Abstract
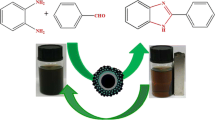
A sulfonated polyethylene glycol-supported nano-magnetite (Fe3O4@PEG-SO3H) has been prepared, characterized, and evaluated as a magnetically recoverable core–shell nanocomposite catalyst for the synthesis of bis(indolyl)methane derivatives through the one-pot reaction of indole and aldehydes. Green and eco-friendly reaction profile, mild reaction conditions (room temperature), simple procedure, short reaction times, and excellent yields make this method versatile and demonstrate its practical merit for the synthesis of the title compounds with potential pharmacological activity. Furthermore, the excellent catalytic performance, thermal stability, and easy separation and reusability of the prepared nanocatalyst without significant deterioration in its catalytic activity make it a good heterogeneous system and a superior alternative to the existing protocols.








Similar content being viewed by others
REFERENCES
Khatami, M., Alijani, H.Q., Nejad, M.S., and Varma, R.S., Appl. Sci., 2018, vol. 8, p. 411. https://doi.org/10.3390/app8030411
Gawande, M.B., Goswami, A., Asefa, T., Guo, H., Biradar, A.V., Peng, D.-L., Zboril, R., and Varma, R.S., Chem. Soc. Rev., 2015, vol. 44, p. 7540. https://doi.org/10.1039/C5CS00343A
Xiao, D., Lu, T., Zeng, R., and Bi, Y., Microchim. Acta, 2016, vol. 183, p. 2655. https://doi.org/10.1007/s00604-016-1928-y
Ghosh Chaudhuri, R. and Paria, S., Chem. Rev., 2012, vol. 112, p. 2373. https://doi.org/10.1021/cr100449n
Kothandapani, J. and Ganesan, S.S., Curr. Org. Chem., 2019, vol. 23, p. 313. https://doi.org/10.2174/1385272823666190312152209
Lisjak, D. and Mertelj, A., Prog. Mater. Sci., 2018, vol. 95, p. 286. https://doi.org/10.1016/j.pmatsci.2018.03.003
Zamani Kouhpanji, M.R. and Stadler, B.J., Sensors, 2020, vol. 20, article no. 2554. https://doi.org/10.3390/s20092554
Zhou, K., Zhou, X., Liu, J., and Huang, Z., J. Pet. Sci. Eng., 2020, vol. 188, article ID 106943. https://doi.org/10.1016/j.petrol.2020.106943
Mokhtary, M., J. Iran. Chem. Soc., 2016, vol. 13, p. 1827. https://doi.org/10.1007/s13738-016-0900-4
Mahmoudi, H., Jafari, A.A., Saeedi, S., and Firouzabadi, H., RSC Adv., 2015, vol. 5, p. 3023. https://doi.org/10.1039/C4RA11605D
Hassani, H., Nasseri, M., Zakerinasab, B., and Rafiee, F., Appl. Organomet. Chem., 2016, vol. 30, p. 408. https://doi.org/10.1002/aoc.3447
Maleki, A., Zand, P., and Mohseni, Z., RSC Adv., 2016, vol. 6, p. 110928. https://doi.org/10.1039/C6RA24029A
Esmaeilpour, M., Javidi, J., Zahmatkesh, S., and Fahimi, N., Monatsh. Chem., 2017, vol. 148, p. 947. https://doi.org/10.1007/s00706-016-1832-5
Abarghooei, M.A., Mohebat, R., Karimi-Jaberi, Z., and Mosslemin, M.H., J. Chem. Res., 2017, vol. 41, p. 408. https://doi.org/10.3184/174751917X14967701767012
