Abstract
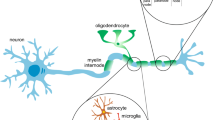
Models of radiation-induced neurogenesis disorders are based on a consideration of the age-related dynamics of the changes in the number of progenitor cells during life. However, for a full-fledged analysis of radiation injuries, it is also necessary to know the age-related dynamics of mature cell types—neurons, astrocytes, and oligodendrocytes formed from the corresponding progenitor cells. To account for the population of both progenitor cells and mature cell types, we have developed a mathematical model of the radiation-induced impairment of adult neurogenesis based on a model of asymmetric division of neural stem cells in the dentate gyrus of the hippocampus of C57BL mice. This model reproduces experimental data on age-related changes in the number of neural stem cells; amplifying neuronal progenitors; neuroblasts; immature neurons; and, for the first time, mature neurons and astrocytes. The dynamics of changes in the number of oligodendrocytes with age is predicted. The proportions of surviving mature neurons, astrocytes, and oligodendrocytes after exposure to X-ray radiation have been calculated.









Similar content being viewed by others
REFERENCES
M. L. Monje and T. Palmer, “Radiation injury and neurogenesis,” Curr. Opin. Neurol. 16, 129 (2003).
M. A. Smith, L. G. Ries, J. G. Gurney, M. L. Bondy, S. E. Plon, D. Malkin, A. T. Look, I. R. Kirsch, C. J. Thiele, and M. B. Kastan, Principles and Practice of Pediatric Oncology, Ed. by P. A. Pizzo and D. G. Poplack (Lippincott Williams and Wilkins, Philadelphia, 2002).
J. Radcliffe, R. J. Packer, T. E. Atkins, G. R. Bunin, L. Schut, J. W. Goldwein, and L. N. Sutton, “Three-and four-year cognitive outcome in children with noncortical brain tumors treated with whole-brain radiotherapy,” Ann. Neurol. 32, 551 (1992).
H. Lackner, M. Benesch, S. Schagerl, R. Kerbl, W. Schwinger, and C. Urban, “Prospective evaluation of late effects after childhood cancer therapy with a follow-up over 9 years,” Eur. J. Pediatr. 159, 750 (2000).
J. R. Crossen, D. Garwood, E. Glatstein, and E. A. Neuwelt, “Neurobehavioral sequelae of cranial irradiation in adults: A review of radiation-induced encephalopathy,” J. Clin. Oncol. 12, 627 (1994).
M. L. Monje, S. Mizumatsu, J. R. Fike, and T. D. Palmer, “Irradiation induces neural precursor-cell dysfunction,” Nat. Med. 8, 955 (2002).
S. Mizumatsu, M. L. Monje, D. R. Morhardt, R. Rola, T. D. Palmer, and J. R. Fike, “Extreme sensitivity of adult neurogenesis to low doses of X-irradiation,” Cancer Res. 63, 4021 (2003).
M. A. Bonaguidi, M. A. Wheeler, J. S. Shapiro, R. P. Stadel, G. J. Sun, G. l. Ming, and H. Song, “In vivo clonal analysis reveals self-renewing and multipotent adult neural stem cell characteristics,” Cell 145, 1142 (2011).
J. M. Encinas, T. V. Michurina, N. Peunova, J. H. Park, J. Tordo, D. A. Peterson, G. Fishell, A. Koulakov, and G. Enikolopov, “Division-coupled astrocytic differentiation and age-related depletion of neural stem cells in the adult hippocampus,” Cell Stem Cell. 8, 566 (2011).
E. Cacao and F. A. Cucinotta, “Modeling impaired hippocampal neurogenesis after radiation exposure,” Radiat. Res. 185, 319 (2016).
E. Cacao, S. Kapukotuwa, and F. A. Cucinotta, “Modeling reveals the dependence of hippocampal neurogenesis radiosensitivity on age and strain of rats,” Front. Neurosci. 12, 980 (2018).
F. Ziebell, A. Martin-Villalba, and A. Marciniak-Czochra, “Mathematical modelling of adult hippocampal neurogenesis: Effects of altered stem cell dynamics on cell counts and bromodeoxyuridine-labelled cells,” J. R. Soc. Interface 11, 20140144 (2014).
