different ecological conditions, in Ekologicheskie problemy obtogeneza ryb: fiziologo-biokhimicheskie aspekty (Ecological Problems of Fish Ontogenesis: Physiological and Biochemical Aspects), Moscow: Mosk. Gos. Univ., 2001, p. 188.
Boys, C.A., Rowland, S.J., Gabor, M., et al., Emergence of epizootic ulcerative syndrome in native fish of the Murray-Darling River System, Australia: hosts, distribution and possible vectors, PLoS One, 2012, vol. 7, no. 4, p. e35568. https://doi.org/10.1371/journal.pone.0035568
Bugaev, V.F. and Kirichenko, V.E., Nagul’no-nerestovye ozera aziatskoi nerki (vklyuchaya nekotorye drugie vodoemy areala) (Fattening and Spawning Lakes of Asian Sockeye Salmon: Including Some Other Reservoirs of the Range), Petropavlovsk-Kamchatskiy: Kamchatpress, 2008.
Buoro, M., Olden, J.D., and Cucherousset, J., Global Salmonidae introductions reveal stronger ecological effects of changing intraspecific compared to interspecific diversity, Ecol. Lett., 2016, vol. 19, no. 11, p. 1363. https://doi.org/10.1111/ele.12673
Busarova, O.Yu. and Esin, E.V., Parasites and diet of the kokanee salmon Oncorhynchus nerka Walb. from Tolmachevskoe Reservoir (Kamchatka), Issled. Vodn. Biol. Resur. Kamchat. Sev.-Zap. Chasti Tikhogo Okeana, 2017, no. 44, p. 39. https://doi.org/10.15853/2072-8212.2017.44.39-43
Busarova, O.Yu., Butorina, T.E., Markevich, G.N., and Anisimova, L.A., Parasite fauna of the kokanee salmon Oncorhynchus nerka of Kronotskoye Lake (Kamchatka), Parazitologiya, 2016, vol. 50, no. 3, p. 212.
Bykhovskaya-Pavlovskaya, I.E., Parazity ryb. Rukovodstvo po izucheniyu (Fish Parasites: Manual), Leningrad: Nauka, 1985.
Conover, W.J., Practical Nonparameteric Statistics, 3rd ed., Chichester: Wiley, 1999, pp. 396–406.
Crawford, S.S. and Muir, A.M., Global introductions of salmon and trout in the genus Oncorhynchus: 1870–2007, Rev. Fish Biol. Fish., 2008, vol. 18, no. 3, p. 313. https://doi.org/10.1007/s11160-007-9079-1
Crawford, W.W., Colorado trematodes studies. I. A further contribution to the life history of Crepidostomum farionis (Muller), J. Parasitol., 1943, vol. 29, no. 6, p. 379.
Delyamure, S.L., Skryubin, A.S., and Sedyukov, A.M., Osnovy tsestodologii. Tom 11. Difillobotriidy–lentochnye gel’minty cheloveka, mlekopitayushchikh i ryb (Basic Cestodology, Vol. 11: Diphyllobothrium Species are the Human, Mammal, and Bird Tapeworms), Moscow: Nauka, 1985.
Eby, L.A., Roach, W.J., Crowder, L.B., and Stanford, J.A., Effects of stocking-up freshwater food webs, Trends Ecol. Evol., 2006, vol. 21, no. 10, p. 576. https://doi.org/10.1016/j.tree.2006.06.016
Eloranta, A.P., Nieminen, P., and Kahilainen, K., Trophic interactions between introduced lake trout (Salvelinus namaycush) and native Arctic charr (S. alpinus) in a large Fennoscandian subarctic lake, Ecol. Freshwater Fish., 2015, vol. 24, no. 2, p. 181.https://doi.org/10.1111/eff.12132
Esin, E.V. and Markevich, G.N., Gol’tsy roda Salvelinus aziatskoi chasti Severnoi Patsifiki: proiskhozhdenie, evolyutsiya i sovremennoe raznoobrazie (Chars of Genus Salvelinus in the Asian Part of the North Pacific: Origin, Evolution, and Modern Diversity), Petropavlovsk-Kamchatskiy: Kamchatpress, 2017.