Maleki, A. and Firouzi-Haji, R., Inorg. Nano-Met. Chem., 2019, vol. 49, p. 132. https://doi.org/10.1080/24701556.2019.1577258
Bonyasi, F., Hekmati, M., and Veisi, H., J. Colloid Interface Sci., 2017, vol. 496, p. 177. https://doi.org/10.1016/j.jcis.2017.02.023
Hayati, P., Eghbali, K., and Rezaei, R., Res. Chem. Intermed., 2019, vol. 45, p. 5067. https://doi.org/10.1007/s11164-019-03881-6
Ashraf, M.A., Liu, Z., Peng, W.-X., and Zhou, L., Catal. Lett., 2020, vol. 150, p. 1128. https://doi.org/10.1007/s10562-019-02973-7
Sharma, V., Kumar, P., and Pathak, D., J. Heterocycl. Chem., 2010, vol. 47, p. 491. https://doi.org/10.1002/jhet.349
Taber, D.F. and Tirunahari, P.K., Tetrahedron, 2011, vol. 67, p. 7195. https://doi.org/10.1016/j.tet.2011.06.040
Zhang, M.-Z., Chen, Q., and Yang, G.-F., Eur. J. Med. Chem., 2015, vol. 89, p. 421. https://doi.org/10.1016/j.ejmech.2014.10.065
Heravi, M.M., Rohani, S., Zadsirjan, V., and Zahedi, N., RSC Adv., 2017, vol. 7, p. 52852. https://doi.org/10.1039/C7RA10716A
Dadashpour, S. and Emami, S., Eur. J. Med. Chem., 2018, vol. 150, p. 9. https://doi.org/10.1016/j.ejmech.2018.02.065
Wan, Y., Li, Y., Yan, C., Yan, M., and Tang, Z., Eur. J. Med. Chem., 2019, vol. 183, article ID 111691. https://doi.org/10.1016/j.ejmech.2019.111691
Bhuiyan, M.M., Li, Y., Banerjee, S., Ahmed, F., Wang, Z., Ali, S., and Sarkar, F.H., Cancer Res., 2006, vol. 66, p. 10064. https://doi.org/10.1158/0008-5472.CAN-06-2011
Rahman, K.W., Ali, S., Aboukameel, A., Sarkar, S.H., Wang, Z., Philip, P.A., Sakr, W.A., and Raz, A., Mol. Cancer Ther., 2007, vol. 6, p. 2757. https://doi.org/10.1158/1535-7163.MCT-07-0336
Nachshon‐Kedmi, M., Fares, F.A., and Yannai, S., Prostate, 2004, vol. 61, p. 153. https://doi.org/10.1002/pros.20092
Rahman, K.W., Banerjee, S., Ali, S., Ahmad, A., Wang, Z., Kong, D., and Sakr, W.A., Cancer Res., 2009, vol. 69, p. 4468. https://doi.org/10.1158/0008-5472.CAN-08-4423
Cho, H.J., Park, S.Y., Kim, E.J., Kim, J.K., and Park, J.H.Y., Mol. Carcinog., 2011, vol. 50, p. 100. https://doi.org/10.1002/mc.20698
Fares, F., Azzam, N., Appel, B., Fares, B., and Stein, A., Eur. J. Cancer Prev., 2010, vol. 19, p. 199. https://doi.org/10.15430/JCP.2014.19.3.199
Bhatnagar, N., Li, X., Chen, Y., Zhou, X., Garrett, S.H., and Guo, B., Cancer Prev. Res., 2009, vol. 2, p. 581. https://doi.org/10.1158/1940-6207.CAPR-08-0142
Ali, S., Banerjee, S., Ahmad, A., El-Rayes, B.F., Philip, P.A., and Sarkar, F.H., Mol. Cancer Ther., 2008, vol. 7, p. 1708. https://doi.org/10.1158/1535-7163.MCT-08-0354
Banerjee, S., Wang, Z., Kong, D., and Sarkar, F.H., Cancer Res., 2009, vol. 69, p. 5592. https://doi.org/10.1158/0008-5472.CAN-09-0838
Ichite, N., Chougule, M.B., Jackson, T., Fulzele, S.V., Safe, S., and Singh, M., Clin. Cancer Res., 2009, vol. 15, p. 543. https://doi.org/10.1158/1078-0432.CCR-08-1558
