Abstract
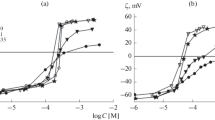
Electrostatic effects in lysine and polylysine adsorption at lipid membrane surface were studied. Electrokinetic measurements demonstrated that lysine induces similar dose-dependent changes of zeta-potential in suspensions of liposomes made from cardiolipin (CL) and phosphatidylserine (PS). These changes correlate well with numerical description of diffuse part of electric double layer by Gouy-Chapman-Stern model in assumption that both potassium cation and lysine molecules determine the ionic strength of the media. Good agreement with the electrokinetic data was found with the isotherm constructed for lysine distribution between bilayer and water with low constant (K d = 1.2 × 10−3 M−1) independently of potassium adsorption (K = 1 M−1) on lipid molecules or for their competitive adsorption with the same constants. Lysine adsorption induces total boundary potential changes of planar bilayer lipid membranes (BLM) from the same lipids registered by the method of intramembranous field compensation. In contrast to surface potential of liposomes in electrokinetic experiments the total boundary potential of BLM remains unchanged up to concentration of lysine that is about 1.5 orders of magnitude higher. This fact corresponds to changes in opposite directions of surface and dipole components of boundary potential. They compensate each other to some extent when lysine adsorbs at the surface. This explanation was supported by molecular dynamic simulation of bilayers from DOPS in the presence of lysine. According to the MD-simulations, the compensation effect can be attributed to lysine effect on hydrogen bonds of water molecules with phosphate groups of lipids but not with carboxylic groups. A similar “compensation effect” was expected and observed with membranes from CL and PS. The amplitude of dipole effect was about 40 mV due to the lysine-lipid interactions and corresponded well to the amplitude of the slow phase in the boundary potential changes induced by polylysine adsorption on planar BLM. This phase can be attributed to polypeptide conformational changes and/or lipid bilayer restructuring phenomena.
Similar content being viewed by others
References
Finogenova O., Filinsky D., Ermakov Yu. 2008. Electrostatic effects upon adsorption and desorption of polylysines on the surface of lipid membranes of different composition. Membr. Cell Biol. 25, 217–226.
Schwieger C., Blume A. 2007. Interaction of poly(L-lysines) with negatively charged membranes: An FT-IR and DSC study. Eur. Biophys. J. 36, 437–450.
Yaroslavov A.A, Efimova A.A., Lobyshev V.I., Ermakov Yu.A., Kabanov V.A. 1997. Reversibility of structural rearrangements in lipid membranes induced by adsorption desorption of a polycation. Membr. Cell Biol. 10, 683–688.
Pashkovskaya A.A., Lukashev E.P., Antonov P.E., Finogenova O.A., Ermakov Y.A., Melik-Nubarov N.S., Antonenko Y.N. 2006. Grafting of polylysine with polyethylenoxide prevents demixing of O-pyromellitylgramicidin in lipid membranes. Biochim. Biophys. Acta. 1758, 1685–1695.
de Kruijff B., Rietveld A., Telders N., Vaandrager B. 1985. Molecular aspects of the bilayer stabilization induced by poly(L-lysines) of varying size in cardiolipin liposomes. Biochim. Biophys. Acta. 820, 295–304.
Diederich A., Baehr G., Winterhalter M. 1998. Influence of polylysine on the rapture of negatively charged membranes. Langmuir. 14, 4597–4605.
Reuter M., Schwieger C., Meister A., Karlsson G., Blume A. 2009. Poly-L-lysines and poly-L-arginines induce leakage of negatively charged phospholipid vesicles and translocate through the lipid bilayer upon electrostatic binding to the membrane. Biophys. Chem. 144, 27–37.
Ermakov Y.A., Averbakh A.Z., Yusipovich A.I., Sukharev S. 2001. Dipole potentials indicate restructuring of the membrane interface induced by gadolinium and beryllium ions. Biophys. J. 80, 1851–1862.
