Abstract
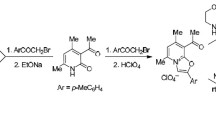
The reaction of NH2OH with the derivatives of 2,2,5,5-tetramethylpyrrolin-1-oxyl-3-carboxylic acid in all events led to the formation of a mixture of the corresponding nitroxylhydroxamic acid with a stable O-acylhydroxylamine. The ratio between the products depends on the nature of the acylating agent and under the studied conditions varies from ∼5.5: 1 to 1: 3 indicating the comparable nucleophilicity in this reaction of N and O atoms in the hydroxylamine. The most active chloride of the mentioned acid alongside the indicated products afforded in a considerable yield N,O-diacylhydroxylamine and the triacylated hydroxylamine, 3-{[(2,2,5,5-tetramethyl-1-oxylopyrrolin-3-yl)carbonyloxyimino][(2,2,5,5-tetramethyl-1-oxylopyrrolin-3-yl)carbonyloxy]-methyl}-2,2,5,5-tetramethylpyrrolin-1-oxyl. The structure of both latter compounds was established by XRD analysis.
Similar content being viewed by others
References
Sen’, V.D., Shilov, G.V., and Golubev, V.A., Zh. Org. Khim., 2008, vol. 44, p. 1193.
Marmion, C.J., Griffith, D., and Nolan, K.B. Eur. J. Inorg. Chem., 2004, p. 3003.
Soule, B.P., Hyodo, F., Matsumoto, K., Simone, N.L., Cook, J.A., Krishna, M.C., and Mitchell, J.B., Free Rad. Biol. Med., 2007, vol. 42, p. 1632.
Giacomelli, G., Porcheddu, A., and Salaris, M., Org. Lett., 2003, p. 2715.
Barlaam, B., Hamon, A., and Maudet, M., Tetrahedron Lett., 1998, vol. 39, p. 7865.
Pirrung, M.C. and Chau, J.H.-L., J. Org. Chem., 1995, vol. 60, p. 8084.
Sen’, V.D. and Golubev, V.A., J. Phys. Org. Chem., 2009, vol. 22, p. 138.
Litvin, E.F., Kozlova, L.M., Shapiro, A.B., Rozantsev, E.G., and Freidlin, L.Kh., Izv. Akad. Nauk SSSR, Ser. Khim., 1975, p. 1353.
Bauer, L. and Exner, O., Angew Chem., Int. Ed., 1974, vol. 13, p. 376.
Zhungietu, G.I. and Artemenko, A.I., Gidroksamovye kisloty i ikh proizvodnye (Hydroxamic Acids and Its Derivatives), Kishinev: Shtinitsa, 1986, p. 9.
Pilipenko, A.T. and Zul’figarov, O.S., Gidroksamovye kisloty (Hydroxamic Acids), Moscow: Nauka, 1989, p. 25.
Goettlicher, S. and Ochsenreiter, P., Chem. Ber., 1974, vol. 107, p. 398.
Schraml, J., Mindl, J., Roithova, J., Blechta, V., Sykora, J., Soukupova, L., Karban, J., Bartlova, M., and Exner, O., Organometallics, 2004, vol. 23, p. 2157.
Schraml, J., Sykora, J., Fiedler, P., Roithova, J., Mindl, J., Blechta, V., Cnsarova, I., and Exner, O., Org. Biomol. Chem., 2004, vol. 2, p. 2311.
Grassi, G., Cordaro, M., Bruno, G., and Nicolo, F., Helv. Chim. Acta, 2002, vol. 85, p. 196.
Buchachenko, A.L. and Vasserman, A.M., Stabil’nye radikaly (Stable Radicals), Moscow: Khimiya, 1973, 408 p.
Hudson, A. and Luckhurst, G., Chem. Rev., 1969, vol. 69, p. 191.
Jencks, W.P., J. Am. Chem. Soc., 1958, vol. 80, p. 4581.
Usova, E.M. and Voronin, E.M., Dokl. Akad. Nauk SSSR, 1957, vol. 113, p. 1306.
Exner, O., Coll. Czech. Chem. Commun., 1962, vol. 27, p. 2284.
Rozantsev, E.G. and Sholle, V.D., Organicheskaya khimiya svobodnykh radikalov (Organic Chemistry of Free Radicals), Moscow: Khimiya, 1979, p. 195.
Artemenko, A.I., Tikunova, I.V., Anufriev, E.K., Ehlicka, V., and Exner, O., Coll. Czech. Chem. Commun., 1981, vol. 46, p. 729.
Mazera, D.J., Gesser, J.C., and Pliego, J.R., ARKIVOC, 2007, vol. 15, p. 199.
Scott, A.W. and Wood, B.L., J. Org. Chem., 1942, vol. 7, p. 508.
Rozantsev, E.G. and Krinitskaya, L.A., Tetrahedron, 1965, vol. 21, p. 491.
Krinitskaya, L.A., Buchachenko, A.L., and Rozantsev, E.G., Zh. Org. Khim., 1966, vol. 2, p. 1301.
Hoffman, B.M., Schofield, P., and Pich, A., Proc. Nat. Acad. Sci. USA, 1969, vol. 62, p. 1195.
Griffith, O.H., Keana, J.F., Noall, D.L., and Ivey, J.L., Biochem. Biophys. Acta, 1967, vol. 148, p. 583.
Brauer, G., Ed., Handbuch der Praparativen Anorganischen Chemie, Stuttgart: Ferdinand Enke Verlag, 1978.
Sheldrick, G.M., SHELXTL, v. 6.14. Structure Determination Software Suite, Bruker AXS, Madison, WI, USA.
Sen’, V.D., Golubev, V.A., and Efremova, N.N., Izv. Akad. Nauk SSSR, Ser. Khim., 1982, p. 61.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.D. Sen’, G.V. Shilov, V.A. Golubev, 2009, published in Zhurnal Organicheskoi Khimii, 2009, Vol. 45, No. 8, pp. 1199–1208.
Rights and permissions
About this article
Cite this article
Sen’, V.D., Shilov, G.V. & Golubev, V.A. Synthesis and structure of products of hydroxylamine acylation with 3-carboxy-2,2,5,5-tetramethylpyrrolinoxyl derivatives. Russ J Org Chem 45, 1189–1199 (2009). https://doi.org/10.1134/S1070428009080132
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428009080132