Esin, E.V. and Markevich, G.N., Parallel late ontogeny transformations in contrasting landlocked phenotypes of Salvelinus malma (Salmonidae) from small volcanic lakes, Ecol. Freshwater Fish., 2019, vol. 28, no. 4, p. 624. https://doi.org/10.1111/eff.12480
Folch, J., Lees, M., and Sloan-Stanley, G.H., A simple method for the isolation and purification of total lipides from animal tissues, J. Biol. Chem., 1957, vol. 226, no. 1, p. 497.
Gherardi, F., Biological invasions in inland waters: an overview, in Biological Invaders in Inland Waters: Profiles, Distribution, and Threats, Dordrecht: Springer-Verlag, 2007, vol. 2, p. 3. https://doi.org/10.1007/978-1-4020-6029-8_1
Ginetz, R.M.J., On the Risk of Colonization by Atlantic Salmon in BC Waters, New Westminster, BC: BC Salmon Farm. Assoc., 2002.
Horn, H.S., Measurement of “overlap” in comparative ecological studies, Am. Nat., 1966, vol. 100, no. 914, p. 419. https://doi.org/10.1086/282436
Houlihan, D.F., Mathers, E.M., and Foster, A., Biochemical correlates of growth rate in fish, in Fish Ecophysiology, London: Chapman and Hall, 1993, p. 45.
Johnsen, B.O. and Jensen, A.J., Introduction and establishment of Gyrodactylus salaris Malmberg, 1957, on Atlantic salmon, Salmo salar L., fry and parr in the River Vefsna, northern Norway, J. Fish Dis., 2006, vol. 11, no. 1, p. 35. https://doi.org/10.1111/j.1365-2761.1988.tb00521.x
Jorgensen, E.H. and Johnsen, H.K., Rhythmic life of the Arctic charr: adaptations to life at the age, Mar. Genomics, 2014, vol. 14, p. 71. https://doi.org/10.1016/j.margen.2013.10.005
Knicht, J.A., Anderson, S., and Rawle, J.M., Chemical basis of the sulfo-phospho-vanillin reaction for estimating total serum lipids, Clin. Chem., 1972, vol. 18, no. 3, p. 199.
Knudsen, R. and Klemetsen, A., Infections of Diphyllobothrium dendriticum, D. ditremum (Cestoda), and Cystidicola farionis (Nematoda) in a north Norwegian population of Arctic charr (Salvelinus alpinus) during winter, Can. J. Zool., 1994, vol. 72, no. 11, p. 1922. https://doi.org/10.1139/z94-261
Knudsen, R., Curtis, M.A., and Kristoffersen, R., Aggregation of helminths: the role of feeding behavior of fish hosts, J. Parasitol., 2004, vol. 90, no. 1, p. 1. https://doi.org/10.1645/GE-3184
Kurenkov, S.I., Two reproductively isolated groups of land-locked sockeye salmon Oncorhynchus nerka kennerlyi (Suckley) of Lake Kronotskoye, Vopr. Ikhtiol., 1977, vol. 17, no. 4, p. 597.
Lymbery, A.J., Morine, M., Kanania, H.G., et al. Co-invaders: the effects of alien parasites on native hosts, Int. J. Parasitol., 2014, vol. 3, no. 2. https://doi.org/10.1016/j.ijppaw.2014.04.002
MacNeil, C., Dick, J.T., and Elwood, R.W., The trophic ecology of freshwater Gammarus spp. (Crustacea: Amphipoda): problems and perspectives concerning the functional feeding group concept, Biol. Rev., 1997, vol. 72, no. 3, p. 349. https://doi.org/10.1111/j.1469-185X.1997.tb00017.x
Markevich, G.N., Naturalization of living forms of the kokanee salmon Oncorhynchus nerka Walb. into in Kamchatka reservoirs with fishes, Extended Abstract of Cand. Sci. (Biol.) Dissertation, Moscow, 2008.
