Abstract
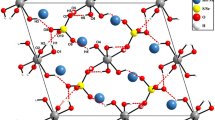
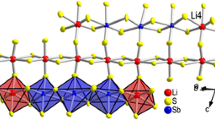
A new compound β-Tl6Si2O7 has been investigated using the methods of IR spectroscopy and microprobe and X-ray diffraction analysis. The elementary unit parameters were as follows: P63/m, a = 9.673(2) Å, c = 3.9169(9) Å, and V = 317.4(1) Å3. The structure was resolved by direct methods and corrected until R 1 = 0.029 (wR 2 = 0.047) for 240 reflections [F o] ≥ 4σ F . The crystal structure of β-Tl6Si2O7 contains one symmetrically independent position of the Tl+ cation that forms three short (〈Tl-O〉 = 2.54 Å) bonds and one weak bond (2.93 Å) with the oxygen atom. One can observe a strong disordering of silicon atoms (Si-Si = 0.64 Å). The distorted tetragonal pyramids TlO4 are linked through silicate tetrahedra into a three-dimensional framework. The interesting feature of the crystal structure of β-Tl6Si2O7 is in the presence of wide channels occupied by lone electron pairs on Tl+ cations. One can also mention that in most of the structures of Tl+ oxygen-containing salts the lone pairs are associated into separate parts in the form of micelles in channels of frameworks or other structural cavities.
Similar content being viewed by others
References
Friberg, S. and Smith, P., Nonlinear Optical Glasses for Ultrafast Optical Switches, IEEE J. Quantum Electron., 1987, vol. 23, pp. 2089–2094.
Hall, D.W., Newhouse, M.A., Borrelli, N.F., Dumbaugh, W.H., and Weidman, D.L., Nonlinear Optical Susceptibilities of High-Index Glasses, Appl. Phys. Lett., 1989, vol. 54, pp. 1293–1295.
Fargin, E., Berthereau, A., and Cardinal, T., Le Flem G., Ducasse L., Canioni L., and Segonds P., Optical Non-Linearity in Oxide Glasses, J. Non-Cryst. Solids, 1996, vol. 203, pp. 96–101.
Jeansannetas, B., Blanchandin, S., Thomas, P., Marchet, P., Champarnaud-Mesjard, J.C., Merle-Mejean, T., Frit, B., Nazabal, V., Fargin, E., Le Flem, G., Martin, M.O., Bousquet, B., Canioni, L., Le Boiteux, S., Segonds, P., and Sarger, L., Glass Structure and Optical Nonlinearities in Thallium(I) Tellurium(IV) Oxide Glasses, J. Solid State Chem., 1999, vol. 146, pp. 329–335.
Kang, I., Krauss, T.D., Wise, F.W., Aitken, B.G., and Borrelli, N.F., Femtosecond Measurement of Enhanced Optical Nonlinearities of Sulfide Glasses and Heavy-Metal-Doped Oxide Glasses, J. Opt. Soc. Am., 1995, vol. B12, pp. 2053–2059.
Touboul, M. and Feutelais, Y., Système Tl2O-GeO2 Etétude Structurale des Germanates de Thallium(I), J. Solid State Chem., 1980, vol. 32, p. 167.
Panek, L.W. and Bray, P.J., NMR of 205Tl in Tl2O-SiO2, Tl2O-GeO2, and Tl2O-B2O3 Glasses, J. Chem. Phys., 1977, vol. 66, pp. 3822–3831.
Touboul, M. and Feutelais, Y., Structure du Germanate de Thallium(I) Tl8Ge5O14, Acta Crystallogr., 1979, vol. B35, pp. 810–815.
Aitken, B.G., Hall, D.W., and Newhouse, M.A., Thallium Germanate, Tellurite, and Antimonite Glasses, US Patent 5283212, 1994.
Edahiro, T., Inagaki, N., and Kurosaki, Sh., Process for Producing Optical Glass Product, US Patent 4528010, 1985.
