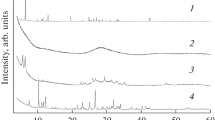
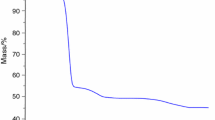
Abstract
Reactions of solid trihydroxopentaoxohexabismuth(III) pentanitrate trihydrate [Bi6O5(OH)3](NO3)5·3H2O and monoclinic bismuth oxide α-Bi2O3 with a succinic acid solution were studied with the aim of preparing a pharmaceutical substance, basic bismuth succinate С2Н4(СООBiO)2, meeting high purity requirements concerning the content of impurity metals. A procedure for preparing basic bismuth(III) succinate was suggested; it involves hydrolytic treatment of a process solution of bismuth in nitric acid to obtain high-purity [Bi6O5(OH)3](NO3)5·3H2O, its treatment with a NaOH solution to obtain finely crystalline α-Bi2O3, and the subsequent reaction of bismuth oxide with a succinic acid solution. The product composition was confirmed by X-ray diffraction, chemical analysis, IR and Raman spectroscopy, and thermal gravimetric analysis; the content of impurity metals was determined by the atomic absorption method.




Similar content being viewed by others
Notes
Pharmacopee Francaise. Bismuth (sous-succinate de).
REFERENCES
Yang, Y., Ouyang, R., Xu, L., Guo, N., Li, W., Feng, K., Ouyang, L., Yang, Z., Zhou, S., and Miao, Y., J. Coord. Chem., 2015, vol. 68, no. 3, pp. 379–397. https://doi.org/10.1080/00958972.2014.999672
Wang, R., Li, H., Ka-Yan Ip, T., and Sun, H., Adv. Inorg. Chem., 2020, vol. 75, pp. 183–205. https://doi.org/10.1016/bs.adioch.2019.10.011
Dore, M.P., Lu, H., and Graham, D.Y., Gut, 2016, vol. 65, no. 5, pp. 870–878. https://doi.org/10.1136/gutjnl-2015-311019
Loh, A., Ching Ong, Y., Blair, V.L., Kedzierski, L., and Andrews, P.C., J. Biol. Inorg. Chem., 2015, vol. 20, pp. 1193–1203. https://doi.org/10.1007/s00775-015-1299-6
Murphy, M.P. and O’Neill, L.A.J., Cell, 2018, vol. 174, no. 4, pp. 780–784. https://doi.org/10.1016/j.cell.2018.07.030
Manna, S.C., Zangrando, E., Bencini, A., Benelli, C., and Chaudhuri, N.R., Inorg. Chem., 2006, vol. 45, no. 22, pp. 9114–9122. https://doi.org/10.1021/ic060807d
Karthik, A.D. and Geetha, K., J. Appl. Pharm. Sci., 2013, vol. 3, no. 5, pp. 16–21. https://doi.org/10.7324/JAPS.2013.3504
Patent RU 2174508, Publ. 2001.
Galachiev, S.M., Makoeva, L.M., Dzhioev, F.K., and Khaeva, L.Kh., Izv. Samarsk. Nauchn. Tsentra Ross. Akad. Nauk, 2011, vol. 13, no. 1 (7), pp. 1678–1680.
Korostelev, P.P., Fotometricheskii i kompleksometricheskii analiz v metallurgii (Photometric and Complexometric Analysis in Metallurgy), Novosibirsk: Sib. Otdel. Ross. Akad. Nauk, 1984.
Yukhin, Yu.M. and Mikhailov, Yu.I., Khimiya vismutovykh soedinenii i materialov (Chemistry of Bismuth Compounds and Materials), Moscow: Sib. Otdel. Ross. Akad. Nauk, 2001.
Yukhin, Y.M., Mishchenko, K.V., and Daminov, A.S., Theor. Found. Chem. Eng., 2017, vol. 51, pp. 495–502. https://doi.org/10.1134/S0040579517040303
De Gelder, J., De Gussem, K., Vandenabeele, P., and Moens, L., J. Raman Spectrosc., 2007, vol. 38, pp. 1133–1147. https://doi.org/10.1002/jrs.1734
Krishnan, S., Raj, C.J., Priya, S.N., Robert, R., Dinakaran, S., and Das, S.J., Cryst. Res. Technol., 2008, vol. 43, no. 8, pp. 845–850. https://doi.org/10.1002/crat.200711102
Dhanya, V.S., Sudarsanakumar, M.R., Suma, S., Prasanna, S., Babu, K.R., Kumar, B.S., and Roy, S.M., J. Cryst. Growth, 2011, vol. 319, pp. 96–101. https://doi.org/10.1016/j.jcrysgro.2011.01.073
Arankumar, A., Ramasamy, P., Vishnu, K., and Jayaray, M.K., J. Mater. Sci., 2014, vol. 49, pp. 3598–3607. https://doi.org/10.1007/s10853-013-7858-8
Caires, F.J., Lima, L.S., Carvalho, C.T., and Ionashiro, M., Thermochim. Acta, 2010, vol. 500, pp. 6–12. https://doi.org/10.1016/j.tca.2009.11.015
Deacon, G.B. and Phillips, R.J., Coord. Chem. Rev., 1980, vol. 33, pp. 227–250. https://doi.org/10.1016/S0010-8545(00)80455-5
Matskevich, N.I., Semerikova, A.N., Gelfond, N.V., Matskevich, M.Yu., Tkachev, E.N., Anufrieva, O.I., and Martynetz, V.G., Russ. J. Inorg. Chem., 2020, vol. 65, pp. 743–746. https://doi.org/10.1134/S0036023620050162
Yokobayashi, H., Nagase, K., and Muraishi, K., Bull. Chem. Soc. Jpn., 1975, vol. 48, pp. 2789–2792. https://doi.org/10.1246/bcsj.48.2789
Patent RU 27402592, Publ. 2021.
ACKNOWLEDGMENTS
The authors are grateful to the Center for Shared Use High Technologies and Nanosystems Analytics at the Novosibirsk State University for the opportunity to use the equipment for recording the Raman spectra.
Funding
The study was performed in accordance with the thematic plan of the Institute of Solid State Chemistry and Mechanochemistry, Siberian Branch, Russian Academy of Sciences (121032500064-8) and Novosibirsk State Technical University (TP-KhKhT-1_21).
Author information
Authors and Affiliations
Contributions
E.V. Timakova: development of the concept of the study and experimental procedure, collection, analysis, and interpretation of the results, and participation in manuscript preparation; E.I. Bun’kova: all the syntheses, collection of literature data, preparation of literature review, and participation in manuscript preparation; L.I. Afonina: studies of the chemical composition of the samples obtained and participation in processing and interpretation of the results and in manuscript preparation; Yu.M. Yukhin: consulting on planning of enlarged laboratory trials and their guidance, participation in interpretation of the results, and major contribution to writing the section on the procedure for preparing basic bismuth succinate; V.A. Volodin: Raman and IR studies of the samples and interpretation of the spectroscopic data obtained.
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Translated from Zhurnal Prikladnoi Khimii, No. 7, pp. 857–866, January, 2021 https://doi.org/10.31857/S0044461821070069
Rights and permissions
About this article
Cite this article
Timakova, E.V., Bun’kova, E.I., Afonina, L.I. et al. Synthesis of High-Purity Basic Bismuth(III) Succinate as a Pharmaceutical Substance. Russ J Appl Chem 94, 911–919 (2021). https://doi.org/10.1134/S1070427221070077
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427221070077