Abstract
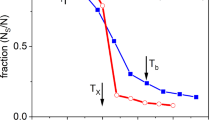
A new version of the STRUCTON-1.2 computer program (2009) has been presented. The program combines the algorithm for calculating real distributions of Q n structons in binary silicate melts (with allowance made for their disproportionation) and the statistical simulation of molecular-mass distributions of polymerized ions at different temperatures. This model has been used to perform test calculations for two melts in the Na2O-SiO2 system (Na6Si2O7, Na6Si3O9). The results of the calculations have made it possible to trace variations in the set and concentrations of chain and ring silicon-oxygen complexes with a decrease in the temperature in the order: stochastic molecular-mass → distribution molecular-mass distribution at T = 2000 K → molecular-mass distribution at the liquidus temperature. The main result of these calculations is that the dominant species of silicon-oxygen anions at the liquidus temperatures (in contrast to the stochastic distributions) exactly correspond to the stoichiometry of the initial melts: the Si2O 6−7 chain anions and (Si n O3n )3n− ring complexes are dominant in the Na6Si2O7 and Na6Si3O9 melts, respectively. It has been established that, with a decrease in the temperature, the average size of polymer complexes varies weakly in the Na6Si2O7 melt but increases by a factor of approximately 1.5 in the metasilicate system.
Similar content being viewed by others
References
Lentz, C.W., Silicate Minerals as Sources of Trimethylsilyl Silicates and Silicate Structure Analysis of Sodium Silicate Solutions, Inorg. Chem., 1964, vol. 3, pp. 574–579.
Kolb, K.E. and Hansen, K.W., Silica Structure in Borate Glasses, J. Am. Ceram. Soc., 1965, vol. 48, p. 439.
Gotz, J., Masson, C.R., and Castelliz, L.M., Crystallization of Lead Silicate Glass as Studied by Trimethylsilylation and Chromatographic Separation of the Anionic Constituents, in Amorphous Materials, Douglas, R.W. and Ellis, B., Eds., New York: Wiley, 1972, pp. 317–325.
Dalby, K.N. and King, P.L., A New Approach to Determine and Quantify Structural Units in Silicate Glasses Using Micro-Reflectance Fourier-Transformed Infrared Spectroscopy, Am. Mineral., 2006, vol. 91, pp. 1783–1793.
Masson, C.R., Ionic Equilibria in Liquid Silicates, J. Am. Ceram. Soc., 1968, vol. 51, pp. 134–143.
Esin, O.A., Polymer Model of Molten Silicates, in Itogi nauki i tekhniki: Rastvory. Rasplavy (Advances in Science and Technology: Solutions and Melts), Moscow: VINITI, 1975, vol. 2, pp. 76–107 [in Russian].
Fraser, D.G., Thermodynamic Properties of Silicate Melts, in Thermodynamics in Geology, Fraser, D.G., Ed., Dordrecht, Holland: D. Reidel, 1977, pp. 301–325.
Hess, P.C., Structure of Silicate Melts, Can. Mineral., 1977, vol. 15, pp. 162–178.
Masson, C.R., Anionic Composition of Glass-Forming Melts, J. Non-Cryst. Solids, 1977, vol. 25, pp. 3–41.
Lacy, E.D., A Statistical Model of Polymerization/Depolymerization Relationships in Silicate Melts and Glasses, Phys. Chem. Glasses, 1965, vol. 6, pp. 171–180.
Falk, M. and Thomas, R.E., Molecular Size Distribution in Random Polyfunctional Condensation with or without Ring Formation: Computer Simulation, Can. J. Chem., 1974, vol. 52, pp. 3285–3295.
Machacek, J., Gedeon, O., and Liska, M., Group Connectivity in Binary Silicate Glasses, J. Non-Cryst. Solids, 2006, vol. 352, pp. 2173–2179.
Sen, S. and Tangeman, J., Evidence for Anomalously Large Degree of Polymerization in Mg2SiO4 Glass and Melt, Am. Mineral., 2008, vol. 93, pp. 946–949.
Pretnar, V.B., Beitrag zur Ionentheorie der Silikatmelzen, Ber. Bunsen Ges. Phys. Chem., 1968, vol. 72, pp. 773–778.
Baes, C.F., Jr., A Polymer Model for BeF2 and SiO2 Melts, J. Solid State Chem., 1970, vol. 1, pp. 159–170.
Masson, C.R., Smith, I.B., and Whiteway, S.G., Activities and Ionic Distributions in Liquid Silicates: Application of Polymer Theory, Can. J. Chem., 1970, vol. 48, pp. 1456–1464.
Esin, O.A., Comparison of Methods for Evaluating the Degree of Polymerization of Silicate Melts, in Fizicheskaya khimiya metallurgicheskikh rasplavov (Physical Chemistry of Metallurgical Melts), Trudy Inst. Metall., 1972, issue 27, part IV, pp. 27–44 [in Russian].
Esin, O.A., On the Anionic Complexes in Molten Slags, in Stroenie i svoistva metallurgicheskikh rasplavov (Structure and Properties of Metallurgical Melts), Trudy Inst. Metall., 1974, issue 28, pp. 76–90 [in Russian].
