Abstract
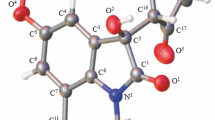
Reactions of alkyl acetoacetates, aromatic aldehydes, ammonium acetate, and 1,3-cyclohexanedione afford alkyl 4-aryl-2-methyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylates. The structure of compounds obtained was established with the help of IR, 1H NMR, and mass spectra and by X-ray diffraction analysis.
Similar content being viewed by others
References
Sapkal, S.B., Shelke, K.F., Shingate, B.B., and Shingare, M.S., Tethrahedron Lett., 2009, vol. 50, p. 1754.
By Yang, Xiao-Hui, Zhang, Ping-Hu, Zhou, Yong-Hong, Liu, Cheng-Guo, Lin, Xiao-Yu, and Cuia, Jing-Fang, ARKIVOC (United States), 2011, vol. 10, p. 327.
CrysAlis Software Package, Oxford Diffraction Ltd., Version 1.171.29.9.
Sheldrick, G.M., Acta Cryst., 2008, vol. A64, p. 112.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original English Text © V.L. Gein, M.I. Kazantseva, L.F. Gein, P.A. Slepukhin, 2014, published in Zhurnal Organicheskoi Khimii, 2014, Vol. 50, No. 2, pp. 247–250.
Rights and permissions
About this article
Cite this article
Gein, V.L., Kazantseva, M.I., Gein, L.F. et al. Synthesis of alkyl 4-aryl-2-methyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylates. Russ J Org Chem 50, 240–243 (2014). https://doi.org/10.1134/S107042801402016X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042801402016X