F. Ziebell, S. Dehler, A. Martin-Villalba, and A. Marciniak-Czochra, “Revealing age-related changes of adult hippocampal neurogenesis using mathematical models,” Development 145, dev153544 (2018).
J. M. Long, A. N. Kalehua, N. J. Muth, M. E. Calhoun, M. Jucker, J. M. Hengemihle, D. K. Ingram, and P. R. Mouton, “Stereological analysis of astrocyte and microglia in aging mouse hippocampus,” Neurobiol. Aging 19, 497 (1998).
D. Keller, C. Erö, and H. Markram, “Cell densities in the mouse brain: A systematic review,” Front. Neuroanat. 12, 83 (2018).
B. Steiner, G. Kronenberg, S. Jessberger, M. D. Brandt, K. Reuter, and G. Kempermann, “Differential regulation of gliogenesis in the context of adult hippocampal neurogenesis in mice,” Glia 46, 41 (2004).
R. Rola, J. Raber, A. Rizk, S. Otsuka, S. R. van den Berg, D. R. Morhardt, and J. R. Fike, “Radiation-induced impairment of hippocampal neurogenesis is associated with cognitive deficits in young mice,” Exp. Neurol. 188, 316 (2004).
J. D. Rieskamp, P. Sarchet, B. M. Smith, and E. D. Kirby, “Stereological characterization of the major cell lineages in the mouse dorsal dentate gyrus,” bioRxiv (2019). https://doi.org/10.1101/847350
M. B. Nada, L. Slomianka, A. L. Vyssotski, and H. P. Lipp, “Early age-related changes in adult hippocampal neurogenesis in C57 mice,” Neurobiol. Aging. 31, 151 (2010).
A. A. Glebov, E. A. Kolesnikova, and A. N. Bugai, “Mathematical modeling of adult neurogenesis, taking into account the progenitors of oligodendrocytes,” in Medical and Radiation Biophysics: Proceedings of the 28th International Conference on Mathematics. Computer. Education, Jan. 25–30, 2021, Pushchino, 2021, p. 126.
A. A. Glebov, E. A. Kolesnikova, and A. N. Bugai, “Mathematical model of adult neurogenesis, taking into account the population of mature neurons,” in Mathematics and Mathematical Modeling: Proceedings of the 15th All-Russian Youth Scientific and Innovation School, April 13–15, 2021 (Sarov, 2021), pp. 123–124.
A. A. Glebov, E. A. Kolesnikova, and A. N. Bugai, “Modeling of neurogenesis impairment after acute X‑ray irradiation of mice,” Aktual. Vopr. Biol. Fiz. Khim. 6, 280 (2021).
O. A. Mineyeva, D. V. Bezriadnov, A. V. Kedrov, A. A. Lazutkin, K. V. Anokhin, and G. N. Enikolopov, “Radiation induces distinct changes in defined subpopulations of neural stem and progenitor cells in the adult hippocampus,” Front. Neurosci. 12, 1013 (2019).
M. Andres-Mach, R. Rola, and J. R. Fike, “Radiation effects on neural precursor cells in the dentate gyrus,” Cell Tissue Res. 331, 251 (2008).
M. E. Calhoun, D. Kurth, A. L. Phinney, J. M. Long, J. Hengemihle, P. R. Mouton, D. K. Ingram, and M. Jucker, “Hippocampal neuron and synaptophysin-positive bouton number in aging C57BL/6 mice,” Neurobiol. Aging 19, 599 (1998).
J. Vinet, P. Lemieux, A. Tamburri, P. Tiesinga, J. Scafidi, V. Gallo, and A. Sík, “Subclasses of oligodendrocytes populate the mouse hippocampus,” Eur. J. Neurosci. 31, 425 (2010).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Smirnova
Rights and permissions
About this article
Cite this article
Glebov, A.A., Kolesnikova, E.A. & Bugai, A.N. Mathematical Model of a Radiation-Induced Neurogenesis Impairment. Phys. Part. Nuclei Lett. 19, 422–433 (2022). https://doi.org/10.1134/S1547477122040124
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1547477122040124