Abstract
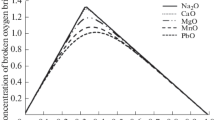
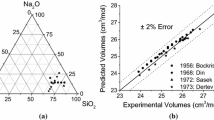
A strict scientific analysis of thermodynamics, kinetics, and mechanism of oxide melts, as well as metal and gas phase interaction, cannot be possible without studying the effect of wire former quantities and nature on the ionic composition and physicochemical properties of those melts. Quantitative evaluation of the ionic composition and thermodynamic properties of silicate systems can be based on the polymer theory. This paper concerns the methods of calculating components’ activity and ionic compositions in silicate melts, as based on the anion volume ratio. The design and experimental data on component activity in binary silicate melts are provided.
Similar content being viewed by others
References
Baes, C.F., A Polymer Model for BeF2 and SiO2 Melts, J. Solid State Chem., 1970, vol. 1, no. 2, pp. 159–169.
Esin, O.A. and Geld, P.V., Physical-Chemistry of Pyrometallurgical Processes, Moscow: Metallurgiya, 1966 [in Russian].
Esin, O.A., To Polymer Model of Ionic Melts, Phys. Chem. Metall. Proc., 1977, vol. 5, no. 76, pp. 4–24.
Elliot, J., Gleiser, M., and Ramakrishna, V., Thermochemistry for Steelmaking, Moscow: Metallrugiya, 1969 [in Russian].
Atlas of Slags (Handbook), Moscow: Metallrugy, 1985 [in Russian].
Author information
Authors and Affiliations
Additional information
This paper was presented at the Topical Meeting of the European Ceramic Society “Structural Chemistry of Partially Ordered Systems, Nanoparticles, and Nanocomposites,” St. Petersburg, Russia, June 27–29, 2006.
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Zalomov, N.I., Boronenkov, V.N., Zinigrad, M. et al. On calculating the activity of components in binary silicate melts. Glass Phys Chem 33, 459–463 (2007). https://doi.org/10.1134/S1087659607050045
Issue Date:
DOI: https://doi.org/10.1134/S1087659607050045