Kuan-Han Sun, Thallium-Silicate Glass, US Patent 2472448(A), 1949.
Otto, K. and Millberg, M.E., Ionic Conduction and Structure in Cesium and Thallium Silicate Glasses, J. Am. Ceram. Soc., 1967, vol. 50, pp. 513–516.
Loshagin, A.V., NMR Study of Sodium Borosilicate Glasses Containing Thallium Oxide, Glass Phys. Chem., 1997, vol. 23, no. 4, pp. 287–292.
Milberg, M.E. and Peters, C.R., Cation Distribution in Thallium Silicate Glasses, Phys. Chem. Glasses, 1969, vol. 10, pp. 46–49.
Piffard, Y., Marchand, R., and Tournoux, M., Structure et Filiation Structurale des Phases Tl6Si2O7 et Tl3BO3, Rev. Chim. Miner., 1975, vol. 12, pp. 210–217.
Britvin, S.N., Siidra, O.I., Krivovichev, S.V., and Depmeier, W., Synthesis and Crystal Structure of the First Thallium Hydrous Nesosilicate Tl4SiO4 · 0.5H2O, Z. Anorg. Allg. Chem., 2009, vol. 635, pp. 518–522.
Siidra, O.I., Britvin, S.N., and Krivovichev, S.V., Hydroxocentered [(OH)Tl3]2+ Triangle as a Building Unit in Thallium Compounds: Synthesis and Crystal Structure of Tl4(OH)2CO3, Z. Kristallogr., 2009, vol. 224, pp. 563–567.
Siidra, O.I., Britvin, S.N., Krivovichev, S.V., and Depmeier, W., Polytypism of Layered Alkaline Hydroxides: Crystal Structure of TlOH, Z. Anorg. Allg. Chem., 2010, vol. 636, pp. 595–599.
Khimiya i tekhnologiya redkikh i rasseyannykh elementov (Chemistry and Technology of Rare-Earth and Scattered Elements), Bol’shakov, K.A., Ed., Moscow: Vysshaya Shkola, 1976 [in Russian].
Sheldrick, G.M., A Short History of SHELX, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, pp. 112–122.
Dai, Y.-S. and Hughes, J.M., Crystal-Structure Refinements of Vanadinite and Pyromorphite, Can. Mineral., 1989, vol. 27, pp. 189–192.
Dai, Y.-S., Hughes, J.M., and Moore, P.B., The Crystal Structures of Mimetite and Clinomimetite, Pb5(AsO4)3Cl, Can. Mineral., 1991, vol. 29, pp. 369–376.
Krivovichev, S.V., Armbruster, T., and Depmeier, W., One-Dimensional Lone Electron Pair Micelles in the Crystal Structure of Pb5(SiO4)(VO4)2, Mater. Res. Bull., 2004, vol. 39, pp. 1717–1722.
Pyykö, P., Strong Closed-Shell Interactions in Inorganic Chemistry, Chem. Rev., 1997, vol. 97, pp. 597–636.
Jansen, M., Zur Kristallstruktur von Kaliumdisilicat, Z. Kristallogr., 1982, vol. 160, pp. 127–133.
Hoch, C. and Roehr, C., Alkalimetall-Oxosilicate A 6(Si3O9) und A 6(Si2O7) (A = Rb, Cs): Darstellung und Kristallstruktur, Z. Naturforsch., B: Chem. Sci., 2001, vol. 56, pp. 423–430.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.I. Siidra, S.N. Britvin, S.V. Krivovichev, D.A. Klimov, W. Depmeier, 2012, published in Fizika i Khimiya Stekla.
Rights and permissions
About this article
Cite this article
Siidra, O.I., Britvin, S.N., Krivovichev, S.V. et al. Synthesis and crystal structure of the disordered modification of Tl6Si2O7 . Glass Phys Chem 38, 473–477 (2012). https://doi.org/10.1134/S1087659612060053
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659612060053