Polyakov, V.B. and Ariskin, A.A., Simulation of the Composition and Proportions of Anions in Polymerized Silicate Melts, Fiz. Khim. Stekla, 2008, vol. 34, no. 1, pp. 66–80 [Glass Phys. Chem. (Engl. transl.), 2008, vol. 34, no. 1, pp. 50–62].
Ariskin, A.A. and Polyakov, V.B., Simulation of Molecular Mass Distributions and Evaluation of O2- Concentrations in Polymerized Silicate Melts, Geokhimiya, 2008, no. 5, pp. 467–486 [Geochem. Int. (Engl. transl.), 2008, vol. 46, no. 5, pp. 429–447].
Fraser, D.G., Acid-Base Properties and Structons: Towards a Structural Model for Predicting the Thermodynamic Properties of Silicate Melts, Ann. Geophys., 2005, vol. 48, nos. 4–5, pp. 549–559.
Henderson, G.S., The Structure of Silicate Melts: AGlass Perspective, Can. Mineral., 2005, vol. 43, pp. 1921–1958.
Mysen, B.O. and Richet, P., Silicate Glasses and Melts, Properties and Structure, Amsterdam: Elsevier, 2005.
Anfilogov, V.K, Bykov, V.K, and Osipov, A.A., Silikatnye rasplavy (Silicate Melts), Moscow: Nauka, 2005 [in Russian].
Malfait, W.J., Zakaznova-Herzog, V.P., and Halter, W.E., Quantitative Raman Spectroscopy: High-Temperature Speciation of Potassium Silicate Melts, J. Non-Cryst. Solids, 2007, vol. 353, pp. 4029–4042.
Brandiss, M.E. and Stebbins, J.E., Effect of Temperature on the Structures of Silicate Liquids: 29Si NMR Results, Geochim. Cosmochim. Acta, 1988, vol. 52, pp. 2659–2669.
Schneider, J., Mastelaro, V.R., Zanotto, E.D., Shakhmatkin, B.A., Vedishcheva, N.M., Wright, A.C., and Panepucci, H., On Distribution in Stoichiometric Silicate Glasses: Thermodynamic Calculations and 29Si High Resolution NMR Measurements, J. Non-Cryst. Solids, 2003, vol. 325, pp. 164–178.
Halter, W.E. and Mysen, B.O., Melt Speciation in the System Na2O-SiO2, Chem. Geol., 2004, vol. 213, pp. 115–123.
Koroleva, O.N., Thermodynamic Simulation and Raman Scattering Spectroscopy of Silicate Melts, Abstract of Cand. Sci. (Chem.) Dissertation, Yekaterinburg: Institute of Metallurgy, Ural Branch of the Russian Academy of Sciences, 2009 [in Russian].
Zakaznova-Herzog, V.P., Malfait, W.J., Herzog, F., and Halter, W.E., Quantitative Raman Spectroscopy: Principles and Application to Potassium Silicate Glasses, J. Non-Cryst. Solids, 2007, vol. 353, pp. 4015–4028.
Bale, C.W., Bélisle, E., Chartrand, P., Decterov, S.A., Eriksson, G., Hack, K., Jung, I.-H., Kang, Y.-B., Melançon, J., Pelton, A.D., Robelin, C., and Petersen, S., FactSage Thermochemical Software and Databases—Recent Developments, CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 2009, vol. 33, pp. 295–311.
Shakhmatkin, B.A. and Vedishcheva, N.M., A Thermodynamic Approach to the Modeling of Physical Properties of Oxide Glasses, Fiz. Khim. Stekla, 1998, vol. 24, no. 3, pp. 333–344 [Glass Phys. Chem. (Engl. transl.), 1998, vol. 24, no. 3, pp. 229–236].
Lin, C.-C, Chen, S.-F., Liu, L.-G., and Li, C.-C., Anionic Structure and Elasticity of Na2O-MgO-SiO2 Glasses, J. Non-Cryst. Solids, 2007, vol. 353, pp. 413–425.
Vedishcheva, N.M., Shakhmatkin, B.A., and Wright, A.C., The Structure-Property Relationship in Oxide Glasses: A Thermodynamic Approach, Adv. Mater. Res., 2008, vols. 39–40, pp. 103–110.
Ariskin, A.A., Shil’dt, A.A., and Polyakov, V.B., Simulation of Molecular-Mass Distributions of Si Anions on the Liquidus of the Na2O-SiO2 System with the Inclusion of Q Structons, Geokhimiya, 2009 (in press) [Geochem. Int. (Engl. transl.), 2009 (in press)].
Toop, G.W. and Samis, C.S., Activities of Ions in Silicate Melts, Trans. Metall. Soc. AIME, 1962, vol. 224, pp. 878–887.
Ottonello, G., Moretti, R., Marini, L., and Zuccolini, M.V., Oxidation State of Iron in Silicate Glasses and Melts: A Thermochemical Model, Chem. Geol., 2001, vol. 174, pp. 159–179.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.B. Polyakov, A.A. Ariskin, A.V. Shil’dt, 2010, published in Fizika i Khimiya Stekla.
Rights and permissions
About this article
Cite this article
Polyakov, V.B., Ariskin, A.A. & Shil’dt, A.V. Analysis of disproportionation of Q n structons in the simulation of the structure of melts in the Na2O-SiO2 system. Glass Phys Chem 36, 579–588 (2010). https://doi.org/10.1134/S108765961005007X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S108765961005007X