Abstract
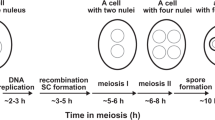
Intranuclear distribution of actin was studied in mouse embryos in the middle and end of the four-cell stage of development, as well as in the blastomeres of uncompacted eight-cell embryos. Actin was visualized with direct fluorescence, indirect immunofluorescence and confocal microscopy. It was found that nuclei at these stages of development contained monomeric, as well as polymeric actin, differing from conventional filamentous. The pattern of actin distribution in the nuclei at the middle-stage four-cell embryos and uncompacted eight-cell embryos was similar. Comparison of this pattern with nuclear actin distribution in the two-cell embryos that we examined previously showed that at the early stages of embryo cleavage the intranuclear actin localization was not stage-specific and was stably reproduced in the nuclei of the embryos during transition from earlier to later stage of embryogenesis. Filamentous actin that had not been found in the nuclei of middle-stage four-cell embryos was detected by fluorescent phalloidin in the nuclei of the embryos at the end of a four-cell stage of development, shortly before the beginning of the third cleavage division. The appearance of nuclear filamentous actin is possibly related to the preparation of embryos to the following cleavage division.

Similar content being viewed by others
REFERENCES
Baarlink, C., Plessner, M., Sherrard, A., Morita, K., Misu, S., Virant, D., Kleinschnitz, E.-M., Harniman, R., Alibhai, D., Baumeister, S., Miyamoto, K., Endesfel-der, E., Kaidi, A., and Grosse, R., A Transient pool of nuclear F-actin at mitotic exit controls chromatin organization, Nat. Cell Biol., 2017, vol. 19, pp. 1389–1399.
Belin, B.J., Cimini, B.A., Blackburn, E.H., and Dyche Mullins, R., Visualization of actin filaments and monomers in somatic cell nuclei, Mol. Biol. Cell, 2013, vol. 24, pp. 982–994.
Biachi, E., and Sette, K., Post-transcriptional control of gene expression in mouse early embryo development: a view from the tip of the iseberg, Genes, 2011, vol. 2, pp. 345–359.
Bogolyubov, D.S., Batalova, F.M., Kiselyov, A.M., and Stepanova, I.S., Nuclear structures in Tribolium castaneum oocytes, Cell Biol. Int., 2013, vol. 37, pp. 1061–1079.
Bogolyubova, N.A. and Bogolyubova, I.O., Actin localization in nuclei of two-cell mouse embryos, Cell Tissue Biol., 2009, vol. 3, no. 5, pp. 417–422.
Bogolyubova, I.O. and Parfenov, V.N., Immunofluorescent detection of nuclear actin in early mouse embryos, Cell Tissue Biol., 2012, vol. 6, nos. 5–6, pp. 458–464.
Bogolyubova, I., Stein, G., and Bogolyubov, D., FRET analysis of interactions between actin and exon-exon junction complex proteins in early mouse embryos, Cell Tiss. Res., 2013, vol. 352, pp. 277–285.
Bohnsack, M.T., Stuven, T., Kuhn, C., Cordes, V.C., and Gorlich, D., A selective block of nuclear actin export stabilizes the giant nuclei of Xenopus oocytes, Nat. Cell Biol., 2006, vol. 8, pp. 257–267.
Castano, E., Philimonenko, V.V., Kahle, M., Fukalová, J., Kalendová, A., Yildirim, S., Dzijak, R., Dingová-Krásna, H., and Hozák, P., Actin complexes in the cell nucleus: new stones in an old field, Histochem. Cell Biol., 2010, vol. 133, pp. 607–626.
Fujimori, T., Preimplantation development of mouse: a view from cellular behavior, Dev. Growth Differ., 2010, vol. 52, pp. 253–262.
Funaki, K., Katsumoto, T., and Iino, A., Immunocytochemical localization of actin in the nucleolus of rat oocytes, Biol. Cell, 1995, vol. 84, pp. 139–146.
Gard, D.L., Confocal microscopy and 3-D reconstruction of the cytoskeleton of Xenopus oocytes, Microsc. Res. Tech., 1999, vol. 44, pp. 388–414.
Gounon, P. and Karsenti, E., Involvement of contractile proteins in the changes in consistency of oocytes nucleoplasm of the newt Pleurodeles waltlii, J. Cell Biol., 1981, vol. 88, pp. 410–421.
Hamatani, T., Carter, M.G., Sharov, A.A., and Ko, M.S.H., Dynamics of global gene expression changes during mouse preimplantation development, Dev. Cell, 2004, vol. 6, pp. 117–131.
Hendzel, M.J., The F-act’s of nuclear actin, Curr. Opin. Cell Biol., 2014, vol. 28, pp. 84–89.
Maître, J.-L., Mechanics of blastocyst morphogenesis, Biol. Cell, 2017, vol. 109, pp. 323–338.
Maslova, A. and Krasikova, A., Nuclear actin depolymerization in transcriptionally active avian and amphibian oocytes leads to collapse of intranuclear structures, Nucleus, 2012, vol. 3, pp. 300–311.
Misu, S., Takebayashi, M., and Miyamoto, K., Nuclear actin in development and transcriptional reprogramming, Front. Genet., 2017, vol. 8, p. 27. https://doi.org/10.3389/fgene.2017.00027
Nguyen, E., Besombes, D., and Debey, P., Immunofluorescent localization of actin in relation to transcription sites in mouse pronuclei, Mol. Reprod. Dev., 1998, vol. 50, pp. 263–272.
Pochukalina, G.N. and Parfenov, V.N., Actin and mRNA export factors distribution in the nucleus of preovulatory mouse oocytes, Cell Tissue Biol., 2012, vol. 6, nos. 5–6, pp. 423–434.
Rawe, V.Y., Payne, C., and Schatten, G., Profilin and actin-related proteins regulate microfilament dynamics during early mammalian embryogenesis, Hum. Reprod., 2006, vol. 21, pp. 1143–1153.
Rübsam, R. and Büning, J., F-actin is a component of the karyosome in neuropteran oocyte nuclei, Arthropod Struct. Dev., 2001, vol. 30, pp. 125–133.
Uchiyama, S., Kobayashi, S., Takata, H., Ishihara, T., Naoto, N., Higashi, T., Hayashihara, K., Sone, T., Higo, D., Nirasawa, T., Takao, T., Matsunaga, S., and Fukui, K., Proteome analysis of human metaphase chromosomes, J. Biol. Chem., 2005, vol. 280, pp. 16 994–17 004.
Viita, T. and Vartiainen, M., From cytoskeleton to gene expression: actin in the nucleus, Handb. Exp. Pharmacol., 2017, vol. 235, pp. 311–329.
Wesolowska, N. and Lenart, P., Nuclear roles for actin, Cromosoma, 2015, vol. 124, pp. 481–489.
Zheng, B., Han, M., Bernier, M., and Wen, J.-K., Nuclear actin and actin-binding proteins in the regulation of transcription and gene expression, FEBS J., 2009, vol. 276, pp. 2669–2685.
ACKNOWLEDGMENTS
This study was supported by state order 0124_2018_003, IITIC АААА-А17-117032350035-4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Translated by I. Fridlyanskaya
Abbreviations: AbC—antibodies to actin C-terminus, AbN—antibodies to actin N-terminus.
Rights and permissions
About this article
Cite this article
Bogolyubova, N.A. Intranuclear Actin Distribution at the Four- and Eight-Cell Stages of Mouse Embryo Development. Cell Tiss. Biol. 13, 42–46 (2019). https://doi.org/10.1134/S1990519X19010036
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X19010036