Markevich, G.N. and Esin, E.V., Trout and char of Russia, in Trout and Char of the World, Bethesda, MD: Am. Fish. Soc., 2019, p. 603.
Markevich, G.N., Esin, E.V., Leman, V.N., et al., Possible fishery in the Kronotskoye Lake (Kamchatka Peninsula), Vopr. Rybolov., 2019, vol. 20, no. 1, p. 5.
Measures, L. and Bosse, L., Gammarus lawrencianus (Amphipoda) as intermediate host of Echinorhynchus salmonis (Acanthocephala) in an estuarine environment, Can. J. Fish. Aquat. Sci., 1993, vol. 50, no. 10, p. 2182. https://doi.org/10.1139/f93-244
Mikhailova, E.I., Worms of genus Neoechinorhynchus (Acanthocephales: Neoechinorhynchidae) from the northeastern Asia: taxonomy, zoogeography, and ecology, Extended Abstract of Cand. Sci. (Biol.) Dissertation, St. Petersburg, 2015.
Minagawa, M. and Wada, E., Stepwise enrichment of 15N along food chains: further evidence and the relation between δ15N and animal age, Geochim. Cosmochim. Acta, 1984, vol. 48, no. 5, p. 1135. https://doi.org/10.1016/0016-7037(84)90204-7
Murza, I.G. and Khristoforov, O.L., Opredelenie stepeni zrelosti gonad i prognozirovanie vozrasta dostizheniya polovoi zrelosti u atlanticheskogo lososya i kumzhi. Metodicheskie ukazaniya (Determination of Maturity Degree of Gonads and Forecasting of Maturity Age of Atlantic Salmon and Brown Trout: Practical Guide), Leningrad: Gos. Nauchno-Issled. Inst. Ozern. Rechn. Rybn. Khoz., 1991.
Nasegava, K., Invasions of rainbow trout and brown trout in Japan: a comparison of invasiveness and impact on native species, Ecol. Freshwater Fish, 2020, vol. 29, no. 3, p. 419. https://doi.org/10.1111/eff.12534
Okamura, B., Gruhl, A., and Bartholomew, J.L., Myxozoan Evolution, Ecology, and Development, Cham: Springer-Verlag, 2015. https://doi.org/10.1007/978-3-319-14753-6
Olden, J.D., Poff, N.L., Douglas, M.R., et al., Ecological and evolutionary consequences of biotic homogenization, Trends Ecol. Evol., 2004, vol. 19, no. 1, p. 18. https://doi.org/10.1016/j.tree.2003.09.010
Opredelitel’ parazitov presnovodnykh ryb fauny SSSR. Tom 1. Paraziticheskie prosteishie (Guide for Identification of Freshwater Fishes of Fauna of the Soviet Union, Vol. 1: Parasitic Protozoa), Leningrad: Nauka, 1984.
Opredelitel’ parazitov presnovodnykh ryb fauny SSSR. Tom 3. Chast’ 2. Paraziticheskie mnogokletochnye (Guide for Identification of Freshwater Fishes of Fauna of the Soviet Union, Vol. 3, Part 2: Parasitic Metazoa), Leningrad: Nauka, 1987.
Peterson, B.J. and Fry, B., Stable isotopes in ecosystem studies, Annu. Rev. Ecol., Evol. Syst., 1987, vol. 18, no. 1, p. 293. https://doi.org/10.1146/annurev.ecolsys.18.1.293
Plokhinskii, N.A., Biometriya (Biometry), Moscow: Vysshaya Shkola, 1970.
Pogodaev, E.G. and Kurenkov, S.I., Naturalization of the land-locked sockeye salmon Oncorhynchus nerka kennerlyi (Suckley) into the lakes of Kamchatka, Vopr. Rybolov., 2007, vol. 8, no. 3, p. 394.
