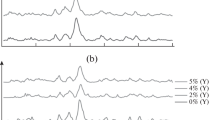
Abstract
Synthetic hydroxylapatites are prepared with additives, such as Mg2+, CO 2−3 , and C2O 2−4 . An increase in the concentration of magnesium leads to the formation of struvite. In the Ca(NO3)2-(NH4)HPO4-Na2CO3-NH4OH-H2O system, an excess of carbonate ions leads to the formation of calcite. When the synthesis is performed using oxalate ions as additives, calcium oxalate does not form the inherent phase. Calcium oxalate monohydrate is synthesized with additives, such as CO 2−3 , HPO 2−4 , and SO 2−4 ions and urea, glycine, and glutamic acid. X-ray powder diffraction analysis has revealed that the composition of the CaC2O4 · H2O precipitate remains unchanged under these conditions and in the presence of the aforementioned additives.
Similar content being viewed by others
References
Smolegovskii, A.M., Istoriya kristallokhimii fosfatov (History of the Crystal Chemistry of Phosphates), Moscow: Nauka, 1986 [in Russian].
Dorozhkin, S.V. and Epple, M., Biological and Medical Significance of Calcium Phosphates, Angew. Chem., Int. Ed. Engl., 2002, vol. 41, pp. 3130–3146.
Ferguson, J.F. and McCarty, P., Effects of Carbonate and Magnesium on Calcium Phosphate Precipitation, Environ. Sci. Technol., 1971, vol. 5, no. 6, pp. 534–540.
Pal’chik, N.A., Golovanova, O.A., Kachesova, P.A., Stolpovskaya, V.N., Grigor’eva, T.N., Nizovskii, A.I., and Shkuratov, S.S., The Phase and Elemental Compositions and the Occurrence of Urinary Stones (The Novosibirsk and Omsk Regions), Ural. Geol. Zh., 2002, no. 4, pp. 191–195.
Pyatanova, P.A., Physicochemical Investigation of Kidney Stones: The Formal Genesis, Candidate’s Dissertation in Chemistry, Omsk: Omsk State University, 2004.
Khamskii, E.V., Zelenkova, L.V., and Novikova, E.P., On the Initial Stage of the Deposition of Calcium Oxalate from Urea-Containing Solutions, Zh. Prikl. Khim. (St. Petersburg), 1990, no. 9, pp. 1976–1980.
Veresov, A.G., Putlyaev, V.I., and Tret’yakov, Yu.D., The Chemistry of Inorganic Biomaterials Based on Calcium Phosphates, Ross. Khim. Zh., 2004, vol. 48, no. 4, pp. 52–64.
Rabinovich, A.A., Golovanova, O.A., Bubnov, A.V., Trenikhin, M.V., and Ponomareva, E.A., Synthesis of Hydroxylapatite and the Influence of Additives on Its Structure, Khim. Khim. Tekhnol., 2007, vol. 50, no. 2, pp. 36–40.
Rabinovich, A.A., Golovanova, O.A., Izatulina, A.R., and Blinov, V.I., Simulation of the Formation of Calcium Oxalate Monohydrate in Living Organisms: The Influence of the Experimental Conditions on the Phase Composition of the Precipitate, Vestn. Omsk. Univ., 2006, no. 3, pp. 48–50.
Achkasova, E.Yu., Golovanova, O.A., Kogut, V.A., and Zhelyaev, E.V., Crystallization of Calcium Oxalate in the Presence of Organic Additives, in Mineralogiya tekhnogeneza (Mineralogy of Technogenesis), Miass: Institute of Mineralogy of the Ural Division of the Russian Academy of Sciences, 2005, pp. 137–139 [in Russian].
Tiktinskii, O.L. and Aleksandrov, V.P., Mochekamennaya bolezn’ (The Urinary Calculosis), St. Petersburg: Meditsina, 2000 [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Rabinovich, O.A. Golovanova, V.I. Blinov, 2008, published in Fizika i Khimiya Stekla.
Rights and permissions
About this article
Cite this article
Rabinovich, A.A., Golovanova, O.A. & Blinov, V.I. Influence of inorganic and organic additives on the composition of the precipitate of synthetic hydroxylapatite and calcium oxalate monohydrate. Glass Phys Chem 34, 332–335 (2008). https://doi.org/10.1134/S1087659608030152
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659608030152