Marukovich N., McMurray M., Finogenova O., Nesterenko A., Batishchev O., Ermakov Yu. 2013. Interaction of polylysines with the surface of lipid membranes: The electrostatic and structural aspects. In: Advances in planar lipid bilayers and liposomes. A tribute to Marin D. Mitov. Eds A. Iglic and J. Genov. Amsterdam, London, San Diego: Elsevier, Academic press, p. 139–166.
Mueller P., Rudin D.O., Tien H.T., Wescott W.C. 1963. Methods for the formation of single bimolecular lipid membranes in aqueous solution. J. Phys. Chem. 67, 534–535.
Ermakov Yu.A., Sokolov V.S. 2003. Boundary potentials of bilayer lipid membranes: Methods and interpretations. In: Planar lipid bilayers (BLM) and their applications. Eds Tien H.T., Ottova A. Amsterdam: Elsevier, Academic press, p. 109–141.
Jorgensen W. 1996. Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236.
Hess B., Kutzner C., Van Der Spoel D., Lindahl E. 2008. GROMACS 4: Algorithms for highly efficient, load-balanced, and scalable molecular simulation. J. Chem. Theory Comput. 4, 435–447.
Nesterenko A.M., Ermakov Yu.A. 2012. Molecular dynamic simulation of phospholipid bilayers: Ion distribution at the surface of neutral and charged bilayer in the liquid crystalline state. Membr. Cell Biol. 6, 320–328.
Nesterenko A.M., Krasilnikov P.M., Ermakov Yu.A., Molecular-dynamic simulation of DPPC bilayer in different phase state: Hydration and electric field distribution in the presence of Be2+ cations. Biochem. (Moscow) Suppl. Ser. A Membr. Cell Biol. 5, 370–378.
Ermakov Yu.A. 2000. Ion equilibrium near lipid membranes: empirical analysis of the simplest model. Colloid J. 62, 389–400.
Grahame D.C. 1947. The electrical double layer and the theory of electrocapillarity. Chem. Rev. 41, 441–501.
McLaughlin S. 1989. The electrostatic properties of membranes. Annu. Rev. Biophys. Biophys. Chem. 18, 113–136.
Ermakov Yu. A., Cherniy V.V., Sokolov V.S. 1992. Beryllium adsorption on neutral and charged lipid membranes. Membr. Cell Biol. 9, 201–213.
Diaz S., Amalfa F., Lopez A.C.B., Disalvo E.A. 1999. Effect of water polarized at the carbonyl groups of phosphatidylcholines on the dipole potential of lipid bilayers. Langmuir. 15, 5179–5182.
Blume A., Hubner W., Messner G. 1988. Fourier transform infrared spectroscopy of 13C=O-labeled phospholipids hydrogen bonding to carbonyl groups. Biochemistry. 27, 8239–8249.
Diaz S., Lairion F., Arroyo J., de Lopez A.C.B., Disalvo E.A. 2001. Contribution of phosphate groups to the dipole potential of dimyristoylphosphatidylcholine membranes. Langmuir. 17, 852–855.
Ermakov Yu.A. 1990. The interaction of polyelectrolytes with bilayer lipid membranes. In: Elektrokhimiya polimerov (Electrochemistry of polymers). Ed. Tarasevich M.R., Chruscheva E.I. M: Nauka, p. 93–101.
Ermakov Yu. A., Fevraleva I.S., Ataullahanov R.I. 1985. The influence of polycations on the boundary potential of BLM. Membr. Cell Biol. 2, 1094–1100.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.I. Marukovich, A.M. Nesterenko, Yu.A. Ermakov, 2014, published in Biologicheskie Membrany, 2014, Vol. 31, No. 6, pp. 401–409.
Rights and permissions
About this article
Cite this article
Marukovich, N.I., Nesterenko, A.M. & Ermakov, Y.A. Structural factors of lysine and polylysine interaction with lipid membranes. Biochem. Moscow Suppl. Ser. A 9, 40–47 (2015). https://doi.org/10.1134/S1990747814060038
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990747814060038