Post, D.M., Layman, C.A., Arrington, D.A., et al., Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses, Oecologia, 2007, vol. 152, no. 1, p. 179. https://doi.org/10.1007/s00442-006-0630-x
Pronin, N.M., Zhaltsanova, D.-S.D., Pronina, S.V., et al., Dinamika zarazhennosti zhivotnykh gel’mintami (Dynamics of Infestation of Animals with Helminthes), Ulan-Ude: Buryat. Nauchn. Tsentr, Sib. Otd., Akad. Nauk SSSR, 1991, p. 3.
Reist, J.D., Pover, M., and Dempson, B., Arctic charr (Salvelinus alpinus): a case study of the importance of understanding biodiversity and taxonomic issues in northern fishes, Biodiversity, 2013, vol. 14, no. 1, p. 45. https://doi.org/10.1080/14888386.2012.725338
Stolbunov, I.A., Gusakov, V.A., Dien Tran Duc, and Thanh Nguyen Thi Hai, Food spectrum, trophic and length-weight characteristics of non-indigenous suckermouth armored catfishes Pterygoplichthys spp. (Loricariidae) in Vietnam, Inland Water Biol., 2021, vol. 14, no. 5, pp. 597–605. https://doi.org/10.1134/S1995082921050163
Sudarikov, V.E., Shigin, A.A., Kurochkin, Yu.V., et al., Metatserkarii trematod—parazity gidrobiontov Tsentral’noi Rossii (Metacercaria of Trematodes are the Parasites of Hydrobionts of Central Russia), Moscow: Nauka, 2002, vol. 1.
Tveiten, H., Johnsen, H.K., and Jobling, M., Influence of the maturity status on the annual cycles of feeding and growth in Arctic charr reared at contrast temperature, J. Fish Biol., 1996, vol. 48, no. 5, p. 910. https://doi.org/10.1111/j.1095-8649.1996.tb01486.x
Vitousek, P.M., Mooney, H.A., Lubchenco, J., and Melillo, J.M., Human domination of Earth’s ecosystems, Science, 1997, vol. 277, no. 5325, p. 494. https://doi.org/10.1007/978-0-387-73412-5_1
Vitule, J.R.S., Freire, C.A., and Simberloff, D., Introduction of non-native freshwater fish can certainly be bad, Fish Fish., 2009, vol. 10, no. 1, p. 98. https://doi.org/10.1111/j.1467-2979.2008.00312.x
Wada, E., Stable δ15N and δ13C isotope ratios in aquatic ecosystems, Proc. Jpn. Acad., Ser. B, 2009, vol. 85, no. 3, p. 98. https://doi.org/10.2183/pjab.85.98
Willemse, J.J., The genus Proteocephalus in the Netherlands, J. Helminthol., 1969, vol. 43, no. 1, p. 207. https://doi.org/10.1017/S0022149X00004041
ACKNOWLEDGMENTS
We thank M.N. Kozhin (Moscow State University, Moscow) for help in carrying out expeditionary work and to the Dr. Sci. (Biol.) A.V. Tiunov (Severtsov Institute of Ecology and Evolution, Russian Academy of Sciences, Moscow) for providing access to the Instrumental Methods in Ecology Shared Use Center.
Funding
This work was partly supported the Russian Foundation for Basic Research, project no. 16-04-01687.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests. The authors declare that they have no conflicts of interest.
Statement on the welfare of animals. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Translated by T. Kuznetsova
Abbreviations: MS, maturity stage; FL, Smith’s body length (Fork length); Q, body weight.
Rights and permissions
Open Access. This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Busarova, O.Y., Esin, E.V. & Markevich, G.N. Trophic Relations between Native Salvelinus malma Walb. and Introduced Oncorhynchus nerka Walb. in the Landlocked Lake Sevo, Kamchatka. Inland Water Biol 15, 160–169 (2022). https://doi.org/10.1134/S1995082922020031
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995082922020031