Amare, D.E., Nutr. Diet. Suppl., 2020, vol. 12, p. 123. https://doi.org/10.2147/NDS.S261577
Rieck, G.C. and Fiander, A.N., Mol. Nutr. Food Res., 2008, vol. 52, p. 105. https://doi.org/10.1002/mnfr.200700138
Yu, T.-Y., Pang, W.-J., and Yang, G.-S., J. Pharmacol. Sci., 2015, vol. 127, p. 75. https://doi.org/10.1016/j.jphs.2014.11.006
Xue, L., Pestka, J.J., Li, M., Firestone, G.L., and Bjeldanes, L.F., J. Nutr. Biochem., 2008, vol. 19, p. 336. https://doi.org/10.1016/j.jnutbio.2007.05.004
Bharate, S.B., Bharate, J.B., Khan, S.I., Tekwani, B.L., Jacob, M.R., Mudududdla, R., Yadav, R.R., Singh, B., Sharma, P., and Maity, S., Eur. J. Med. Chem., 2013, vol. 63, p. 435. https://doi.org/10.1016/j.ejmech.2013.02.024
Shiri, M., Chem. Rev., 2012, vol. 112, p. 3508. https://doi.org/10.1021/cr2003954
Shiri, M., Zolfigol, M.A., Kruger, H.G., and Tanbakouchian, Z., Chem. Rev., 2010, vol. 110, p. 2250. https://doi.org/10.1021/cr900195a
Wu, Z., Wang, G., Yuan, S., Wu, D., Liu, W., Ma, B., Bi, S., Zhan, H., and Chen, X., Green Chem., 2019, vol. 21, p. 3542. https://doi.org/10.1039/C9GC01073D
Patil, M., Karhale, S., Bhenki, C., Kumbhar, A., and Helavi, V., React. Kinet. Mech. Catal., 2020, vol. 130, p. 993. https://doi.org/10.1007/s11144-020-01828-2
Kokare, N.D., Sangshetti, J.N., and Shinde, D.B., Chin. Chem. Lett., 2008, vol. 19, p. 1186. https://doi.org/10.1016/j.cclet.2008.07.015
Handy, S. and Westbrook, N.M., Tetrahedron Lett., 2014, vol. 55, p. 4969. https://doi.org/10.1016/j.tetlet.2014.07.024
Banari, H., Kiyani, H., and Pourali, A., Res. Chem. Intermed., 2017, vol. 43, p. 1635. https://doi.org/10.1007/s11164-016-2720-7
Hemmat, K., Nasseri, M.A., and Allahresani, A., Iran. J. Catal., 2019, vol. 9, p. 329. http://ijc.iaush.ac.ir/article_666535.html
Veisi, H., Mohammadi, P., and Ozturk, T., J. Mol. Liq., 2020, vol. 303, article ID 112625. https://doi.org/10.1016/j.molliq.2020.112625
Karimi-Jaberi, Z. and Hooshmandpour, A., Polycyclic Aromat. Compd., 2020, vol. 40, p. 432. https://doi.org/10.1080/10406638.2018.1441876
Bahrani, A. and Karimi-Jaberi, Z., Monatsh. Chem., 2019, vol. 150, p. 303. https://doi.org/10.1007/s00706-018-2313-9
Abarghooei, M.A., Mohebat, R., Karimi-Jaberi, Z., and Mosslemin, M.H., Catal. Commun., 2018, vol. 105, p. 59. https://doi.org/10.1016/j.catcom.2017.11.016
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interest.
Rights and permissions
About this article
Cite this article
Mardani, Y., Karimi-Jaberi, Z. & Soltanian Fard, M.J. Application of Magnetically Recoverable Core–Shell Nanocomposite in the Synthesis of Bis(indolyl)methanes at Room Temperature. Russ J Org Chem 57, 1740–1747 (2021). https://doi.org/10.1134/S1070428021100249
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428021100249